Abstract
Mixed urinary incontinence accounts for 33% of all incontinence and is the involuntary loss of urine associated with the sensation of urgency; it is also associated with exertion, sneezing or coughing. Riskfactors include vaginal delivery, obesity, age and possible genetic factors. Treatment includes lifestyle changes, behavioral therapies, medication and nerve modulation. Surgery with midurethral slings can cure both stress and urge components in 40–50% of cases. Future therapies may include new medications adapting potassium and calcium channels and more widespread use of sacral neuromodulation. This reviewfocuses on the investigation and optimal management of mixed urinary incontinence.
The International Continence Society defines urinary incontinence as ‘involuntary leakage of urine’ [1]. Mixed urinary incontinence (MUI) is defined as the ‘involuntary loss of urine associated with the sensation of urgency and also associated with exertion, effort, sneezing or coughing’ [1]. MUI accounts for 33% of the incontinence that is seen in daily clinical practice [2] and affects women of all ages. It has been estimated that there are more than 3.5 million sufferers in the UK [3]. Using the Kings Health Questionnaire, Duggan found that of the three main types of incontinence, MUI had the greatest adverse effect on quality of life [4]. Treatment of the predominant and most troublesome component of MUI usually achieves good results and most women respond to conservative measures. The group of women nonresponsive to standard treatment pose a challenge.
Pathophysiology
The cause of MUI is unclear, but it is likely to be due to multiple etiologies that contribute in varying degrees to the condition. Prevalence of MUI increases after middle-age. It is unclear whether this is a cause-and-effect relationship but Resnick created the mnemonic ‘DIAPPERS’ to describe possible associations of geriatric incontinence: delirium, infection, atrophic change, pharmacological, psychological, excess urine production, restricted mobility and stool impaction [5].
Brown et al. found that both stress incontinence and mixed incontinence were more common during pregnancy than urgency incontinence alone [6]. Prevalence of incontinence during pregnancy varies from 6 to 67% and prevalence 2–3 months after delivery varies from 3 to 38% [7]. There is an acknowledged link between stress incontinence and pregnancy. During pregnancy, this may be due to pressure and stretching of the pelvic floor and changes in hormone levels. After delivery, this damage is secondary to pudendal neuropathy in addition to damage of the pelvic floor and urethral sphincter muscles during labor.
There is an association between adult urinary incontinence and childhood enuresis shown by D'Ancona et al. [8]. The study confirmed that individuals with MUI and urge urinary incontinence (UUI) in adult life were more likely to have a history of enuresis in childhood than those with stress urinary incontinence.
It has been suggested that there is a genetic contribution to pelvic floor disorders, such as incontinence and prolapse. A twin study conducted in Denmark evaluated 1168 female twins and found that heritability of urgency incontinence was 42% among middle-aged women and 49% among the elderly group. It also found MUI had a substantial genetic factor [9]. There is evidence that stress incontinence has a genetic factor, but the influence of environmental factors is substantial [10].
Genetic susceptibility seems to be due to differences in elastin metabolism and remodeling [11,12] and collagen fiber make-up, which have been found in women with stress urinary incontinence when compared with women not presenting with any sign or history of urinary incontinence or overactive bladder (OAB). There is also evidence that androgen receptor polymorphism is linked to the development of incontinence [13], although larger studies to explore genetic links are needed.
Urgency in an older person with restricted mobility can cause anxiety as they worry if they will be able to reach the bathroom before leaking. This can lead to a cycle of voiding more frequently to prevent urgency incontinence, leading to reduced bladder capacity and possible worsening of their symptoms [14].
Several studies have shown a link between obesity and incontinence. In 1988 it was shown by Dwyer et al. that obesity is more common in patients with urinary stress incontinence and detrusor overactivity (DO) than in the asymptomatic population [15]. In a recent study, women who lost between 5 and 10% of their bodyweight showed significant improvement in urinary continence for both stress and urge episodes [16].
There is a possibility that mixed symptoms may be due to a more severe form of stress rather than two separate mechanisms for urge and stress incontinence [17]. Another theory is that DO is caused by a weak urethral sphincter mechanism, resulting in funneling of the proximal urethra. When intra-abdominal pressure is increased, urine enters the proximal urethra, producing sensory stimulation and resulting in a reflex bladder contraction. Other mechanisms could be that increased intra-abdominal pressure results in stretching of the pelvic nerves, and this triggers an involuntary bladder contraction [18]. There is also a possibility of an urethral element that induces bladder contraction rather than a primary bladder abnormality [19].
Investigations
Initial diagnosis of urinary incontinence can be made by most health professionals, but a detailed urogynecological history should be taken to assess if the incontinence should be classified as urgency, stress or mixed incontinence, taking note of the definitions laid out by the International Continence Society [1]. A good clinical history will include enquiries about the impact of the disease on quality of life including personal, social and sexual relationships.
Clinical examination includes abdominal palpation to exclude abdominal mass and should involve a digital assessment of the pelvic floor muscle contraction and examination for pelvic organ prolapse. Neurological assessment is essential if a neurological cause is suspected.
A urine dipstick should be performed to look for evidence of infection, glycosuria and hematuria. A mid-stream urine specimen should be sent leukocytes and nitrites are detected in the urine dipstick and if the patient shows symptoms of a urinary tract infection [101]. If hematuria is found, urgent investigations should be undertaken as this could indicate an underlying malignancy or calculi.
Postvoid residual volumes of urine in the bladder should be measured. Ultrasound is the preferred method as it is noninvasive and does not carry the risk of introducing infection. A postvoid residual volume of more than 50 ml indicates there is a voiding dysfunction; causes can include nerve dysfunction, constipation and various medications, including opiates and anticholinergics.
The NICE suggests the use of at least a 3-day frequency–volume chart to aid clinicians in making a diagnosis and direct treatment accordingly [101]. This diary should cover the patient's normal day-to-day activities to give a realistic representation of how bothersome their symptoms are.
Quality of life questionnaires, such as the Kings Health Questionnaire and International Consultation on Incontinence Modular Questionnaire offer a chance to clinicians to gain insight into the impact of the condition from the patient's perspective.
Treatment
In MUI, management should focus on the predominant symptom and should be treated initially with conservative measures. If conservative management fails to yield the expected results, medication can be added in or urodynamics carried out prior to considering surgical management.
Conservative treatment
Lifestyle modifications are recommended as first-line treatment for urinary incontinence, which include weight loss, changes to diet and medication, pelvic floor exercises and bladder training. Weight loss is recommended in women with a BMI greater than 30 kg/m2 as a first-line treatment for urinary incontinence [10]. In a recent study, women who lost between 5 and 10% of their bodyweight showed significant improvement in urinary continence for both stress and urge episodes [16].
Evidence for recommendations, such as altering fluid and caffeine intake and avoiding constipation, is sparse. A cohort study conducted by Jura et al. showed that a high intake of caffeine, more than 450 mg per day, which is the equivalent of nine cups of tea and five cups of coffee on average, is associated with a modest increase in the incidence of urgency incontinence [20]. Arya et al. also found an association between high caffeine intake and DO, but was unable to ascertain if the link was causal [21].
Pelvic floor exercises are recommended in all types of urinary incontinence. Studies have shown up to 70% improvement in symptoms of stress incontinence following intensive pelvic floor muscle training (PFMT) [22,23]. NICE guidelines suggest that a trial of supervised PFMT of at least 3 months duration should be offered as first-line treatment to women with MUI [16].
There is evidence to suggest that this improvement is not maintained long term as Agur et al. showed at an 8 year follow-up of their randomized controlled trial (RCT), when they found there was no significant difference in quality of life domains between the study group and the control group, despite more than 60% of the study group claiming they still performed pelvic floor exercises regularly [24].
Bladder training aims to increase the times between voids and increase the capacity of the bladder. In this way, it helps people to control symptoms of urgency. Methods used include Kegel exercises, scheduled bathroom trips and delaying urination techniques. It can be carried out on an in patient basis, but this is costly and is usually reserved for those patients in which all other treatment has failed. It is used as an outpatient therapy by most practitioners and has been shown to be beneficial, reducing the number of incontinent episodes by up to 57% [25].
There have been two studies examining the outcome of the combination of tolterodine, an antimuscarinic drug (see below), and bladder training [26,27]. Both have shown combination therapy is better than either one treatment alone, with Mattiasson et al. showing that 76% of patients in the treatment arm receiving tolterodine and bladder training reported an improvement in their bladder symptoms relative to baseline, compared with 71% in the arm receiving tolterodine alone [26].
Pharmacological treatment
It is known that the bladder urothelium and detrusor muscle contain muscarinic receptors and the most abundant are subtypes M2 and M3. While the detrusor expresses far more M2 than M3 receptors, it appears that detrusor contraction is largely mediated by M3 receptors [28,29]. Antimuscarinic drugs block muscarinic receptors, some more selectively than others, and seem to act more in the storage phase to decrease urgency and urgency incontinence and increase bladder capacity than on detrusor muscle overactivity [30]. Antimuscarinics also act on the iris, lacrimal gland, salivary glands, heart and bowel, which gives the common side effects of dry mouth and eyes, blurred vision secondary to pupil dilatation, fatigue and constipation.
NICE recommends immediate-release (IR) oxybutynin as first-line treatment as ‘there is no evidence of a clinically important difference in efficacy between antimuscarinic drugs’ [18] and it is the most cost-effective method of the available options [31]. A meta-analysis showed that IR tolterodine had a more favorable profile of adverse events than IR oxybutynin, while the extended-release formulations of the two drugs had similar efficacy and safety profiles [32]. Dry mouth was the most commonly reported adverse event and no drug was associated with an increase in any serious adverse event. There were significant differences between the antimuscarinics in rates of withdrawal and rates and range of adverse events and efficacy outcomes. In essence, the antimuscarinics have different tolerability and safety profiles, which are clinically significant. If oxybutynin is not well tolerated, solifenacin, extended-release tolterodine, trospium or a transdermal formulation of oxybutynin can be considered [19].
The extended-release oxybutynin tablets and transdermal patches have the advantage of more convenient dosing regimens and a better side-effect profile [33,34]. The transdermal patch has the advantage of a reduced incidence of adverse events, such as dry mouth [35], but does have the effect of application site reaction.
Solifenacin succinate is available in 5 or 10 mg tablets. Both doses of solifenacin are significantly more efficacious than placebo. Indeed, comparing the two dosages, 10 mg solifenacin was significantly better in terms of the percentage of patients having a ≥50% reduction in incontinence episodes (p = 0.006) [32]. However, withdrawals due to adverse events (p = 0.0005), dry mouth (p < 0.0001), and constipation (p < 0.0001) were significantly more common among the patients treated by 10 mg solifenacin [36].
Darifenacin is a relatively selective muscarinic M3 receptor antagonist available in two doses 7.5 and 15 mg. Trials found a dose-related significant reduction in the median number of incontinence episodes per week (7.5 mg: −68.4%; 15 mg: −76.8%: both p < 0.01 vs placebo), and significant decreases in the frequency and severity of urgency, voiding frequency and number of significant leaks together with an increase in bladder capacity (both p < 0.01 vs placebo). The most common adverse events were dry mouth and constipation, although, together, these resulted in few discontinuations (darifenacin 7.5 mg, 0.6% of patients; 15 mg, 2.1%; placebo, 0.3%). The incidence of peripheral/CNS and cardiovascular adverse events were comparable with those on placebo [37].
Tolterodine has no selectivity for muscarinic receptor subtypes, but is claimed to have functional selectivity for the bladder over the salivary glands [38]. It is available as IR and extended-release formulations and the extended-release form seems to have advantages in terms of both efficacy and tolerability [39]. Transdermal oxybutynin has been compared with extended-release tolterodine for women with urgency and mixed incontinence. It has been shown that both medications reduced the number of daily incontinence episodes and improved quality of life. They had similar efficacy but there was shown to be more anticholinergic side effects with oral tolterodine [40]. Fesoterodine at an 8 mg dose has been compared with 4 mg extended-release tolterodine and was found to be superior for UUI episodes, micturitions and urgency episodes, as well as for self-reported patient assessments of bladder-related problems, urgency, symptom bother and health-related quality of life as early as 3 weeks into treatment at that dose [41].
In conclusion, there are many antimuscarinic medications, each with side effects that lead to low compliance. Brostrom has shown persistence to be 25% at 1 year and less than 10% beyond 2 years [42].
Duloxetine is a combined serotonin and noradrenaline reuptake inhibitor that increases sphincter activity during the storage phase. It is licensed for the treatment of moderate-to-severe stress incontinence. It has exhibited favorable effects on bladder capacity, suggesting possible benefits in mixed incontinence [43]. In a study by Norton et al., the withdrawal rate from the study, despite good outcomes for urinary incontinence parameters, was 16%, mostly due to the side effects of nausea and vomiting [44].
It is known that estrogen has a physiological effect on the lower urinary tract in women. Estrogen deficiency, which commonly occurs after the menopause, is thought to worsen symptoms of stress and urgency and to play a role in recurrent urinary tract infections. A Cochrane systematic review [45] concluded that local estrogen treatment for incontinence may improve or cure it, but there is little evidence from the trials on outcome after estrogen treatment has finished and on long-term effects of therapy. The risk of endometrial and breast cancer after long-term use suggests that estrogen treatment should be for limited periods, especially in those with an intact uterus. There were too few data to reliably address the question of dosage and type of estrogen therapy.
Botulinum toxin
Intravesical botulinum toxin is being increasingly used to treat severe OAB refractory to standard management. An increasing body of literature is forming that supports this technique as effective, well tolerated, and safe in patients with OAB. At present, there is little research into the effect on patients with MUI. A recent Cochrane systematic review and meta-analysis included 19 studies where most patients in the studies had neurogenic OAB, but some included patients with idiopathic OAB. All studies demonstrated the superiority of botulinum toxin to placebo (

Forest plot of comparison of intravesical botulinum toxin A versus placebo for change in frequency.

Forest plot of intravesical botulinum toxin A versus placebo for outcome of change in incontinence episodes.
Percutaneous tibial nerve stimulation
The exact mechanism of percutaneous tibial nerve stimulation (PTNS) is not understood, but the posterior tibial nerve contains mixed sensory motor nerve fibers that originate from the same spinal segments as the nerves to the bladder and pelvic floor. PTNS is thought to act by retrograde stimulation of the sacral nerve plexus and is a treatment for urgency and its associated problems.
Most studies outline a regimen of 12 outpatient sessions lasting 30 min each, usually a week apart [47,48], but there has been a study by Yoong et al. suggesting that a 6-week course of therapy still yields good results and may be more convenient for patients [49].
A meta-analysis showed significant improvement in OAB symptoms using PTNS, which is comparable to antimuscarinic treatment but has a better side-effect profile. Pooled subjective success rates from nonrandomized studies was 61.4% (95% CI: 57.5–71.8) and objective success rate was 60.6% (95% CI: 49.2–74.7) [50].
Sacral neuromodulation
Sacral neuromodulation (SNM) has been approved by the NICE in the UK for treatment of refractory urge/frequency syndrome and urge incontinence as well as fecal incontinence. Its use has not been approved for women with MUI and there are no studies that have examined its efficacy. It is a minimally invasive procedure performed under local anesthetic. An electrode is implanted into the S3 or S4 sacral foramen and linked to a temporary control unit for a trial period of a number of days to weeks. This period is called the peripheral nerve evaluation test. If the trial period indicates that the treatment will be successful, a more permanent stimulator device is embedded in the lower back. An external control unit is used to control the frequency, pulse width and amplitude of electrical pulses produced by the device. It is thought to work by modulating the medullary reflexes and brain centers by peripheral afferents.
Reported outcomes from this procedure are very good, with most trials reporting success as a >50% improvement from baseline in symptoms. Efficacy from RCTs and case series studies shows approximately 56–85% of people are dry or show improvement with follow-up of 36–60 months [51–54]. A case series by Leong et al. for 207 patients from their center showed a satisfaction rate with SNM of 90%, despite 56% of patients mentioning side effects of pain at the insertion site and change in bowel habit [55]. The most frequent adverse event in the literature seems to be loss of efficacy, which was found in 17% of the 1418 patients from the French National Registry [54].
There have been advances in the field [56], including improved placement technique using fluoroscopy and local anesthetic use of tined lead electrodes that have reduced operating time and tissue inflammation. The implant has also become smaller, improving cosmesis and decreasing local discomfort and need for revision. The role of SNM continues to expand, as does the sophistication of techniques and technology involved.
Surgery
Surgery is a common treatment for stress incontinence and can be used effectively in women with mixed incontinence. The cure rate of the stress component is 85–97% with the use of midurethral slings (MUS) in women with MUI (

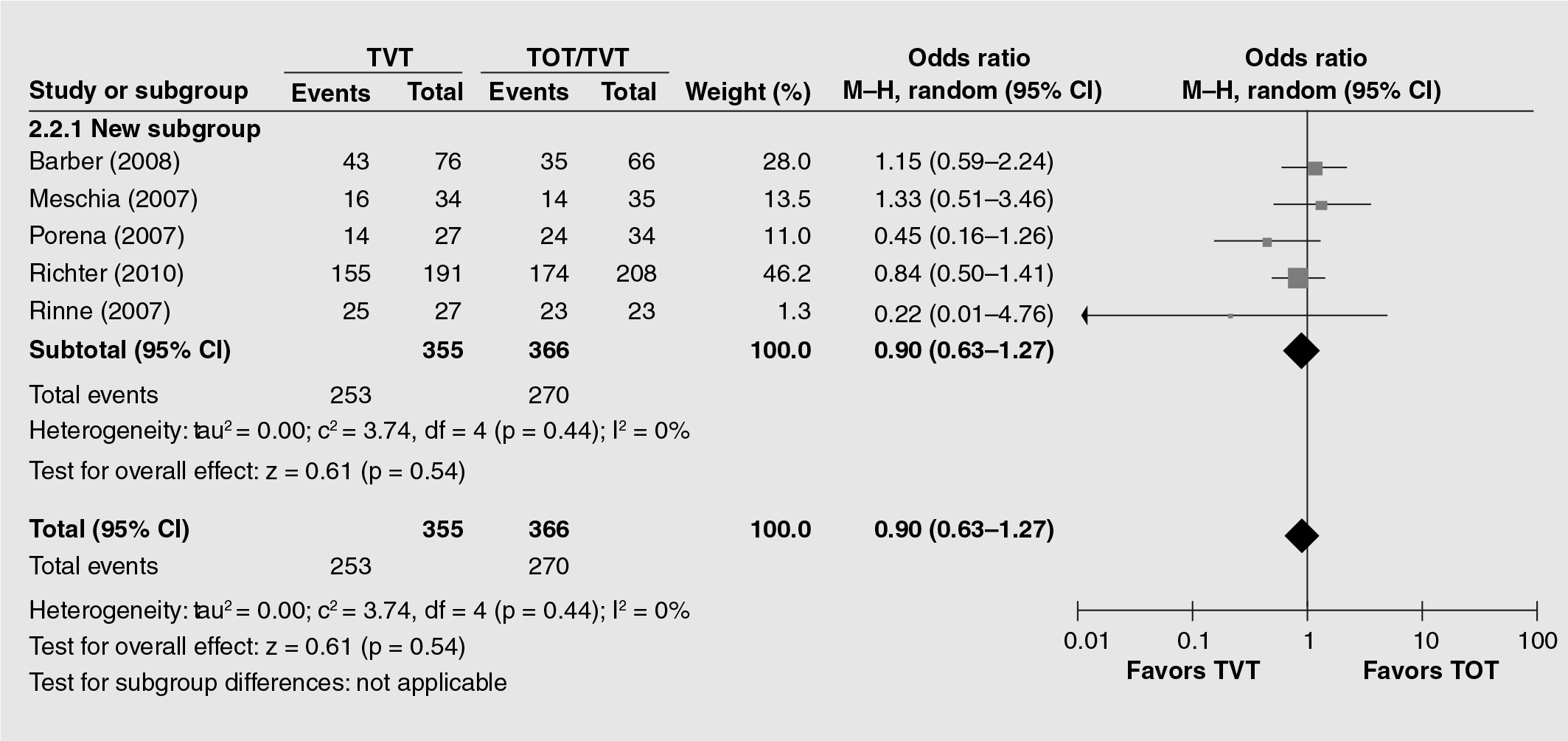
Overall subjective cure in women with mixed urinary incontinence symptoms with or without urodynamic study confirmation (randomized controlled trials).
Periurethral or transurethral injection of bulking agents is a minimally invasive surgical procedure used for the treatment of stress or MUI in adult women. It can be offered to the older woman with multiple comorbidities, but the effectiveness is much lower when compared with MUS and it can cost as much or more. A comparison of periurethral and transurethral methods of injection found similar outcomes but a higher (although not statistically significant) rate of early complications in the periurethral group [60]. The available evidence base remains insufficient to guide practice.
Factors that predict outcome of surgery in women with MUI
There are variable data available regarding cure rates of UUI/OAB following sling surgery. The postoperative course of urgency incontinence after surgery is unpredictable because it may resolve, persist or worsen. There are no consistent predictors of persistent or worsening urgency symptoms after sling surgery. The inconsistencies in outcomes in various studies make it difficult to counsel patients preoperatively on their risk of worsening or persistence of UUI. These surgeries also carry a risk of de novo urgency/urgency incontinence.
Various factors are described in many studies to predict the outcome of surgeries in women with MUI. A recent meta-analysis did not find any significant difference in overall cure rate of urinary incontinence between tapes used by retropubic or transobturator routes [57]. Results are better in stress predominant mixed incontinence (UUI was cured in ten out of 23 [43.5%] patients, improved in ten [43.5%] and persistent in three [13%] patients) when compared with urgency predominant mixed incontinence (UUI was cured in nine out of 21 [43%] patients, improved in six [28.5%] and persistent in six [28.5%]) patients [61].
Presence of specific urodynamic criteria preoperatively is more likely to be associated with persistence of UUI/DO. These include low maximum urethral closure pressure, low maximum urinary flow rate, higher opening detrusor pressure, low maximum cystometric capacity, presence of uninhibited detrusor contraction during cystometry, lower volume at DO and higher detrusor pressure [59].
Prevention
There is some evidence that PFMT in women having their first baby can prevent urinary incontinence in late pregnancy and postpartum [7]. Avoiding constipation, obesity, excessive caffeine and such lifestyle changes may help, although there is no definitive evidence.
Conclusion
Management of MUI should be based on focusing on the most bothersome symptom first. Conservative treatment should be started before any invasive testing is needed and should include lifestyle changes, physiotherapy and bladder training. A quality of life questionnaire should be used to direct treatments.
Antimuscarinic medications and serotonin and noradrenaline reuptake inhibitors can be used for pharmacological treatment. Some patients struggle to cope with the side effects and alternative routes of administration, such as patches, may be of use to these women. Extended-release formulations are preferred to IR formulations. With regard to IR formulations, dose escalation may yield some improvements in the efficacy with significant increase in the adverse events. More clinical studies are needed to indicate which of the drugs should be used as first-, second- or third-line treatment [32].
Surgical management is an effective option for patients with stress-predominant MUI. The stress element of their condition is very likely to be cured or improved by surgery, but they may still need alternative treatment for their urgency and related symptoms.
A supervised course of pelvic floor exercises in the antepartum or postpartum period may be beneficial in preventing the development of urinary incontinence for some women.
Further studies are needed to clarify which factors make cure of incontinence more likely after surgery and the role of newer pharmacological therapies in MUI.
Future perspective
Our population is aging and obesity has become a pandemic in western society. It is logical to presume that pelvic floor dysfunction will become a more common problem and long-term solutions are constantly being sought. Further research needs to be carried out to conclude which treatments have sustained benefit over long-term use. There is some literature on research priorities in this area [62].
Several families of potassium channels are expressed and functional in the detrusor muscle of the bladder. Defects of these channels or in the substances involved in their regulation may be the underlying cause of DO. Chapple et al. conducted a Phase II trial on the potassium ATP opener ZD0947, but found that although there were no problems with hypotension or other vasodilator effects, urgency and frequency did not improve significantly over placebo [63]. Other trials are currently being carried out to investigate other subtypes of potassium channels [64].
Future classes of drugs under investigation for use in patients with an element of OAB, include calcium channel blockers, phosphodiesterase-5 inhibitors, β-agonists and neurokinin receptor antagonists [65]. It is postulated that they may have the potential for reduced side effects and equivalent or better efficacy due to different mechanisms of action. More research needs to be carried out examining those factors that predispose us to incontinence, such as differences in extracellular matrix proteins and androgen receptors. This could help in the development of new therapies.
There is a need for methodologically robust RCTs to evaluate the effectiveness of MUS versus conservative treatment in women with MUI. The studies should be powered enough to enable identification of any patient and/or surgical variables responsible for higher overall cure/improvement rates in MUI.
Executive summary
Mixed urinary incontinence (MUI) accounts for 33% of all incontinence and is diagnosed when symptoms of stress incontinence and urgency coexist.
Pregnancy, vaginal delivery, obesity and increasing age predispose women to incontinence. Genetic differences in extracellular matrix proteins and androgen receptors may also play a role.
Clinicians should focus on medical, neurological and genitourinary history, explore symptoms in terms of duration, frequency and most bothersome symptoms and explore precipitants and treatments.
Simple investigations, including a urine dipstick, postvoid residual bladder scan and a bladder diary, will aid diagnosis.
Access to a quality of life questionnaire will aid clinicians to assess the impact of the condition on the patient's life.
Treatment should focus on the predominant symptom first.
Conservative measures include lifestyle modifications, pelvic floor exercises and bladder training.
Pharmacological treatment is with antimuscarinic agents in the form of tablets, immediate or extended release; patches and gel are alternative vehicles of administration.
Local estrogen has the potential to improve irritative symtoms, such as frequency and dysuria.
When midurethral sling is offered, counsel that cure rates are better for stress-predominant MUI than for urgency-predominant MUI, and both have lower cure rates than surgery for pure stress urinary incontinence. Tension-free vaginal tape and transobturator tape have similar subjective cure rates at a 6–33 month follow-up.
Failure of surgery is more likely for women with previous bladder neck surgery, low maximum urethral pressure closure, low maximum cystometric capacity and higher detrusor pressures preoperatively on urodynamic testing.
Sacral neuromodulation is used for refractory patients with overactive bladder.
Women in the antepartum and postpartum period should practice pelvic floor exercises as part of a supervised course to prevent urinary incontinence.
Research into genetic factors may help to direct new therapies.
Drug development of potassium channel agonists, calcium channel blockers, phosphodiesterase-5 inhibitors, β-agonists and neurokinin receptors antagonists is underway.
Footnotes
P Latthe receives speaker fees and conference sponsorship from Astellas, Pfizer and European Pharma and grants from Health Technology Assessment and Department of Health. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
