Abstract
Keywords
Women who experience cyclic heavy menstrual bleeding (HMB), or menorrhagia, will often present to a healthcare professional because of the subsequent negative impact on their daily lives and activities [1,2]. HMB has been shown to adversely affect mood, energy/vitality, work productivity, social interactions, family life and sexual functioning [3–5]. The impact on health-related quality of life (HRQoL) due to cyclic HMB derives from both the effort associated with managing menstrual bleeding and the consequences of excessive blood loss such as fatigue and iron deficiency anemia [5].
Previous HMB clinical trials have emphasized reduction in menstrual blood loss as the key efficacy end point, without measuring the effects of treatment on patient-reported outcomes. Thus, comparative data are lacking in this area and a standard HRQoL instrument has not been established [6]. Studies involving oral contraceptives, progestins and other pharmacological therapies for abnormal uterine bleeding have demonstrated some improvement in patient HRQoL using various instruments; however, these improvements were less than the HRQoL improvements noted with other treatments for HMB (i.e., ablation or hysterectomy) [5]. Recently, a study by Kouides
Tranexamic acid has been used for decades outside the USA to reduce bleeding in surgical applications [8] and to treat HMB [9]. Although generally well tolerated, the standard immediate-release formulation of tranexamic acid has been associated with nausea, vomiting, dyspepsia and diarrhea [8,9]. In Phase III trials of the novel, oral formulation of the competitive plasminogen inhibitor tranexamic acid (TA; Lysteda®, Ferring Pharmaceuticals, NJ, USA), women with cyclic HMB for three and six cycles experienced clinically significant improvements in menstrual blood loss and HRQoL, with a low incidence of treatment-related gastrointestinal (GI) adverse events (AEs) [10,11]. The current study was designed to assess long-term (up to 27 cycles) safety and evaluate HRQoL-based measures during treatment of cyclic HMB with TA.
Methods
Patients
Women (aged 18–49 years) with a history of cyclic HMB were enrolled in the study at 62 sites throughout the USA. Diagnosis of cyclic HMB was based on the medical judgment of each investigator. The diagnosis of HMB was made after review of the patient's medical history, a patient's self-report of the impact menstrual bleeding had on her normal activities, and then evaluation of a screening period, evaluation of results of a screening physical and gynecologic examination and laboratory results. Women with at least 6 months of regularly occurring menstrual cycles (21–35 days apart) with menstrual periods lasting no more than 10 days and normal findings on pelvic examination, normal cervical cytology results and no abnormal findings on transvaginal ultrasonography (fibroids or ovarian cysts were not considered abnormalities) were eligible to participate in the study. Women of childbearing potential were required to use an acceptable method of birth control other than hormonal contraception.
Women were excluded from study participation if they had a history or presence of clinically significant health problems (including thromboembolic disease or coagulopathy), endometrial abnormalities, cervical carcinoma, anovulatory dysfunctional uterine bleeding, metrorrhagia, menometrorrhagia, polymenorrhea, severe anemia (hemoglobin <8 g/dl), known acquired defective color vision, glaucoma, ocular hypertension, macular degeneration, retinopathy, or were pregnant or lactating.
Study design
Following one menstrual cycle screening phase, women who enrolled in this multicenter, open-label, long-term safety study received TA 1.3 g orally three-times daily for up to 5 days per menstrual cycle for a total of 27 menstrual cycles. Study visits were scheduled 1–7 days after the last day of the preceding menstrual period and occurred monthly through the third menstrual cycle, and thereafter at 3-month intervals.
Use of other medications or herbal remedies to relieve HMB were not allowed during the study. NSAIDs, vitamins and oral iron therapy were permitted.
The study received initial and annual Institutional Review Board approval at each site and was conducted in accordance with the Declaration of Helsinki and applicable regulatory requirements. All study participants provided written informed consent.
Measures
Safety was the primary end point of the study. Safety was assessed by the incidence of AEs; study discontinuation information; changes in physical examinations, vital sign measurements and laboratory test results (hematology, blood chemistry and urinalysis); ophthalmologic examinations (acuity, color blindness, intraocular pressure and dilated fundoscopic examination); and findings on 12-lead ECGs. AEs were coded using version 7.1 of the Medical Dictionary for Regulatory Activities and summarized for the number of subjects reporting the AE and the number of AEs reported. AEs were recorded from the time informed consent was signed until the participant completed the study.
Subjects' HRQoL was assessed via administration of the Ruta Menorrhagia Questionnaire (RMQ); the Short Form 36 Health Survey, Version 2.0 (SF-36v2®, Acute US Version 2.0, QualityMetrics Inc., RI, USA); and the Menorrhagia Impact Questionnaire (MIQ). The RMQ is disease specific and is scored from 0 to 100, with a higher score indicating more severe HMB symptoms [12]. The SF-36v2 is a generic HRQoL measure that provides two summary scores and eight individual concept scores, each measured on a scale from 0 (poor) to 100 (favorable). The MIQ is a disease-specific HRQoL measure that was completed by a subset of women following the pretreatment cycle and at treatment cycles 1, 2, 3 and 6. The RMQ and SF-36v2 were administered after the pretreatment cycle and at treatment cycles 1, 2, 3, 6, 9, 12 and 15. Change-from-baseline values were calculated for summary measures and individual domains, as appropriate.
Statistical analysis
A sample size of 865 women was determined to ensure that at least 200 women would receive study drug for 1 year or longer, with a minimum of 10,000 treatment cycles. Safety and efficacy analyses were conducted on the intent-to-treat population (all study participants who received at least one dose of study drug). The summaries for quantitative variables included appropriate descriptive statistics. Frequency counts were compiled for classification of qualitative variables. Normal–abnormal shift tables were generated for ophthalmologic examinations, with statistical significance determined using a Stuart–Maxwell test. All hypothesis testing and safety evaluations were two-sided with an α = 0.05 level of significance. Statistical analyses were conducted using statistical software (SAS®, Version 9.1.3, SAS Institute, NC, USA).
Results
A total of 1051 women were screened and 784 women were enrolled in the study (
Patient disposition of the women enrolled in the study.

The number of patients enrolled was used as the denominator for calculated percentages.
The number of patients withdrawn was used as the denominator for calculated percentages.
‘Other event’ category included noncompliance, withdrawal of consent, cessation of heavy menstrual bleeding, pregnancy or trying to conceive and surgical intervention or ablation, among other reasons for discontinuation.
ITT: Intent-to-treat.
Summary of demographic and baseline characteristics
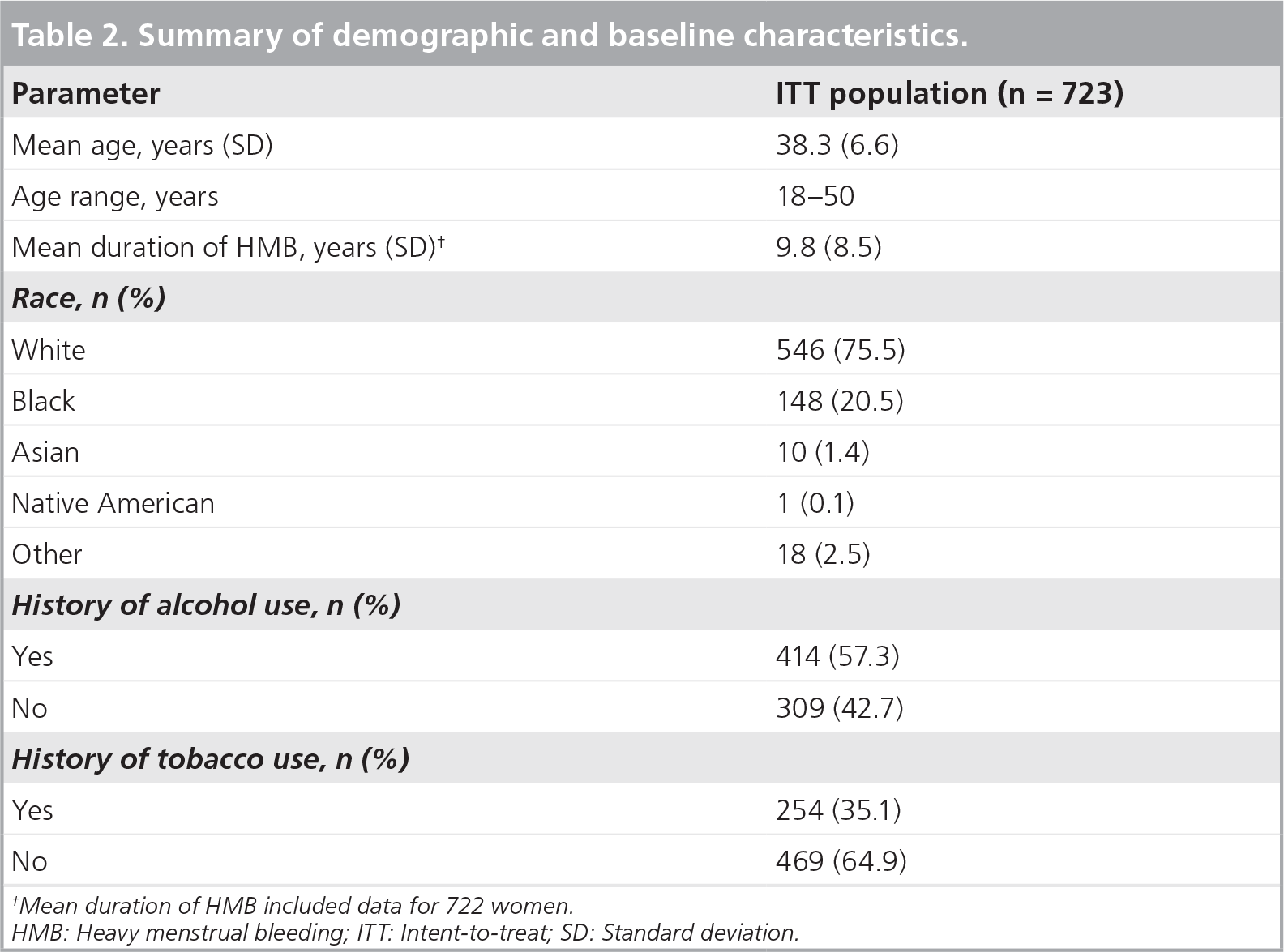
Mean duration of HMB included data for 722 women.
HMB: Heavy menstrual bleeding; ITT: Intent-to-treat; SD: Standard deviation.
A total of 239 women remained in the study throughout all 27 on-treatment menstrual cycles (
Women enrolled in the study accumulated 29,767 days of TA exposure, with an average per patient exposure of 41 days. At least one dose of study drug was taken during 10,213 treatment cycles and 387 women received at least one dose of TA during 12 or more menstrual cycles. The mean daily dose was 3.81 g/day; the average period of drug taking was 2.91 days per menstrual cycle.
Safety
A total of 678 (93.8%) study participants experienced at least one treatment-emergent AE (TEAE; i.e., an AE that occurred after receipt of at least one dose of study drug, regardless of causality). The majority (94.6%) of TEAEs were mild or moderate in severity. Frequently reported TEAEs included headache, menstrual discomfort and back pain (
Frequently occurring treatment-emergent adverse events†

Events that occurred in ≥10% of patients.
Menstrual discomfort includes but is not limited to menstrual cramps, breakthrough bleeding, menstrual pain and spotting.
ITT: Intent-to-treat.
A total of 28 women experienced a total of 42 serious TEAEs; none of these were considered by the investigator to be related to study treatment. The most common serious TEAEs were categorized as reproductive system and breast disorders (1.2% of patients), infections and infestations (1.1% of patients) or nervous system disorders (1.0% of patients). Two women had life-threatening TEAEs: one woman experienced cardiac arrest, pneumonia and pneumococcal sepsis (this woman later died of pneumonia and pneumococcal sepsis); and one woman experienced menorrhagia and was discontinued from the study. This patient was subsequently hospitalized and then underwent elective total abdominal hysterectomy.
A total of 14 pregnancies occurred during the study. There were no fetal abnormalities for those women who carried to full term.
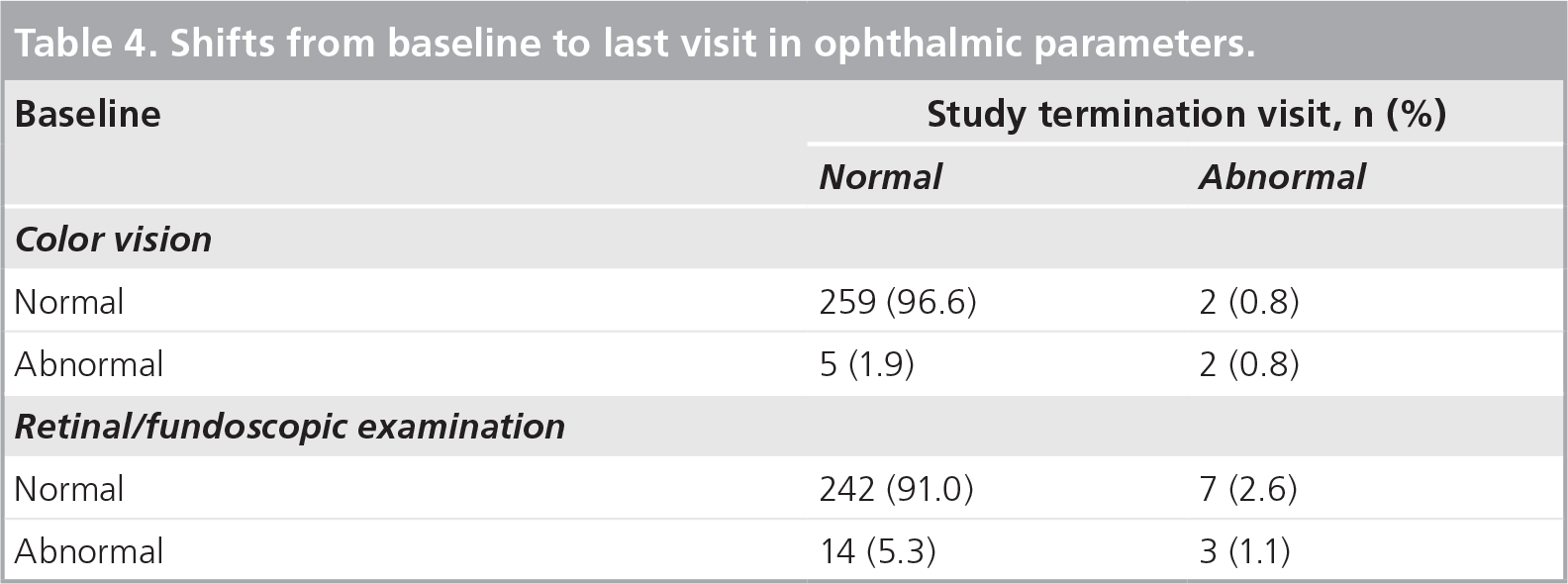
Ophthalmologic examinations
A total of 82 women reported eye disorders during the study. Investigators considered these eye disorders definitely not related to study treatment in 51 women and probably not related to study treatment in 31 women. A few shifts in visual acuity, color vision and retinal/fundoscopic findings occurred during the study, although the majority of women remained within the normal range for these parameters throughout the study (
Shifts from baseline to last visit in ophthalmic parameters
Laboratory parameters
Mean change from baseline and shifts from within the normal range for laboratory parameters were variable and not considered clinically significant. A nonsignificant trend toward a mean decrease in platelet count from baseline (298,791) was observed at visit five (296,237; change from baseline: 1660), visit six (294,000; 4933), visit 9 (288,177; 11,595) and study termination (282,689; 13,233).
Other safety parameters
Changes in blood pressure, pulse rate and weight, and minor changes noted at physical and gynecological examination and on ECGs were not considered to be clinically significant.
Quality of life
Significant improvements in HRQoL, as measured by the SF-36v2 and RMQ scales, were observed over the course of the study (p < 0.05) (

Improvements in health-related quality of life measures.
Data from the MIQ analysis were collected from 131 women who participated in the study and 131 age-matched controls. The RMQ global score highly correlated with each item of the MIQ (range: 0.757 to 0.809), whereas low-to-moderate correlation was observed between MIQ components and SF-36v2 domains (range: −0.303 to −0.118). The strongest correlations between SF-36v2 and MIQ occurred for the ‘Physical’ and ‘Role – Physical’ domains.
Discussion
A novel formulation of oral TA was found to be safe and was well tolerated during long-term administration. Long-term administration was up to 27 weeks with the dosing of TA only during heavy days of menstrual bleeding at 3.9 g/day for up to 5 days. The results of this study are consistent with the established safety profile of TA. As expected, in this study of nonhormonal TA treatment for women with HMB, the commonly reported TEAEs observed were different from the TEAEs reported in recent studies of hormonal treatments for HMB [13,14]. No serious AEs were reported that were considered related to study treatment. To our knowledge, this is the longest duration of tranexamic acid use within a clinical trial or research study.
In the current study, the number of subjects reporting treatment-related GI AEs is comparable to reports of treatment-related GI AEs in previous short-term studies of this novel oral formulation of TA [10]. The subject discontinuation rate due to AEs was 12.4% (97/784), which is comparable to discontinuation rates seen in other studies of tranexamic acid as reviewed by Wellington and Wagstaff (range: 0–12%) [9]. Serious AEs occurred in less than 4% of women, and none of the serious AEs were considered to be related to study treatment.
Ophthalmologic disorders were considered events of interest in this trial because of preclinical findings that indicated a potential for retinal degeneration with markedly high tranexamic acid doses using the immediate-release formulation, as well as in previous reports of color vision or visual acuity disturbances in patients treated with tranexamic acid [8,15]. Visual changes reported in the literature have typically been transient and resolved after drug cessation [15]. In the current study of a novel oral TA formulation, one subject experienced blurred vision that subsequently completely resolved, but resulted in this individual's withdrawal from the study. Otherwise, no significant ophthalmic impairment was evident in subjects.
Although speculation has been raised about the theoretical possibility of increased risk for thromboembolic events with antifibrinolytics such as tranexamic acid, the frequency of thrombosis over 19 years (238,000 patient years) in women treated with tranexamic acid for HMB was similar to the frequency of spontaneous thrombosis in women within the general population [16]. The lack of thromboembolic events in this study confirms data from prior studies demonstrating no increased risk with tranexamic acid treatment [9–11,16]. A recent case–control study detected a nonsignificant increase in risk of venous thromboembolism among patients who had previously been exposed to tranexamic acid; however, significantly increased risk of venous thromboembolism was observed with other HMB therapies as well, including mefenamic acid and noretruster one [17]. In this study it was speculated that HMB may be a prothrombotic condition.
Unlike the pivotal TA efficacy and safety studies, NSAIDs were allowed during dosing in this study, thereby better reflecting a real-world treatment scenario. Certain NSAIDs, such as mefenamic acid, naproxen, ibuprofen and flurbiprofen, have been explored as treatment options for HMB, although their efficacy in reducing menstrual blood loss is generally low [18]. NSAIDs are among the most commonly used prescription or over-the-counter medications in the USA, and for women, they are often used for pain relief during menstruation [19]. Consequently, in the general population, there is a high likelihood that NSAIDs and TA would be used concomitantly. In our study, use of NSAIDs during TA dosing did not adversely affect the safety or tolerability of TA.
Women treated with TA in this study experienced significant improvements in HRQoL measures. In women with cyclic HMB, poorer performance in all eight concept scores of the SF-36v2 has been observed compared with population norms, with the largest deficits in ‘Role – Physical’, ‘Bodily Pain’ and ‘Role – Emotional’ concept scores [20]. Significant improvements from baseline with TA treatment were observed for all three of these domains, which, notably, include measures of interference with work, social or other activities. In an open, uncontrolled study of women with HMB, Winkler reported that subjects described improvements in social activity and work limitations, and increases in alertness, productivity, cleanliness, mood and overall well-being with TA treatment [4]. One of the strengths of the current study is the use of both generic (SF-36v2) and disease-specific (RMQ) HRQoL instruments. As might be predicted, findings from the disease-specific MIQ correlated more strongly with the RMQ than did findings from the SF-36v2.
Some of the observed HRQoL improvement may be linked to the improvements in hemoglobin concentrations observed during the study. In a recent study, de Souza
One limitation of the current study is the lack of a placebo or control group. Without a placebo comparator, the true frequency of AEs associated with TA versus treatment-independent background events is not determinable. Interpretation of HRQoL is also hindered by the lack of a control arm, although baseline data and population norms aid in assessing the importance of observed changes in these parameters.
Conclusion
During a long-term treatment study, women received TA (Lysteda), which was well tolerated when used for up to 5 days during their menstrual cycles. Noticeable and sustained improvements in HRQoL as noted by study participants support TA as a beneficial addition to the current armamentarium available to treat cyclic HMB. TA should be considered as a first-line, nonhormonal treatment option for cyclic HMB.
Future perspective
Pharmacokinetic and clinical assessments of modified-release TA in an adolescent population with evidence of HMB would provide treating physicians with much-needed data regarding the treatment of HMB in this population. Also, studies of modified-release TA used in surgical procedures and dental procedures, and in the treatment of hemorrhages and hemophilia would allow for comparisons against immediate-release tranexamic acid currently being used for these purposes.
Prior presentations
Interim data from this clinical trial were presented at the American Congress of Obstetricians and Gynecologists (ACOG), LA, USA, 3–7 May 2008 and some of these data were presented at the American Society for Reproductive Medicine Annual Meeting, 23–27 October 2010. NIH clinical trials registry: NCT00113568.
Executive summary
The goal of this multicenter, long-term, open-label study was to assess the safety and health-related quality of life (HRQoL) of an oral tranexamic acid (TA; Lysteda®) formulation across 27 menstrual cycles in women with cyclic heavy menstrual bleeding (HMB).
Safety was assessed by treatment-emergent adverse event (TEAE) monitoring, physical examinations, laboratory results, ophthalmologic examinations and electrocardiography. HRQoL was evaluated using both generic and HMB-specific instruments.
Most of the TEAEs were mild to moderate in severity and were largely considered unrelated to study treatment.
The most frequently reported adverse events were consistent with those observed with the use of an immediate-release formulation of TA and included menstrual discomfort, headache and back pain.
The incidence of treatment-related gastrointestinal TEAEs was low, and no evidence of ocular toxicity or prothrombotic effects was observed.
Improvements in generic and disease-specific HRQoL measures were evident during the first treatment cycle and were maintained throughout the 15 cycles of measurement for most domains.
Long-term TA treatment was well tolerated and improved measures of HRQoL in women with cyclic HMB.
During a long-term treatment study, treatment with TA was well tolerated when used for up to 5 days during menstrual cycles and was consistent with the established safety profile.
This formulation of TA adds an oral, nonhormonal therapeutic option for the treatment of cyclic HMB.
