Abstract
Overactive bladder syndrome is a chronic condition that requires long-term management and is associated with a significant impairment in the quality of life of the individual as well as having an economic impact on healthcare. Behavioral interventions, along with lifestyle modifications, are effective, but medical management remains the mainstay of treatment. Challenges to improving the overactive bladder syndrome burden and outcomes include underdiagnosis, undertreatment and patient nonadherence to medication. Recent pharmacological advances, along with behavioral interventions, have helped to improve patient compliance. Healthcare providers should acknowledge the need for education regarding overactive bladder syndrome symptoms, its diagnosis and its management options. This article gives an overview of overactive bladder syndrome, its diagnosis and the different treatment options available, as well as discussing recent advances in the field.
Overactive bladder (OAB) syndrome is the term used to describe the symptom complex of urgency, with or without urgency incontinence, usually with frequency and nocturia. The urge syndrome, or urgency frequency syndrome, are synonyms of OAB syndrome, which can be further classified into OAB-wet and OAB-dry [1]. This combination of symptoms is suggestive of detrusor overactivity (DO) in which the bladder contracts, spontaneously or with provocation, during bladder filling while the subject is trying to inhibit micturition. Hashim et al. reported that 69% of men and 44% of women with urgency (OAB-dry) had DO, while 90% of men and 58% of women with urgency and urge-incontinence (OAB-wet) had DO [2]. Epidemiological studies have reported the overall prevalence of OAB syndrome in women to be 16.9% [3], suggesting there could be 17.5 million women who suffer from OAB syndrome in the USA. The prevalence increases with age and is reported to be 4.8% in women under 25 years of age, increasing to 30.9% in those over the age of 65 years (according to data from the National Overactive Bladder Evaluation [NOBLE] program) [3]. The most recent epidemiological study examining lower urinary tract symptoms in Canada, Germany, Italy, Sweden and the UK has reported an overall prevalence of 11.8% in men and women (the European Prospective Investigation Into Cancer and Nutrition [EPIC] study) [4].
Overactive bladder syndrome has a significant impact on quality of life (QOL), often requiring coping mechanisms such as restricting fluids and voiding to a timed schedule, as well as adjusting one's habits and lifestyle in order to accommodate the management of symptoms. Box 1 demonstrates the impact of OAB syndrome on the domains of QOL. This has been further confirmed in the EPIC study [5].
Assessment
Assessment includes a full medical history, including obstetric, gynecological, surgical and neurological histories. The medical history should include information regarding daytime frequency, urgency, nocturia, presence/absence of nocturnal enuresis, pattern of micturition, details of incontinence episodes (e.g., when, how often and how severe) and any associated discomfort, as well as voiding difficulty and bowel habits. In the elderly, a mental status evaluation and assessment of mobility as well as social and environmental factors should be performed. Several drugs can affect bladder function. Therefore, it is essential to elicit a comprehensive history of concurrent medication (Box 2). There are several conditions that can exacerbate or cause symptoms of OAB syndrome and these should be excluded (Box 3) [6].
Physical examination
Physical examination should include:
Abdominal examination for a mass (e.g., uterine fibroids) since they may cause extrinsic bladder compression, giving rise to the symptoms of OAB syndrome;
Gynecological examination to evaluate the strength of the muscles of the pelvic floor and to assess for pelvic organ prolapse. The presence of a cystocele may cause urinary urgency and frequency as it drags on the trigone and causes a sensation of bladder fullness [7]. For women with vaginal atrophy and symptoms of OAB syndrome, estrogen deficiency may be a contributing factor to their symptoms [8];
Rectal examination to rule out fecal impaction or prostate enlargement in males;
Focused neurologic examination in order to determine any underlying neurological cause for OAB syndrome.
Impact of overactive bladder syndrome on quality of life.
Avoidance of sexual contact and intimacy
Limitation or cessation of physical activities (e.g., sports or aerobics)
Absence from work
Decreased productivity
Requirements for specialized underwear and bedding
Specialized precautions with clothing
Reduction in social interaction and limitation of travel
Depression
Loss of self-esteem
Fear of being a burden
Urine odor
Investigations
Urinalysis
Urinalysis should be carried out in order to rule out microhematuria, pyuria, urinary tract infection and glycosuria.
Frequency volume chart/bladder diary
Frequency volume charts/bladder diaries can be used to record the time of micturitions, voided volumes, incontinence episodes, pad usage and other information such as fluid intake, the degree of urgency and incontinence. These charts help to quantify total fluid intake and identify patients who are drinking too much. Various lengths of time have been advocated for the completion of a diary. A 3-day diary may be optimal for diagnostic accuracy [9].
List of drugs that affect bladder function.
Anticholinergics
Sedatives
Antipsychotics
Alcohol
Hypnotics
α-adrenergic agonists
Diuretics
β-adrenergic agonists
Caffeine
Calcium-channel blockers
Post-micturition residual
Post-micturation residual can be measured by ultrasound or by an ‘in and out’ catheter in order to exclude overflow incontinence or incomplete bladder emptying, which can cause symptoms of OAB syndrome.
Specialist investigations
For some women, referral to a specialist unit for further investigation may be necessary. At these centers, diagnostic tests, such as uroflowmetry, urodynamics (video and ambulatory), cystoscopy and radiological imaging (e.g., intravenous urethrography and MRI) may be undertaken, especially in complex cases, in order to dictate a plan of management.
Management
There are many different ways to manage the symptoms of OAB syndrome. These include behavioral and lifestyle strategies, pharmacological management and surgical options. The NICE in the UK has published guidelines on the management of OAB syndrome [101], summarized as:
Caffeine reduction;
6 weeks of behavioral therapy;
Generic oxybutynin should be recommended as a first-line pharmacotherapy;
Tolterodine extended release (ER), oxybutynin ER, transdermal oxybutynin, darifenacin, solifenacin or trospium can all be used for those intolerant of oral oxybutynin immediate release (IR);
Botulinum toxin (Botox®) can be offered to patients who are unresponsive to drugs.
Nonpharmacological strategies
Lifestyle modification
Lifestyle modifications can include moderation of fluid intake to approximately 1–1.5 l/day and avoiding alcohol, caffeine, carbonated and artificially sweetened drinks. A 25% reduction in fluid intake can cause a significant reduction in urgency, frequency and nocturia [10]. Reducing caffeine intake to less than 100 mg/day has been demonstrated to lead to a reduction in urgency episodes, voiding frequency and the number of incontinence episodes [11].
There exists some evidence that suggests that obesity contributes to the onset of OAB syndrome symptoms [12] and weight loss has been demonstrated to improve the number of urgency episodes [13]. Medical problems, such as a chronic cough and constipation, should be carefully managed. Women who report straining at stool are 1.7-times more likely to report urgency [14].
Bladder retraining
Bladder retraining involves a program of patient education and a scheduled voiding regimen. The goals of bladder retraining are to normalize urinary frequency, improve control over bladder urgency, increase bladder capacity, decrease incontinence episodes, prolong voiding intervals and to improve the patient's confidence in bladder control. This is achieved through teaching patients urge suppression techniques (e.g., distraction, curling toes and perineal pressure) [15].
The mechanism by which bladder training works is not clear; however, several theories have been postulated, such as improved central modulation of sensory afferent impulses and improved cortical inhibition over detrusor contraction. Bladder retraining has been reported to reduce the number of incontinence episodes in 50–80% of cases [16].
Conditions that can cause or contribute to the symptoms of overactive bladder syndrome.
Urinary tract infection, inflammation with activation of sensory afferent nerves and obstruction can contribute to detrusor overactivity and/or urinary retention.
Bladder abnormalities of inflammation (e.g., tumors, calculi or interstitial cystitis) may precipitate detrusor overactivity.
Estrogen deficiency, atrophic vaginitis and urethritis may contribute to symptoms.
Sphincter weakness can cause leakage of urine into the proximal urethra and thus precipitate urgency. The ability to inhibit detrusor by sphincter contraction may be diminished.
Conditions of the brain include stroke, Alzheimer's disease, dementia, Parkinson's disease and multiple sclerosis, which can cause higher cortical inhibition of bladder impairment, causing detrusor overactivity of neurogenic origin.
Conditions of the spinal cord include multiple sclerosis, cervical or lumbar stenosis or disc herniation and spinal cord injury, which can cause neurogenic detrusor overactivity or urinary retention.
Peripheral innervations, such as diabetic neuropathy and nerve injury, can result in urinary retention and low functional bladder capacity.
Congestive heart failure and venous insufficiency with edema can cause volume overload, contributing to urinary frequency and nocturia when the person is lying down.
Diabetes mellitus, characterized by poor blood glucose control, can contribute to osmotic diuresis and polyuria.
Hypercalcemia may result in polyuria.
Excess intake of caffeine, alcohol or fluids may result in polyuria and urinary frequency.
Poor bowel function and constipation may result in fecal impaction, which can contribute to symptoms.
Impaired mobility or dexterity (e.g., joint disease or muscle weakness) interferes with toileting ability and may precipitate urgency incontinence.
Chronic anxiety and learned voiding dysfunction can cause symptoms of overactive bladder syndrome.
Data from [5].
Pelvic floor muscle training
Pelvic floor muscle training (PFMT) involves exercises designed to improve the function of pelvic floor muscles. The rationale is that the contraction of the muscles can reflexively or voluntarily inhibit contraction of detrusor muscle. It is important to ensure that patients are seen by a qualified women's health physiotherapist. Physiotherapists may incorporate different treatment modalities in order to help improve pelvic floor muscle function, including electrical stimulation and biofeedback. PFMT significantly increases the pelvic floor muscle strength and reduces the frequency of urinary incontinence episodes [17]. Most women do not appear to demonstrate a post-treatment benefit from biofeedback-assisted PFMT. However, for individuals who have trouble identifying and contracting their pelvic floor muscles, biofeedback may be useful.
Pharmacological treatment
Antimuscarinics
Anticholinergic drugs (also known as antimuscarinics) are the mainstay of treatment for OAB syndrome symptoms. They inhibit the binding of acetylcholine at muscarinic receptors in the detrusor muscle, thus reducing the contractions of the detrusor muscle and controlling involuntary detrusor contractions without disturbing normal voiding.
Tolterodine
Tolterodine is a tertiary amine that is rapidly absorbed and extensively metabolized by the cytochrome P450 system. It demonstrates no specificity for receptor subtypes but does appear to have relative functional selectivity for the bladder over salivary glands [18]. The drug is metabolized in the liver to an active metabolite 5-hydroxymethyl derivative that has a similar pharmacokinetic profile, and this derivative is thought to significantly contribute to the therapeutic effect of the drug.
Tolterodine is available as immediate release (TOLT-IR; 2 mg twice-daily dosing) and ER (detrusitol XL; 4 mg/day dose). The ER form seems to have advantages over IR preparations in terms of both efficacy and tolerability [19]. Oxybutynin ER and tolterodine ER have also been compared in the Overactive Bladder: Performance of Extended Release Agents (OPERA) study; improvements in episodes of urge incontinence were similar for the two drugs, although oxybutynin ER was significantly more effective in reducing the frequency of micturition [20]. The Overactive Bladder: Judging Effective Control and Treatment (OBJECT) trial compared oxybutynin ER 10 mg daily with tolterodine 2 mg daily. Overall, oxybutynin ER was much more effective [21]. Various aspects of efficacy and tolerability have been further documented in a number of randomized controlled trials (RCTs) [22,23].
Solifenacin
Solifenacin is a tertiary amine and is easily absorbed from the gastrointestinal tract (bioavailability: 90%). It is a potent M3-receptor antagonist that has selectivity for the M3 receptors over M2 receptors, has a much higher potency against M3 receptors in smooth muscle than it does against M3 receptors in salivary glands and is available in 5 mg and 10 mg daily dosages.
The Solifenacin versus Tolterodine (STAR) multinational trial, a prospective, double-blind study comparing solifenacin with tolterodine, has demonstrated the efficacy of solifenacin [24,25]. The antimuscarinic adverse effects of dry mouth, constipation and blurred vision were the most frequently reported adverse events overall. These were also found to be dose-related. A number of other studies and reviews have further documented the effects of solifenacin [26]. Pooled analyses of four RCTs have demonstrated positive responses for nocturia, urgency and frequency [27]. Michel et al. studied the cardiovascular safety of solifenacin and concluded that it does not increase the heart rate or blood pressure [28]. The Solifenacin in the Treatment of Urgency and Other Symptoms of Overactive Bladder (SUNRISE) trial demonstrated that solifenacin significantly reduced the number of urge incontinence episodes at both the 5 and 10 mg doses [29]. Recently, the Study of Vesicare in Overactive Bladder Subjects to Evaluate Symptom Bother and Health Related Quality of Life (VIBRANT) confirmed the efficacy of solifenacin [30].
Darifenacin
Darifenacin is a tertiary amine with moderate lipophilicity and is a highly selective M3-receptor antagonist that has been found to have a fivefold higher affinity for the human M3-receptor compared with the M1 receptor. It has been developed as a controlled release formulation, which allows for once-daily dosing. The recommended dosages are 7.5 mg and 15 mg per day. The efficacy of darifenacin has been investigated in a multicenter, double-blind, placebo-controlled, parallel-group study that enrolled 561 patients with symptoms of OAB syndrome [31]. The most common adverse events were mild-to-moderate dry mouth and constipation, with a CNS and cardiac safety profile comparable to placebo. Trials have demonstrated that darifenacin does not cause impairment of memory or other cognitive functions and, therefore, may be particularly useful in vulnerable populations, such as the elderly and CNS-compromised neurogenic bladder patients [32]. Zinner et al. found that patient satisfaction was high during treatment with darifenacin in patients who were previously dissatisfied with antimuscarinic treatment [33]. Over the long-term, darifenacin has been associated with significant improvements in health-related QOL (HRQOL) in patients with urge urinary incontinence over 2 years [34].
Fesoterodine
Fesoterodine is a new and novel derivative of 3,3-diphenylpropyl-amine, which is a potent antimuscarinic agent and is available in 4 mg and 8 mg daily dosages. In clinical studies, both doses have been found to be superior to placebo [35]. The most common side effect reported was dry mouth. Compared with TOLT-ER 4 mg, fesoterodine 8 mg had statistically significant advantages in improving incontinence episodes and severe urgency with incontinence [36]. Recently, an open-label study has been performed to evaluate the efficacy and tolerability of flexible-dose fesoterodine in patients with OAB syndrome who were dissatisfied with tolterodine treatment, demonstrating a significant improvement in OAB syndrome symptoms and HRQOL [37].
Trospium chloride
Trospium chloride is a quaternary ammonium compound that is nonselective for muscarinic receptor subtypes and demonstrates low biological availability [38]. It crosses the blood–brain barrier to a limited extent and hence would appear to have few cognitive effects [39]. Trospium is available in a 20 mg twice-daily dosage, but recently an ER formulation (60 mg) has been introduced for once-daily dosing. RCTs have demonstrated similar efficacy for the ER formulation as was found with previous formulations [40,41]. The most frequent side effects were dry mouth and constipation. Intravesical application could be an interesting alternative [42].
Drugs with mixed actions
Oxybutynin
Oxybutynin is a tertiary amine that under-goes extensive first-pass metabolism to an active metabolite, N-desmethyl oxybutynin, which occurs in high concentrations [43] and is thought to be responsible for a significant part of the action of the parent drug. It has a mixed action consisting of both an antimuscarinic and a direct muscle relaxant effect, in addition to local anesthetic properties. The latter properties are important when given intravesically but probably have no effect when given systemically. Oxybutynin has been found to have a high affinity for muscarinic receptors in the bladder [44] and has a higher affinity for M1 and M3 receptors compared with M2 receptors [45]. Oxybutynin is effective for the management of patients with DO but the antimuscarinic adverse effects of oxybutynin are well-documented and are often dose-limiting [46]. Using an intravesical route of administration, higher local levels of oxybutynin can be achieved whilst minimizing the systemic adverse effects. Using this method, oxybutynin has been found to increase bladder capacity and lead to significant clinical improvement [47]. A controlled release oxybutynin preparation using an osmotic system (OROS) has been developed that has been found to have comparable efficacy to oxybutynin IR and is associated with fewer adverse effects [48]. These findings are in agreement with a further study of controlled release oxybutynin (Lyrinel XL®, Janssen-Cilag, UK) that reported the incidence of moderate-to-severe dry mouth to be 23%, and only 1.6% of participants discontinued the medication owing to these adverse effects [49].
An oxybutynin transdermal delivery system (Kentera®, UCB Pharma) has also been developed and was compared with tolterodine ER in 361 patients with mixed urinary incontinence [50]. Both agents significantly reduced incontinence episodes, increased voided volume and led to an improvement in QOL when compared with placebo; in addition, there was no significant difference in efficacy between the oxybutynin patch and tolterodine ER. The most common adverse event in the oxybutynin patch arm was application site pruritis in 14% of participants; however, the incidence of dry mouth was reduced to 4.1% compared with 7.3% in the tolterodine arm [50]. The Multicenter Assessment of Transdermal Therapy in Overactive Bladder Syndrome with Oxybutynin (MATRIX) study has demonstrated significant improvements in work productivity with the transdermal system [51]. Recently, oxybutynin has also been developed in the form of a gel, the efficacy and safety of which has been investigated in a randomized, double-blind, placebo-controlled, multicenter study in which women were assigned to receive 1 gm oxybutynin chloride gel once-daily or matching placebo for 12 weeks. There was a significant decrease in the number of urge incontinence episodes and urinary frequency, and a significant increase in voided volumes. Application site reactions were infrequently observed in the oxybutynin chloride and placebo groups (5.4 and 1.0%, respectively) [52]. Oxybutynin hydrochloride gel is currently available only in the USA.
Propiverine hydrochloride
Propiverine hydrochloride has a calcium modulating property (it causes an inhibition of calcium influx resulting in musculotrophic spas-molysis), as well as anticholinergic properties. Propiverine is available in IR (15 mg twice-daily) and ER (30 mg once-daily) preparations. A head-to-head comparison with tolterodine has demonstrated that both have equal efficacy in reducing the number of incontinence episodes [53]. There are strong data in support of the efficacy of propiverine in reducing the frequency of micturition. Recently, the effect of the propiverine 20 mg daily dose has been evaluated, demonstrating its efficacy on OAB syndrome by reducing urgency [54]. Table 1 summarizes the doses and side effects of antimuscarinics.
Side effects and frequency of antimuscarinics and their dosages.
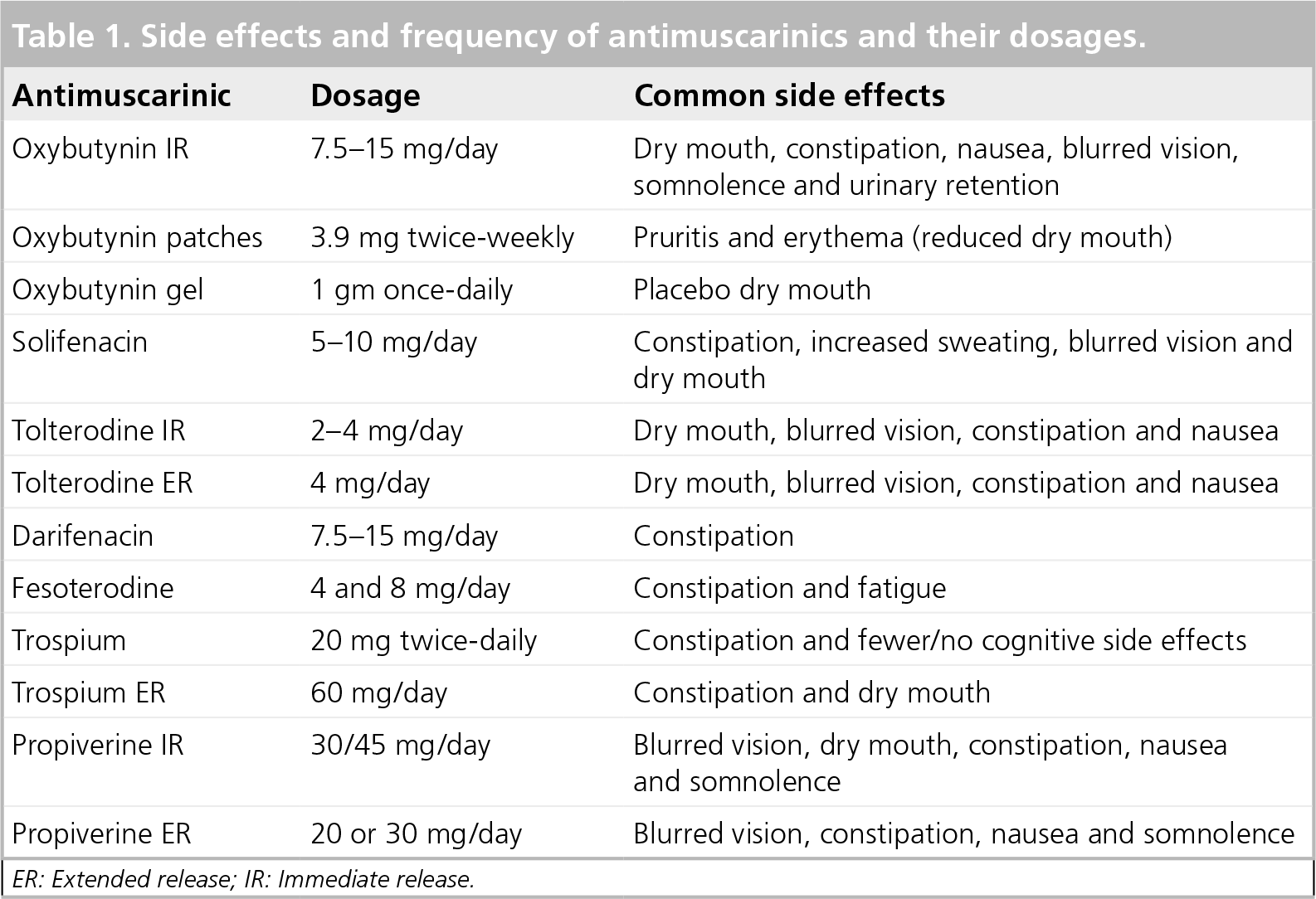
ER: Extended release; IR: Immediate release.
Antidepressants
Imipramine
Imipramine has been shown to have systemic anticholinergic effects, to blocks the reuptake of serotonin and to have some β-agonist activity [55]. Some studies have found a significant effect in the treatment of patients with DO [56], although others report little effect [57]. In light of this evidence and the serious adverse effects associated with tricyclic antidepressants, their role in DO remains of uncertain benefit, although they are often useful in patients complaining of nocturnal enuresis or bladder pain and in countries where the more modern and more expensive agents are not available.
Prostaglandin synthetase inhibitors
The bladder mucosa has been found to have the ability to synthesize eicosanoids, although it is uncertain whether they contribute to the pathogenesis of unstable detrusor contractions. However, prostaglandin synthetase inhibitors may have a role in sensitizing sensory afferent nerves by increasing the afferent input produced by a given bladder volume. A double-blind, controlled study of flurbiprofen in women with DO was found to have an effect, although this was associated with a high incidence of adverse effects (43%) including nausea, vomiting, headache and gastrointestinal symptoms [58]. The few controlled clinical trials and the limited number of drugs tested makes it difficult to evaluate the therapeutic value of eicosanoids. At present, this evidence does not support the use of eicosanoids in treating DO.
Hormones
Estrogen
Although estrogen has an important physiological effect on the lower urinary tract, the current level 2 evidence does not support the use of systemic estrogens in the treatment of urinary incontinence, as demonstrated by the Women's Health Initiative (WHI) study [59]. However, there is evidence to suggest a beneficial role for topical vaginal estrogen in treating OAB syndrome [60].
Desmopressin
Desmopressin is a synthetic vasopressin that has a potent antidiuretic effect and is used in the management of diabetes insipidus and nocturnal enuresis. More recently, it has been found to be effective in reducing nocturia in patients with both neuropathic and non-neuropathic OABs [61]. Nasal desmopressin has been reported as a ‘designer drug’ for the treatment of daytime urinary incontinence in a multicenter, multinational, randomized study [62]. Overall, there were higher incidences of periods without leakage in the first 4 h while taking desmopressin compared with placebo. Furthermore, there was a higher frequency of dry days on desmopressin [62]. Desmopressin is safe; however, the drug should be prescribed with care in the elderly owing to the risk of hyponatremia [63], and the current recommendations are that serum sodium should be checked on days 3 and 5 following the start of treatment and should not be prescribed for those over 65 years of age. Recently, a double-blind, placebo-controlled, prospective, randomized cross-over study was conducted using 0.2 mg of oral desmopressin in adults suffering from OAB syndrome. There was an increase in the time to first urgency episode and a decrease in the number of urgency episodes in the drug group compared with placebo. There was a subjective improvement in frequency, urgency and overall QOL. A total of 27 people reported adverse events, all of which were mild (headache being the most common) and no hyponatremia was recorded. Antidiuresis, using oral desmopressin tablets, is a novel, feasible and safe (on a short-term basis) method of treatment for adults with OAB syndrome and can be considered in the range of drugs available for the treatment of OAB syndrome [64].
α-adrenoreceptor antagonists
The lower urinary tract is innervated by both the parasympathetic and the sympathetic nervous systems, which act via muscarinic and adrenergic receptors, respectively. The adrenergic receptors found at the bladder neck are α1 adrenergic receptors. Consequently, α1 adrenergic-blocking agents may be useful in the management of lower urinary tract dysfunction, and it has been speculated that the α1 adrenergic receptors may mediate the OAB symptoms. Anecdotal evidence has demonstrated that tamasulosin, a selective α1-adrenoreceptor antagonist, may improve urinary symptoms that are secondary to DO in men, but the results from a randomized, double-blind, placebo-controlled study suggest that α-adrenoreceptor antagonists alone are not clinically useful in the management of OAB syndrome in women [65].
Drugs acting on membrane channels
At present, there is insufficient evidence to suggest that calcium channel-blocking agents are effective in the treatment of DO, although the development of a selective calcium channel-blocking agent that eliminates spontaneous contractions without affecting micturition may prove to be of use in the treatment of DO [66].
At present, despite promising preclinical efficacy data for potassium channel-opening agents, these are not a viable therapeutic option owing to a lack of selectivity for the bladder over cardiovascular tissues [67].
Intravesical drugs
Botulinum toxin
Botulinum toxin is a neurotoxin that interferes with neural transmission by blocking the calcium-dependent release of acetylcholine, causing the affected muscle to become weak and atrophic. The use of intravesical Botox was first described in the treatment of intractable neurogenic DO in 31 patients with traumatic spinal cord injury [68]. Whilst the role of Botox has been established in the treatment of neurogenic DO (NDO), the data regarding its use in intractable idiopathic DO (IDO) are less robust. When compared with NDO, information regarding IDO patients is more limited and based on smaller trials. Studies using Botox that were performed in IDO patients have reported an increase in cystometric capacity with a reduction in the number of incontinence episodes but with high incidence of voiding dysfunction requiring self-catheterization [69,70]. The side effects include bladder pain [71], urinary retention requiring self-catheterization (20–30%) [69] and rarely transient muscle weakness. The main problem is dose-related side effects and there is no clear evidence for the optimal dose of Botox regarding either the safety of or the number of repeated injections patient can have in the treatment of IDO. The dose of Botox can vary from 100 IU to 300 IU. Higher doses are required for the treatment of NDO, while lower doses are advised for IDO.
A recent multicenter study has been performed assessing the dose-response effect of Botox. Botox demonstrated durable efficacy in patients with IDO at doses greater than 100 IU. The only adverse effect noted was an increase in postvoid residual in 36.4% compared with 18.6% of IDO patients taking placebo [72]. This is seen more when higher doses were used. At present, the evidence would suggest that intravesical administration of Botox may offer an alternative to surgery in those women with intractable DO but is not currently licensed in the UK since there is no agreement by regulatory agencies for its use in patients with OAB syndrome.
Capsaicin
Capsaicin is the pungent ingredient found in red chilies and is a neurotoxin of substance P-containing nerve fibers [73]. Intravesical application of capsaicin dissolved in 30% alcohol solution appears to be effective and is used for NDO only. The pungency of alcoholic capsaicin solutions leading to extensive bladder irritation and pain has prevented the widespread use of this compound.
Resiniferatoxin
Resiniferatoxin is a potent analogue of capsaicin that appears to have a similar efficacy but with fewer side effects of pain and burning during intravesical administration [74]. Current evidence does not support the routine clinical use of this agent, although it may prove to have a role as an intravesical preparation in NDO.
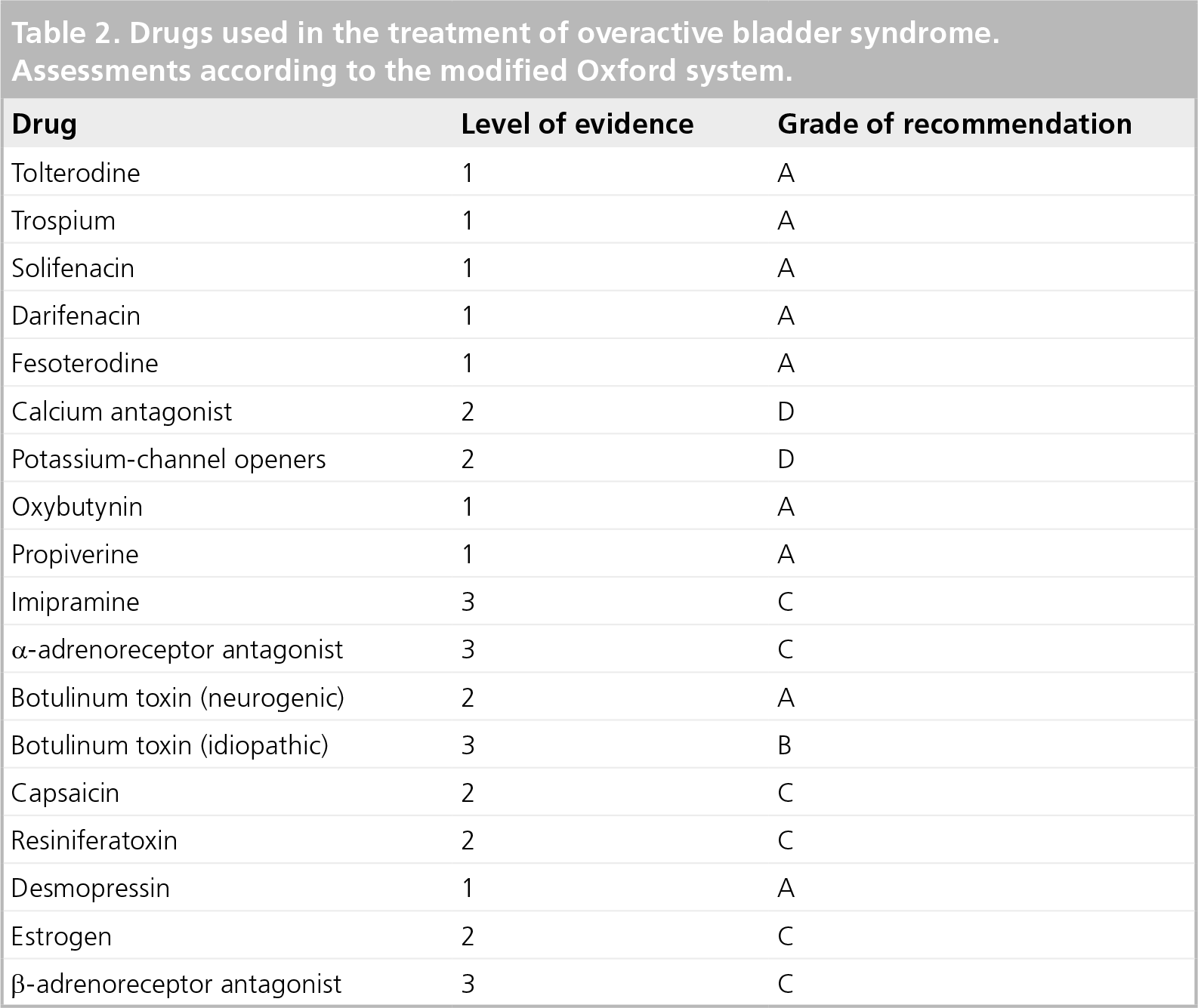
The recommendations regarding drugs from the International Continence Society are based on evaluations made using a modification of the Oxford system, which is shown in Table 2.
Drugs used in the treatment of overactive bladder syndrome. Assessments according to the modified Oxford system.
Considerations in the elderly
The pathophysiology of OAB syndrome in the elderly is thought to be multifactorial, with an abnormality occurring in the nervous system and/or the structure/function of the urothelium or bladder smooth muscle, leading to bladder hypersensitivity, abnormalities in bladder sensation (urgency) and involuntary detrusor contraction. Coexisting conditions that need consideration during the management of OAB syndrome are numerous and include (but are not limited to) cardiovascular conditions, neurological disease, dementia and sleep disorders. Cognitive impairment is an important issue when considering the use of pharmacological treatment, particularly as there is a spectrum of disability that can range from age-related cognitive impairment to true dementia, which increases with age [75]. Data have been presented on the efficacy and tolerability of some therapeutic agents in older patients, including trospium [39] and darifenacin [32].
Future possibilities
Vitamin D3 receptor antagonists
Rat and human bladders have been found to express receptors for vitamin D3, which makes it conceivable that the bladder may also be a target for vitamin D3. One of the vitamin D3 analogues, BXL-628 (also called elocalcitol) was demonstrated to have an inhibitory effect on the RhoA/Rho kinase pathway. Upregulation of this pathway has been associated with bladder changes, which in turn are associated with diabetes, outflow obstruction and DO [76]. In an RCT enrolling women with OAB syndrome, a significant increase was observed in the mean voided volume [77]. Further studies are required to prove elocalcitol as a useful alternative for the treatment of OAB syndrome.
NK1-receptor antagonists
The main endogenous tachykinins, namely substance P, neurokinin A and neurokinin B and their respective receptors, NK1, NK2 and NK3, have been found in various CNS regions, including those involved in micturition control. Aprepitant, an NK1-receptor antagonist used for the treatment of chemotherapy-induced nausea and vomiting, significantly improved symptoms of OAB syndrome in postmenopausal women with a history of urgency incontinence or mixed incontinence (with predominantly urgency urinary incontinence), as demonstrated in a well-designed pilot RCT [78]. The results of this initial proof-of-concept study suggest that NK1-receptor antagonism holds promise as a potential treatment approach for OAB syndrome.
β-adrenoreceptor agonists
β-adrenoreceptors induce bladder relaxation by activation of adenylyl cyclase with subsequent formation of c-AMP. The in vivo effects of β3-adrenergic receptor agonists on bladder function have been studied in several animal models. It has been demonstrated that, compared with other agents, including antimuscarinics, β3-adrenergic receptor agonists increase bladder capacity with no change in micturition pressure and the residual volume [79]. A controlled trial was undertaken using YM178 (mirabegron) in patients with OAB syndrome in which tolterodine and placebo served as controls. Compared with placebo, there was a significant reduction in the mean micturition frequency, number of incontinence episodes and urgency episodes per 24 h. Mirabegron was well-tolerated, and the most commonly reported side effects were headache and gastrointestinal side effects [80]; however, demonstrating that this drug is a useful alternative or complement to the current treatments of OAB syndrome will require further well-designed RCTs. Phase III studies have just been completed that confirm its efficacy and safety profile [102].
Gonadotrophin-releasing hormone antagonist
The effect of gonadotrophin-releasing hormone antagonist has been demonstrated in the treatment of male lower urinary tract symptoms [81]. No study has demonstrated any beneficial effect in females.
Gabapentin
Gabapentin is one of the new first-generation antiepileptic drugs that expanded its use into the treatment of a broad range of neurologic and psychiatric disorders [82]. The mechanism of action remains controversial but it has been postulated that it acts by binding an α2S subunit of voltage-dependent calcium channels [83]. The effect of gabapentin on NDO has been reported [84]; however, gabapentin should be further explored in controlled studies in order to prove the drug as a new therapeutic alternative.
Sacral nerve neuromodulation
Sacral nerve neuromodulation is an established treatment for OAB syndrome and its associated incontinence. Studies now exist with greater than 10 years of follow-up. Neuromodulation of the sacral nerve roots at S3 and S4 have been demonstrated to alter bladder and sphincter function and to be useful in the treatment of urgency incontinence [85]. Work with animal models suggests this may occur via selective stimulation and alteration of C-fibers, thus sparing the C-fibers [86]. The C-fibers may mediate DO whereas C-fiber afferents probably confer the ability of the bladder to relax reflexively as part of the normal micturition cycle. However, there are problems with this treatment, such as it being expensive and the fact that its efficacy has not been determined on a population basis, since none of the trials were able to determine absolute positive outcome measures. Another issue is whether unilateral or bilateral stimulation gives better results. These questions require further research [87].
Peripheral-posterior tibial nerve stimulation
Peripheral-posterior tibial nerve stimulation is a minimally invasive outpatient neuromodulation procedure for refractory OAB syndrome. As the tibial nerve contains L4-S3 fibers that also innervate the bladder and pelvic floor, stimulation at this point can have beneficial effects on bladder function [88].
Surgical treatment
Sugery is reserved for patients who have IDO that is unresponsive to medication and behavioral therapy since all surgical procedures are accompanied by associated risks and complications. With the increasing popularity of Botox, the surgery option is rarely used. The procedures in use include detrusor myomectomy, augmentation cystoplasty and urinary diversion.
Conclusion
Overactive bladder syndrome is a common and distressing condition that is known to adversely affect QOL. Lifestyle advice includes moderation of fluid intake and avoiding alcohol and caffeine. Behavioral therapy, such as bladder retraining, is important, as is PFMT, although drugs remain the mainstay of treatment. The development of new drugs, treatment regimens and delivery systems should improve patient persistence with treatment and acceptability. Intravesical injection of Botox has a role in refractory DO, although there are still concerns regarding its long-term efficacy and side effects. Surgery remains the last resort for IDO.
Ultimately, perhaps a better understanding of the causes and pathophysiology of OAB syndrome may result in the development of new treatments for such a common and distressing condition.
Future perspective
With an increasingly elderly population, the impact of OAB syndrome will continue to have an important effect on the QOL of many, and may become an increasing economic burden on healthcare. While the etiology of OAB syndrome remains to be determined, a better understanding of the pathophysiology may improve the efficacy of treatment. Many parts of the brain appear to be activated during storage and voiding, and there is increasing interest in drugs modulating the micturition reflex by a central action. Some of the central mechanisms involved in micturition, including the periaqueductal gray and pontine micturition center, could be involved with the problems of OAB syndrome or urinary incontinence. Whereas the development of the newer longer-acting agents has made only a small impact on patient compliance, the introduction of more bladder-specific agents, such as solifenacin and darifenacin, may lead to greater acceptability to patients. Similarly, the use of alternative delivery systems and flexible dosing regimens will also offer physicians and women greater choice. In addition, the concept of short-acting agents to be used ‘as required’ may become increasingly popular in those women who do not want to take medication every day for something that only troubles them occasionally.
Currently, the majority of agents used in the treatment of OAB syndrome are antimuscarinic drugs. To date, there has been little success with calcium-channel antagonists and potassium-channel opening agents, although there remains a need for alternative types of medication. Agents acting on the CNS could influence OAB syndrome; however, target selection and adverse effects are an issue. Current research has implied that the efferent response to pain may be partially responsible for detrusor contractions and there is now considerable interest in assessing the use of drugs that block the pain pathways, such as the neurokinin antagonists. Vitamin D3-receptor analogues require further evaluation. β3-adrenergic receptor agonists may work by reducing the contractility or excitability of bladder muscle. These may become increasingly important and offer an alternative to antimuscarinics in the future.
Executive summary
Overactive bladder (OAB) syndrome is a common and distressing condition that has a significant impact on the quality of life of the individual, as well as having an economic impact on healthcare.
Lifestyle modifications, behavioral therapy and pelvic floor muscle training are important aspects in the management of OAB syndrome.
Pharmacological therapy, used synergistically with behavioral therapy, remains integral in the management of women with OAB syndrome.
Over the last 10 years, we have seen the development of many new antimuscarinic agents, in addition to those with more novel modes of action.
At present, antimuscarinic agents remain the most widely used preparations. Fesoterodine is the most recently launched drug of this kind.
Transdermal oxybutynin and oxybutynin gel now provide a viable therapeutic alternative for those women who are unable to tolerate oral antimuscarinic agents.
Botulinum toxin may offer an alternative to surgery in women with intractable detrusor overactivity.
Neuromodulation is an interesting approach and holds promise for the future.
A further alternative approach is to use combination therapy. While currently there is no evidence to support the use of α-adrenergic antagonists alone, it is possible that when used concomitantly with an antimuscarinic agent there may be a synergistic effect leading to the possibility of reduced dosing and, consequently, reduced adverse effects. Botox holds promise in the management of OAB syndrome. Neuromodulation, especially peripheral-posterior tibial nerve stimulation, may prove to be an interesting and innovative outpatient approach in the management of refractory OAB syndrome.
Footnotes
Mr Dudley Robinson has acted as an investigator and consultant for Astellas, Pfizer, Uroplasty, Gynaecare, Riccordati, Novonordisk, Allergan and Ferring. Professor Linda Cardozo is a consultant for Astellas, Pfizer, Rhottapharm and Schering-plough, as well as an investigator for Astellas and Bioxella. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
