Abstract
Atrial fibrillation (AF) is the most common clinically significant cardiac arrhythmia. The most serious complication of AF is thromboembolic stroke. The individual risk of stroke in the setting of AF varies. Several clinical factors have been identified as independent predictors of stroke in AF, including prior stroke, age, hypertension and diabetes. The bulk of available data identifies female gender as another independent predictor of stroke risk in AF. In this article, we review the link between AF and an elevated stroke risk in women, explore the potential pathophysiologic basis for this association and examine the data regarding the effectiveness of anticoagulation in reducing this risk.
Atrial fibrillation (AF) is the most common clinically significant cardiac arrhythmia [1]. It is well established that AF increases the risk of thromboembolism and stroke. However, individual risk of stroke in the setting of AF varies and may be influenced by the presence or absence of several clinical risk factors [2,3]. Prior thromboembolic events (stroke or transient ischemic attack [TIA]), advanced age, hypertension, heart failure and diabetes have been most consistently identified as predictors of stroke associated with nonvalvular AF [3–5]. Dissenting opinions exist as to whether female gender represents another risk factor. In this article, we explore the evidence for an association between AF and an increased risk of stroke in women. If female gender truly represents an independent predictor for stroke in AF, enhanced clinician awareness and understanding of gender-specific differences is essential to mitigate this risk.
Evidence for female gender as a clinical risk factor for stroke in AF
Several early observational studies pointed to a significant association between AF, female gender and increased risk of thromboembolism [6–8]. Other studies argued against such a relationship [9,10]. A recent systematic review of clinical predictors of stroke in paroxysmal and chronic AF identified 11 studies that assessed female gender as a risk [11]. Five of these 11 studies identified female gender as an independent predictor of stroke [6,12–15], five studies showed no significant interaction between female sex and stroke risk [9,10,16–18], and one identified males as being at a higher risk of stroke in the setting of paroxysmal AF [19]. Notably, several of the studies identified in this analysis were relatively small observational cohorts [11].
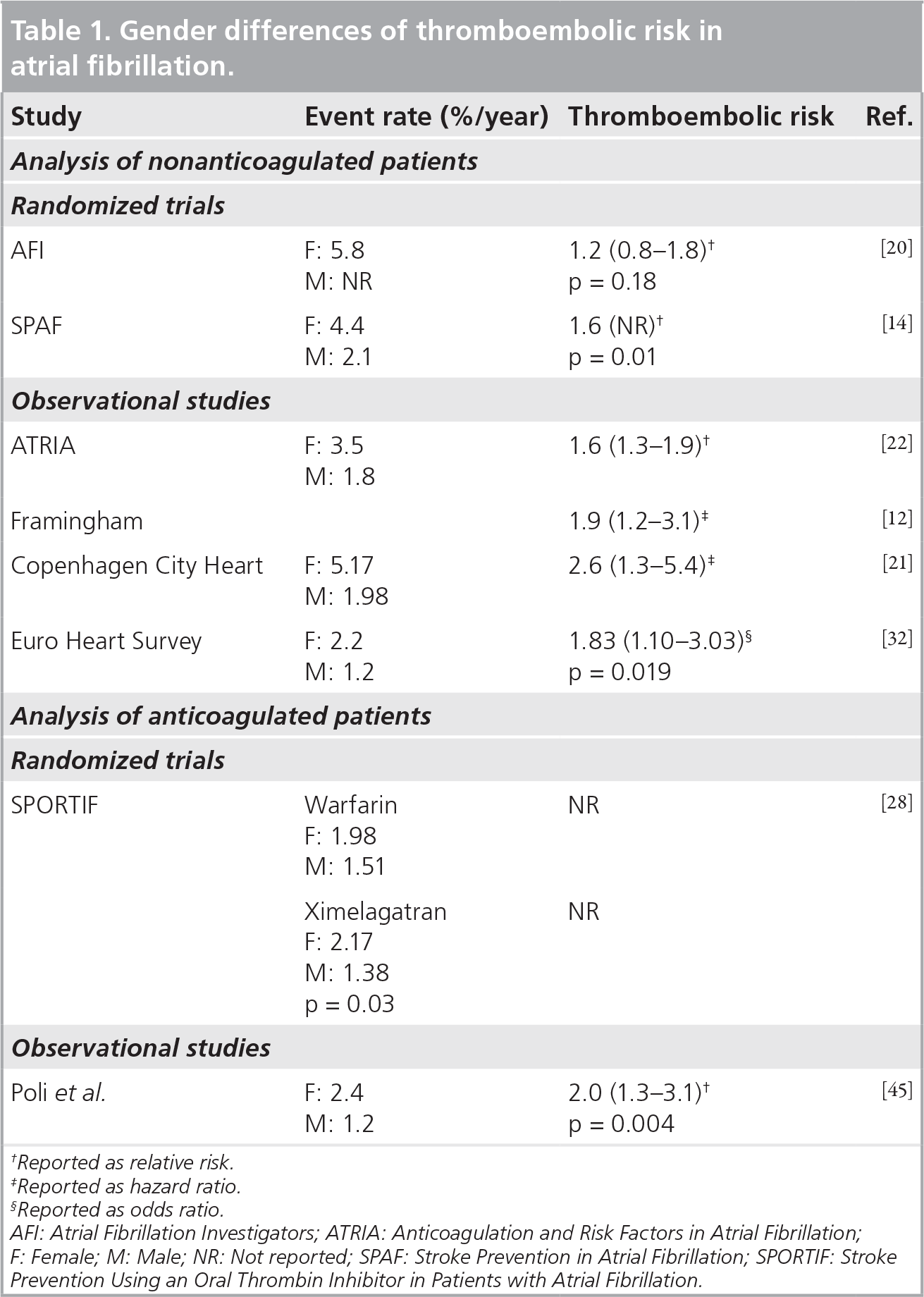
Women are under-represented in randomized trials of stroke prevention in AF, and data regarding female gender as a risk is somewhat contradictory. In a collaborative effort led by the Atrial Fibrillation Investigators (AFI), data from five early randomized trials evaluating the efficacy of warfarin therapy for the prevention of stroke in AF patients were pooled and risk factors for stroke were evaluated [20]. In multivariate analysis of patients assigned to the control group, previous stroke or TIA, old age, hypertension and diabetes proved to be independent predictors of stroke. Women assigned to the control groups in these studies had a slightly elevated, but nonsignificant, stroke risk compared with male controls (relative risk [RR]: 1.2; 95% CI: 0.8–1.8; p = 0.18) [20].
In the Stroke Prevention in Atrial Fibrillation (SPAF) I–III trials, female gender proved to be one of the five independent predictors for stroke (along with age, hypertension, systolic blood pressure >160 mmHg and prior stroke or TIA) in patients with sustained or recurrent AF receiving aspirin therapy (RR: 1.6; p = 0.01) [14]. In addition, a strong interaction between gender and age was noted in this analysis. Although there was little influence on stroke rate in younger women, the stroke rate in elderly women (aged >75 years) was substantially higher than in elderly men (9.7 vs 3.2%/year, respectively; p < 0.001). For participants aged >75 years without other identifiable thromboembolic risk factors, the stroke rate for women was 7.8 versus 1.2%/year for men (p = 0.002) [14].
Female gender was also found to be a significant risk factor for stroke in the analysis of large prospective observational cohort studies. In the Framingham Heart Study, in multivariate analysis among warfarin-censored patients with AF, female gender was identified as an independent predictor of stroke (hazard ratio [HR]: 1.92; 95% CI: 1.20–3.07) [12]. Other independent predictors of stroke risk were hypertension, age, diabetes, and prior stroke or TIA [12]. In the Copenhagen City Heart Study, AF increased the risk of first stroke significantly in both genders; however, the effect of AF on the risk of stroke was over four-times greater in women than in men (HR: 4.5; 95% CI: 2.2–9.2) [21]. In analysis of only the participants with AF, women showed a higher age-adjusted risk of stroke than men (HR: 2.6; 95% CI: 1.3–5.4). In addition, AF was a significant risk factor for overall mortality in both genders; however, the effect of AF continued to be almost twice as large in women as in men (HR: 1.9; 95% CI: 1.2–2.9) [21].
The large Anticoagulation and Risk Factors in Atrial Fibrillation (ATRIA) study cohort included 5795 women and 7764 men [22]. Rates of thromboembolism between male and female patients not taking anticoagulants were assessed, while controlling for other known risk factors of stroke. Women had a higher annual incidence of ischemic stroke (RR: 1.5; 95% CI: 1.2–1.8). Notably, the interaction between older age and female gender seen in the SPAF studies was not observed in the ATRIA cohort. Higher rates of thromboembolism in women were observed at both younger and older ages and across all stroke risk factor categories. The RR for stroke was 1.6 (95% CI: 1.032.3) for those aged ≤75 years and 1.8 (95% CI: 1.4–2.3) for those aged >75 years [22].
In summary, early observational data revealed conflicting evidence for a higher risk of stroke in women. However, the bulk of data from larger, contemporary cohorts and randomized trials identifies female gender as an independent predictor of stroke in the setting of AF. Female gender appears to confer a relative stroke risk of 1.5–1.9
Gender differences of thromboembolic risk in atrial fibrillation.
Reported as relative risk.
Reported as hazard ratio.
Reported as odds ratio.
AFI: Atrial Fibrillation Investigators; ATRIA: Anticoagulation and Risk Factors in Atrial Fibrillation
F: Female; M: Male; NR: Not reported; SPAF: Stroke Prevention in Atrial Fibrillation; SPORTIF: Stroke
Prevention Using an Oral Thrombin Inhibitor in Patients with Atrial Fibrillation.
Female gender & thromboembolism in AF: biology, bias or both?
Prothrombotic biomarkers
Mechanisms for sex-related differences in stroke risk remain uncertain. It is possible that women exhibit increased thrombogenicity in the setting of AF owing to hormonal differences or other procoagulant traits [23]. Elevated markers of endothelial dysfunction and prothrombotic factors have been demonstrated in women with AF [24,25]. Plasma von Willebrand factor level is a marker of endothelial dysfunction, and elevated levels have been linked to atherosclerosis and thrombosis. Recently, elevated von Willebrand factor levels were shown to increase the risk of stroke in the Rotterdam Study, a prospective, elderly population-based cohort [26]. Furthermore, in the Rotterdam Study, the presence of AF was significantly associated with increased levels of von Willebrand factor in women but not in men [24]. The relationship between female gender and elevated von Willebrand factor suggests a greater prothrombotic state and might contribute to the higher stroke risk that is apparent in women with AF.
Hormone replacement therapy
The issue of procoagulant effects of hormone replacement therapy has also been assessed. Among healthy postmenopausal women evaluated in the Women's Health Initiative, estrogen replacement therapy was shown to increase the risk of stroke [27]. In the SPAF trials, hormone replacement therapy was associated with higher rates of ischemic stroke in women with AF (RR: 3.2; p = 0.007) [14]; however, in the ATRIA study, oral estrogen replacement therapy did not result in increased thromboembolic risk [22]. Owing to small numbers, the effect of estrogen replacement in the Stroke Prevention Using an Oral Thrombin Inhibitor in Patients with Atrial Fibrillation (SPORTIF) trials was inconclusive [28].
Thyrotoxicosis
The link between thyrotoxicosis, AF and heightened thromboembolic risk remains controversial [2]. Reports suggest that the risk for thromboembolic stroke is elevated in hyperthyroidism, but a causal relationship is not proven [2,29]. A higher risk of thromboembolic stroke has recently been described in young adults with hyperthyroidism [30]. Hyperthyroidism is known to be associated with an increased risk of AF and is more common in women with AF than in men [31,32]; however, the overall contribution of hyperthyroidism to the increased risk of stroke in women with AF is largely uncertain.
Sex differences in medical management
Risk factor profiles appear to differ significantly between men and women with AF
Gender differences in stroke risk factors in selected atrial fibrillation trials.

Data not reported separately for males and females.
Congestive heart failure or myocardial infarction.
AFI: Atrial Fibrillation Investigators; ATRIA: Anticoagulation and Risk Factors in Atrial Fibrillation; F: Female; M: Male; NR: Not reported; SPAF: Stroke Prevention in Atrial Fibrillation; SPORTIF: Stroke Prevention Using an Oral Thrombin Inhibitor in Patients with Atrial Fibrillation.
Finally, there exists the possibility that other risk factors that have not been systematically evaluated in the setting of AF could account for gender differences. For example, in one study, obesity as measured by increasing waist circumference has been shown to be an independent predictor of stroke in women [34]. Most studies that have identified female gender as an independent risk for stroke in AF do not correct for weight or BMI.
Anticoagulant therapy for stroke prevention in women with AF
The benefit of anticoagulation for the prevention of thromboembolism in AF is clear [42,43]. Current guidelines recommend oral anticoagulation for patients with either paroxysmal or chronic AF who have stroke risk factors [2,102]. This is supported by data from the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) trial, in which a comparison was performed of rhythm control versus rate control strategies for AF [44]. In both groups, the majority of strokes occurred after cessation of warfarin or during times of subtherapeutic international normalized ratio (INR) [44]. Thus, maintenance of sinus rhythm has no bearing on stroke prevention. However, women with AF are often more symptomatic than men [32]. Because of women's proclivity for QT prolongation and torsades de pointes, restoration and maintenance of sinus rhythm with specific antiarrhythmic agents is reserved for symptomatic relief [1]. Regardless of whether a rate or rhythm control strategy is used, anticoagulation remains pivotal in patients with risk factors for stroke.
Few studies have examined gender differences in response to anticoagulation therapy. In pooled data from five randomized trials evaluating the efficacy of warfarin for stroke prevention in AF, anticoagulation decreased the risk of stroke by 84% in women (95% CI: 55–95%; p < 0.001) compared with a 60% reduction in men (95% CI: 35–76%; p < 0.001) [20]. In the ATRIA study, despite similar distribution of INR intensities, the reduction in rates of thromboembolism on warfarin was again larger in women than in men [22]. Anticoagulation with warfarin reduced annual rates of thromboembolism from 3.5 to 1.5% in women versus reduction from 1.8 to 1.2% in men (p = 0.001 for the interaction of sex and warfarin use) [22].
Interestingly, despite the previously mentioned data suggesting that anticoagulation is at least as effective – if not more so – at reducing thromboembolic event rates in women, a recent study suggests that anticoagulated women might still remain at a higher overall risk than men. In a recent analysis of AF patients followed prospectively in an outpatient anticoagulation clinic, women remained at higher risk for stroke than men with no difference in the intensity of anticoagulation [45]. Thromboembolic event rates were 1.2 × 100 patient/years in men versus 2.43 × 100 patient/years in women (RR: 2.0; 95% CI: 1.3–3.1; p = 0.004). A report from the Euro Heart Survey also showed that women were at higher risk for stroke than men, despite no difference in oral anticoagulation prescription rates [32]. However, this difference appeared to be largely driven by women who did not receive oral anticoagulation and were managed with a rhythm control strategy.
Bleeding risks of anticoagulant therapy in women with AF
Despite the apparent efficacy of anticoagulation in women, data suggest that anticoagulant therapy might be underused in women, particularly in the elderly [43,46]. It is believed that this disparity is due to a presumed higher bleeding risk in elderly women. In the ATRIA study, women had similar rates of major hemorrhage compared with men (1.0 vs 1.1%; adjusted RR: 0.8; 95% CI: 0.6–1.1) and were less likely to develop intracranial hemorrhage on warfarin therapy [22]. In the SPORTIF trial, there was again no difference in major bleeding between men and women (2.25 vs 2.15%/year; p = 0.765), although women had higher rates of combined major and minor bleeding (41.3 vs 33.9%/year; p < 0.001) [28]. These findings were in contrast to those of the Euro Heart Survey, which reported a higher major bleeding risk in women (2.2 vs 1.3%; p = 0.028). The Canadian Registry of Atrial Fibrillation (CARAF), also reported that women on warfarin were 3.35-times more likely to experience a major bleed than were men [33]. Of note, the mean INR of the women who experienced major bleeds in this trial was 4.02 ± 2.96. More recently, Poli
In summary, anticoagulation clearly reduces stroke risk in women. The available evidence does not justify withholding this therapy based on an unproven bleeding risk.
Incorporation of gender in risk stratification schemes
Several risk stratification schemes have been developed to help clinicians gauge individual patient thromboembolic risk in AF and to guide the use of anticoagulant therapy. Predictors of thromboembolic risk derived from both randomized trials and large community cohorts have been used to construct these risk schemes. Prior stroke, old age, hypertension and diabetes have most consistently been identified as predictors of stroke associated with nonvalvular AF and have been incorporated into all major risk schemes [2,12,20,48,49]. In addition, history of heart failure or low ejection fraction is also commonly included. Based on risk scheme scores, the individual thromboembolic risk is often categorized as low, intermediate or high risk.
Pooled data from control patients in five stroke prevention trials were analyzed by the AFI [20]. Female gender was associated with a slightly elevated risk of stroke, but was not a significant predictor and thus, was not incorporated into this risk scheme. Analysis of patients on aspirin therapy in the SPAF trials revealed women over 75 years of age to be at increased risk of thromboembolism [14]. AFI and SPAF were used to develop the congestive heart failure, hypertension, age, diabetes, previous stroke (double point; CHADS2) risk score, which owing to its ease of use has become the most widely utilized risk scheme [48]. Female gender is not included among the clinical risk predictors in CHADS2. The 2006 American College of Cardiology/American Heart Association/European Society of Cardiology (ACC/AHA/ESC) guidelines include female gender among a list of ‘less validated or weaker risk factors' [2]. However, use of anticoagulation is given a class I recommendation for women aged >75 years with AF.
The UK NICE national guidelines for AF management evolved from that of the AFIs [37]. The NICE guidelines were further refined into the Birmingham 2009 risk scheme that includes female gender as a risk factor [49]. This point-based scheme, cardiac failure or dysfunction, hypertension, age ≥75 (double point), diabetes, previous stroke (double point), vascular disease, age 65–74 and female sex (CHA2DS2-VASc), was recently validated in the Euro Heart Survey on Atrial Fibrillation cohort [49]. In comparison with other contemporary risk stratification schemes, the CHA2DS2-VASc score improves identification of patients at high risk for stroke and minimizes classification of patients in the intermediate risk category [50–52]. It has been suggested that the CHA2DS2-VASc score be applied as a complement to the more straightforward CHADS2 scheme, affording a more comprehensive assessment of thromboembolic risk to patients with a CHADS2 score of 0–1 [51]. In doing so, CHA2DS2-VASc might more clearly guide physicians in the use of oral anticoagulant therapy in those patients who might otherwise have been labeled at intermediate risk by CHADS2.
Conclusion
Women with AF appear to have a higher thromboembolic stroke risk than men. The mechanism of an elevated stroke risk in women with AF remains unclear and could be multifactorial. Women with AF tend to be older and to have more comorbidities than men with AF. Whether the elevated stroke risk in women with AF is due to a higher clinical risk profile and undertreatment of known cardiovascular risk factors, or rather that female gender itself is associated with an inherent thromboembolic risk, requires further elucidation. Anticoagulation therapy clearly reduces stroke risk in women with AF, with bleeding risks that appear to be similar to those seen in men. Clinicians should be aware of the elevated stroke risk associated with female gender, and manage women aggressively as per guidelines, targeting effective anticoagulation as well as the management of all modifiable cardiovascular risk factors. Gender differences in response to newer oral anticoagulants will need to be monitored closely in the coming years (see following section).
Future perspective
Although vitamin K antagonists have proven to be the agents of choice for reducing stroke risk in AF, the recent introduction of newer anticoagulants makes it likely that the field of stroke prevention will change substantially during this decade. In the Randomized Evaluation of Long-term anticoagulation therapy (RE-LY) trial, the direct thrombin inhibitor, dabigatran, at a dose of 150 mg twice daily was associated with lower rates of stroke and systemic thromboembolism and similar rates of major hemorrhage as compared to adjusted-dose warfarin [53]. Of the 18,113 participants in this trial, 36% were women. Subgroup analysis revealed no significant interaction between sex and treatment effect of dabigatran (p = 0.24) [53]. Interestingly, gender analysis of the SPORTIF trial revealed that treatment with another direct thrombin inhibitor, ximelagatran, was associated with higher rates of stroke or systemic embolism in women as compared to men (2.17%/year; 95% CI: 1.48–2.87%/year vs 1.38%/year; 95% CI: 1.0–1.75%/year; p = 0.03) [28]. Ximelagatran has been removed from the market owing to high incidence of hepatotoxicity; however, the use of other new anticoagulants is on the horizon. As compared to aspirin, the novel factor Xa inhibitor, apixaban, reduced the risk of stroke and systemic embolism without significantly increasing the bleeding risk in patients with AF who were deemed unsuitable for management with vitamin K antagonists [54]. Subgroup analysis revealed no significant gender difference in rates of stroke or systemic embolism (1.9%/year female vs 1.4%/year male; p = 0.42) or in rates of major bleeding (1.5%/year female vs 1.4%/year male; p = 0.97) [54]. In addition, in a Phase III trial, rivaroxaban, an oral direct factor Xa inhibitor, was recently proven noninferior to warfarin in management of AF [55].
Introduction of these newer agents is likely to challenge the role of warfarin as the primary agent for stroke prevention in AF [56]. Although these new anticoagulants appear at least as effective, safer and more convenient than warfarin, the choice to use such agents must take into account potential disadvantages. These include the need for twice-daily dosing with certain agents, the lack of a laboratory test to assess patient compliance, higher cost and the lack of a specific antidote in the case of severe hemorrhage [57,58]. In the coming years, any gender differences in response to these agents will have to be monitored closely.
Executive summary
Women with atrial fibrillation (AF) have a higher thromboembolic stroke risk than men.
The mechanism of an elevated stroke risk in women with AF is likely to be multifactorial; however, women with AF tend to be older and to have more comorbidities than men with AF.
Anticoagulation therapy clearly reduces stroke risk in women with AF, with bleeding risks that appear to be similar to those seen in men.
Clinicians should be aware of the elevated stroke risk associated with female gender, and manage women aggressively as per the guidelines, targeting effective anticoagulation as well as the management of all modifiable cardiovascular risk factors.
Gender differences in response to newer oral anticoagulants will need to be monitored closely in the coming years.
Footnotes
