Abstract

Nipple discharge is a common complaint among women. It is classified as normal or abnormal depending on features such as laterality, cycle variation, quantity, color or presentation (i.e., induced vs spontaneous). It can be related to benign conditions, such as intraductal papilloma, duct ectasia, plasma cell mastitis or galactorrhea; or to malignant conditions such as ductal, lobular or papillary carcinoma. Techniques used in nipple discharge evaluation include mammography, ultrasound, cytology (which could be assisted by a mammary pump), duct endoscopy, ductography, immunochemical methods and at least surgical excision of the pathological ducts for diagnosis and treatment in the same procedure.
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of MedscapeCME and Future Medicine Ltd. MedscapeCME is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to provide continuing medical education for physicians. MedscapeCME designates this educational activity for a maximum of
Learning objectives
Upon completion of this activity, participants should be able to:
Describe the prevalence of normal and abnormal nipple discharge
Identify features of abnormal nipple discharge
Describe the differential diagnosis of abnormal nipple discharge
Identify features of duct ectasia
List diagnostic tests appropriate for identifying malignant causes of nipple discharge
Nipple discharge is a common presenting symptom among women. The evaluation and management of nipple discharge can be undertaken with minimal difficulty by performing a careful history and examination and following a logical thought process linking the type of discharge with the suitable mode of treatment [1]. However, nipple discharge could be a sign of serious abnormality within the breast [2]. Nipple discharge is 7–10% of all breast symptoms, and galactorrhea is a relatively common problem that occurs in approximately 20–25% of women [3]. However, abnormal nipple dis-charge (with a probable local lesion) is rare, constituting only 3–5% of mammary consultations [4]. Finally, nipple discharge accounts for 3–7% of all breast-related surgical referrals [5]. Although most nipple discharge is due to a benign etiology, approximately 7–15% is due to breast carcinoma [6,7]. Common features of nipple discharge include:
Unilateral or bilateral discharge;
Cycle variation;
Induced or spontaneous discharge;
Discharge may be clear, serous, milky, yellow, green, pink or slightly bloody, brown or black (old blood);
Discharge may originate from one or multiple ducts;
Discharge may be of small, moderate or large quantity.
The discharge may be accompanied by other clinical findings, such as:
Breast findings: mastodynia, palpable mass, palpable lymph node, pruritus or skin depression;
Local: herpes zoster, herpes simplex [8], tuberculosis, trauma or previous surgery of the breast or the thoracic cage;
General: fever, headache, myxoedema or visual disturbances.
Spontaneous or induced nipple discharge may be identified either accidentally by the patient (e.g., coagulated discharge found on the bra) or during clinical examination. Less commonly, it may present during mammography as a result of pressure.
Not all nipple discharges are abnormal. Therefore, it is necessary to categorize it as normal (i.e., causally connected to lactation and not related to an underlying histologic abnormality) or abnormal, or to attribute it to galactorrhea according to history and physical examination.
Normal discharge
During pregnancy and postpartum (until after 2 years) nipple discharge is normal and milky in nature. The same applies after a spontaneous abortion or an intentional termination of pregnancy in the second trimester. In the remaining cases, nipple discharge may or may not be associated with malignancy.
Abnormal discharge
Nipple discharge can be nonspontaneous, bilateral and serous, originating in many ducts. Multiple duct discharges are rarely caused by cancer [9]. In cases not related to the breast, mechanical stimulation, neoplastic processes, systemic diseases, thoracic trauma and medications are included in the differential diagnosis. Medications linked to nipple discharge include antihypertensives, phenothiazines, tranquilizers, antidepressants, antipsychotics, contraceptives and others, as summarized in Box 1. Diagnosis is made from a good history and is confirmed by the discontinuation of the medication.
The patient who is curious to check for discharge may cause continuous mechanical stimulation. In such cases, discontinuation of the stimulation will discontinue the discharge. Women should be advised to avoid checking themselves for discharge since benign discharge may resolve when the nipple is left alone [10].
Causes of galactorrhea.
Medications.
Antidepressants and anxiolytics, including tricyclic antidepressants and selective serotonin-reuptake inhibitors.
Antihypertensives, including methyldopa (Aldomet®) and atenolol (Tenormin®).
Antipsychotics [51].
Histamine H2-receptor blockers, including cimetidine (Tagamet®) and ranitidine (Zantac®).
Hormonal medications, including conjugated estrogen plus medroxyprogesterone acetate, medroxyprogesterone acetate contraceptive injection (Depo-Provera) and oral contraceptives.
Phenothiazines, including chlorpromazine.
Others, including amphetamines, anesthetics, arginine, cannabis, danazol, isoniazid, metoclopramide, opiates and valproic acid [3].
Moreover, nipple discharge may decrease or increase depending of the day of the cycle and, thus, hormonal levels. In these cases, the discharge is milky, bilateral, it originates in many ducts and is copious in quantity. No further workup is indicated in these cases and the patient should be reassured. Ductal discharge must be differentiated from discharge from the surface of the nipple–areola complex.
Abnormal nipple discharge is often nonlactational (i.e., the patient is not pregnant or is more than 2 years from the last breast feeding), persistent, spontaneous and unilateral. It is usually located in one duct. Since it is usually spontaneous, it may also disappear. It may be green or gray and serous in appearance. The most common cause of this discharge is usually benign breast disease. Abnormal nipple discharge is associated with an underlying malignancy in 1.2–15% of patients, and evidence of blood in discharge from one breast and from one duct is an indication to operate for most surgeons [11]. One should identify the quadrant that is involved by applying pressure on the breast, and in this way the affected duct can be easily localized.
Lesions that may be linked to abnormal discharge are intraductal papillomas, duct ectasia, papillomatosis, mastitis, fibrocystic changes, breast carcinoma and Paget's disease of the nipple (Figure 1).

Paget's disease of the nipple.
Bloody, unilateral and discharge originating from one duct are features associated with suspicious lesions [12]. The possibility of an associated cancer increases when the discharge is accompanied by a lump, and when the patient is over 50 years of age [13]. In such cases, a intraductal malignancy cannot be excluded and therefore surgical intervention should not be delayed. According to various reports, cancer is diagnosed in 5–12% of the cases [14–16].
Galactorrhea
Galactorrhea is typically bilateral and spontaneous. It usually originates from many ducts and the discharge is milky. This is typical in pharmacological galactorrhea and discontinuation of the medication will eliminate the discharge. If the cause cannot be found and eradicated, bromocriptine is the drug of choice [13]. Table 1 may be used to categorize nipple discharge according to its nature.
Nipple discharge features based on its nature.

Relatively infrequent causes of nipple discharge.
Etiology
Benign diseases that may be involved in nipple discharge include intraductal papilloma, duct ectasia, fibrocystic changes and mastitis, while malignant diseases include intraductal carcinoma and Paget's diseas of the nipple. Tumors may be identified in small and bigger ducts.
Benign conditions
Intraductal papilloma
Intraductal papilloma is a term that describes an intraductal papillary configuration of the mammary epithelium on macroscopic or microscopic examination [17]. In fact, intraductal papilloma is a local intraductal hyperplasia with simultaneous formation of a vascular and neural axis that eventually becomes macroscopically visible. Solitary or multiple papillomas are the most common cause of nipple discharge reported (35–62%).
A total of 60–80% of intraductal papillomas are accompanied by spontaneous or induced bloody, serous, aqueous or serobloody discharge, which is one of its main clinical findings. A 5% increased risk of developing invasive carcinoma has been reported.
Papillomas affect women of 35–50 years of age, they vary in size, are round or lobular, have a broad base or are pedunculated, may be located in any quadrant (although the more frequent location is in a big duct behind the nipple), may be single or multiple and are not usually accompanied by a palpable mass. Nevertheless, if the papilloma is greater than 1 cm and close to the nipple, it may be palpable.
The presence of multiple papillomas is called papillomatosis, a common finding in menopausal women. In intraductal papillomas, there is bloody flow only if there is a communication between the cyst and the duct. The remaining clinical findings are those typical of a cyst. When bloody (or brown) fluid is aspirated from the cyst, a differential diagnosis between the benign intracystic papilloma and cancer must be made by the cytological evaluation of the specimen [12].
Papillomas may be diagnosed by ductography, in which there is one or multiple contrast filling defects of smooth contour in the duct or interruption of contrast flow in the duct where the papilloma exists. The presentation of ductal cancer may also be similar. The diagnosis is confirmed by cytology, where papillomatous formations are visualized, and by ductography, where interruption of contrast flow in the duct is identified where the papilloma is located. Ductography will facilitate the surgical management and simplify the differential diagnosis between benign intraductal papilloma and papillary carcinoma.
The management of a papilloma requires a surgical procedure: this could include intraductal biopsy with mammary duct endoscopy for diagnosis and treatment [18], sonographically guided mammotome excision of ducts [19], and microdochectomy or pyramidectomy (which consist of dissection of the affected duct). Periareolar incision may be performed, in which a small sound is used to catheterize the duct from which the discharge is originating, and methylene blue dye is injected in order to visualize the duct branches. Staining with methylene blue due (which is both safe and inexpensive [20]) identifies the major discharging duct, as well as its side branches, which allows the surgeon to resect that specific ductal system only. This method allows neighboring major ducts to remain intact, preserving nipple function and permitting future cancer detection. Methylene blue-enhanced major duct excision allows the surgeon to more accurately determine the precise location and volume of tissue that needs to be excised [20]. After the nipple has been stabilized, the dye-colored duct is dissected from the nipple to its depth; the specimen has a triangular shape. After meticulous hemostasis has been obtained, rebuilding of the retro-areolar area is performed and usually a small drain is placed in the wound. It must be noted that although allergic reactions to blue dye are rare, they may be life-threatening, and there is immunologic cross-reactivity between methylene blue and patent blue [21]. Nevertheless, methylene blue dye has been replaced in some countries with patent blue since cases of anaphylaxis and complications have been reported for the former. Generally, the use of blue dye in identifying papilloma is controversial. It tends to make the surgical field messy, and it carries a risk of extravasation, and most surgeons in the UK have abandoned its use intraoperatively for microductectomies. Modified techniques were described, such as transnipple pyramidectomy [15], with a very small incision in the nipple. However, it must be noted that a negative specimen after minimal procedures does not exclude malignancy, and major duct excision appears to detect a higher percentage of occult carcinoma when compared with minimally invasive micro-ductectomy, a finding that may be related to the larger sample size of the resection specimen [22].
Duct ectasia
Duct ectasia is the cause of nipple discharge, with different percentages recorded in different articles (>10% [23], >30% [2] and >50% [24]). There is a broad range of terminology used in the bibliography for the same entity: ‘varicocele-like tumor’, ‘plasma-cell mastitis’, ‘obstructive mastitis’, ‘galactophoritis’, ‘duct ectasia’ and ‘periductal mastitis’ are some of the terms used. The last two terms are widely used and are the most representative of the process. The other terms mentioned earlier represent different stages of the same entity. Duct ectasia usually accompanies benign breast conditions (e.g., epithelial hyperplasia and papillomatosis), but may also be associated with breast cancer.
The diagnosis is set by high sensitivity ultrasonic evaluation together with a cytological assessment of the discharge and, if the discharge originates from one duct, by the ductogram.
Duct ectasia differs from cystic disease since it is an inflammatory process, usually affecting the ducts below the nipple. It develops gradually, either through the ductal system or the breast lobes. Its presentation also varies depending on the duration of the process.
Clinically, it presents as a firm, stable mass under the nipple, similar to a malignant mass. It is more common in the perimenopausal years. It is interesting to note that in a third of mastectomy specimens in women aged over 50 years of age, subclinical duct ectasia was diagnosed. The cause is unknown, although breastfeeding may be related. It is unknown and contradictive whether the inflammatory process proceeds or follows the dilatation of the duct. We believe that the primary process is the dilatation of the duct and the accumulation of amorphous ‘cheese-like’ secretion. The acute inflammation may be caused by epithelial rupture and diffusion of the contents into the fibrous duct-wall and the underlying structures. These contents consist of neutral fat and lipid crystals that are typical of duct ectasia. The chronic granulation-type reaction may develop foreign body-like giant cells and a multiform inflammatory cell population. Since the whole process in duct ectasia is long in duration, plasma cells are sometimes dominant in the inflammatory infiltrations. This finding caused some pathologists to use the term ‘plasma cell mastitis’ in order to describe duct ectasia. Other findings in duct ectasia include the following:
Serous or green-white nipple discharge in 20% of cases;
Nipple inversion that leads to fibrosis and development of ring or tubular calcification (this finding is typical in the mammogram and especially when the disease is in its advanced stages);
Apocrine metaplasia with or without epi-thelial hyperplasia. The epithelial layer consists of widened atrophic cells.
Causal therapy of duct ectasia is not available [25]. The condition may be managed symptomatically and/or with antibiotics, but in persistent or recurrent cases it is managed with surgical excision of the ducts below the nipple. A focused excision is preferred to a complete subareolar excision since the later technique is associated with higher rates of seroma formation, nipple numbness and nipple inversion. The Hadfield's procedure (major duct excision) is an option [26].
Plasma cell mastitis
Plasma cell mastitis is a rare type of chronic mastitis characterized by the presence of multiple plasma cells in microscopical evaluation. It is usually found in multiparous women and is caused by condensation of the breast discharge. Typical findings include thickened ducts and fibrosis that may be multicentric. In palpation, there is a firm mass impression, and fibrosis may lead to nipple inversion. It may be accompanied by serobloody and thick nipple discharge. Cancer should be included in the differential diagnosis.
Other causes
Other syndromes that are indirectly related to the breast discharge are neurohormonal causes such as the amenorrhea-galactorrhea syndrome Chiari-Frommel, pituitary adenomas, hypothyroidism and diabetes mellitus. Investigations of thyroid-stimulating hormone, T3, T4 and prolactin levels are necessary in such cases. Hyperprolactinemia must be ruled out with computed tomography and visual field tests. In a few selected cases, tests should include measurement of estradiol levels, progesterone and androgens.
Malignant conditions
Carcinomas
All types of breast carcinoma (e.g., ductal, lobular, tubular and medullar) are sometimes diagnosed as a result of the nipple discharge symptom. Special consideration should be given to papillary carcinoma, despite it being a rare malignancy (<1%). It is usually inside a duct and may present initially as a unilateral nipple discharge that originates in one duct.
The incidence of intraductal carcinomas of the breast has increased dramatically since 1980 when screening mammography was widely adopted in Western nations [27]. In more than 50% of cases, it appears mammographically as a high-density lesion with accompanying calcifications. Ductogram and cytology are used to reach the diagnosis. Since almost 50% of lesions showing papillary features on cytology prove to be malignant, all cases reported as papillary on cytology should be excised urgently for histologic assessment [28].
Today, it may be diagnosed by high sensitivity ultrasonic examination by an experienced physician.
Evaluation & techniques
Although it was proposed that patients presenting with nipple discharge should undergo biopsy or subareolar exploration based on the presence or absence of a palpable tumor [29], the evaluation could be a simple process. Since the majority of nipple discharge cases are more frequently due to benign conditions, less operative, nonsurgical methods can be applied to limit the need for surgical intervention [16].
The woman that presents with nipple discharge must be managed according to her age by tests such as mammography, ultrasound and cytology. Early cancer cases are difficult to identify, especially in young women with dense breasts.
Mammography
The clinical examination and mammography are first-line diagnostic approaches. Mammography may reveal microcalcifications that must be evaluated (Figure 2). Scattered areas of increased density with microcalcifications could be a mammographic finding in papillomatosis (although this presentation is not typical of the disease).

Microcalcifications in mammography.
Mammography is associated with a 9–10% false-negative rate and a 1–2% false-positive rate for the detection of breast cancer in patients with nipple discharge.
Mammography is recommended to any patient presenting with abnormal nipple discharge, although it has poor positive predictive value (16.7%). This percentage reveals that it is not reliable in the diagnosis of the underlying cause of nipple discharge. Any mammographic abnormality should correspond with the quadrant of the breast from which the discharge originates in order for it to be considered relevant to the cause of the discharge. Owing to its low sensitivity (59%) in the diagnosis of malignant duct pathology, it has limited value as a screening method in the management of nipple discharge. Almost 50% of the patients who are diagnosed with malignant breast disease and who underwent mammography did not have exact diagnosis of the corresponding duct pathology, and there are studies confirming that mammography is inadequate in the diagnosis of abnormal nipple discharge [30]. Only half of the patients presenting with nipple discharge who were found to have cancer had an abnormal mammography. However, most experts recommend that a mammogram be per-formed in women with nonlactational spontaneous discharge [31], usually in women over 30 years of age. It may be that tomosynthesis will be more sensitive; however, experience with this technique is very low at present.
Breast ultrasonography
Breast ultrasonography is complementary to mammography, but as lesions could be in the ducts, this exam could be useful in many cases, such as in intraductal papillomas (Figure 3), particularly with high frequency probes (11-13 MHz) and with 3D–4D technology.

Intraductal papilloma identified with breast ultrasonography.
In addition, ultrasonography can guide fine needle aspiration (FNA) and will help in obtaining cytology specimens from the abnormal area. Finally, since ultrasonography allows visualization of ductal pathology as small as 0.5 mm in diameter, it can be used for ultrasound-guided percutaneous biopsy of lesions [101]. The ultrasonographical findings of the most common causes of nipple discharge are presented in Table 2.
Ultrasonographical findings in common nipple discharge etiology.

In contrast: *To 2 or 3 gentle lobulations; ‡of the intense, uniform hyperechogenicity; and §of thin, echogenic capsule of benign conditions.
Magnetic resonance imaging
It seems that magnetic resonance imaging (MRI) has a moderate sensitivity (>75%), a low-to-moderate specificity (<65%) and a low positive predictive value (< 60%) in the evaluation of nipple discharge [32]. Therefore, the role of MRI is limited in the evaluation of nipple discharge and nowadays it should be restricted to facilities with MRI biopsy capabilities.
Cytology
Cytology is the major complementary technique to clinical examination; it is a good approach to diagnosis in many cases (Figure 4). Cytological evaluation of nipple discharge can be performed directly from the nipple. Cytology has been improved by new techniques such as ‘duct lavage’ and ‘duct endoscopy’ (ductoscopy) [33]. These newer techniques are designed to check the abnormal cells that travel from the ducts to the nipple. Biological markers also analyze the liquid specimen. Nevertheless, this technique has high false-negative findings; it is unrevealing and is not helpful in localizing a tumor. It should be noted that in previous investigations in asymptomatic women with nipple discharge, a diagnosis of malignancy was made in 35% of cytology specimens (70% were suspicious for malignancy).

Cytology of nipple discharge material.
Duct endoscopy (ductoscopy)
Mammary ductoscopy allows visualization of the breast ductal wall and sampling of the abnormal area for diagnostic purposes [34]. First developed in Asia during the 1990s, this technique was performed under local anesthesia. Recent rapid and groundbreaking developments in the field of optics have made the labyrinth of mammary ducts more easily accessible to direct visualization and examination [35]. Ductoscopy identifies intraductal lesions in a high proportion of women with spontaneous nipple discharge, and it may contribute to more accurate resection of these [36]. Ductoscopy may be used for direct diagnosis of papillomas, cancer and direct cytological evaluation. Endoscopy may also be combined with local techniques aiming to manage the lesion, such as radiofrequencies, lasers and microwaves.
Duct endoscopy is performed under local, or even better general, anesthesia by direct visual-ization of the lesion with a special instrument, the microendoscope (0.1-0.2 mm in diameter), which is connected to a source of light (Figure 5). Tumors up to 0.1 mm in diameter can be visualized at a maximum distance of 10 cm.
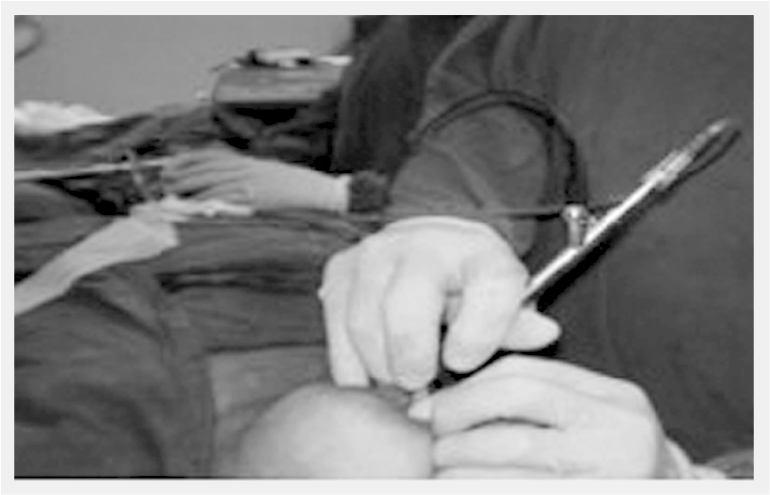
Duct endoscopy (ductoscopy) is performed with a special instrument, the microendoscope.
The first step of the technique consists of an irrigation lavage during which the breast is gently massaged in order to identify the duct where the discharge originates. After catheterization of the duct by a fine cannula, approximately 10 cc of normal saline is injected, and the liquid is then aspirated and sent to cytology. The duct is then catheterized by an endoscope ranging from 1.1 to 0.5 mm in external diameter with a working channel of 0.35 to 0.45 mm that provides depth of perception during visualization.
According to our experience and published data [37,38], the findings can be classified as:
Obstructing endoluminal lesions, such as duct papillomas (Figure 6);
Papillomatous lesions;
Intraductal scarring/adhesions and duct obliteration;
Intraductal calcifications.

Obstructing endoluminal lesion (papilloma) identified by ductoscopy.
Practical and technically feasible, duct endoscopy can even be used in the absence of nipple discharge or in normal nonectatic ducts. According to our experience, ductoscopy is a very promising technique. When better biopsy instruments are developed it will be improved further, since the findings will be correlated to histopathology.
Mammary pump
Zervoudis has developed a simple tool – the mammary pump [39] – in order to obtain additional cytological specimen from tumors that are in close proximity to the nipple (Figure 7). This instrument can also be used to increase spontaneous nipple discharge that originates from benign breast lesions for cytology. The principle of this technique is to suction cells from the breast ducts.

Zervoudis' ‘mammary pump’ (adapted to a fine-needle aspiration gun).
The mammary pump is made of a 20 cc syringe with its front end cut open by scissors. The syringe is adapted to a FNA gun that is used for cytology sampling. The suctioning technique begins after the skin has been prepared with antiseptic solution in circular massage movements over the breast from the periphery to the nipple for approximately 1 min. The mammary pump is then placed on the nipple and between eight and ten suctioning attempts are performed with a negative pressure of 3 cm (FIGURE 8).

Mammary pump placed on the nipple and suctioning attempt with a negative pressure of 3 cm.
The technique is, in general, very well tolerated by the patients. The mammary pump collects fluid from the nipple and the smear is applied and stained on slides by using the Papanicolaou and Diff-Quick techniques. The Masood semiquantitative cytology [40] score is used to classify the breast lesions (Table 3). Classical cytology and liquid-cytology with centrifuge techniques can also be used.
Masood cytologic classification.
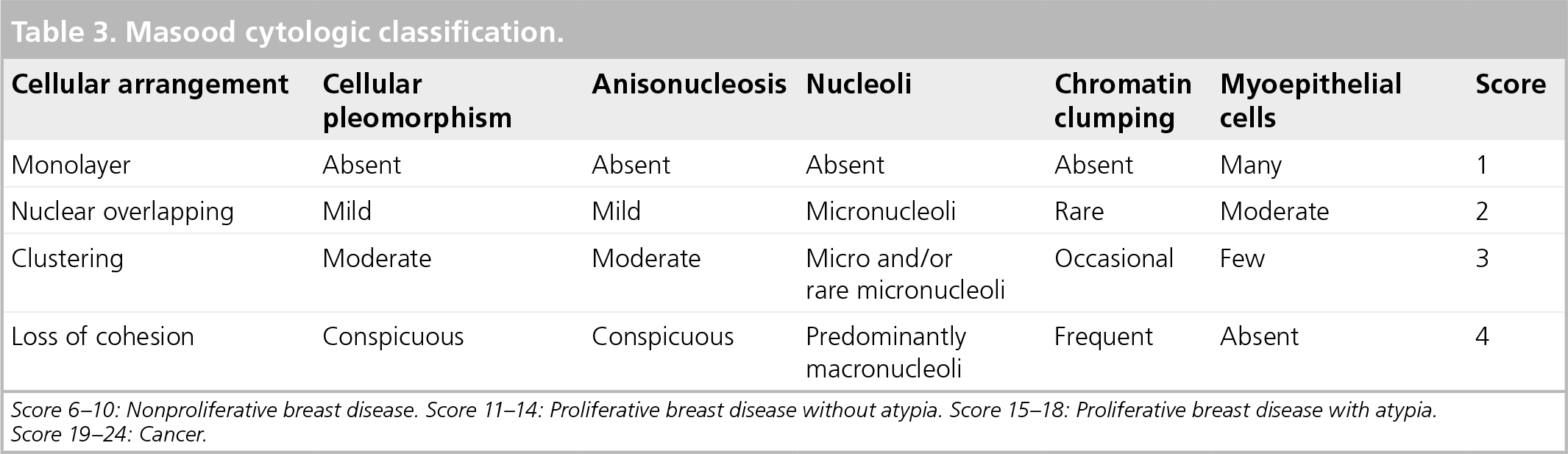
Score 6–10: Nonproliferative breast disease. Score 11–14: Proliferative breast disease without atypia. Score 15–18: Proliferative breast disease with atypia. Score 19–24: Cancer.
The mammary pump is a complimentary technique that increases the numbers of cells used in the diagnosis of breast disease. It applies to the evaluation of any nipple discharge and tumors in close proximity to the nipple, and may be used as an adjunct to FNA. The accuracy of the mammary pump in the detection of abnormal cells is 84% for typical hyperplasia lesions, 61 % for atypical hyperplasia and 70% for intraductal carcinoma [39,41]. These findings represent the correct diagnosis of the cytology specimen obtained by the mammary pump compared with the histological findings of the surgical biopsy. The technique has an accuracy of greater than 80% in the diagnosis of invasive ductal and lobular cancers, when the lesion is less than 4–5 cm from the nipple. Therefore, the technique can be used in the early diagnosis of breast cancer and the positive results may be used in the design of future studies.
Ductography
Ductography (galactography) is probably useful in the evaluation of spontaneous discharge from the nipple of a nonlactating breast [42]. It was previously suggested that patients with abnormal nipple discharge should undergo routine ductography and dye localization before surgery [43]. However, ductography can only be performed if the nipple discharge is reproducible on physical examination and if the duct can be cannulated [31]. This technique developed many decades ago was abandoned, but recently has been put back into use owing to improved mammographic technology. Tabar et al. recommended that all patients with a spontaneous bloody or serous discharge from a single lactiferous orifice undergo galactography in addition to physical, cytological and mammographic examination [23]. In case of abnormal nipple discharge, galac-tography can detect variations of ductal caliber, intraductal alterations and ductal discontinuities [44]. Moreover, the important advantage of this technique is that it gives the exact topography and the depth of the lesion, in contrast with cytology, which is unable to localize it.
Ductography is performed by dilatation, cath-eterization and injection of a water-soluble contrast by a 2 cc Telebrix® syringe. Craniocaudal, lateral and compression views are obtained. The results may be characterized as:
Normal
Ductal dilatation
Filling defect
Cut-off sign (Figure 9)

Cut-off sign in ductography (papilloma).
Ductography has been shown to be accurate in providing the location and depth of ductal abnormalities when a single duct is identified as the source. Data regarding the location of the lesion greatly facilitate biopsy, especially with deep lesions. Galactography-aided wire or coil localization is a practical localization method for intraductal lesions not detectable on mammography or sonography [45]. It improves the diagnostic yield of surgical biopsy from 67% in nonstudied patients to 99% in patients receiving a ductogram. After the lesion has been localized, excision will follow. In conclusion, galactogra-phy is accurate in identifying the location of the ductal abnormality and it allows a focused surgical approach to the pathologic lesion [46]. Nevertheless, ductography cannot be effectively used for the differential diagnosis of a benign versus a malignant lesion [47,48], and it is not useful as a predictor of underlying pathology [32].
Immunochemical analysis/Hemoccult®
The identification of blood in the fluid (blood in small quantities) may be potentially useful with concomitant cytological review. Blood identification kits with the Hemoccult® (Beckman-Coulter, CA, USA) technique are useful, but are not neces-sarily useful in the prediction of the final pathology findings. The positive predictive value of the technique is less than 10% [11] to 20%. However, in 5–28% of cases, bloody or Hemoccult-positive discharge is more likely to be associated with cancer. In general, it should be underlined that mammography, ductograms, cytology and Hemoccult staining, used separately, are inadequate in the correct diagnosis [16].
Surgical diagnosis
Biopsy-excision of the pathological duct(s) should be performed if nipple discharge persists (pathological nipple discharge), or when it is bloody. Biopsy should be preformed by terminal duct excision [14,49].
Microdochectomy
Preoperative workup and imaging may not be suspicious in patients with single-duct pathological nipple discharge and underlying malignancy; therefore, microdochectomy should be considered in such cases [50].
After identifying the orifice of the discharging duct by gentle pressure on the nipple, the duct is probed with a fine lacrimal probe that is passed gently peripherally as far as possible without disrupting the duct. After gentle dilatation the probe is temporarily removed and a 22 G intravenous cannula is inserted into the duct in order to inject 1–2 ml of methylene blue dye. The probe is then reinserted and the affected duct is marked. A limited circumareolar incision is made to raise the areolar skin flap. The probed duct is then identified close to its attachment on the underside of the nipple and carefully dissected free of the surrounding tissues and the unaffected ducts over a length of 5–6 cm towards its proximal end. Finally, the duct is removed by transaction and is marked with a single suture to orientate the specimen.
We have published a minimally invasive technique of microdochectomy, the ‘transnipple pyramidectomy’ [15]. With this technique, a pyramid-shaped specimen through a transnipple approach excises the pathological duct(s) that cause the pathological discharge. The initial steps described previously in micro-dochectomy are identical. After catheterization of the pathological duct (Figure 10), an injection of methylene blue dye is given (Figure 11) and a horizontal transnipple incision is made; only the nipple is opened, not the areola. After the incision, the two nipple flaps are grasped (Figure 12). Next, with small forceps, the proximal part of the marked duct is pulled (Figure 12) and the pathological duct is excised with the periductal tissue as a pyramide shape (Figure 13). The technique has excellent aesthetic and functional results and preserves nipple sensation.

Catheterization of the pathological duct.

Injection of methylene blue dye.

Nipple flaps grasped and small forceps pulling the proximal part of the marked duct.

The pathological duct is excised with the periductal tissue resembling a pyramide shape.
In older patients, irrespective of whether the discharge is localized to one or multiple ducts, major duct excision is preferred in order to provide comprehensive histology and avoid probable further discharge from another duct. In young patients where the option of breastfeeding must be preserved and when the discharge is localized to one duct, microdochectomy can be performed.
In cases where the the excision of many pathological ducts is necessary, we extend the nipple's incision to the areola horizontally 1 cm on each side, and perform a transnipple–transareolar pyramidectomy.
The specimen resembles a pyramid: the peak represents the beginning of the pathological duct marked by a stitch; the base of the pyramid with the corresponding lobules is marked with methylene blue colorant. The choice of technique depends on the anatomy of the breast, on the age of the patient, and on the necessity to excise fewer or more ducts. In general, in older patients, irrespective of whether the discharge is localized to one or multiple ducts, major duct excision is preferred in order to provide comprehensive histology since breast cancer is more frequent (i.e., ductal carcinoma in situ or invasive cancer), and to avoid probable further discharge from other ducts. In young patients when the discharge is localized to one or two ducts, owing to aesthetics and because the option of breastfeeding must be preserved, microdochectomy can be performed.
Conclusion
Nipple discharge is a common complaint among women (7–10% of all breast symptoms). It is necessary to determine the exact location (origin) and the cause of the discharge. Despite the fact that the most frequent causes are duct ectasia and benign papilloma, we should remember that breast cancer is found in 6–8% of cases involving pathological nipple discharge. Cytology remains a good indicative tool for diagnosis, but in the majority of cases it is necessary to obtain a histological diagnosis by surgical procedure. Moreover, the surgery will definitively treat the lesion in cases of benign cause. Recently, the development of endoscopic tools and fiber optics will allow safer diagnosis and treatment without any sacrifice in function, as well as excellent aesthetic results. Today, there are simple noninvasive or minimally invasive techniques that can be used in the evaluation and management of this condition that provide very good results in the majority of the cases. Nipple discharge screening is very important during breast examination; a pathological discharge imposes complementary explorations, and cytology is fundamental; however, in the majority of cases, histology after surgical excision will establish the final diagnosis. However, it must be noted that nipple discharge on squeezing (and/or with small amounts of suction) is found in a relatively high percentage of cases and ‘over-reacting’ in investigations is not always the golden rule. Attempting to forcefully express the nipple for discharge during breast examination may increase the number of unwanted investigations that would otherwise not be performed in these patients.
Executive summary
Nipple discharge is a common complaint among women. Evaluation and management of nipple discharge can be undertaken with minimal difficulty by performing a careful history and examination, and by logically linking the type of discharge with a suitable mode of treatment.
Normal discharge is nonspontaneous and is usually bilateral. It is serous and originates in many ducts.
Galactorrhea is typically bilateral, it originates in many ducts and the discharge is milky. This is typical in pharmacological galactorrhea, and discontinuation of the medication will eliminate the discharge.
Abnormal nipple discharge is defined as nonlactational, persistent, spontaneous and unilateral. It is usually located in one duct.
Benign diseases that may be involved in nipple discharge include intraductal papilloma, duct ectasia, fibrocystic changes and mastitis; while malignant diseases include intraductal carcinoma and Paget's disease of the nipple.
Solitary or multiple papillomas are the most common cause of reported nipple discharge. The presence of multiple papillomas is called papillomatosis, a common finding in menopausal women.
Duct ectasia is the cause of nipple discharge in less than 15% of patients.
A broad range of terminology are used for the same entity, including plasma-cell mastitis since it is an inflammatory process.
Plasma cell mastitis is a rare type of chronic mastitis characterized by the presence of multiple plasma cells in microscopical evaluation.
All types of breast carcinoma (including ductal and lobular) are sometimes diagnosed as a result of a nipple discharge symptom. Papillary carcinoma (although rare) is usually inside a duct and may present initially as a unilateral nipple discharge that originates in one duct.
Mammography: the clinical examination and mammography are first-line diagnostic approaches. Mammography may reveal microcalcifications that must be evaluated, and it should be performed in women with nonlactational spontaneous discharge, usually in those aged over 30 years.
Ultrasonography: this is complementary to mammography. This exam could be useful in many patients, particularly with high-frequency probes (11–13 mHz).
Magnetic resonance imaging (MRI): at present, MRI should be restricted to facilities with MRI biopsy capabilities.
Cytology: cytological evaluation of nipple discharge can be done directly from the nipple, and the method can be improved by ‘duct lavage’ and ‘duct endoscopy’. Nevertheless, cytology has a high rate of false-negative findings.
Mammary pump: a simple tool in order to obtain additional cytological specimen from tumors that are in close proximity to the nipple.
Ductoscopy: this identifies intraductal lesions in a high proportion of women with spontaneous nipple discharge, and it may contribute to more accurate resection of these.
Ductography: this can detect variations of ductal caliber, intraductal alterations and ductal discontinuities, although it can only be performed if the nipple discharge is reproducible and the duct can be cannulated.
Immunochemical analysis/Hemoccult®: the identification of blood which is in small quantities may be potentially useful with concomitant cytological review.
Biopsy-excision of the pathological duct(s) should be performed if nipple discharge persists or when it is bloody.
The removal of the duct by a technique that leaves intact the surrounding tissues and the unaffected ducts. The procedure can also be carried out by transnipple pyramidectomy.
Endoscopic appearances of ductoscopy should be improved in order to achieve accurate diagnoses in more cases.
Biopsy tools need further improvements.
Further evaluation of the potential therapeutic role of guided intraductal biopsy, and of cytology assisted by ductoscopy as a screening tool, is required.
The mammary pump could be prove to be an essential instrument in nipple discharge screening.
Footnotes
CME Author
Désirée Lie, MD, MSEd
Clinical Professor, Department of Family Medicine, University of California, Irvine; Director, Division of Faculty Development, UCIMedical Center, Irvine, California, USA
Disclosure: Désirée Lie, MD, MSEd, has disclosed no relevant financial relationships.
Authors and Disclosures
All authors from: Department of Gynaecology, Department of Mastology, Lito Maternity Hospital, Athens, Greece Stefanos Zervoudis, MD
Disclosure: Stefanos Zervoudis, MD, has disclosed no relevant financial relationships.
Georgos latrakis, MD
Disclosure: Georgos latrakis, MD, has disclosed no relevant financial relationships.
Panagiotis Economides, MD
Disclosure: Panagiotis Economides, MD, has disclosed no relevant financial relationships.
Demetrios Polyzos, MD
Disclosure: Demetrios Polyzos, MD, has disclosed no relevant financial relationships.
Iordanis Navrozoglou, MD
Disclosure: Iordanis Navrozoglou, MD, has disclosed no relevant financial relationships.
Editor
Elisa Manzotti, Editorial Director, Future Science Group, London, UK.
Disclosure: Elisa Manzotti has disclosed no relevant financial relationships.
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Nipple discharge screening
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions and earn continuing medical education (CME) credit, please go to http://www.medscape.com/cme/futuremedicine. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.com. If you are not registered on Medscape.com, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,
