Abstract
Despite recent advances in the treatment of ovarian cancer, a large majority of women with this diagnosis will die from recurrence of their disease. Targeted therapies, in the form of monoclonal antibodies and small molecule tyrosine kinase inhibitors have significantly altered the management of many solid tumors and hematologic malignancies. No such agents have been approved by the US FDA for use in ovarian cancer, although Phase II data suggests excellent single-agent activity of some of these drugs. Antiangiogenic agents in combination with chemotherapy are being evaluated in Phase III clinical trials, both in the adjuvant setting and in recurrent platinum-sensitive disease. Poly-ADP-ribose polymerase inhibitors are promising agents in BRCA1/2-mutated breast and ovarian cancers. Ongoing clinical trials are exploring the anti-tumor effect of poly-ADP-ribose polymerase inhibitors administered as single agents and in combination with chemotherapy. Many other new drugs are in earlier grades of development. In this article, we review the state of the art in targeted therapies for ovarian cancer and identify future directions for their development in the management of this often devastating disease.
Keywords
Ovarian cancer is the leading cause of death among malignancies of the female reproductive system throughout the developed world and the fifth most common cause of cancer-related mortality among women in the USA [1]. Although progress has been made in the treatment of ovarian cancer leading to improved overall survival (OS) rates compared with historic rates, 5-year survivals remain poor with only 46% of patients living beyond that time point [2]. This is due, in part, to a lack of accurate screening and early detection methods, resulting in a majority of ovarian cancers being diagnosed at advanced grades. The limited availability of effective systemic therapy and the development of chemotherapy-resistant disease also contribute to the poor survival figures. Only 20% of patients present with early-grade disease [2]. These patients have an excellent prognosis with a 5-year survival rate exceeding 80% [3].
The optimal postsurgical chemotherapeutic approach for patients with epithelial ovarian, fallopian tube or primary peritoneal cancer includes chemotherapy with a platinum-taxane doublet [4–6]. The addition of a third cytotoxic agent to the standard carboplatin/paclitaxel combination fails to improve the OS [7]. Three randomized trials have demonstrated superiority of the combination of intraperitoneal (ip.) and intravenous (iv.) chemotherapy over iv. chemotherapy alone [8–10]; however, challenges posed by its delivery and concerns over increased toxicity have resulted in the failure of this approach to gain widespread acceptance [11]. Disease relapse in ovarian cancer remains a common and ominous development, with median survivals of only 12–24 months following recurrence [12].
Improved understanding of tumor biology, cell signaling pathways and tumorigenesis has led to new insights into cancer therapy. The development of more effective systemic treatment strategies promises to substantially improve the poor prognosis for women with advanced ovarian cancer. Targeted agents have been successfully incorporated into the standard management algorithms for many tumor types. In this article, we will discuss the major advancements in targeted therapies for ovarian cancer and identify potential future directions of this broad category of anticancer treatments.
Targeting angiogenesis
Angiogenesis, the process of new blood vessel formation, is essential to sustainable tumor growth and metastasis, and is an important target in the treatment of gynecologic malignancies [13,14]. VEGF is a major contributor to angiogenesis. VEGF-A is the most important member of this subfamily of growth factors, which also includes VEGF-B, VEGF-C, VEGF-D, VEGF-E and PlGF [13].
Preclinical and
Anti-VEGF therapy
Clinical experience with single-agent bevacizumab
Bevacizumab is a humanized monoclonal antibody that inhibits VEGF and has shown promising activity in early-Phase clinical trials in ovarian cancer
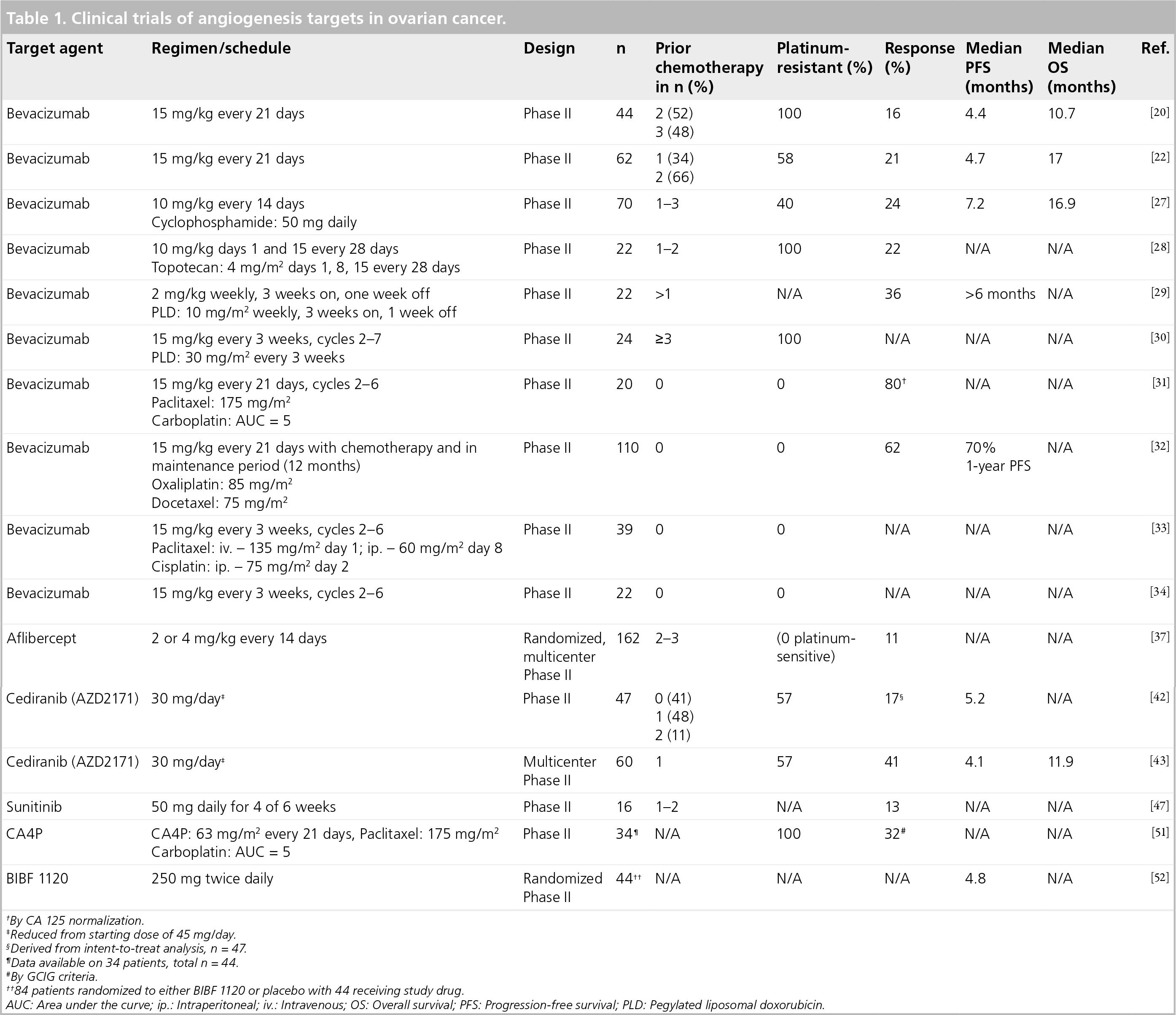
Clinical trials of angiogenesis targets in ovarian cancer.
By CA 125 normalization.
Reduced from starting dose of 45 mg/day.
Derived from intent-to-treat analysis, n = 47.
Data available on 34 patients, total n = 44.
By GCIG criteria.
84 patients randomized to either BIBF 1120 or placebo with 44 receiving study drug.
AUC: Area under the curve; ip.: Intraperitoneal; iv.: Intravenous; OS: Overall survival; PFS: Progression-free survival; PLD: Pegylated liposomal doxorubicin.
Two Phase II clinical trials with single-agent bevacizumab have been conducted in ovarian cancer. In an industry-sponsored Phase II trial of 44 patients with platinum-refractory epithelial ovarian cancer (EOC) or peritoneal serous carcinoma, bevacizumab, 15 mg/kg every 21 days, has demonstrated a response rate of 15.9% and a median OS of 10.7 months [20]. This trial was closed prematurely because of a high rate of GIP (11%) compared with previous experience in other tumor types (1.5–2.4%) [21]. Analysis of the toxicity data from this trial suggested that incidences of GIP were seen primarily in more heavily pretreated patients (those receiving more than two prior regimens), in platinum-refractory EOC and in patients who demonstrated radiographic evidence of bowel wall thickening or bowel obstruction. This study also reported an unexpected rate of arterial thromboembolic events. Other bevacizumab-related complications such as grade 3 or 4 hypertension, proteinuria, bleeding, wound healing complications and venous thromboembolic events were seen at the anticipated rates.
A similar Gynecologic Oncology Group (GOG) study utilizing the same schedule of single-agent bevacizumab in persistent or recurrent EOC and primary peritoneal cancer showed promising clinical activity without the GIP complications reported in the previous clinical trial [22]. GOG 170-D enrolled patients with both platinum-sensitive and platinum-resistant disease who had received two or fewer previous chemotherapy regimens. A RR of 21% and a median OS of 17 months were reported in 62 patients treated on this study. Expected toxicities were seen; however, no GIP was observed. The differences in the complication rates reported by these two studies might be explained by differences in eligibility. The industry trial included only women with primary or secondary platinum-resistant disease who had a higher number of prior regimens (half of the study population had received three prior regimens). The GOG study enrolled both platinum-sensitive and platinum-resistant patients and allowed up to two prior regimens.
A recent meta-analysis suggests that bevacizumab remains a significant risk factor in the development of GIP in cancer treatment, occurring in 0.9% of treated patients with a mortality rate of 22% [23]. The rate of GIP may be as high as 4.8% in ovarian cancer [24] and continued vigilance is recommended when incorporating bevacizumab into future clinical trials in ovarian cancer. A small series of 13 patients with recurrent ovarian and fallopian tube cancer combining bevacizumab with the EGFR inhibitor, erlotinib reported an increased rate (15%) of bowel perforations [25].
Bevacizumab in combination with chemotherapy
When added to chemotherapy, bevacizumab has demonstrated improved outcomes and tolerability in both metastatic colorectal cancer and non-small-cell lung cancer [21,26]. Phase II trials of bevacizumab in combination with chemotherapy in recurrent ovarian cancer have shown encouraging clinical activity and acceptable toxicity profiles.
The combination of bevacizumab and low-dose metronomic oral cyclophosphamide in 70 patients with EOC who received 1–3 previous lines of chemotherapy showed a RR of 24% and a median OS of 16.9 months [27]. Four episodes of GIP (6%) were observed, but no predictive factors were identified.
A Phase II trial of weekly topotecan and bevacizumab in 22 patients with platinum-refractory ovarian or primary peritoneal cancer showed similar results, with a partial response (PR) rate of 22% and stable disease (SD) in 28% of patients [28]. Study participation was limited to patients who had previously received a maximum of two chemotherapy regimens. No patients discontinued the study owing to treatment-related toxicity and no bowel perforations were observed.
Small trials of bevacizumab in combination with pegylated liposomal doxorubicin (PLD) have also been recently reported. In a trial of 22 patients with recurrent ovarian cancer previously treated with chemotherapy receiving weekly PLD and bevacizumab, a complete response (CR) rate of 9% and a PR rate of 27% were reported [29]. GIP was observed in one patient and hypertension was reported as being manageable. Preliminary toxicity data from another Phase II study of PLD and bevacizumab in platinum-resistant ovarian cancer also suggest that this doublet is well-tolerated [30].
Bevacizumab in combination with first-line chemotherapy
The combination of carboplatin and paclitaxel plus bevacizumab as a first-line strategy was reported in a trial of 20 patients with advanced grade epithelial ovarian cancer, following cytoreductive surgery [31]. Bevacizumab was administered with cycles two through to six. The overall RR, defined by normalization of CA-125 levels, was observed in 80% of patients with 30% showing CRs and 50% showing PRs. No GIP or wound complications were observed, and grade 3 hypertension was observed in only 10% of the study subjects. More recently, the preliminary results of a Phase II study of a novel chemotherapeutic regimen of oxaliplatin and docetaxel plus bevacizumab followed by maintenance bevacizumab every 3 weeks for a total of 12 months of therapy after debulking surgery for ovarian, primary peritoneal or fallopian tube cancer has been reported [32]. A total of 110 subjects were included in the toxicity analysis; 55 patients had measurable disease and 61% of patients were optimally debulked. The CR rate was 32.8% and the PR rate was 29.1% with a 1-year progression-free survival (PFS) estimated at 70.1%. One episode (1%) of GIP was reported and 9% of patients experienced grade 3 or 4 hypertension.
Data are also emerging from trials of bevacizumab and ip. chemotherapy regimens in the setting of first-line therapy for optimally debulked patients with ovarian or primary peritoneal cancer. In a Phase II study of 39 women with optimally debulked grade II or III ovarian cancer, patients received iv. and ip. paclitaxel, ip. cisplatin and iv. bevacizumab as first-line therapy [33]. This was followed by the continuation of bevacizumab every 3 weeks for 17 treatments. This regimen appears to be feasible and appropriate for further study. One patient (2.5%) died following dehiscence of a colonic anastamosis and three episodes (8%) of grade 3 small bowel obstructions were noted. Another Phase II trial (n = 22) of iv. bevacizumab, iv. paclitaxel and ip. cisplatin followed by bevacizumab consolidation in optimally debulked ovarian cancer reported no additional acute toxicities related to bevacizumab [34].
Ongoing Phase III trials of bevacizumab in ovarian cancer
The encouraging results of these Phase II clinical trials have prompted the GOG to initiate Phase III trials of bevacizumab in combination with chemotherapy both in first-line treatment (GOG 218 and GOG 252) and in recurrent platinum-sensitive disease (GOG 213)
Bevacizumab Phase III trial summary.

Patients determined not to be candidates for secondary cytoreductive surgery will be randomized to receive carboplatin plus paclitaxel chemotherapy or the same chemotherapy plus bevacizumab.
AUC: Area under the curve; GI: Gastrointestinal; GOG: Gynecologic Oncology Group; ICON7: The seventh International Collaborative Ovarian Neoplasm study
ip.: Intraperitoneal; iv.: Intravenous; OCEANS: Ovarian Cancer Evaluation of Avastin and Safety; OS: Overall survival; PFS: Progression-free survival; QOL: Quality of life.
In platinum-sensitive recurrent ovarian or primary peritoneal carcinoma, GOG 213 will explore two principal hypotheses. First, this study has been designed to evaluate the role of secondary surgical cytoreduction in recurrent ovarian cancer; and second, it has been designed to determine the utility of adding bevacizumab to second-line carboplatin and paclitaxel to improve OS.
The seventh International Collaborative Ovarian Neoplasm (ICON7) study, comparing standard chemotherapy with and without bevacizumab, completed accrual of 1528 patients in February 2009. This large, international trial randomized high-risk early grade (FIGO grade I or IIA, clear cell or grade 3 carcinoma) and advanced grade (FIGO grade IIB or greater, all grades and all histological subtypes) ovarian cancer patients to receive standard carboplatin and paclitaxel versus chemotherapy plus bevacizumab at 7.5 mg/kg every 3 weeks for six cycles followed by maintenance bevacizumab for an additional 36 weeks. This study, along with GOG 218 and 252, promises to further define the role of first-line bevacizumab.
Another ongoing Phase III study will explore the combination of bevacizumab with carboplatin and gemcitabine (Ovarian Cancer Evaluation of Avastin and Safety-AVF4095s [OCEANS trial]).
Other targets in the VEGF pathway: VEGF trap
Other strategies to target the VEGF pathway include the administration of VEGF-trap, a fusion protein designed to bind VEGF-A. VEGF-trap binds and neutralizes all forms of VEGF-A as well as PlGF and has been shown to decrease tumor burden, inhibit ascites formation and initiate vascular remodeling in preclinical ovarian cancer models [35]. Preliminary results of a multicenter, randomized, Phase II study of VEGF-trap (aflibercept) given as a single agent at 2 or 4 mg/kg iv. every 14 days, were reported [36]. The study accrued 162 heavily pretreated patients including those with two or three previous chemotherapy regimens. Five patients (11%) achieved a PR. Serious adverse events included 4% of patients with hypertension and 2% with GIP. Final results are pending.
Inhibition of angiogenesis by small molecule kinase inhibitors
Several agents, including tyrosine kinase inhibitors of the VEGF pathway and multikinase inhibitors with VEGF inhibitory properties, have shown promise in early Phase studies in ovarian cancer.
Cediranib (AZD2171) is a potent orally bioavailable tyrosine kinase inhibitor (TKI) of VEGFR-2 (kinase insert domain receptor [KDR]) activity, an important mediator of VEGF-induced effects [37]. Phase I trials of cediranib showed evidence of clinical activity and tolerability at the maximally tolerated doses of 30 or 45 mg/day [38–40].
A Phase II study of cediranib enrolling 47 patients with recurrent EOC demonstrated a clinical benefit rate (defined as CR, PR, SD >16 weeks or CA-125 nonprogression of greater than 16 weeks) of 30% [41]. The initial starting dose of 45 mg/day was decreased to 30 mg/day after the first 11 patients experienced increased toxicity. The adverse events reported on this study were similar to other TKIs of the VEGF pathway, including hypertension (46%), fatigue (24%) and diarrhea (13%). No bowel perforations were observed. The stated overall RR of 17% may be underestimated as this figure incorporated an intent-to-treat analysis. A total of 11 patients who were treated at 45 mg/day were censored from response analysis. The median PFS of 5.2 months was comparable to Phase II studies of bevacizumab in EOC [20,22] and the median OS has yet to be determined.
Another early trial of cediranib in relapsed or persistent ovarian cancer demonstrated that a daily dose of 30 mg was better tolerated than 45 mg/day [42]. Overall response and prolonged stable disease rates were 41 and 29% for platinum-sensitive and platinum-resistant disease respectively, suggesting potential activity. The median time to progression and median survivals were 4.1 and 11.9 months respectively with no significant differences between groups.
Pazopanib (Votrient®) is an orally bioavailable multitargeted receptor TKI of VEGFR-1, VEGFR-2, VEGFR-3, PDGFR-α/β and c-kit approved by the FDA in October 2009 in the treatment of patients with advanced renal cell carcinoma. Early Phase II trial results of pazopanib in advanced, previously treated ovarian cancer showed activity as a single agent with response by reduction in CA-125 levels noted in 47% of patients [43]. The most common toxicities were diarrhea and elevation of liver enzymes.
Ongoing Phase III trials
A randomized Phase III international study in patients with platinum-sensitive ovarian cancer (ICON6) will compare standard carboplatin and paclitaxel with and without cediranib (AZD 2171), given concurrently and as maintenance therapy. The accrual goal is 2000 patients randomized to one of three treatment arms, including the reference arm (A) which is chemotherapy alone, versus chemotherapy with concurrent cediranib (arm B), versus chemotherapy with concurrent cediranib plus maintenance cediranib (arm C).
A Gynecological Cancer Intergroup (GCIG) trial of pazopanib (AGO-OVAR 16) is a randomized, double-blind, placebo-controlled study in ovarian cancer designed to evaluate pazopanib monotherapy in patients who have not progressed following standard first-line chemotherapy. The goal duration of study-drug therapy is 12 months. This study opened in 2009 with an accrual goal of 900 subjects.
Other agents under investigation
Sunitinib malate is an oral multitargeted TKI with antitumor and antiangiogenic activity via inhibition of the VEGF and platelet-derived growth factor receptor pathways [44,45]. Sunitinib has undergone small-scale early-phase trials in EOC. Preliminary results from the National Cancer Institute of Canada Clinical Trials Group IND 185 study showed activity and tolerability in advanced ovarian cancer [46].
Combretastatin A-4 phosphate (CA4P) is a novel tubulin-binding vascular-disrupting agent that has been shown to decrease tumor blood flow in animal models and Phase I human subject trials [47,48], and may potentiate antitumor activity of cytotoxic chemotherapy [49]. CA4P has been studied in combination with carboplatin and paclitaxel in a Phase II study of patients with platinum-resistant EOC and appears to increase the RR of chemotherapy without adding significant toxicity [50]. A Phase III trial of CA4P is anticipated.
BIBF 1120 (Vargatef™) is a triple angiokinase inhibitor that targets VEGFR, PDGFR and FGF. A randomized Phase II placebo-controlled study of BIBF 1120 in relapsed EOC showed delayed disease progression with a 36-week PFS rate of 15.6% in the study arm compared with 2% in the placebo group [51]. Gastrointestinal toxicities and transaminitis occurred more frequently in the BIBF 1120 arm.
Vandetanib (ZD6474), a multitargeted TKI with activity against KDR/VEGFR-2 and EGFR [52], is currently undergoing Phase II evaluation in EOC (NCT00445549). CP 547,632 is an orally bioavailable multikinase inhibitor with activity against VEGFR-2 and basic FGFR kinase [53] and is also undergoing Phase II trials in recurrent EOC (NCT00096239).
Inhibition of Erb family pathways
The Erb family of transmembrane receptors includes EGFR or HER1/ErbB1, HER2/ErbB2, HER3/ErbB3 and HER4/ErbB4 [54]. Increased EGFR expression has been reported in 19–98% of epithelial ovarian cancers [55–57] and has been shown to be a predictor of poor-prognosis disease [58–60]. However, clinical trials of EGFR inhibitors in ovarian cancer, either in the form of small molecule TKIs or monoclonal antibodies directed at cell surface receptors, have been mostly disappointing.
Erlotinib (Tarceva®), an orally available small molecule EGFR/HER1 TKI, showed minimal antitumor single-agent activity, with 6% overall response noted in a Phase I–II study in recurrent advanced ovarian cancer [61]. A total of 44% of patients on this trial had stable disease and the 1-year survival rate was 35%, suggesting that erlotinib, similar to other targeted agents, may contribute to disease stabilization rather than inducing a response. However, valid interpretation of such findings in small trials is difficult and may simply reflect patient selection and natural disease course. Patients on this study had EGFR-positive disease by immunohistochemistry, but there was no correlation between EGFR expression and response to therapy. The treatment was well tolerated, with the dose-limiting toxicity being diarrhea. Rash was a commonly encountered toxicity and, as has been noted in studies of erlotinib in other tumor types [62], increased severity of the rash was associated with improved survival.
A feasibility study of erlotinib in combination with first-line docetaxel 75 mg/m2 and carboplatin area under the curve of 5 in patients with ovarian cancer, established an maximally tolerated dose (MTD) for erlotinib of 75 mg/day, with dose-limiting toxicities including persistent diarrhea and emesis [63]. The MTD of 75 mg/day was lower than that established in other combination trials, perhaps owing to the additive toxicities of carboplatin and docetaxel. The overall RR was modest at 52%.
Gefitinib (Iressa®), another TKI of EGFR, was evaluated as a single agent in Phase II trials in recurrent ovarian cancer and has shown disappointing results [64,65]. As observed in similar studies with erlotinib, a small number of patients had prolonged (>6 months) PFS [65], which on this study was correlated with EGFR expression. A Phase II trial of gefitinib and tamoxifen in platinum-resistant or refractory disease did not demonstrate any clinically significant activity [66].
Cetuximab (Erbitux®) is a monoclonal antibody that inhibits the activity of EGFR by effectively competing for cell surface receptor binding with EGF ligands. Cetuximab demonstrated only minimal activity as a single agent in epithelial ovarian cancer and failed to improve the response and the survival rates when added to chemotherapy [67–69], despite attempts to dose cetuximab based on the skin acneiform rash, which correlated with activity in other tumor types [70].
Targeting HER2
Abnormal expression of HER2 has been observed in a number of solid tumor types, most notably in breast cancer where
A Phase II trial of single-agent trastuzumab in recurrent ovarian cancer showed minimal activity, with a reported overall RR of 7% [73]. Of the screened population, only 11% demonstrated HER2 overexpression. Another Phase II trial investigating the addition of trastuzumab to carboplatin and paclitaxel in patients with resistant advanced ovarian cancer demonstrated similar low rates of HER2 overexpression in only 6.4% of 320 screened patients [74]. Of the 20 with HER2-positive disease, only seven met eligibility criteria. However, three of the seven patients achieved a CR, suggesting that the addition of trastuzumab to platinum-based chemotherapy may reverse platinum resistance in HER2-positive ovarian cancer.
Pertuzumab (Omnitarg™) is a humanized monoclonal antibody that inhibits dimerization of HER2 with other ligand-activated HER receptors, independently of HER2 expression levels [75,76]. This suggests a more promising role of pertuzumab compared with trastuzumab in ovarian cancer, where HER2 expression is limited. Single-agent pertuzumab has minimal activity (RR 4%) [77], but its addition to gemcitabine chemotherapy appears to significantly improve activity. In a randomized Phase II trial of 135 patients with relapsed, platinum-resistant ovarian cancer, gemcitabine plus pertuzumab showed an improved RR and a trend toward improved PFS compared with gemcitabine plus placebo [78]. The study also suggests that low HER3 mRNA expression, a possible marker of aggressive chemoresistant disease, may predict for response to pertuzumab.
A Phase II trial of lapatinib, a TKI of EGFR and HER2 in combination with topotecan in recurrent ovarian cancer was terminated early owing to poor activity [79].
In summary, EGFR and HER2/neu inhibitors have not demonstrated significant single-agent activity in ovarian cancer and there is little to suggest that combining these agents with chemotherapy will be an effective strategy.
PARP inhibition
Poly ADP-ribose polymerase-1 (PARP1) is an enzyme that serves a key role in the repair of DNA single-strand breaks via base-excision repair [80]. Loss of PARP1 activity leads to accumulation of double-stranded DNA breaks, which, in normal cells, may be repaired via homologous recombination [81].
A Phase I study of the oral PARP inhibitor olaparib (AZD2281) has been reported [83]. This study was initially open to patients with advanced solid tumors, but in the expansion phase, only patients with
A Phase II trial of olaparib in BRCA-deficient advanced ovarian cancer has also been recently reported [84]. This study enrolled patients with chemotherapy-refractory BRCA-mutated ovarian cancer. The study design included two patient cohorts treated with olaparib, 400 mg twice daily (n = 33) and 100 mg twice daily (n = 24). Patients received continuous oral olaparib in 28-day cycles. The rationale for the treatment dose and schedule was based on the Phase I study mentioned above. At the interim analysis, the overall response rate (ORR), which represented the primary efficacy end point, was 33% at the 400-mg dose level compared with 12.5% at the 100-mg dose level. The clinical benefit rate, defined as ORR and/or more than 50% decline in CA-125 level, was 57.6% at 400 mg b.i.d. compared with 16.7% at 100 mg. Olaparib was well tolerated with primarily grade ½ toxicity observed.
In addition to olaparib, multiple PARP inhibitors including AG0146999 (Pfizer), ABT-888 (Abbott) and BSI-201 (BI Par) are undergoing evaluation in Phase I and Phase II clinical trials. These studies will explore PARP inhibitors as a monotherapy and in combination with chemotherapy. For some of these agents, the optimal treatment schedule (i.e., intermittent vs continuous administration) and the most effective doses have yet to be defined. In ovarian cancer, PARP inhibitors are being investigated both in BRCA-associated and sporadic ovarian cancer.
There has also been increasing interest in PARP inhibitors in treating platinum-resistant disease. Preclinical data suggest that inhibition of PARP can increase platinum chemosensitivity or reverse platinum resistance [85]. In a BRCA-associated mouse breast cancer model, the addition of the PARP inhibitor AZD2281 to cisplatin has been shown to significantly increase recurrence-free and OS compared with carboplatin or cisplatin alone [86]. However, early clinical trials have produced unclear or contradictory results with regard to platinum-resistant and platinum-sensitive ovarian cancer [84,85]. One reason may be the development of secondary BRCA mutations, consistent with restoration of BRCA2-mediated DNA repair function [87]. Ongoing clinical trials may help to better define the target populations of women with ovarian cancer that would benefit from PARP inhibitors.
Folate receptor-α
Folate receptor-α (FRα) is a folate-binding protein that is overexpressed in greater than 70% of ovarian cancers but is only rarely present in normal tissue [88,89]. FRα is implicated in tumor proliferation, possibly owing to growth advantages provided by upregulation of folate transporters in rapidly dividing cells [89]. MORAb-003 is a humanized monoclonal antibody with strong affinity for FRα that inhibits growth of FRα overexpressing ovarian cancer cells
PI3K/AKT/mTOR cell signaling pathway
Mammalian target of rapamycin (mTOR) is a protein kinase, which, as part of the mTORC1 and mTORC2 complexes, is thought to have a key role in the proliferation and angiogenesis of human cancers via the phosphoinositide-3-kinase (PI3K)/AKT cell signaling pathway [91,92]. Dysregulation of this pathway appears to be important in the development of many tumors, including ovarian cancer, and is an attractive target for novel therapy [93,94]. The mTOR inhibitors temsirolimus (Torisel®) and everolimus (Afinitor, RAD001) have been shown to be active in the treatment of renal cell carcinoma and are approved by the FDA for that indication [95,96].
The overexpression of mTOR or activation of the PI3K/AKT/mTOR pathway is not observed in normal ovarian tissue, but is common in ovarian malignancies and may be of prognostic significance [97,98]. Preclinical studies in ovarian cancer xenograft models showed that everolimus inhibited tumor growth, angiogenesis and tumor invasiveness and enhanced cisplatin-induced apoptosis in cells with high AKT/mTOR activity [99]. Other preclinical data suggest that dual targeting of VEGF-A and mTOR may produce additive antitumor effects [100,101]. The results of GOG 248, a randomized Phase II trial of temsirolimus with and without concomitant hormonal therapy, are pending. Other clinical trials involving mTOR and PI3K-targeting agents are also ongoing. A multi-institutional Phase II trial sponsored by the National Cancer Institute Clinical Trials Evaluation Program of temsirolimus and bevacizumab in combination is currently ongoing.
Hormonal therapy
Endocrine therapy is a mainstay of breast cancer treatment, owing to the sensitivity of hormone receptor-expressing tumors to hormonal manipulations. A majority of ovarian cancers are known to express estrogen receptors, and response to hormonal therapy may be predicted by the degree of estrogen receptor expression [102].
A Cochrane review updated in 2002 compiled data from 11 nonrandomized series and two randomized trials [103]. A total of 60 of 623 patients treated on these protocols (9.6%) had an objective RR and 31.9% had stable disease of 4 weeks or more. The RRs reported by the individual studies varied widely, from zero to 56%. Despite this activity, the authors concluded that there is no reliable evidence that tamoxifen either prolongs survival or leads to an improved quality of life. Another review of tamoxifen in ovarian cancer reported an ORR of 13%, but suggested that this figure may be an underestimation of the activity of antiestrogen therapy since many patients who received tamoxifen had been heavily pretreated with chemotherapy [104].
Other interest in tamoxifen has grown from preclinical evidence that hormonal blockade may be important in reversing resistance to chemotherapeutic agents, particularly to platinum compounds vital to current ovarian cancer management [105–107]. Two studies of combination therapy with platinum chemotherapy and tamoxifen have shown conflicting results. One Phase II trial of platinum (cisplatin or carboplatin) plus tamoxifen in 50 patients with relapsed or persistent ovarian cancer following platinum-based chemotherapy reported a 50% ORR (30% CR) with a median duration of 8.5 months [108]. A similar trial of carboplatin plus tamoxifen found no activity, either by measurable disease or CA-125, among 14 patients with relapsed or persistent disease [109]. This disparity may have been due to the fact that the latter study consisted of more heavily pretreated subjects with 71% deemed to have ‘taxane-resistant’ disease.
Aromatase inhibitors have also been studied in ovarian cancer. Initial trials with letrozole (Femara™) and anastrazole (Arimidex™) demonstrated the approach to be well-tolerated but with minimal antitumor activity [110,111]. However, these studies did not prospectively select patients with hormone receptor-positive disease. A study of letrozole in preselected ER-positive relapsed ovarian cancer showed a RR of 17% by CA-125 (>50% decrease) and 9% by radiographic criteria [112]. Radiographic SD was observed in 42% of patients and 26% had PFS greater than 6 months. Response was predicted by low HER2 expression.
A Phase II trial of exemestane (Aromasin®) in refractory ovarian cancer reported stable disease of greater that 14 weeks in eight of 22 patients (36%), with a median duration of 23 weeks [113]. One patient had prolonged disease stabilization exceeding 95 weeks. All patients on this trial received prior chemotherapy with platinum/taxane combinations; 68% had received two prior lines of chemotherapy.
A small Phase II study of fulvestrant (Faslodex®), an estrogen receptor antagonist without agonist effect, in multiply recurrent ER-positive ovarian cancer has been reported [114]. In this heavily pretreated cohort (median number of previous chemotherapeutic regimens: 5; range: 2–13), activity was low, with an overall RR of 8%; however, stable disease was observed in 50% of patients (by Response Evaluation Criteria in Solid Tumors [RECIST] criteria) and the median time to progression was 62 days.
Future perspective
With the pace of development of novel targeted therapeutics for all cancer types increasing, the need to identify new strategies for their application becomes ever more important. The term ‘targeted therapy’ implicitly suggests that molecular biomarkers may be used, at least theoretically, to predict for clinical response to agents targeting those molecules. In breast cancer, ER, PR and HER2 have been prospectively validated as predictive biomarkers for both response and survival and are used routinely in clinical settings as a means of determining appropriate therapy [115–117]. Retrospective studies in non-small-cell lung cancer have shown mutations in the
Such clinically validated predictive biomarkers for ovarian cancer have yet to be incorporated into clinical practice. Although VEGF expression has been shown to have prognostic value in preclinical and retrospective studies in ovarian cancer, it has not been correlated prospectively to clinical benefit from VEGF inhibitors such as bevacizumab. Similarly, germline mutations in BRCA promise to predict for response to PARP inhibitors; however, these data have yet to be substantiated.
The results of pivotal trials of bevacizumab, both in front-line treatment (GOG 218, 252 and ICON7) and recurrent platinum-sensitive disease (GOG 213) will help to determine the role of this agent in ovarian cancer management. Based on results from Phase II studies of single-agent bevacizumab, this drug is perhaps the most promising of the investigational agents discussed in this review to win approval from the FDA in the near future. However, its role either as an additive to first-line chemotherapy or as maintenance therapy has yet to be substantiated; in addition, the identification of factors that may predict for serious adverse toxicities, such as gastrointestinal perforation, is pending.
PARP inhibitors have also demonstrated clear promise, particularly with respect to BRCA½-mutated ovarian cancers. However, as a class their path to final approval is, at present, in its early grades, since optimal doses and treatment schedules still have to be determined.
Another matter that merits discussion is that of determining how targeted therapies will be evaluated in the future. In addition to measures of survival, the RR, based on the RECIST, has traditionally been used to assess the efficacy of cytotoxic agents. However, clinical trials evaluating targeted therapies in solid tumors have demonstrated that some agents may produce high rates of prolonged disease stabilization in the absence of significant RRs [121–123]. Such findings suggest that tumor RRs may be less important in assessing the efficacy of targeted agents and highlight the need for establishing more appropriate statistical end points. Other data, such as pharmacological effects and disease stabilization may be more relevant in determining the utility of targeted therapies.
Executive summary
Increased VEGF expression is associated with poor prognosis and decreased survival in ovarian cancer.
VEGF-A, or simply VEGF, is the most important of a family of growth factors including VEGF-B, VEGF-C, VEGF-D, VEGF-E and PlGF.
Phase II trials of single-agent bevacizumab demonstrated response rates of 15.9 and 21%.
Early trials of bevacizumab in combination with chemotherapy have shown tolerability and encouraging activity in both front-line and recurrent disease settings.
Higher than expected incidences of life-threatening gastrointestinal perforation with bevacizumab suggest that this agent should be avoided in heavily pretreated, platinum-resistant disease.
VEGF-trap (aflibercept) has shown significant activity (overall response rate: 11%) in a Phase II trial.
Cediranib has shown activity as a single-agent (overall response rate: 17%).
Sunitinib malate, combretastatin A-4 phosphate (CA4P), BIBF 1120 and vandetanib (ZD6474) are other promising novel agents currently under investigation.
Erlotinib has limited activity as a single agent but acceptable toxicity profile when added to chemotherapy.
Gefitinib, an oral EGFR TKI, has not shown significant activity as a single agent therapy.
Cetuximab, either alone or in combination with chemotherapy, has also shown disappointing activity.
HER2 overexpression in ovarian cancer is associated with poor prognosis and decreased survival but occurs with variably reported frequency.
Trastuzumab has minimal activity as a single agent but may reverse platinum resistance.
Pertuzumab has shown poor activity as a single agent but may improve response in platinum-resistant relapse.
A trial of lapatinib was terminated early owing to poor activity.
Trials of EGFR- and HER2-targeted agents have been disappointing.
Loss of PARP1 activity leads to accumulation of dsDNA breaks normally repaired by homologous recombination – a function that is lost in BRCA½-related cancers.
Tumor cells with loss of BRCA½ function, as in BRCA½-mutated cancers, are sensitive to inhibition of PARP1 via synthetic lethality.
A Phase II trial of the PARP inhibitor, olaparib, demonstrated a 57% clinical benefit rate.
Folate receptor-α is a folate-binding protein overexpressed in more than 70% of ovarian cancers but rarely presents in normal tissue and is implicated in tumor proliferation.
A Phase II trial of MORAb-003 plus platinum/taxane chemotherapy showed increased overall response rate compared with historical rates.
mTOR overexpression and/or activation of the PI3K/AKT/mTOR pathway is common in ovarian malignancies and may be of prognostic significance.
Preclinical studies of the mTOR inhibitor RAD001 showed inhibition of tumor growth, angiogenesis and tumor invasiveness, with enhanced chemotherapy-induced apoptosis in cells with high AKT/mTOR activity.
The activity of tamoxifen in ovarian cancer has been reported with meta-analyses showing response rates from 9.6 to 13%.
Tamoxifen and the aromatase inhibitors (e.g., letrozole and exemestane) show minimal rates of tumor shrinkage but may produce prolonged disease stabilization in a subset of patients.
The findings of the large Phase III trials of bevacizumab (GOG 213, 218, 252; ICON7), which have now completed or nearly completed accrual, will help to determine the role of this agent in ovarian cancer management.
PARP inhibitors are promising agents in BRCA-mutated ovarian cancers. Improved patient selection and individualized assessment of tumor biology will aid in identifying the appropriate targets for these novel therapies.
Further evolution of the concept of targeted therapy in ovarian cancer, both in the development of active targeted agents as well as the incorporation of robust and clinically validated predictive biomarkers, is required to fulfill the promise of truly personalized cancer care.
In the future, improved patient selection and individualized assessment of tumor biology will serve to aid in identifying the appropriate targets for these current novel therapies and others that are sure to come. Further development of the concept of targeted cancer therapy promises to usher in an era of truly personalized cancer care.
Footnotes
