Abstract
Background:
Endometriosis is a common gynecological condition that in sporadic cases can mimic an ovarian malignancy. We report a unique case of endometriosis characterized by massive hemorrhagic ascites and a mass-like plaque on the uterine surface. Atypical endometriosis is recognized as a potential precursor to ovarian malignancies.
Case presentation:
A 51-year-old woman with no significant medical history presented with abdominal pain, massive ascites and abnormal findings on ultrasound and CT. This, together with an elevated CA-125 level pre-operatively raised suspicion for ovarian malignancy. Diagnostic laparoscopy with frozen section biopsy was executed and the uterus and adnexa were removed. The final diagnosis however revealed an atypical presentation of endometriosis.
Conclusion:
This case highlights a rare manifestation of endometriosis that closely mimics ovarian malignancy. Clinicians should consider this differential diagnosis when encountering similar presentations.
Introduction
Endometriosis is a benign disease characterized by the presence of endometrium-like epithelium and/or stroma outside the endometrium and myometrium, typically accompanied by an associated inflammatory process. 1 It affects up to 10% of women of reproductive age and can cause symptoms such as dysmenorrhea, dyspareunia, abdominal pain and/or infertility. 1 Although most manifestations are confined to the pelvis, extra pelvic presentations have been described. One of the rarest clinical manifestations is hemorrhagic ascites, a condition that can mimic ovarian malignancy and therefore poses a significant diagnostic challenge.2,3
Case description
A 51-year-old Dutch multiparous woman was referred to our tertiary hospital with abdominal pain, massive ascites, abnormal findings of the uterus on the ultrasound and an elevated CA-125, leading to the pre-operative clinical differential diagnosis of an ovarian malignancy, endometriosis or an infectious episode. Informed consent was obtained from the patient.
Over the past 3–4 months, she had experienced multiple episodes of abdominal pain, accompanied by fatigue and weight loss of 10 kg. She had been absent from work over the last 3 months, primarily due to fatigue. Additionally, during the past few days she experienced nausea and vomiting.
She used to have a regular menstrual cycle and commenced oral contraceptive therapy 2 years ago to address severe (hemoglobin-depleting) menstrual hemorrhage. After initiating oral contraception, she developed hypertension, prompting a switch to a Mirena intrauterine device (IUD) 2 years ago. This change resulted in amenorrhea. Prior to this, she did not experience dysmenorrhea or other symptoms suggestive for endometriosis. The current pain appeared to be cyclic (every 4 weeks) and manifested centrally in the abdomen, with no associated dysuria or dyschezia.
Upon physical examination, we observed a non-ill, fatigued yet physically fit patient, with normal vital parameters (blood pressure 117/75 mmHg, heart frequency 80/min, saturation 99%). Abdominal examination revealed a slightly distended, but non-tender abdomen with normal peristalsis. There were no palpable inguinal lymph nodes. Vaginal examination revealed a normal length of the vagina with a high-positioned, normal cervix and a moderately mobile uterus. A small irregularity was palpable in the posterior fornix. Speculum examination showed a normal cervix with the threads of the Mirena IUD in situ. In the fornix posterior was a small, currently bleeding, irregularity observed, but a biopsy could not be obtained due to pain. The Mirena IUD was removed and cultured for actinomyces, with the culture yielding negative results. Vaginal swabs for routine cultures and PCR testing for sexually transmitted infections were also negative. Transvaginal ultrasound showed a uterus in anteversion flexion with an IUD in situ and thin endometrium, with a small intramural myoma in the anterior wall. Focally, a plaque with an echodensity similar to the uterus extended toward both fallopian tubes/adnexa, poorly demarcated from the uterus (Figure 1). Both the right and left adnexa were solidly enlarged with poor demarcation from the uterus and contralateral adnexa. Acoustic shadowing was absent and a central echolucent clearance was present, with moderate blood flow (IOTA flow score 3). Additionally, a significant amount of free pelvic fluid with echodensity was noted. The lab results showed a hemoglobin (Hb) level of 8.1 mmol/L, a leukocyte count of 10.0 billion/L, a C-reactive protein (CRP) level of 24 and normal renal function with an eGFR of 83.3 mL/min/1.73 m.
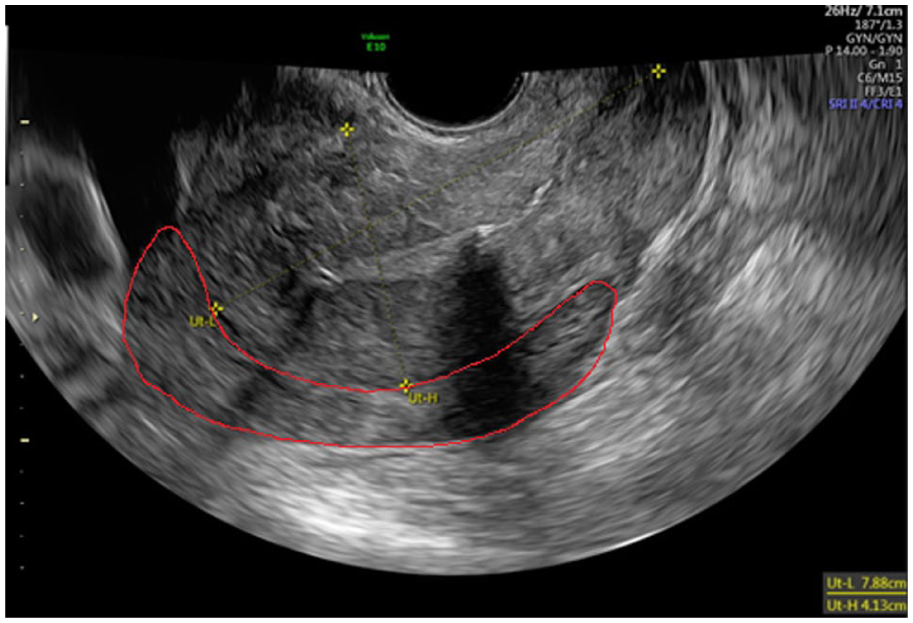
Ultrasound image showing the uterus with a plaque on the fundus and posterior side with an echodensity similar to the myometrium of the uterus (indicated in red).
CT thorax/abdomen showed heterogeneous tissue surrounding the uterus, with both ovaries medially displaced (Figure 2). Given the presence of peritoneal fluid and suspicion of peritoneal implants and omental infiltrations, malignancy with peritoneal carcinomatosis could not be ruled out. The cervical cytology revealed Pap3a2 (based on endometrial cells; Kopac-B A4) and the CA-125 level was elevated at 1003 kU/L. Cytology examination of ascites fluid demonstrated inflammation and reactive mesothelium, with a single group of atypical cells with slight atypical features (more rounded nucleus and prominent nucleolus). These cells weren’t present in the additional staining’s.
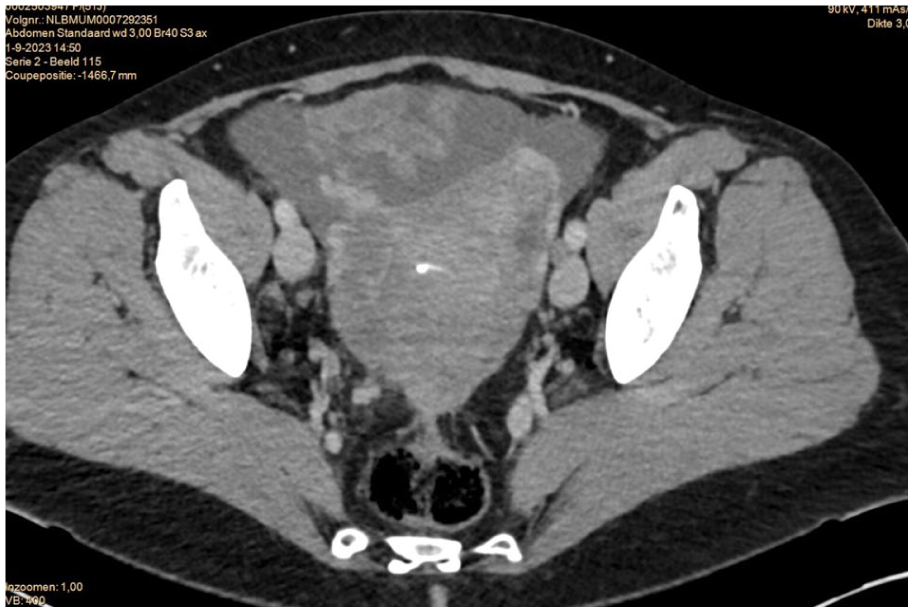
Transversal CT-scan image with ascites and heterogeneous tissue suspicious for peritoneal implants surrounding the enlarged uterus with Mirena IUD in situ. Lymph nodes are not enlarged.
The patient underwent diagnostic laparoscopy with frozen section analysis. During laparoscopy, 2.8 l of hemorrhagic ascites were drained. The uterus and ovaries were unrecognizable, with only the fimbrial ends visualized. Extensive peritoneal involvement was observed throughout the abdomen, along with adhesions to the anterior abdominal wall and the liver. Tumor manipulation resulted in easy bleeding. The clinical presentation was most suggestive of an extensive infection. Multiple biopsies were taken for frozen section analysis and turned out to be suspicious for carcinosarcoma, but no certain diagnosis could be reached during the procedure on frozen section. Subsequently, a laparotomy was performed for further exploration and debulking. Numerous adhesions were present throughout the abdomen, along with scattered diffuse peritoneal implants and implants on bowel mesenteric and small bowel serosa (multiple implants measuring <1 cm and 3–4 implants measuring >1 cm). Despite efforts, the ovaries and uterus remained poorly identifiable and therefore it was decided to resect the uterus and adnexa completely en-bloc. Due to the diffuse spread of intraperitoneal lesions, complete resection would require resection of the recto-sigmoid and three resections of small bowl. Since the interpretation of the frozen section biopsy turned out to be inconclusive, no bowel resection was performed and rest disease up to 1 cm was accepted. Therefore, complete debulking was not performed. The total blood loss was 1.2 L.
Final pathology, as seen in Figure 3, revealed findings consistent with endometriosis with an exceptionally unusual presentation. It was characterized by plaques with endometrial glandular structures and stroma across all examined organs. These plaques exhibited partially pseudodecidualized changes in the stroma, potentially resulting from exogenous hormone use. Combined with inflammation, this has led to an atypical cellular appearance. The overall presentation was extraordinarily uncommon and severe. Additionally, there were areas with architectural disruption and increased atypia, indicative of hyperplasia with atypia, without evidence of malignancy.
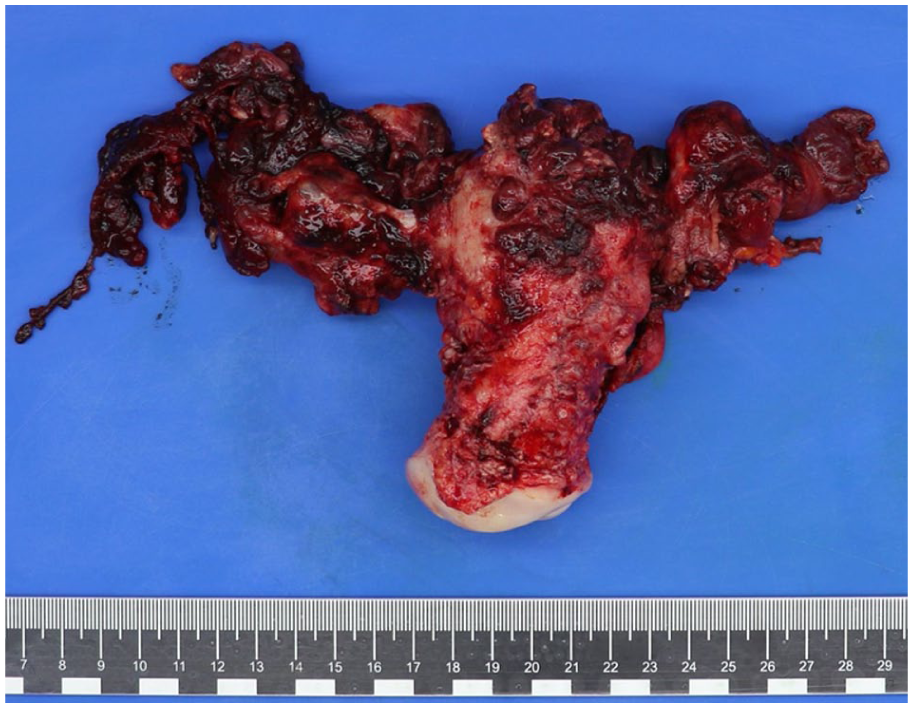
Surgical specimen illustrating the massive hemorrhagic plaques at the surface.
Post-operatively, an estrogen level was assessed to exclude exogenous estrogen production, revealing a low level (<6 pmol/L; normal level in postmenopausal women < 26 pmol/L). Treatment with an aromatase inhibitor and follow-up were considered since the presence of possible rest endometriosis lesions and the finding of atypia. Ultimately, no aromatase inhibitor therapy was initiated and clinical follow-up was commenced. At 4 months post-operatively, the patient remained significantly fatigued and had not yet resumed work duties. Due to ongoing complaints, a MRI was executed. The MRI revealed spiculated soft tissue with low signal intensity on T2-weighted sequences at the surgical site, with additional traction on the sacro-uterine ligaments and adjacent rectosigmoid. This imaging pattern may be consistent with fibrosis, with underlying endometriosis not definitively excluded. Importantly, all histopathological examinations excluded malignancy. There were no signs of ascites. The year following the surgery, the patient slowly recovered.
Conclusion
Massive hemorrhagic ascites with mass-like endometriosis on the surface of the uterus is extremely rare and may be mistaken for a malignancy, particularly in peri-menopausal women who have not previously presented with symptoms suggestive of endometriosis. In our case, the patient exhibited B-symptoms like fatigue and weight loss and recent developed cyclic pain. The diagnostic work-up initially suggested malignancy, given the elevated CA-125, the presence of a solid mass with ascites and omental infiltrations on imaging and atypical cells in the ascites.
In the differential diagnosis of massive hemorrhagic ascites in women, ovarian malignancy remains the primary concern. Other possible causes include benign ovarian cyst rupture, tubo-ovarian abscess, Meigs’ syndrome, peritoneal tuberculosis or cirrhosis-related ascites with secondary hemorrhage. In addition to ovarian malignancy, benign gynecological tumors such as degenerative uterine leiomyomas may occasionally mimic ovarian neoplasms. 4
Only a few similar cases have been described in the literature. 3 These case reports most often involved younger patients and they were treated with hormonal therapy, such as GnRH agonists or combined oral contraceptives or surgery. Only one previously published case involved mass-like endometriosis on the uterine surface, where the patient presented with symptoms characteristic of endometriosis, but was also suspected of an ovarian malignancy and therefore underwent laparoscopic surgery with frozen section analysis, ultimately leading to removal of uterus and both ovaries due to ongoing bleeding after the biopsy. 5
Our case underscores the importance for gynecologists to recognize this diagnostic challenge and communicate atypical intraoperative findings to the pathologist. While frozen section analysis for ovarian tumors is relatively accurate (range 86%–97%), 6 the analysis in this case turned out to be inconclusive. Secondly, as our case concerned a peri-menopausal woman, the uterus and adnexa were removed. Nevertheless, it is important to preserve the ovaries in younger patients and consider treatment to suppress the menstrual cycle and thereby progression of endometriosis. Lastly, it is important to be aware of the prevalence of malignancy in patients with endometriosis, which is about 0.7%–1.6%. 7 Specifically, atypical endometriosis is recognized as a potential precursor to ovarian malignancies. 8 However, long-term follow-up studies indicate low recurrence and malignant transformation rates. Nevertheless, uncertainties remain about its precise malignancy risk and optimal management. 8
Although several theories about the pathophysiology of endometriosis exist, the exact etiology of endometriosis and thereby mass-like endometriosis and massive hemorrhagic ascites is still unknown. 9 Smaller volumes of hemorrhagic ascites may result from rupture of an endometriotic cyst, leading to peritoneal inflammation. In very rare occasions, pregnant patients with endometriosis might develop spontaneous haematoperitoneum in pregnancy (SHiP). 10 However, in this case, the mass-like plaque on the surface of the uterus might be the source of the hemorrhagic ascites.
In conclusion, it is important for gynecologists and pathologists to be aware of this rare manifestation of endometriosis, potentially mimicking ovarian malignancy.
Footnotes
Acknowledgements
The authors would like to thank the patient for allowing this case to be published.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
MUMC+ does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Consent for publication
Written consent of patient.
Data availability statement
The data are available from the corresponding author upon reasonable request and with permission of the patient.
