Abstract
Letrozole is a type 2 aromatase inhibitor, which reduces availability of estrogen in postmenopausal women, thereby decreasing its ability to stimulate breast cancer cells. Phase III trials in both the advanced and early breast cancer setting have shown an improvement in disease-free survival compared with other compounds, including tamoxifen. Letrozole is well-tolerated, with the main adverse effects reported as hot flushes, arthritis, arthralgia and myalgia, and a trend towards increased risk of fracture.
Breast cancer comprises 22.9% of all cancers (excluding nonmelanoma skin cancers) in women [1]. Estrogen deprivation was first reported to cause regression of breast tumors in 1896 [2]. Since then, hormone receptors have been found and targeted as part of conventional treatment of breast cancer. Letrozole is part of a family of compounds called the aromatase inhibitors (AIs) that reduce the availability of estrogen in the serum and tissue, and hence decrease stimulation of hormone-driven tumor cells. This review will examine the mechanism of action of letrozole, as well as its pharmacodynamics, efficacy and safety.
Pharmacology
Aromatase is an enzyme of the CYP450 family and catalyzes the conversion of estrone from androstenedione and estradiol from testosterone [3]. It is present in high concentrations in the granulosa cells of ovarian tissue and in lower concentrations in adrenal gland, fat, liver, muscle, brain and breast tissue [4]. In postmenopausal women, estrogen production is reliant on synthesis from these non-ovarian tissues and it is therefore possible, through inhibition of aromatase, to decrease estrogen levels [4].
The first AI to be investigated in the treatment of breast cancer was aminoglutethimide in the 1970s [5]. However, it lacked selectivity for aromatase, requiring concomitant steroid replacement, and had significant toxicities including lethargy and rash [6]. Therefore, it was not widely used, but its discovery paved the way for newer, more selective compounds. There are two different classes of AIs (
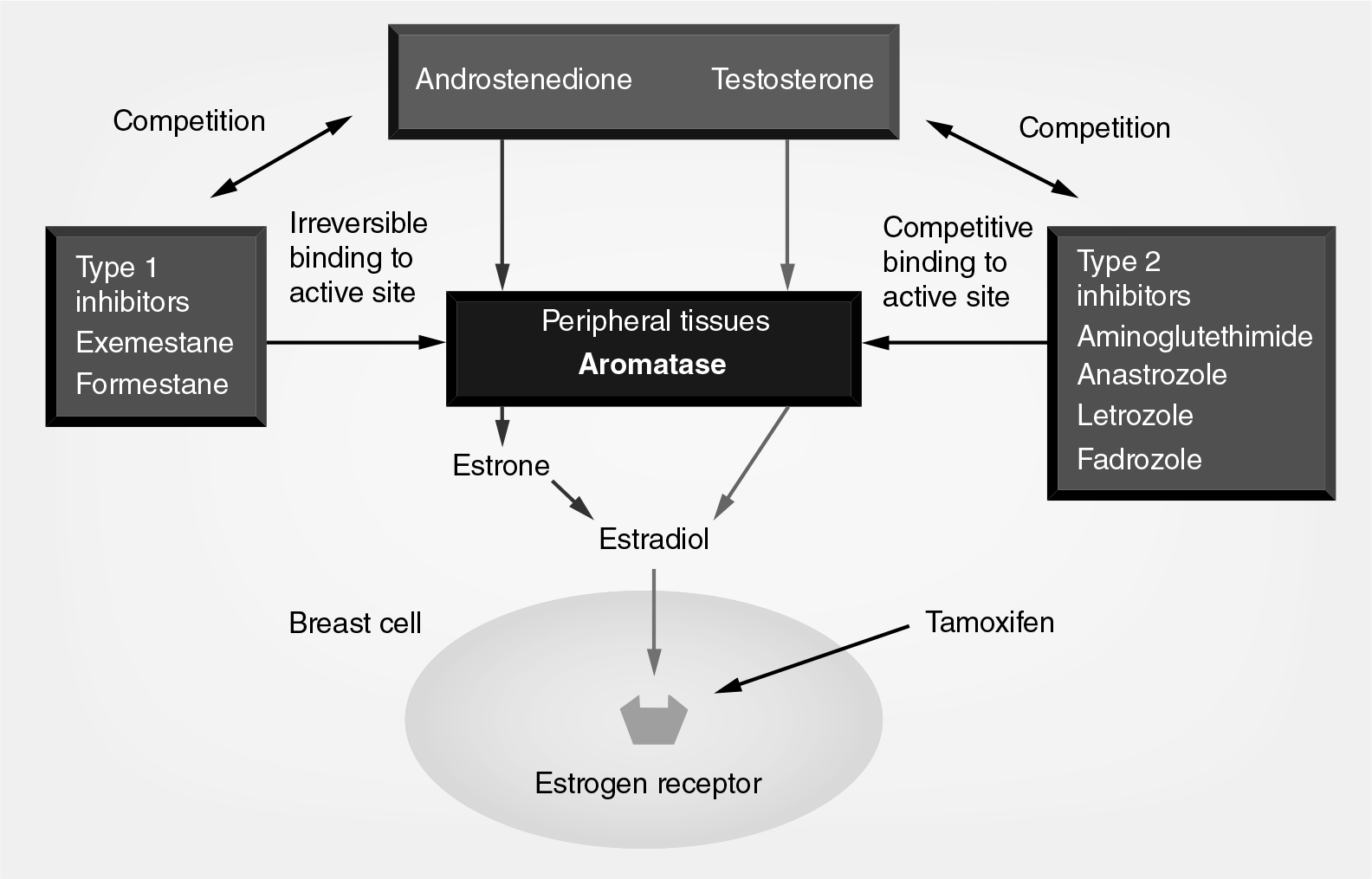
Mechanism of action and classes of aromatase inhibitors.
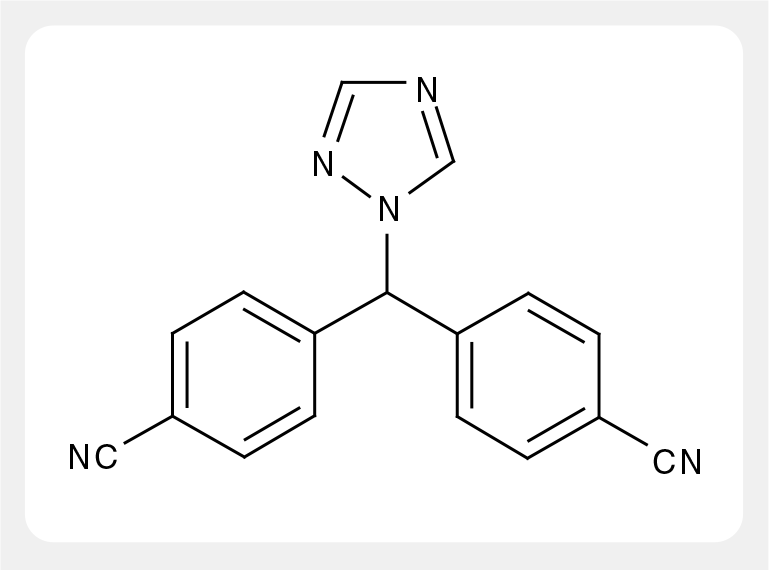
Letrozole.
Animal models
Bhatnagar
Pharmacokinetic properties of letrozole
In two initial Phase I studies, letrozole was assessed in young men and healthy postmenopausal women [8,9]. No significant side effects were reported in either study [8,9]. Doses up to 30 mg have not shown any effect on aldosterone and corticosterone levels, however, higher doses (10–30 mg) increased follicle-stimulating hormone, luteinizing hormone and testosterone in young male volunteers, thought to be due to the longer-lasting aromatase inhibition at these doses [9]. A dose of 2.5 mg was found to be optimal, and at this dose the time taken to reach maximum estradiol suppression was 2–4 days [10]. Letrozole has been found to take up to 60 days to achieve steady-state plasma levels and tends to accumulate over time [10]. One study found that the mean plasma concentration of letrozole increased by 34% after 84 days of treatment at 0.5 mg daily [11]. Therefore, there may be a nonlinear relationship between dose and efficacy [10].
Studies have found letrozole to be a potent suppressor of estrone and estradiol serum levels. Bisagni
Other third generation AIs – anastrozole and exemestane – have been found to decrease estrone plasma levels by 81–94% and 52–72%, respectively [10]. In a randomized double-blind crossover trial, Geisler
Drug–drug interactions
Letrozole has been found to interact with a variety of enzymes within the cytochrome (CYP) system. It strongly inhibits CYP2A6 and moderately inhibits CYP2C19, while its metabolite has moderate affinity for CYP2C19 and CYP2B6 [14]. Therefore, there is potential for interactions with drugs that also interact with these enzymes. However, CYP2A6 does not play a major role in drug metabolism and an
Phase II studies
A number of Phase II trials showed that letrozole had antitumor activity in metastatic breast cancer (MBC) [11,12,16,17]. The largest trial conducted by Ingle
Bajetta
Tominga
Phase III studies
A number of randomized controlled trials in the Phase III setting have been performed; these are summarized in
Phase III trials using letrozole.

At 5 years.
After 5 years of tamoxifen.
At 4 years.
At 30 months with crossover allowed.
Analyzed at 9 months after enrollment of the last patient.
Analyzed at 15 months after enrollment of the last patient.
At 30 months.
DFS: Disease-free survival; OS: Overall survival; TTP: Time to progression.
Locally advanced or metastatic disease
Initial studies compared letrozole versus megesterol acetate (MA) and the first-generation aromatase inhibitor aminoglutethimide in the setting of advanced or progressive disease [18–20]. A further study by the International Letrozole Breast Cancer Group compared letrozole with tamoxifen in this setting [21].
The Letrozole International Trial Group performed an open-label randomized trial comparing letrozole 2.5 and 0.5 mg daily, and aminoglutethimide in postmenopausal women with advanced breast cancer [18]. In total, 555 women were enrolled who had either relapsed on adjuvant therapy (given for >6 months) or had progressed during anti-estrogen treatment for metastatic disease (one line of chemotherapy for advanced disease was allowed). Of the patients, 185 received 2.5 mg letrozole once daily, 192 received letrozole 0.5 mg daily and 178 received aminoglutethimide 250 mg twice daily with corticosteroid support. Overall ORRs of 19.5, 16.7 and 12.4% were seen for letrozole 2.5 mg, 0.5 mg and aminoglutethimide, respectively [18]. These differences were not significant, although a trend of benefit was seen for letrozole 2.5 mg compared to aminoglutethimide (odds ratio: 1.85; 95% CI: 0.97–3.51; p = 0.06 [18]). The median duration of response and SD lasting at least 6 months was longest for letrozole 2.5 mg (21 months) compared with letrozole 0.5 mg (18 months) and aminoglutethimide (14 months) [18].
Two studies looked at letrozole versus MA. Dombernowsky
The International Letrozole Breast Cancer Group performed a Phase III study randomizing 916 women with locally advanced breast cancer or MBC to receive either letrozole 2.5 mg daily (n = 458) or tamoxifen 20 mg daily (n = 458) until disease progression [21]. Optional crossover was permitted, with 233 patients in the letrozole arm and 226 in the tamoxifen arm crossing over at disease progression. Their primary end point of median TTP was 6.0 months for tamoxifen versus 9.4 months for letrozole (HR: 0.72; p = 0.0001) [21]. ORR was significantly higher for the letrozole arm: 32%, compared with 21% for the tamoxifen arm (odds ratio: 1.78; p = 0.0002) [21]. At a median follow-up of 32 months, there was a nonsignificant difference in median overall survival (OS) of 34 months for the letrozole arm versus 30 months for the tamoxifen arm. However, after crossover, median OS for those who received tamoxifen second-line was 19 months (95% CI: 17–24) compared with 31 months (95% CI: 22–40) for those who received letrozole second line [21].
Early-stage breast cancer trials
Two large studies, the Breast International Group (BIG) 1–98 trial and MA17 study, have examined letrozole in the adjuvant, early-stage breast cancer setting [22,23].
BIG 1–98 trial
BIG 1–98 was a randomized, Phase III, double-blind trial that compared 5 years of treatment with regimens containing either letrozole or tamoxifen or both in postmenopausal women with hormone receptor-positive breast cancer [22]. A total of 8010 women were randomly assigned on an ITT basis. Initially, 1828 patients were randomized to receive monotherapy with letrozole or tamoxifen for 5 years, then a further 6182 patients were randomized to receive either tamoxifen monotherapy, letrozole monotherapy each for 5 years or letrozole for 2 years followed by tamoxifen for 3 years, or tamoxifen for 2 years followed by letrozole for 3 years. 133 of the patients were considered to be ineligible after randomization, but were included in the ITT analysis, and a further 47 did not receive any treatment and were excluded from the analysis of safety and toxicity.
The primary end point was met at 25.8 months median follow-up when the disease-free survival (DFS) was significantly greater in the letrozole group than in the tamoxifen group (hazard ratio [HR]: 0.81; 95% CI: 0.70–0.93; p = 0.003 by the log-rank test) [22]. Of particular note, there was a significant decrease in the risk of distant recurrence with letrozole compared with tamoxifen (HR: 0.73; 95% CI: 0.60–0.88; p = 0.001) [22]. In addition, planned subgroup analysis showed that DFS was greater in the letrozole group for women treated with chemotherapy (HR: 0.70; 95% CI: 0.54–0.92; p = 0.01), those who did not receive radiotherapy (HR: 0.77; 95% CI: 0.61–0.98; p = 0.03) and those who had node-positive disease (HR: 0.71; 95% CI: 0.59–0.85; p < 0.001) [22].
In an updated efficacy analysis, at a median follow-up of 8.1 years from randomization (range: 0–12.4), letrozole monotherapy was significantly better than tamoxifen, whether by inverse probability of censoring weighting (IPCW) or ITT analysis [23]. IPCW analysis showed DFS HR of 0.82 (95% CI: 0.74–0.92), OS HR of 0.79 (95% CI: 0.69–0.90), distant recurrence-free interval (DRFI) HR of 0.79 (95% CI: 0.68–0.92) and breast cancer-free interval (BCFI) HR of 0.80 (95% CI: 0.70–0.92) [23]. ITT analysis showed DFS HR of 0.86 (95% CI: 0.78–0.96), OS HR of 0.87 (95% CI: 0.77–0.99), DRFI HR of 0.86 (95% CI: 0.74–0.998) and BCFI HR of 0.86 (95% CI: 0.76–0.98) [23]. At a median follow-up of 8 years from randomization (range: 0–11.2) for the comparison of the sequential groups with letrozole monotherapy, there were no statistically significant differences in any of the four end points for either sequence. The 8-year ITT estimates (each with SE ≤1.1%) for letrozole monotherapy, letrozole followed by tamoxifen and tamoxifen followed by letrozole were: 78.6, 77.8 and 77.3% for DFS; 87.5, 87.7 and 85.9% for OS; 89.9 88.7 and 88.1% for DRFI; and 86.1, 85.3 and 84.3% for BCFI [23].
MA17 trial
This was a double-blind, placebo-controlled trial examining the effectiveness of 5 years of letrozole therapy in postmenopausal women with breast cancer who had completed 5 years of tamoxifen therapy [24]. Of the total 5187 women enrolled, 2593 were assigned to the letrozole arm and 2594 to placebo. The trial was terminated early after the first interim analysis when 171 events had occurred [24]. Following unblinding, 61% of patients in the placebo arm elected to crossover and receive 5 years of letrozole and 31.1% decided to have no further treatment [24]. At the event-driven first analysis at median follow-up of 2.4 years, they estimated 4-year DFS rates of 93% for the letrozole group and 87% for the placebo group [24]. The HR for a local or metastatic recurrence, or a new contralateral breast cancer occurence in the letrozole group as compared with the placebo group was 0.57 (95% CI: 0.43–0.75; p = 0.00008) [24]. There was a trend for increased OS in the letrozole arm; however, this was not statistically significant in the initial data at unblinding (p = 0.25) [24]. Longer-term data with 64 months follow-up with the data adjusted for crossover using IPCW showed a letrozole versus placebo DFS HR of 0.52 (95% CI: 0.45–0.61; p = 0.001), a distant DFS (DDFS) HR of 0.51 (95% CI: 0.42–0.61; p = 0.001) and an OS HR of 0.61 (95% CI: 0.52–0.71; p = 0.001) in favor of letrozole compared to placebo [24]. A second method to adjust for crossover based on a Cox model with a time-dependent treatment covariate (the SCC approach [24]) showed very similar results: HRs for letrozole versus placebo were 0.58 (95% CI: 0.47–0.72; p < 0.001) for DFS, 0.68 (95% CI: 0.52–0.88; p = 0.004) for DDFS and 0.77 (95% CI: 0.61–0.97; p = 0.03) for OS [24].
Neoadjuvant studies
The P024 trial was a Phase IIb–III double-dummy, randomized study comparing daily doses of letrozole 2.5 mg or tamoxifen 20 mg in 337 postmenopausal women with hormonepositive invasive breast cancer in the neoadjuvant setting [25]. Patients were enrolled if they were not considered to be suitable for breast-conserving surgery (BCS) and 14% of the patients were considered inoperable [25]. They found that ORR in terms of clinical palpation was 55% in the letrozole group, compared with 36% for the tamoxifen group, which was statistically significant (p < 0.001). Statistical significance for letrozole was seen in ultrasound response (35 vs 25% in the tamoxifen arm; p = 0.042), mammographic response (34 vs 16%; p < 0.001) and number of patients who were candidates for BCS following treatment (45 vs 35%; p = 0.022) [25].
Comparison with other third-generation AIs
To date, there have been no published head-to-head studies of letrozole, anastrozole and exemestane in the adjuvant setting, although the FACE trial comparing letrozole with anastrozole in postmenopausal women with hormone receptor- and node-positive breast cancer has now finished recruitment and results of follow-up are awaited. There is only one study comparing letrozole versus anastrozole in advanced disease [26]. In addition, there has been one Phase II study comparing anastrozole, exemestane and letrozole in the neoadjuvant setting [27].
Rose
Ellis
Safety
Letrozole has been to found to be generally well-tolerated and to have a relatively favorable safety profile.
In the BIG 1–98 trial, there were more adverse events of any grade reported in the letrozole group (2912 patients) than in the tamoxifen group (2554 patients) but the number of fatal events was similar (67 and 69, respectively) [22]. Fractures were significantly more frequent in the letrozole group than in the tamoxifen group (5.7 vs 4.0%; p < 0.001). Letrozole was associated with fewer thromboembolic events (1.5 vs 3.5%; p < 0.001), a lower rate of vaginal bleeding (3.3 vs 6.6%; p < 0.001) and fewer invasive endometrial cancers (0.1 vs 0.3%; p = 0.18) [22].
Hypercholesterolemia was higher in the letrozole monotherapy group than the tamoxifen monotherapy group (29.9 vs 41.4–53.2%; p < 0.001) [22]. However, the MA17 trial did not show an increased incidence of hypercholesterolemia in women taking letrozole compared with placebo (p = 0.67) [24]. In addition, in the BIG 1–98 trial, the incidence of cardiac events of any type or grade was similar between tamoxifen and letrozole monotherapy groups at 71 months follow-up (6.1–7.0% and 5.7%, respectively; p = 0.45) [22].
In the MA17 trial, grade I/II hot flashes, arthritis, arthralgia and myalgia were more common in the letrozole group than in the placebo group (p < 0.05 for all comparisons) [24]. The difference in rates of discontinuation of treatment due to toxic effects was not significant: 4.5% in the letrozole group compared with 3.6% in the placebo group (p = 0.11) [24]. Although there was a trend towards more bone fractures in women in the letrozole group, this was not significant (p = 0.24). Rates of cardiovascular events were not significantly different (p = 0.40) [24].
Expert commentary
Letrozole is a third-generation, type 2 AI shown to be a potent and selective inhibitor of aromatase
The safety and tolerability of letrozole has been shown to be reasonable with the most frequently reported side effects such as hot flushes, arthralgia, nausea and fatigue being grade I/II. Letrozole was shown to have an increased fracture rate in the BIG 1–98 trial compared with tamoxifen and a trend was seen in the MA17 trial towards increased fractures with letrozole compared with placebo, although this was not significant. This is an expected risk from the mechanism of action as estradiol is a key factor in regulation of bone homeostasis. AI-induced bone loss is well recognized and guidelines are present for its management [29]. Increased rates of hypercholesterolemia seen in the BIG 1–98 relative to tamoxifen need to be viewed in the perspective of the improved OS from letrozole compared with tamoxifen. Available data do not support the concept of a substantial risk from AIs from a cardiovascular perspective [30]. As with bone health, there is a need to ensure a holistic approach and to ensure appropriate monitoring of cholesterol and other cardiac risk factors for patients on letrozole as with all other people.
Conclusion
Letrozole has been found to be an effective option for treatment of postmenopausal women with hormone receptor-positive early breast cancer and MBC. Letrozole improves OS and DFS compared with tamoxifen in early breast cancer. Safety and tolerability are reasonable, although monitoring of bone health and cholesterol levels are advised.
Financial & competing interests disclosure
Executive summary
Letrozole is a type 2 nonsteroidal aromatase inhibitor that prevents synthesis of estrogen from nonovarian tissues.
In the MA17 trial, letrozole had a 4-year disease-free survival for placebo of 87%, compared with 93% for letrozole.
In the BIG 1–98 trial, letrozole improved disease-free and overall survival compared with tamoxifen; hazard ratio: 0.81.
Letrozole is well-tolerated but is associated with hot flashes, arthritis, arthralgia, myalgia and a trend towards increased rates of bone fractures.

