Abstract
Tamoxifen has long been the standard endocrine therapy for postmenopausal women with hormone-sensitive breast cancer. However, data now suggest that the third-generation aromatase inhibitors have emerged as superior alternatives for advanced disease. In early disease evaluation of initial adjuvant therapy, data from the Arimidex®, Tamoxifen, Alone and in Combination trial has shown that anastrozole is more effective than tamoxifen with a better risk–benefit profile. This trial provides the most mature data of any aromatase inhibitor study and suggests that anastrozole should be considered the preferred initial adjuvant therapy for postmenopausal women with hormone-responsive early breast cancer. The emergence of aromatase inhibitors as an alternative to tamoxifen for the treatment of breast cancer is challenging the management of the disease and influencing the change of regulatory guidelines.
Breast cancer is the most prevalent cancer in women (23% of all cancers) and in 2002 was estimated to be the second most common cancer worldwide in terms of new cases (1.15 million) [1]. Despite a reduction in death rates in recent years [101], breast cancer still leads to significant mortality. For the past three decades, tamoxifen has been the standard treatment for estrogen receptor (ER)-positive breast cancer. However, data now suggest that the third-generation aromatase inhibitors (AIs); anastrozole, exemestane and letrozole, are more effective than tamoxifen and have the potential to improve outcomes.
Anastrazole: a third-generation aromatase inhibitor
Overview of the market
Since tamoxifen entered the market for the treatment of breast cancer, its use has broadened from the treatment of metastatic disease to the treatment of early stage breast cancer and prevention of disease in high-risk women. Despite its proven efficacy, tamoxifen therapy is associated with a number of serious and potentially life-threatening adverse events, including an increased risk of endometrial cancer and thromboembolic disorders [2–4]. These shortfalls have led to the search for alternative agents with equal or improved efficacy and better tolerability profiles.
The third-generation AIs first became a potential treatment option for postmenopausal women with advanced and early breast cancer in the 1990s. Compared with first- and second-generation AIs, these agents showed increased potency with respect to both aromatase inhibition and subsequent estrogen suppression [5,6]. Anastrozole and letrozole are both reversible nonsteroidal AIs and exemestane is an irreversible steroidal AI. Anastrozole has the broadest licensing of the third-generation AIs, but letrozole and exemestane have also been investigated and approved in different disease settings. Letrozole is approved as extended adjuvant therapy for women who have already received 5 years of tamoxifen and as first- or second-line treatment for advanced disease. Exemestane, the latest AI to undergo clinical investigation, is approved for the treatment of advanced breast cancer in patients who have progressed following tamoxifen therapy.
In postmenopausal women, the ER antagonist fulvestrant provides an alternative endocrine option for the treatment of hormone receptor-positive, metastatic breast cancer following progression with prior estrogen therapy. Data are accumulating to suggest that fulvestrant is also effective after treatment with nonsteroidal AIs [7–9].
Introduction to anastrozole
Anastrozole (Arimidex®, AstraZeneca) is the most advanced third-generation AI in clinical development. This review summarizes the pharmacology and clinical efficacy of anastrozole with a focus on recent clinical data on the use of anastrozole as an adjuvant therapy for early breast cancer.
Chemistry
Anastrozole (1,3-benzenediacetonitrile, α, α, α′, α′-tetramethyl-5-[1H-1,2,4-triazol-1-ylmethyl]) is a potent and selective benzyltriazole derivative.
It has a molecular weight of 293.4 and a molecular formula C17H19N5 (Figure 1). Anastrozole has moderate aqueous solubility (0.5 mg/ml at 25°C) independent of pH in the physiologic range. It is freely soluble in methanol, acetone, ethanol, and tetrahydrofuran, and very soluble in acetonitrile.

The chemical structure of anastrozole.
Pharmacodynamics
Estrogen stimulates tumor growth via receptors on the breast tumor cells and, in women, estrogens are derived from the conversion of androgens via the aromatase enzyme complex in peripheral tissues. In breast tumor tissue, anastrozole suppresses the formation of estrogen through selective inhibition of aromatase. Anastrozole has a very high intrinsic potency in vitro with an inhibitory concentration 50% (IC50) of 15 nM, and is approximately 200 times more potent than aminoglutethimide at inhibiting aromatase [10].
Pharmacodynamic data from Phase I clinical studies are presented in Table 1. In postmenopausal female volunteers and patients with advanced breast cancer, maximal suppression of plasma estrogens occurred after 3–4 days of anastrozole treatment (0.5–1.0 mg), with reductions in estradiol of approximately 80% of baseline values [11–13]. The long-term effects of doses of anastrozole 1 mg/day on plasma estrogen levels were compared with the effects of the second-generation AI, formestane, in postmenopausal women with advanced breast cancer [14]. Anastrozole provided constant and reliable estradiol suppression over a 4-week period and, compared with formestane, produced greater suppression of serum estradiol, estrone and estrone sulfate levels. Suppression of plasma estrogen levels by anastrozole was also shown to be similar in Japanese and Caucasian women, indicating that the therapeutic benefits of anastrozole in Caucasians are predictive of the drug's effects in Japanese women [15].
Summary of Phase I trial results.
ACTH: Adrenocorticotropic hormone.
Anastrozole is highly effective in inhibiting in vivo aromatization. In a cross-over study including postmenopausal women progressing after tamoxifen treatment, anastrozole 1 mg was shown to inhibit whole body aromatase activity by more than 95% after 28 days' treatment [16]. In another study, the suppression of estrone and estrone sulfate was greater during treatment with letrozole compared with anastrozole [5]. Although this difference in the degree of suppression was significant, the efficacy of AIs does not appear to directly correlate with their potency [18]. The effect of anastrozole on intratumoral aromatase has also been studied in postmenopausal women [17]. This trial demonstrates that suppression of plasma estrogen levels and inhibition of total body aromatization by anastrozole is accompanied by a similar reduction in tumor estrogen levels.
Pharmacokinetics & metabolism
The pharmacokinetic characteristics of anastrozole are presented in Table 2. After oral administration, anastrozole is rapidly and completely absorbed from the gastrointestinal tract, with peak plasma concentrations reached after 2 h [11,13]. The estimated half-life is 40–50 h, which is consistent with a once-daily dosing regimen [11].
Main pharmacokinetic characteristics of anastrozole.
Clinical efficacy
Efficacy data for anastrozole have been derived from Phase III clinical trials in first- and second-line advanced breast cancer and adjuvant treatment settings (Table 3).
Summary of key published efficacy results for anastrozole.
The combination arm was closed because of low efficacy after the analysis at 47 months' median follow-up, results are presented for the monotherapy arms only
Results are presented here only for those patients who were considered inoperable or not eligible for breast-conserving surgery.
CI: Confidence interval; DDFS: Distant disease-free survival; DFS: Disease-free survival; DRFS: Distant recurrence-free survival
EFS: Event-free survival; HR: Hazard ratio; HR+: Hormone receptor-positive; MA: Megestrol acetate; N: Number of patients; NS: Not significant; OR: Odds ratio; ORR: Objective response rate; OS: Overall survival; RFS: Recurrence-free survival; TTDR: Time to distant recurrence; TTP: Time to progression; TTR: Time to recurrence.
Second-line treatment of advanced breast cancer
Two large, multicenter Phase III trials of identical design compared anastrozole (1 and 10 mg/day) with megestrol acetate (40 mg, four times daily) in postmenopausal women with advanced breast cancer who had progressed on tamoxifen [31,32]. A prospectively planned, combined analysis of these trials after a median follow-up of 6 months demonstrated that anastrozole (1 and 10 mg/day) was at least as effective as megestrol acetate for time-to-progression and objective response (complete response [CR] + partial response [PR]) [23]. The estimated progression hazard ratios (HRs) were 0.97 (95% confidence interval [CI]: 0.75–1.24) for anastrozole 1 mg versus megestrol acetate and 0.92 (95% CI: 0.71–1.19) for anastrozole 1 mg versus megestrol acetate. A total of 27 patients (10.3%) in the anastrozole 1 mg group, 22 (8.9%) in the anastrozole 10 mg group and 20 (7.9%) in the megestrol acetate group had either a CR or PR to treatment. A total of 66 (25.1%), 56 (22.6%) and 66 (26.1%) patients in the respective groups had stable disease for 24 weeks or longer. Furthermore, in a survival update at 31 months' median follow-up, a statistically significant survival advantage was reported in the group of women receiving anastrozole 1 mg versus megestrol acetate (HR 0.78; 95% CI: 0.60–1.0; p < 0.025) [24].
In the only head-to-head study directly comparing third-generation AIs as second-line treatment for advanced disease, letrozole conferred no clinical benefit over anastrozole (p = 0.216) [33]. There were no significant differences between treatment in the primary efficacy end point of time-to-progression (p = 0.92) or in the secondary end point of overall survival (p = 0.624). In the overall population, the secondary end point, objective response rate, significantly favored letrozole (19.1 vs 12.3% for anastrozole; p = 0.013), but there was no significant difference between treatments in the more clinically relevant target population of women with hormone receptor-positive disease.
First-line treatment of advanced breast cancer
Two trials, the North American [26] and Tamoxifen or Arimidex Randomized Group Efficacy and Tolerability (TARGET) [25] trials, examined anastrozole (1 mg/day) versus tamoxifen (20 mg/day) as first-line endocrine therapy in postmenopausal women with advanced breast cancer. Efficacy results demonstrated clear advantages for anastrozole compared with tamoxifen. In the North American trial, anastrozole showed a statistically significant advantage over tamoxifen in median time-to-progression (11.1 vs 5.6 months for anastrozole and tamoxifen, respectively; p = 0.005). Anastrozole was as effective as tamoxifen in terms of objective response (21 vs 17% of patients, respectively), with clinical benefit observed in 59% of patients receiving anastrozole and 46% receiving tamoxifen (p = 0.0098). In the TARGET trial, anastrozole was at least as effective as tamoxifen with time-to-progression values of 8.2 and 8.3 months for anastrozole and tamoxifen, respectively. Anastrozole was also as effective as tamoxifen in terms of objective response (32.9 vs 32.6% of patients, respectively). Clinical benefit rates were 56.2 and 55.5% for patients receiving anastrozole and tamoxifen, respectively.
A prospectively planned combined analysis of the hormone receptor-positive patients from both trials demonstrated that anastrozole treatment was associated with a significantly longer median time-to-progression compared with tamoxifen (10.7 vs 6.4 months for anastrozole and tamoxifen, respectively; p = 0.022) [34].
Adjuvant therapy for early breast cancer
Anastrozole is currently the only AI with data available over the full 5-year treatment period in the adjuvant therapy setting [27,35,36]. In the Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial, 9366 postmenopausal women with histologically proven operable invasive breast cancer who had completed primary surgery and chemotherapy (where given), were randomized to receive adjuvant anastrozole (1 mg/day), tamoxifen (1 mg/day) or a combination for a period of 5 years. The first analysis of the ATAC trial, performed at a median follow-up of 33 months, demonstrated that anastrozole was superior to tamoxifen in terms of the primary efficacy end point, disease-free survival, and also with respect to time to recurrence and incidence of contralateral breast cancer [35]. An updated analysis at a median follow-up of 47 months showed that the early efficacy benefits of anastrozole were maintained [36].
At 68 months' median follow-up, the completed treatment analysis of the ATAC trial reinforced previous analyses [35,36] and confirmed that anastrozole provided additional benefits over and above those provided by tamoxifen [27]. In the overall population, treatment with anastrozole led to significant improvements in disease-free survival, time to recurrence and time to distant recurrence, and significantly reduced contralateral recurrence compared with tamoxifen (Figure 2). A greater advantage was seen in disease-free survival and time to recurrence in hormone receptor-positive patients [27]. Notably, the benefits of anastrozole were observed throughout the 68-month follow-up period, including during the first 3 years, when the peak of recurrence well described for tamoxifen was not observed with anastrozole (Figure 3) [37]. The completed treatment analysis also provides robust confirmation that there is no interaction between chemotherapy previously received and anastrozole [38]. It is still too early to know whether anastrozole will demonstrate a long-term survival benefit compared with tamoxifen in the treatment of postmenopausal women with early breast cancer.
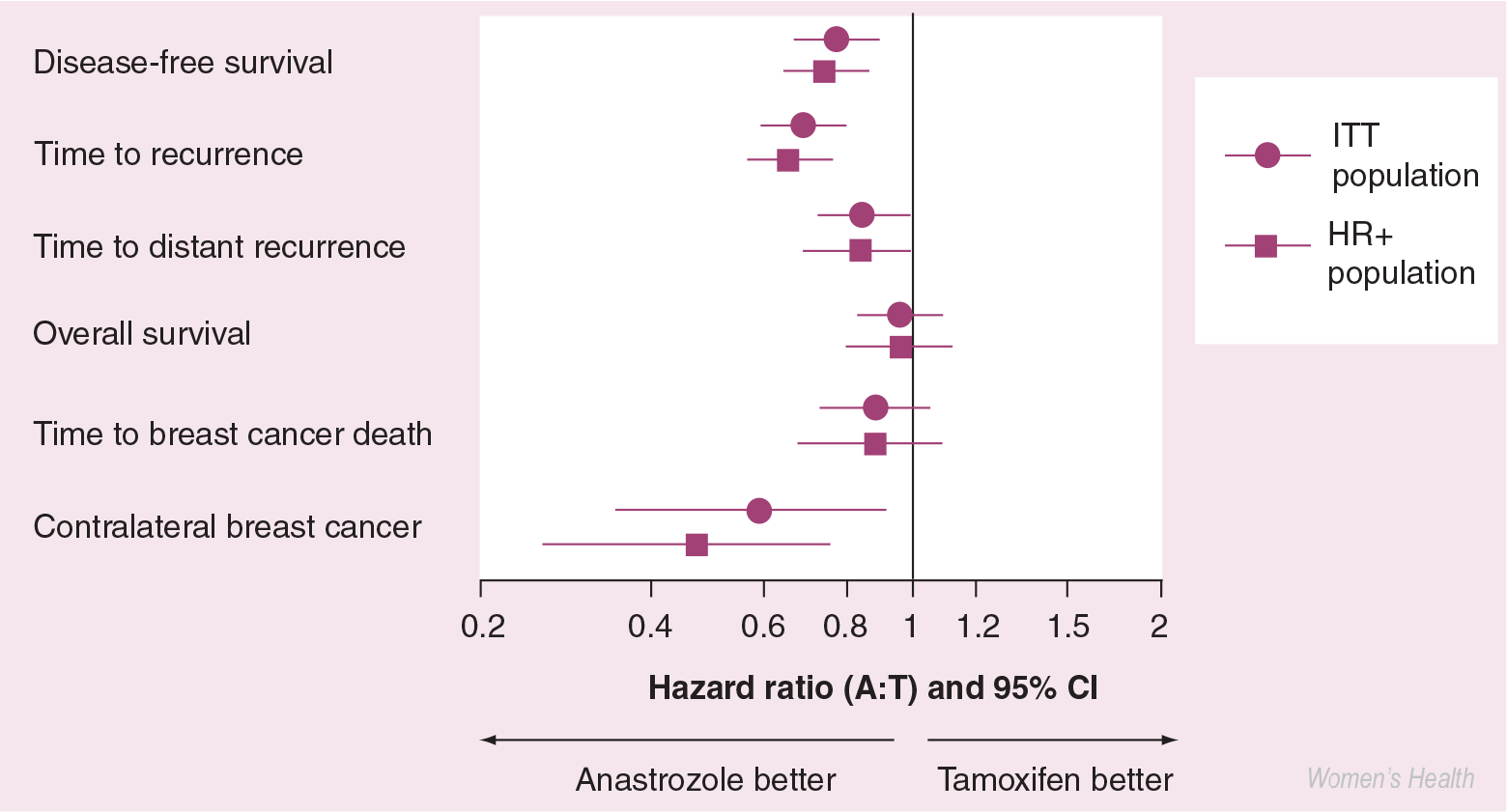
Forest plot showing efficacy end points from the ATAC trial.
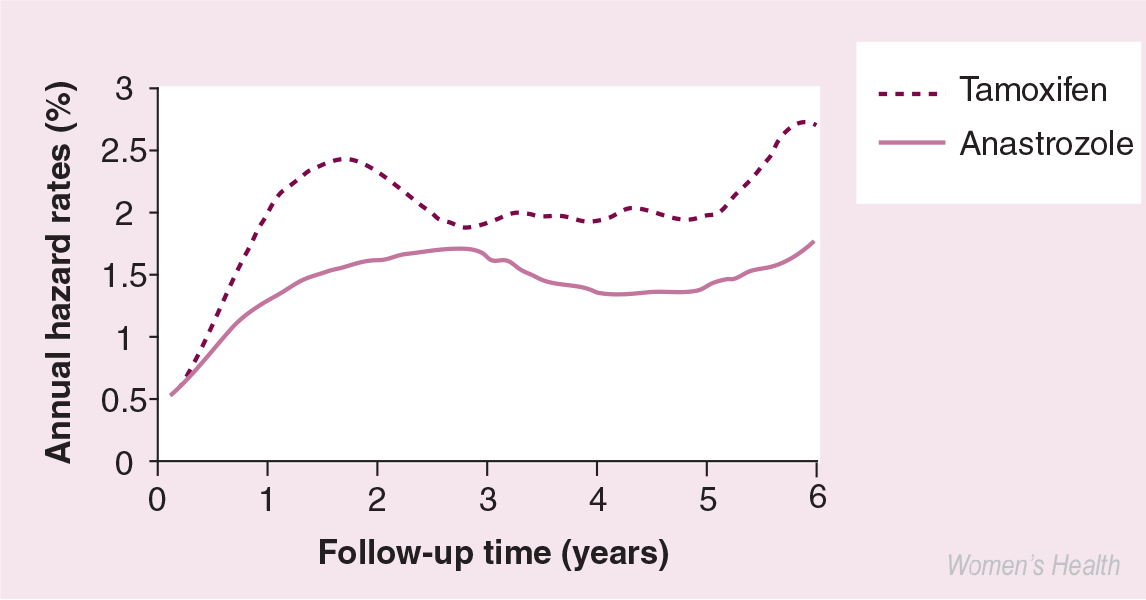
Smoothed hazard rates of recurrence for anastrozole versus tamoxifen treatment in hormone receptor-positive patients from the ATAC trial.
Adjuvant therapy for women who have already received tamoxifen
Until recently, it has not been clear whether the advantages of anastrozole over tamoxifen observed in the ATAC trial can be extrapolated to women already receiving adjuvant tamoxifen. The Italian Tamoxifen Arimidex (ITA) trial, which included 448 postmenopausal women with ER-positive disease, was the first to show that patients can still receive the benefits of anastrozole by changing therapy from tamoxifen to anastrozole after 2–3 years rather than remaining on tamoxifen for the full 5-year treatment period [29]. Although findings from this study were based on a small number of events following tamoxifen therapy, further investigation is warranted to fully establish treatment differences between anastrozole and tamoxifen in this setting.
The results of the Austrian Breast & Colorectal cancer Study Group (ABCSG) 8/ARimidex-NOlvadex (ARNO) 95 study were derived from a combined analysis of more than 3000 patients who, following primary surgery and exposure to 2 years of tamoxifen, received either 3 years of tamoxifen or 3 years of anastrozole [28]. The trial aimed to prospectively assess whether switching postmenopausal women with hormone receptor-positive early breast cancer from adjuvant tamoxifen to anastrozole at 2 years is more effective than continuing on adjuvant tamoxifen. The 3-year event-free survival favored switching to anastrozole (HR 0.60; 95% CI: 0.44–0.81; p = 0.0009). Anastrozole also showed a 39% improvement in distant recurrence-free survival compared with tamoxifen (HR 0.61; 95% CI: 0.42–0.87; p = 0.0067), clearly demonstrating that switching treatment to anastrozole, subsequent to 2 years of tamoxifen, is superior to continuing tamoxifen. The benefits of such treatment were irrespective of nodal status and age.
Patients who have completed a 5-year course of tamoxifen can extend their protection against breast cancer by undergoing a further 3 years of treatment with anastrozole [39]. Data from study ABCSG 6a demonstrated that, following 5 years of adjuvant tamoxifen, extended adjuvant treatment with anastrozole for 3 years reduced the risk of disease recurrence (local, metastatic or contralateral) compared with no treatment (30 vs 56 patients, respectively; HR 0.64; 95% CI: 0.41–0.99; p = 0.047).
Preoperative therapy for early breast cancer
The primary aim of preoperative (neoadjuvant) therapy is to decrease tumor size, thereby enabling breast-conserving surgery in patients with large, operable breast tumors and rendering inoperable tumors resectable. Anastrozole has been compared with tamoxifen alone or with a combination of both anastrozole and tamoxifen in the preoperative treatment of ER-positive operable breast cancer in postmenopausal women in the IMmediate Preoperative Arimidex, tamoxifen or Combined with Tamoxifen (IMPACT) trial [40]. Treatment with preoperative anastrozole was more likely to downstage tumors, enabling more patients in the anastrozole group to have breast-conserving surgery compared with the tamoxifen group (46 vs 22%; p < 0.05). The benefits of preoperative anastrozole have been confirmed in the PReOperative Arimidex Compared with Tamoxifen (PROACT) trial, in which more patients receiving anastrozole than tamoxifen showed improvement with respect to the actual surgery performed (43 vs 31%; p < 0.05) [41]. In a prospectively planned combined analysis of the population common to both trials, the objective response, assessed by caliper, was significantly greater in anastrozole- than in tamoxifentreated patients (47 vs 35%; p < 0.05) [30].
Chemoprevention
On studying the mature 5-year adjuvant anastrozole data, one of the most notable observations is the substantial reduction in incidence of contralateral breast cancer compared with tamoxifen, particularly in the hormone receptor-positive patient population (53% reduction; 95% CI: 25–71; p = 0.001) [27]. Since tamoxifen demonstrates a 50% reduction in contralateral disease in hormone-receptor positive patients compared with placebo [4], it is feasible that anastrozole has the potential to prevent (or delay) up to 80% of these tumors in women at high risk of breast cancer. These observations in the adjuvant setting, in addition to the superiority of anastrozole over tamoxifen in the advanced disease and preoperative settings, have led to the launch of the International Breast Cancer Intervention Study (IBIS) II [42].
Postmarketing surveillance
A postmarketing surveillance program is continuing to assess anastrozole in the adjuvant setting. Ongoing blinded assessment of patients participating in the ATAC trial for 10 years will enhance understanding of the risk–benefit profile of anastrozole. Several trials, including Letrozole, Exemestane and Anastrozole Pharmacodynamics (LEAP) trial and Study of Arimidex with the Bisphosphonate ResidronatE (SABRE), are also being conducted to evaluate safety parameters of anastrozole, including the further investigation of postmenopausal bone loss and lipid parameters.
Safety & tolerability
As long-term safety and tolerability data are particularly important in the adjuvant setting, where patients typically receive 5 or more years of endocrine therapy, this section of the review will concentrate on adverse-event data in this setting. Predefined adverse events in the ATAC trial at 68 months' median follow-up (Table 4) [27], when 90% or more patients had completed treatment, occurred with similar relative frequency to adverse events at the 33– and 47-month analyses [35,36], although the absolute incidence of adverse events increased with longer follow-up.
Predefined adverse events during treatment or within 14 days of discontinuation after 5 years of adjuvant treatment for early breast cancer in the ATAC trial for anastrozole compared with tamoxifen.
n = 222 and n = 2236 for anastrozole and tamoxifen, respectively (excluding patients with hysterectomy at baseline), recorded at any time
Patients with one or more fractures occurring at any time before recurrence (includes patients no longer receiving treatment)
In favor of tamoxifen
Patients may have had one or more fracture at different sites.
ATAC: Arimidex, Tamoxifen, Alone or in Combination trial; CI = confidence interval.
Adapted from [27], reprinted with permission from The Lancet, 365, ATAC Trialists' Group, Results of the ATAC trial after completion of 5 years' adjuvant treatment for breast cancer, 60–62, ©2005 with permission from Elsevier.
Anastrozole was associated with a number of important tolerability benefits over tamoxifen, including significant reductions in the incidence of endometrial cancer (5 vs 17%; p = 0.02), thromboembolic events (venous, 2.8 vs 4.5%, p = 0.0004; deep venous, 1.6 vs 2.4, p = 0.02, respectively) and ischemic cerebrovascular events (2.0 vs 2.8%; p = 0.03). Importantly, and due to the current concern about a link between the use of aromatase inhibitors and cardiovascular toxicity [5,6,43,44], it is reassuring to see that there is no difference in the number of cardiac deaths between anastrozole (n = 49) and tamoxifen (n = 46). The only adverse events with significantly increased risks associated with anastrozole therapy compared with tamoxifen were joint disorders (35.6 vs 29.4; p < 0.0001) and fractures (11.0 vs 7.7; p < 0.0001). However, from a clinical perspective, the increased fracture risk with anastrozole compared with tamoxifen may be considered an acceptable alternative to the increased risk of endometrial malignancy or cerebrovascular/thromboembolic morbidity. Patients at increased risk of fracture can be identified and managed proactively according to consensus guidelines.
It has been postulated that, during long-term use in the adjuvant setting, differences in selectivity between the AIs may affect clinical safety and tolerability. These differences may be due to the different chemical structures of the AIs and, although the clinical significance is unknown, the American Society of Clinical Oncology (ASCO) panel noted that ‘closely related agents with similar mechanisms of action may have different toxicity profiles’ [45]. Indeed, evidence for this is emerging and there now appear to be differences in the safety of AIs in the adjuvant setting (Table 5) [27,43,44]. Anastrozole has the most mature adverse-event data of all the third-generation AIs and, overall, data obtained in clinical studies of anastrozole in all treatment settings show that it has a better tolerability profile than tamoxifen.
Tolerability characteristics of the third-generation aromatase inhibitors.
The trials reported are of different designs and include different patient populations; therefore, any cross-trial comparisons should be interpreted with caution.
−: Significant decrease compared with tamoxifen; +: Significant increase compared with tamoxifen.
ATAC: Arimidex, Tamoxifen, Alone or in Combination trial; BIG: Breast International Group; IES: Intergroup Exemestane Study; NR: Not reported; NS: Not significantly different to tamoxifen.
Regulatory affairs
Anastrozole was first indicated for the treatment of advanced breast cancer in postmenopausal women with disease progression following tamoxifen therapy and then for first-line treatment of postmenopausal women with hormone receptor-positive or hormone receptor-unknown locally advanced or metastatic breast cancer. It is now also approved for adjuvant treatment (treatment following surgery with or without radiation) of postmenopausal women with hormone receptor-positive early breast cancer.
Conclusion
Third-generation AIs are now established as the endocrine treatment of choice for advanced breast cancer, while in the adjuvant setting, results of the ATAC trial have shown that anastrozole is superior to tamoxifen in terms of disease-free survival, time to recurrence, time to distant recurrence and prevention of contralateral breast cancer in postmenopausal women with early breast cancer. These benefits are also coupled with a more favorable safety and tolerability profile than tamoxifen.
As new data emerge, treatment guidelines assess the relevance of the research findings and translate these into recommendations for clinical practice. In 2004, the ASCO Technology Assessment recommended that 5 years of tamoxifen alone was no longer the optimal adjuvant treatment for hormone-sensitive early breast cancer and that the preferred treatment should include the use of an AI to reduce the risk of tumor recurrence [45]. Furthermore, the ASCO assessment favors using the AI that has been most studied in the setting most closely approximating to any individual patient's clinical circumstance. The National Comprehensive Cancer Network (NCCN) practice guidelines [102] recommend using the drug with the best available data. The NCCN recommends 5 years of anastrozole as initial adjuvant therapy or a switch to anastrozole after an initial 2–3 years tamoxifen for a completed course of 5 years' hormonal therapy (Figure 4).

Schema demonstrating the position of anastrozole in NCCN 2005 Practice Guidelines* for adjuvant hormonal therapy of postmenopausal women with early breast cancer.
It is now clear that third-generation AIs have assumed an important role in the optimal management of postmenopausal patients with breast cancer. However, physicians and patients will continually need to reconsider emerging data alongside current data and guidelines when making treatment strategy decisions.
Future perspective
The introduction of potent third-generation AIs has changed the treatment of early and advanced breast cancer. These agents are superior to tamoxifen but many questions remain to be answered regarding their exact place in the sequence of treatments given for hormone receptor-positive breast cancer. Although anastrozole reduces early relapse (Figure 3), there is no proof that 5 years of anastrozole is superior to 2–3 years of tamoxifen followed by a switch to an AI. Initial use, compared with sequencing, is being investigated with letrozole in the Breast International Group (BIG)-1–98 trial, but the final results of this trial will not be available for 2 years. Another important question is how long should the duration of treatment with anastrozole be? Although the National Cancer Institute of Canada MA-17 trial indicates that an average of 2–3 years of letrozole is superior to placebo after 5 years of tamoxifen [46], no data are available now, or will be in the foreseeable future, to indicate whether we should treat for more (or less) than 5 years with an AI, beginning after surgery.
Angela Brodie's group have reported interesting data in a model system where human mammary tumor (MCF)-7 cells transfected with the aromatase gene are grown as tumors in immune-deprived mice treated with various endocrine therapies. In this model, continuous letrozole is superior to the sequence of tamoxifen and letrozole, which may presage the results of the BIG-1–98 trial [47]. In addition, the combination of letrozole and the pure anti-estrogen fulvestrant has been shown to provide markedly better tumor control than either drug alone [48]. The combination of an AI and fulvestrant is currently being tested in advanced breast cancer and, if successful, may be tested as adjuvant therapy, since it is possible that fulvestrant may prevent AI resistance. There is also evidence from in vitro studies that AI resistance may be produced by upregulation of growth factor pathways, and trials are in progress to test the combination of AIs with growth factor inhibitors (e.g., gefitinib) to prevent resistance.
In all adjuvant trials where the incidence of contralateral breast cancer has been reported, the AIs show a reduction of new cancers of approximately 50%. This is in addition to the 50% already obtained with tamoxifen compared with placebo or no treatment [4]. Thus, AIs may reduce contralateral breast cancer by 70–80% and are good candidates for use in prevention trials such as IBIS II (anastrozole) and MA3 (exemestane).
Although AIs show a superior safety and tolerability profile to tamoxifen, there are still problems with vaginal dryness and dyspareunia, hot flashes, joint pains and bone fractures. It appears likely that the bone loss caused by all three modern AIs (anastrozole, exemestane and letrozole) can be prevented or reversed by the use of bisphosphonates. The ABCSG group, for example, demonstrated that 6-monthly treatment with zoledronic acid 4 mg was able to prevent the bone loss induced by treatment with goserelin and anastrozole in premenopausal women [49]. Additional data are required on the protective effect of this and other bisphosphonates in pre-and postmenopausal women, while further investigation is necessary to determine how to treat joint aches that are problematic in a minority of AI-treated women. Long-term safety and tolerability data in the current large trials are also essential: to this end, follow-up beyond 5 years in the ATAC trial remains blinded.
Information resources
American Society of Clinical Oncology is a comprehensive resource covering latest news, meetings and education, policy and practice guidelines
(Accessed October 2005)
Breast Cancer Care is the leading provider of breast cancer information and support across the UK
(Accessed October 2005)
BreastCancerSource is an international information resource for healthcare professionals with an interest in breast cancer treatment, provided by AstraZeneca
(Accessed October 2005)
Breast Cancer Online is an independent, peer-reviewed educational website managed by an editorial board of experts and sponsored by AstraZeneca. It provides education, information, news and support for both clinical and nonclinical professionals working in the field of breast cancer
(Accessed October 2005)
Cancer Research UK provides background information on cancer in general and details of specific types of cancers. The science and research pages cover funding and give details of institutes and centers
(Accessed October 2005)
Cancer.gov is the USA National Institute of Cancer website providing detailed information on all types of cancer. The support and resources sections are US-specific
(Accessed October 2005)
The National Comprehensive Cancer Network (NCCN), an alliance of 19 of the world's leading cancer centers, is an authoritative source of information to help patients and healthcare professionals make informed decisions about cancer care. Through the collective expertise of its member institutions, the NCCN develops, updates and disseminates a complete library of clinical practice guidelines
(Accessed October 2005)
Executive summary
Anastrozole 1 mg results in near maximal suppression of estradiol levels and effectively suppresses estrogens during long-term treatment of breast cancer.
The absorption of anastrozole is rapid and complete. Anastrozole is extensively metabolized and has no known drug interactions.
Anastrozole has demonstrated a statistically significant mature survival benefit compared with megestrol acetate as a second-line therapy for advanced breast cancer.
Anastrozole is an effective first-line alternative treatment option to tamoxifen for hormone receptor-positive advanced breast cancer.
For postmenopausal women with early breast cancer, data from the Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial demonstrate that adjuvant anastrozole treatment provides significant improvements in disease-free survival, time to recurrence and time to distant recurrence as well as reduced contralateral recurrence compared with tamoxifen.
Anastrozole has a favorable tolerability profile and is generally well tolerated as a long-term adjuvant treatment.
The American Society of Clinical Oncology (ASCO) Technology Assessment now recommends that optimal treatment should include the use of an aromatase inhibitor to reduce the risk of tumor recurrence. The assessment favors using the agent that has been most studied in the setting most closely approximating to any individual patient's clinical circumstance.
The ATAC trial provides the most mature data of any aromatase inhibitor trial and suggests that anastrozole should be considered the preferred initial adjuvant therapy for postmenopausal women with hormone-responsive early breast cancer.
Footnotes
Acknowledgements
The author would like to thank Mark Walker PhD from the Cumberland Medical Center (CMC) for his editorial assistance.
