Abstract
Breast cancer is the most common type of cancer diagnosed in women. Anthracyclines and taxanes are the most active and widely used chemotherapeutic agents in the treatment of both early-stage and advanced breast cancer. In the past decade, novel formulations of these cytotoxic agents have been developed to improve efficacy and decrease toxicity. nab-paclitaxel is a solvent-free, albumin-bound 130-nm particle form of paclitaxel (Abraxane®, Abraxis Bioscience, CA, USA), which was developed to avoid toxicities associated with the Cremophor vehicle used in solvent-based paclitaxel. In a Phase III study, nab-paclitaxel demonstrated higher response rates, a better safety profile compared with conventional paclitaxel, and improved survival in patients receiving it as second-line therapy. Based on this pivotal trial, nab-paclitaxel is now approved in the USA for treatment of breast cancer after failure of combination chemotherapy for metastatic disease or relapse within 6 months of adjuvant therapy where prior therapy included an anthracycline unless clinically contraindicated. Recently, several Phase II studies have suggested a role for nab-paclitaxel as a single agent and in combination with other agents for first-line treatment of metastatic breast cancer. Studies are ongoing to explore the use of nab-paclitaxel in other solid tumors such as non-small-cell lung cancer, ovarian cancer and malignant melanoma.
Breast cancer is the most common type of cancer diagnosed in women. It is the leading cause of cancer deaths in Hispanic women, and ranks as the second most common cause of cancer deaths in other ethnic groups [101]. Although the overall death rates have dropped steadily since the early 1990s [102], breast cancer continues to be a major health problem. The American Cancer Society estimates that approximately 192,370 new cases of invasive breast cancer were diagnosed in the USA in 2009 with 40,170 deaths [102]. Metastatic breast cancer remains incurable and the major objectives of treatment are palliation of symptoms, prolongation of survival, reducing toxicities associated with such treatments and improvement of quality of life. A better understanding of the tumor biology has led to the development of several targeted agents and biologic therapies. Additionally, newer chemotherapy agents with an improved efficacy and toxicity profile have been introduced. However, taxanes (e.g., paclitaxel and docetaxel) and anthracyclines (e.g., doxorubicin and epirubicin) remain among the most active and widely used chemotherapy agents in breast cancer, both in adjuvant and metastatic settings [1,2,103]. A recent meta-analysis of 13 randomized clinical trials showed a significant improvement of disease-free and overall survival (OS) rates in high-risk early-stage breast cancer with chemotherapy regimens incorporating a combination of taxanes and anthracyclines [3].
The solvents used for dissolving hydrophobic molecules, paclitaxel and docetaxel are associated with significant risk of hypersensitivity reactions and neuropathy and also impair drug delivery to the tumor, limiting their clinical effectiveness [4,5]. With the advent of nab-technology, a novel formulation of solvent-free 130-nm albumin-bound paclitaxel (nab-paclitaxel, Abraxane®, Abraxis Bioscience, CA, USA) was developed for use as a colloidal suspension intravenously (Box 1). Based on pivotal Phase III clinical trial results, nab-paclitaxel was approved by the US FDA in January 2005 and by the EMA in January 2008 in Europe for use in patients with metastatic breast cancer (MBC) who have failed combination chemotherapy or relapsed within 6 months of adjuvant therapy where prior therapy included an anthracycline unless clinically contraindicated.
This article provides a pharmacologic profile of nab-paclitaxel, an overview of its current state of development and evaluates its potential for use in the treatment of breast cancer.
Solvent-based taxanes: side-effects, drawbacks & limitations
Taxanes are microtubule stabilizers. They bind to the interior surface of the β-microtubule chain and enhance tubulin polymerization. This inhibits mitosis, motility and intracellular transport within (cancerous) cells, leading to apoptotic cell death. Taxanes also block antiapoptotic effects of the BCL-2 gene family and induce p53 gene activation with consequent mitotic arrest leading to cell death [6].
nab-paclitaxel.
ABI-007, albumin-bound paclitaxel Trade name
Abraxane® (Abraxis Bioscience)
Antimicrotubule agent, promote microtubule assembly from tubulin dimers and stabilize microtubules to prevent depolymerization. This stability causes inhibition of the normal dynamic reorganization of the microtubules, which is necessary for important interphase and mitotic functions in the cells
260 mg/m2.
Intravenous infusion over 30 min once every 3 weeks Pharmacokinetics
Distribution: extensive extravascular distribution and/or tissue binding; does not penetrate blood–brain barrier
Protein binding: 89–98%
Metabolism: hepatic, P450 (CYP2C8 and CYP3A4)
Excretion: fecal (20%), renal (4%)
Elimination half-life: 27 h
Cardiovascular: abnormal EKG (60%), edema (10%)
Dermatologic: alopecia (90%)
Gastrointestinal: diarrhea (27%), nausea (30%), vomiting (18%)
Hematologic: anemia (33%), neutropenia (any grade, 80%)
Hepatic: raised transaminases (39%), raised alkaline phosphatase (36%)
Neurologic: asthenia/myalgia/fatigue (47%), sensory neuropathy (any grade, 71 %)
Ophthalmic: visual disturbance (13%)
Renal: raised serum creatinine (11%)
Respiratory: dyspnea (12%)
Cardiovascular: cardiac arrest, cerebrovascular accident, supraventricular tachycardia, transient ischemic attack (3%)
Hematologic: severe anemia (1%), bleeding (2%), febrile neutropenia (2%), grade 4 neutropenia (9%), severe thrombocytopenia (<1 %)
Neurologic: severe sensory neuropathy (10%)
Paclitaxel has been shown to be clastogenic, teratogenic and fetotoxic and should not be used in pregnancy. Men should be advised not to father a child while receiving treatment. It is not known if paclitaxel is excreted in human milk; however, it is recommended that nursing should be discontinued during therapy
From [105].
Paclitaxel, first approved in 1992 for clinical use, is a naturally occurring diterpinoid product extracted from the bark of pacific yew (Figure 1). Docetaxel is a semisynthetic esterified product of 10-deacetyl baccatin III extracted from needles of European yew. Both paclitaxel and docetaxel are highly hydrophobic. Cremaphor EL (CrEL), a nonionic surfactant polyoxyethylated castor oil mixed 1: 1 with dehydrated ethanol, was identified as the most feasible option to solubilize paclitaxel for intravenous administration. Similarly, the solvent used for docetaxel is another polyoxyethylated surfactant, polysorbate 80 [5]. These solvents are both biologically and pharmacologically active and are associated with several significant side effects such as hypersensitivity reactions and neuropathies, and also impair tumor penetration, limiting the clinical effectiveness of solvent-based taxanes [4,5]. The CrEl–paclitaxel formulation requires a special infusion set to minimize exposure to di(2-ethylhexyl)phthalate (DHEP), which may be leached from standard polyvinyl chloride sets. Prolonged infusion times and premedications with corticosteroids and antihistamine agents are required to reduce hypersensitivity reactions. However, minor reactions still occur in approximately 40% of all patients receiving solvent-based taxanes and nearly 3% develop potentially life-threatening reactions [5]. CrEL has also been shown to cause neutropenia and prolonged peripheral neuropathy related to axonal degeneration. Fluid retention, a toxicity commonly seen with docetaxel, has been attributed in part to alteration of membrane fluidity by polysorbate 80 [5,7]. Formation of large polar micelles of CrEL–paclitaxel in the plasma compartment can cause entrapment of the drug leading to nonlinear pharmacokinetics [4]. This alters the pharmacodynamic characteristics of the solubilized drug, resulting in a substantial increase in systemic exposure with concomitantly reduced systemic clearance placing patients at risk for severe systemic toxicities. This drug entrapment phenomenon, which decreases the duration of drug exposure, partly explains why the attempts to improve efficacy of CrEL–paclitaxel by utilizing doses higher than the standard-of-care dose (175 mg/m2 over 3 h every 3 weeks) have been unsuccessful [8]. More frequent dosing (such as weekly paclitaxel administration), which may lead to increased duration of exposure, has demonstrated improved efficacy in both adjuvant/neoadjuvant and metastatic settings [9].
To address these limitations of solvent-based taxanes and to improve their therapeutic index, various solvent-free formulations and delivery systems such as liposomal-encapsulated paclitaxel, paclitaxel–vitamin E emulsion and polymer–microsphere formulation of paclitaxel have been investigated. The first successful attempt to formulate a solvent-free taxane has been the development of nab-paclitaxel. The nanoparticle protein platform utilizes the natural properties of albumin to increase drug delivery to the tumor and eliminates the need for solvents.
Nanomedicine & role of albumin in nab-technology
Nanomedicine is the medical application of molecular nab-technology, a new area of science that involves working with small-scale materials and devices that are at the nanometer level (10−9 of a meter). A few examples of the development by this discipline include liposomes, dendrimers, super paramagnetic nanoparticles and polymer-based platforms [10]. Albumin has a number of features that make it an ideal drug delivery system. It is a natural carrier of endogenous hydrophobic molecules such as vitamins, hormones and other water-insoluble plasma substances that are bound in a reversible noncovalent manner. Albumin partakes in endothelial transcytosis of protein-bound and unbound plasma constituents mainly by binding to a cell surface 60 kDa glycoprotein receptor (gp60) on the endothelial cell membrane. This in turn activates caveolin-1, a major component of membrane vesicles resulting in receptor-mediated internalization of the albumin–drug complex into caveolae (small invaginations of the plasma membrane) with subsequent transcytosis and delivery of the drug to tumor cells [104].
Preclinical & clinical efficacy studies
Comparative intratumoral and antitumoral activity of nab-paclitaxel has been demonstrated to be greater than CrEL–paclitaxel and docetaxel in multiple tumor types using preclinical models [10,11]. Desai et al., using radiolabeled paclitaxel in mice with xenografts, demonstrated that nab-paclitaxel was significantly less toxic: 50% lethal dose (LD50) values and maximum tolerated dose (MTD) for nab-paclitaxel and CrEL–paclitaxel were 47 and 30 mg/kg/day, and 30 and 13.4 mg/kg/day, respectively [10]. At equal doses, intratumoral paclitaxel accumulation was found to be 33% higher for nab-paclitaxel (Figure 2). In live human umbilical vascular endothelial cells (HUVEC), endothelial binding and transport across the endothelial cell monolayer was significantly higher (9.9- and 4.2-fold, respectively) with nab-paclitaxel and this difference was abrogated by methyl β-cyclodextrin, a known inhibitor of endothelial gp60 receptor and caveolar-mediated transport [10]. This data provided the preclinical evidence to advance the drug to clinical studies.

Secreted protein acid rich in cysteine (SPARC), a type of caveolin-1, has a sequence homology with gp60 that leads to its binding to albumin. It is overexpressed in several tumor types including breast cancer. This interaction between SPARC and albumin has been suggested to be the reason for enhanced uptake and intratumoral accumulation, and also a possible role for SPARC as a biomarker for nab-paclitaxel effectiveness [11].
Phase I & pharmacokinetic studies
Three different dose schedules of nab-paclitaxel have been evaluated in Phase I and pharmacokinetic studies. In a study by Ibrahim et al., 19 patients with advanced solid tumors received nab-paclitaxel as a 30-min infusion given every 3 weeks without premedication using doses ranging from 135 to 375 mg/m2 [12]. No infusion-related acute hypersensitivity reactions were noted during the drug administration. Hematological toxicity was mild and not cumulative. At the highest dose studied (level 3: 375 mg/m2), dose-limiting toxicity occurred in three out of six patients and consisted of sensory neuropathy (three patients), stomatitis (two patients) and superficial keratopathy (two patients). The MTD was determined to be 300 mg/m2, substantially higher than the typical dose used with CrEL–paclitaxel. Pharmacokinetic analyses revealed whole-blood paclitaxel concentrations and area under the curve (AUC) values to increase linearly over the dose range of 135–300 mg/m2 unlike the nonlinear kinetics of solvent-based paclitaxel.
In another Phase I and pharmacokinetics trial by Nyman et al., 39 patients with advanced nonhematological malignancies received nab-paclitaxel without premedication at dose levels ranging from 80 to 200 mg/m2 as a 30-min infusion once a week for 3 weeks in each monthly cycle [13]. A third of patients received six or more cycles. After enrollment of the first cohort, patients were enrolled into one of two cohorts: ‘lightly’ and ‘heavily’ pretreated, based on the extent of prior exposure to chemotherapy. MTDs for these two cohorts were 150 and 100 mg/m2; dose-limiting toxicities were grade 3 peripheral neuropathy and grade 4 neutropenia, respectively. The pharmacokinetics were again noted to be linear and there were no dose-dependent changes in plasma clearance. Partial response was observed in patients previously treated with CrEL–paclitaxel.
A randomized, crossover study comparing the pharmacokinetics of nab-paclitaxel and CrEL–paclitaxel was reported by Gardner et al. [14]. A total of 17 patients with locally advanced or metastatic solid tumors that were likely to be responsive to taxanes were randomized to receive nab-paclitaxel (260 mg/m2 as a 30-min infusion) or CrEL–paclitaxel (175 mg/m2 as a 3-h infusion). Patients crossed over to the alternate treatment after the first cycle. Thereafter, patients received treatments with 260 mg/m2 of nab-paclitaxel every 3 weeks. Pharmacokinetic studies were carried out for the first cycle of CrEL–paclitaxel and the first two cycles of nab-paclitaxel. Pharmacokinetic study results showed that although the total drug exposure was comparable between the two formulations, the mean fraction of unbound paclitaxel was significantly higher with nab-paclitaxel compared with CrEL–paclitaxel (0.063 ± 0.021 vs 0.024 ± 0.009; p < 0.001). This study purports that systemic exposure to unbound paclitaxel would lead to increased tumoral uptake, thereby resulting in an augmented antitumor efficacy compared with CrEL–paclitaxel.
Stinchcombe et al. have reported the pharmacokinetics and Phase I study results of three different schedules of nab-paclitaxel in combination with carboplatin [15]. In total, 41 heavily pretreated patients with advanced solid tumors received nab-paclitaxel and carboplatin with an AUC of 6 on day 1. Group A received nab-paclitaxel at doses ranging from 220 to 340 mg/m2 on day 1, every 21 days; group B received nab-paclitaxel 100 or 125 mg/m2 on days 1, 8 and 15 every 28 days; and group C received nab-paclitaxel 25 or 150 mg/m2 on days 1 and 8, every 21 days. MTD of nab-paclitaxel in combination with carboplatin was 300, 100 and 125 mg/m2 in groups A, B and C, respectively. Myelosuppression was the primary dose-limiting toxicity.
In a recent Phase I study reported by Chien et al., vascular-priming chemosensitization with a 2-day pulse of high-dose lapatinib followed by weekly infusion of nab-paclitaxel 100 mg/m2 was investigated in 25 patients with advanced solid tumors [16]. In total, 72% of these patients were previously taxane-refractory. The MTD of lapatinib was defined as 5250 mg/day in divided doses. The dose-limiting toxicities were grade 3 vomiting and grade 4 neutropenia. A total of 65% of evaluable patients had a partial or stable response to this therapy.
Phase II studies
Following the results of the Phase I study [12], Ibrahim et al. investigated nab-paclitaxel in a multicenter Phase II study to evaluate safety and antitumor activity in patients with MBC [17]. In total, 63 women with confirmed and measurable MBC received 300 mg/m2 of nab-paclitaxel over 30 min every 3 weeks. A total of 48 patients had received prior chemotherapy; 39 patients had received no prior treatment for metastatic disease. Median number of treatments was six cycles. Overall response rate (ORR), which was the primary end point of this study, was 48% for all patients and 64% for those receiving nab-paclitaxel as first-line treatment. Median time to progression (TTP) was 26.6 weeks and median OS was 63.6 weeks. No severe hypersensitivity reactions were reported despite lack of premedication. Toxicities noted were typical of paclitaxel and included grade 4 neutropenia (24%) and grade 3 sensory neuropathy (11%) and grade 4 febrile neutropenia (5%).
In another Phase II study, Blum et al. reported the benefit of weekly nab-paclitaxel in patients with MBC whose disease had failed conventional taxane treatment [18]. Taxane failure was defined as metastatic disease progression during taxane therapy or relapse within 12 months of adjuvant taxane therapy. Patients received 100 mg/m2 (n = 106) or 125 mg/m2 (n = 75) on days 1, 8 and 15 of a 28-day cycle. Response rates were 14 and 16% for the 100 and 125 mg/m2 cohorts, respectively, with an additional 12 and 21% of patients, respectively, having stable disease for 16 weeks or longer. Median progression-free survival (PFS) (3 vs 3.5 months) and median survival (9.2 vs 9.1 months) were similar for the two dose cohorts; survival was similar for responding patients and those with stable disease. No severe hypersensitivity reactions were reported and grade 4 neutropenia occurred in less than 5% of patients.
Recently, Roy et al. reported a Phase II study of weekly nab-paclitaxel in combination with gemcitabine in patients with MBC [19]. In this open-label, one-stage trial, 55 patients with previously untreated MBC were treated with 125 mg/m2 of nab-paclitaxel and 1000 mg/m2 of gemcitabine on days 1 and 8 of a 21-day cycle until disease progression. In total, 40 patients (80%) had visceral involvement and 30 patients (60%) had three or more sites of metastases. ORR was 50% (four complete responses, 8%; 21 partial responses; 42%). Median PFS was 7.9 months. PFS and OS at 6 months was 60 and 92%, respectively. Neutropenia was the most common toxicity (grade 3: 43%, grade 4: 12%). Grade 3–4 neuropathy was noted in only four patients (8%).
Gradishar et al. reported a randomized, multicenter Phase II study comparing nab-paclitaxel with docetaxel as first-line treatment in patients with MBC [20]. A total of 300 previously untreated MBC patients were randomized to three different nab-paclitaxel treatment schedules: 300 mg/m2 every 3 weeks (n = 76), 150 mg/m2 weekly (n = 74) and 100 mg/m2 weekly (n = 76). Docetaxel dose was 100 mg/m2 once every 3 weeks (n = 74). A total of 43% of patients had received prior adjuvant or neoadjuvant chemotherapy. Doses of nab-paclitaxel 150 mg/m2 weekly demonstrated significantly longer PFS than docetaxel (12.5 vs 7.5 months). On the basis of independent radiologist review, both 150 mg/m2 (49%) and 100 mg/m2 (45%) weekly nab-paclitaxel demonstrated a higher ORR than docetaxel (35%), but this did not reach statistical significance. This trend was supported by statistically significant investigator ORR for both weekly nab-paclitaxel doses versus docetaxel. Every 3-weekly nab-paclitaxel versus docetaxel was not different in terms of ORR or PFS. Grade 3 or 4 fatigue, neutropenia and febrile neutropenia were less frequent in all nab-paclitaxel arms and the frequency and grade of peripheral neuropathy was similar in all treatment groups, but this resolved more rapidly after treatment withdrawal with nab-paclitaxel compared with patients who received docetaxel.
Nab-paclitaxel administered sequentially with anthracycline was evaluated in a neoadjuvant setting for locally advanced breast cancer (LABC) in a Phase II study by Robidoux et al. [21]. A total of 66 patients with LABC but without prior treatment and regardless of hormone receptor or HER2 status were treated with nab-paclitaxel weekly for 12 weeks followed by 5-fluorouracil, epirubicin and cyclophosphamide (FEC) every 3 weeks for four cycles. Trastuzumab was allowed in HER2-positive (HER2+) patients. A total of 63 patients completed four cycles of albumin-bound paclitaxel, following which 58 completed four cycles of FEC. In total, 17 out of 19 HER2+ patients received trastuzumab. Pathologic complete response (pCR), the primary objective of the study, was 29% (19 out of 65). For the HER2+ subset, the pCR was 58% (11 out of 19). Both nab-paclitaxel and FEC were well tolerated. The most significant toxicities were grade 2/3 neuropathy (16%) with nab-paclitaxel and grade 3/4 febrile neutropenia (7%) with FEC.
These studies suggest that nab-paclitaxel alone or in combination with other therapeutic agents has a significant activity in patients with MBC or locally advanced breast cancer, including those previously treated with taxanes and/or anthracyclines. These studies are summarized in Table 1. Because of its efficacy, ease of administration and a favorable toxicity profile, nab-paclitaxel is currently being evaluated in combination with other cytotoxic or targeted agents in breast cancer and other solid tumors. Table 2 lists the currently active Phase II studies with nab-paclitaxel in breast cancer.
Phase II studies assessing the activity of nab-paclitaxel in metastatic breast cancer.
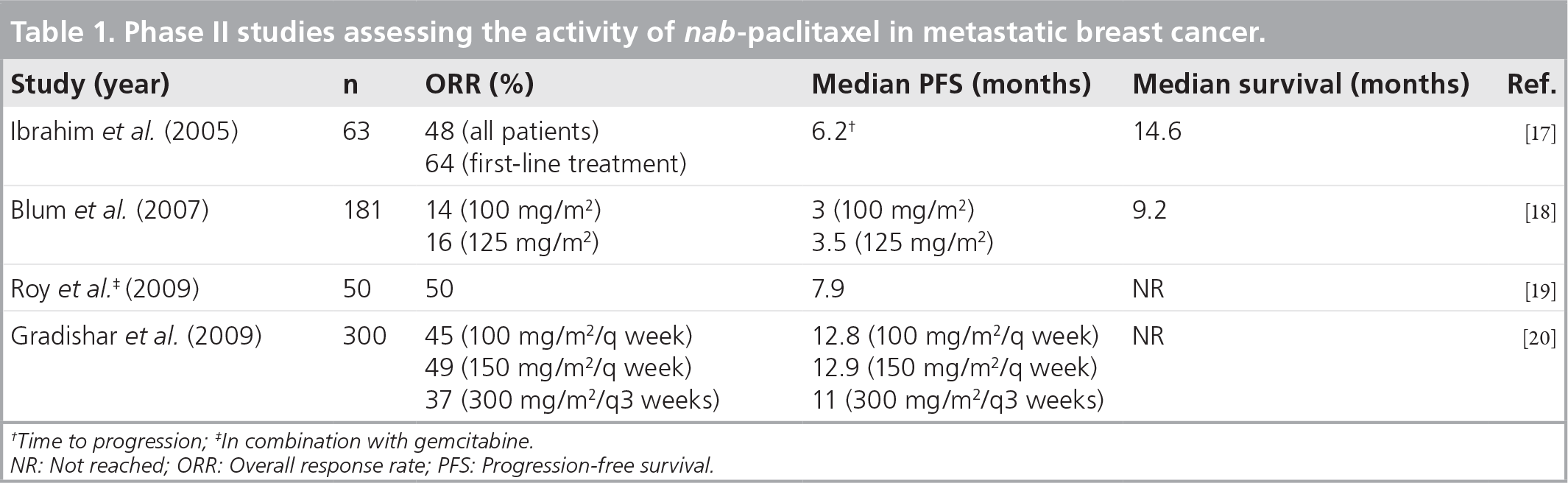
Time to progression; ‡In combination with gemcitabine.
NR: Not reached; ORR: Overall response rate; PFS: Progression-free survival.
nab-paclitaxel in breast cancer: active Phase II trials.

ORR: Overall response rate; pCR: Pathologic complete response; PFS: Progression-free survival; pK: Pharmacokinetics.
Data taken from [106].
Phase III study
Based on the promising Phase I and II studies, nab-paclitaxel's antitumor efficacy and safety was compared to CrEl–paclitaxel in a pivotal, multinational, randomized Phase III trial conducted at 70 sites in five countries [22]. A total of 460 women with MBC were randomized to 3-weekly cycles of either nab-paclitaxel 260 mg/m2 administered intravenously over 30 min without premeditation (n = 229) or CrEL–paclitaxel 175 mg/m2 administered intravenously over 3 h with corticosteroid and antihistamine premedications (n = 225). The large majority of patients had more than three metastatic lesions (76%), visceral disease (79%), prior chemotherapy (86%), and progression after first-line therapy for metastatic disease (59%). Approximately half of the patients in each group received at least six cycles of treatment. Actual delivered paclitaxel dose-intensity was 49% higher in the nab-paclitaxel group than in the CrEL–paclitaxel group (85.13 vs 57.02 mg/m2/week). nab-paclitaxel demonstrated significantly higher response rates compared with CrEL–paclitaxel (33 vs 19%; p = 0.001). Patients who received nab-paclitaxel as first-line and second-line or greater treatment had an ORR of 42 and 27% compared with 27 and 13% with CrEL–paclitaxel, respectively (Figure 3). TTP was also significantly longer with nab-paclitaxel for all patients (23 vs 16.9 weeks; hazard ratio [HR] = 0.75; p = 0.006) and among those receiving second-line therapy or greater (20.9 vs 16.1 weeks; HR = 0.73; p = 0.02). There was no significant difference in median OS among all patients between the nab-paclitaxel and CrEL–paclitaxel groups (65 vs 55.7 weeks; p = 0.374); however, the patients who received nab-paclitaxel as second-line or greater therapy had a significantly longer OS (56.4 vs 46.7 weeks; HR = 0.73; p = 0.024). The incidence of grade 4 neutropenia was lower in the nab-paclitaxel group compared with the CrEL–paclitaxel group (9 vs 22%) despite a 49% higher paclitaxel dose. Febrile neutropenia was uncommon (<2%), and the incidence did not differ between the two study arms. Interestingly, grade 3 sensory neuropathy was more common in the nab-paclitaxel arm than in the CrEL–paclitaxel arm (10 vs 2%) but was easily managed and improved rapidly to grade 1–2 in a median of 22 days. No severe hypersensitivity reactions occurred with nab-paclitaxel despite the absence of premedication and a shorter administration time.

Overall response rates from Phase III trial of nab-paclitaxel versus Cremaphor EL-paclitaxel–paclitaxel in patients with metastatic breast cancer.
Similar results were reported in a Phase III trial comparing nab-paclitaxel with CrEL–paclitaxel in 210 Chinese patients with MBC [23]. Patients were equally randomized to receive either nab-paclitaxel 260 mg/m2 over 30 min every 3 weeks with no premedication or CrEL–paclitaxel 175 mg/m2 over 3 h every 3 weeks with standard premedication. ORR was 54% in the nab-paclitaxel group and 29% in the CrEL–paclitaxel group (p < 0.001). Median TTP (7.6 vs 6.2 months; p = 0.078) and median PFS (7.6 vs 6.2 months; p = 0.118) were higher in the nab-paclitaxel group, although not statistically significant. The most common toxicities reported were alopecia (78%), peripheral neuropathy (75%, 7% grade 3) and neutropenia (65%) and were similar between the two groups.
Therapeutic applicability of nab-paclitaxel is now being tested in a variety of tumor types, including non-small-cell lung cancer (NSCLC), pancreatic cancer and melanoma at multiple stages of disease, as a single agent and in combination with other cytotoxic chemotherapy and/or biologic agents. The currently active Phase III clinical trials in breast cancer with nab-paclitaxel are listed in Table 3.
nab-paclitaxel in breast cancer: active Phase III trials.

Data taken from [106].
Economic analysis of nab-paclitaxel versus docetaxel as first-line therapy in MBC
Dranitsaris et al. has recently published an economic analysis of the Phase II randomized study [24] comparing nab-paclitaxel weekly or every 3 weeks to standard docetaxel as first-line therapy in patients with MBC [20]. Clinical and resource use data was captured from the trial's database and an economic analysis was performed from the perspective of the NHS in the UK. The costs of chemotherapy, drug delivery, monitoring, supportive care drugs and hospitalization due to toxicity were included. A univariate and multivariate regression analysis was performed to compare the total cost of therapy in patients randomized to each of the four arms of the study. When all cost components were combined for the entire study population (n = 300), patients in the nab-paclitaxel 100-mg/m2 once-weekly (GB£15,396) and 300 mg/m2 every-3-weeks arms (£15,809) had comparable costs to the docetaxel arm (£12,923) while the nab-paclitaxel 150 mg/m2 weekly arm had significantly higher overall costs (£27,222). This study proposes that, given its safety profile and better efficacy with comparable costs, nab-paclitaxel could be considered as a reasonable alternative to docetaxel as first-line chemotherapy in MBC.
Tolerability, side effects & dose adjustments
The overall tolerability of nab-paclitaxel 260 mg/m2 was reported to be similar to that of CrEL–paclitaxel 175 mg/m2 in the Phase III trial [22]. No differences in quality of life throughout the study were noted between the two treatment groups despite a higher dose-intensity of paclitaxel in the nab-paclitaxel group (85.13 vs 57.02 mg/m2/week). The most commonly reported toxicities/adverse events during the study were as expected for paclitaxel and included alopecia (90 vs 94%), sensory neuropathy (71 vs 56%), fatigue (47 vs 38%), neutropenia (34 vs 49%), arthralgia (35 vs 33%), myalgia (28 vs 32%), nausea (30 vs 21%), infections (24 vs 20%) and diarrhea (26 vs 15%) for nab-paclitaxel versus CrEL–paclitaxel groups, respectively. Fewer events of neutropenia and skin flushing were reported among nab-paclitaxel recipients than CrEL–paclitaxel recipients, whereas sensory neuropathy and gastrointestinal symptoms were higher with nab-paclitaxel, but this resolved more rapidly after treatment withdrawal. There was no report of grade 4 sensory neuropathy. The side-effects are summarized in Table 4.
Adverse events: nab-paclitaxel versus Cremaphor EL-paclitaxel.

p < 0.05.
AST: Aspartate transaminase; CrEL: Cremaphor EL.
Data taken from [22].
Dose adjustment is recommended in patients with hepatic dysfunction, neutropenia and sensory neuropathy [105]. Nab-paclitaxel is generally avoided in patients with aspartate transaminase (AST) levels greater than ten-times the upper limit of normal (ULN) or bilirubin greater than five-times the ULN. The starting dose for patients with an AST level less than ten-times the ULN and a bilirubin level 2.01- to 5-times the ULN is 130 mg/m2 every 3 weeks with subsequent doses potentially increased up to 200 mg/m2 based on individual tolerance; the starting dose for patients with an AST level less than ten-times the ULN and a bilirubin level 1.26- to 2-times the ULN is 200 mg/m2 with subsequent doses adjusted based on individual tolerance; no dose adjustment is necessary in patients with an AST level less than ten-times the ULN and a bilirubin level greater than the ULN but less than or equal to 1.25-times the ULN. Patients who experience severe neutropenia (neutrophil counts <500 cells/mm3 for a week or longer) or severe sensory neuropathy during treatment with nab-paclitaxel should have dosage reduced to 220 mg/m2 for subsequent courses of nab-paclitaxel. For recurrence of severe neutropenia or severe sensory neuropathy, an additional dose reduction should be made to 180 mg/m2. Treatment should be withheld until neutrophils are greater than 1500 cells/mm3 and platelets recover to a level greater than 100,000 cells/mm3. For grade 3 sensory neuropathy, treatment is held until resolution to grade 1 or 2, followed by a dose reduction for all subsequent courses of nab-paclitaxel. Premedication to prevent hypersensitivity reactions is not required prior to administration of nab-paclitaxel. The use of nab-paclitaxel has not been studied in patients with renal dysfunction. In randomized controlled trials, patients were excluded for serum creatinine greater than 2 mg/dl.
Future perspective
Biomarker studies
Secreted protein acid rich in cysteine is known to be overexpressed in several tumor types including breast cancer and may be associated with a poor prognosis. In preclinical breast cancer models [11] and recently in a retrospective analysis of a clinical study of nab-paclitaxel in head and neck cancer [25], SPARC expression and its interaction with albumin has been suggested to be the reason for enhanced uptake and intratumoral accumulation, indicating a possible role for SPARC as a biomarker for nab-paclitaxel effectiveness. To corroborate these findings, a current Phase III study (NCT00785291) in MBC by Cancer and Leukemia Group B/North Central Cancer Treatment Group (CALGB/NCCTG) is evaluating the serum and tumor biomarkers (caveolin-1 and SPARC) along with circulating tumor cells to assess their possible role as predictive markers of response in MBC.
nab-paclitaxel in lung cancer
Besides breast cancer, nab-paclitaxel is being investigated in a variety of other solid tumors. Results from a Phase II study in advanced NSCLC have demonstrated that nab-paclitaxel at a dose of 260 mg/m2 every 3 weeks is well tolerated; it demonstrated a response rate of 16%, and a disease control rate of 49% with a median TTP of 6 months and median survival of 11 months [26]. In another Phase II study in elderly patients with NSCLC, nab-paclitaxel administered weekly at a dose of 100 mg/m2 for 3 weeks once every 28-day cycle showed a response rate of 30%, and a disease control rate of 50% with a PFS of 5 months and median survival of 11 months [27]. A Phase III study involving 1050 patients with NSCLC comparing nab-paclitaxel versus CrEL–paclitaxel in combination with carboplatin as a first-line treatment has just completed accrual and results are expected in mid-2010.
nab-paclitaxel as a radiosensitizer
Preclinical studies in mice bearing syngeneic ovarian or breast cancer have demonstrated that nab-paclitaxel improved radiotherapy in a supraadditive manner, suggesting that combining nab-paclitaxel with radiotherapy would improve the outcome of taxane-based chemoradiotherapy [28].
Other nab-compounds in the pipeline
nab technology has the potential for improving drug delivery and enhancing the therapeutic ratio of other water-insoluble drugs. Three such drugs with nab-application are currently in clinical studies. nab-docetaxel (ABI-008), a solvent-free nanometer-sized form of docetaxel is being studied in Phase I/II trials for patients with hormone-refractory prostate cancer (NCT00477529) [106]. Rapamycin is an inhibitor of the mammalian target of rapamycin (mTOR), a kinase member of the signaling pathway that promotes tumor growth. nab-rapamycin (ABI-009) is currently being studied in a Phase I trial (NCT00635284) [106], and in various nonhematological malignancies is purported to overcome the poor aqueous solubility and poor chemical stability, which had limited rapamycin's development as an anticancer agent. 17-AAG (17-N-allylamino-17-demethoxygeldanamycin; tanespimycin) is an antineoplastic antibiotic derivative of geldanamycin and is a heat shock protein 90 (hsp90) inhibitor. nab-17-AAG (ABI-010) in combination with nab-paclitaxel administered weekly will be studied in a currently pending Phase I trial (NCT00820768) for various nonhematological malignancies [106].
Conclusion
The current focus in cancer therapeutics including the treatment of breast cancer is to tailor therapy to each patient based on specific tumor phenotype and genomic analysis. Treatment decisions are based not only on predictive and prognostic factors, but also on the safety profile, impact on quality of life and patient preference. In the past decade, several studies have demonstrated that taxanes are an essential component in the treatment of breast cancer both in adjuvant and metastatic settings. Using the nab-technology platform, nab-paclitaxel was developed to overcome the limitations of solvents used in conventional solvent-based taxanes. Clinical data show that nab-paclitaxel has a better safety and side-effect profile with an improved efficacy compared with solvent-based taxanes. Higher doses can be administered over a shorter infusion time without the need for special infusion sets or premedications. The Phase II and III studies showed significant improvement in tumor response rate and progression-free intervals with nab-paclitaxel, but did not demonstrate survival benefit except when used in second-line or greater therapy where the benefit was approximately 9.7 weeks. These encouraging results have prompted initiation of several clinical trials, which are currently underway evaluating the role of nab-paclitaxel as a single agent or in combination with cytotoxic and/or biologic agents in breast cancer and other solid tumors.
Information resources
Executive summary
nab-paclitaxel is a novel albumin-bound paclitaxel formulation that is based on nab-technology for drug delivery without the requirement of synthetic solvents used with other taxanes.
The nab-drug delivery system utilizes the properties of albumin to improve drug uptake into the tumor cells with gp60 receptor and a caveolin-mediated pathway, resulting in better antitumor efficacy and decreased toxicity compared with solvent-based taxanes.
Clinical trials have demonstrated that nab-paclitaxel improves response rates and progression-free survival in patients with metastatic breast cancer including those who have previously been treated with taxanes and anthracyclines.
nab-paclitaxel is currently being studied in several clinical trials for other solid organ tumors including lung and ovarian cancers.
The nab formulation of paclitaxel allows it to be safely administered as a short duration infusion without a need for premedications.
Peripheral sensory neuropathy and myelosuppression are the main adverse effects. Both are manageable and rapidly reversible.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
