Abstract
Polycystic ovary syndrome (PCOS) is one of the most common causes of female infertility, affecting 5–10% of women of reproductive age. The syndrome is characterized by anovulation, hyperandrogenism and polycystic ovaries. Furthermore, PCOS is associated with insulin resistance and obesity, which is present in approximately 50% of women with PCOS. Reproductive function in women with PCOS is strongly dependent on bodyweight and metabolic status. Obesity is associated with an increased risk of infertility and may also have a negative influence on pregnancy outcome. Considering the worldwide epidemic of obesity, clinical problems relating to PCOS may worsen and increase in frequency. Lifestyle interventions resulting in weight loss comprise the most successful strategy to improve symptoms of PCOS. However, many patients fail to lose weight or may quickly regain weight. It is an important challenge to develop effective lifestyle programs and adjuvant pharmacologic treatments in order to improve reproductive and metabolic health among women with PCOS.
Keywords
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of MedscapeCME and Future Medicine Ltd. MedscapeCME is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to provide continuing medical education for physicians. MedscapeCME designates this educational activity for a maximum of
Learning objectives
Upon completion of this activity, participants should be able to:
Describe the diagnostic criteria for polycystic ovary syndrome (PCOS)
Identify clinical consequences of androgen excess in women with PCOS
Describe long-term risks associated with PCOS
Describe the effects of PCOS on pregnancy outcomes
Describe the effect of metformin on PCOS
Polycystic ovary syndrome (PCOS) is one of the most common endocrine disorders in women of fertile age, affecting 5–10% of the female population. The syndrome is characterized by chronic anovulation, hyperandrogenism, polycystic ovaries and decreased fertility [1]. PCOS is furthermore associated with insulin resistance, accumulation of abdominal fat and obesity (BMI >30 kg/m2), which is present in approximately 50% of women with PCOS [2–3]. It is well known that reproductive function in women with PCOS is strongly dependent on bodyweight and the metabolic status of the patient. In the long term, PCOS is associated with an increased risk of Type 2 diabetes, cardiovascular disease, dyslipidemia and endometrial cancer [1]. Therefore, the syndrome is recognized as having a major impact throughout life on the gynecological and metabolic health of women.
Polycystic ovary syndrome is a heterogeneous disorder that presents with a spectrum of symptoms and manifestations that vary over time. This leads to difficulties in diagnosing the syndrome. The diagnostic criteria for PCOS were revised at an International Expert Conference in Rotterdam in 2003 [4]. According to the Rotterdam criteria, diagnosis of PCOS requires at least two of the three following features [4]:
Oligo-/anovulation
Clinical and/or biochemical hyperandrogenism
Polycystic ovaries on ultrasound
However, this definition has been criticized owing to the fact that it broadens the population of women who will meet the criteria for PCOS, since it includes women with regular menses who also have polycystic ovaries. Furthermore, hyperandrogenism is not required for the diagnosis. Recently, the Androgen Excess Society recommended that PCOS should be considered a disorder of androgen excess and that the original NIH criteria of 1990 should be accepted with some modifications [5]. Thus, the criteria for a PCOS diagnosis are still under debate.
The etiology of PCOS is largely unknown, even though the syndrome was first described in 1935 by Stein and Leventhal [6]. Several etiological factors have been postulated, including genes involved in androgen and/or insulin actions, androgen programming in utero, as well as environmental factors [7]. There is strong evidence that PCOS has a genetic background, which is supported by increased familiar occurrence and by twin studies [7–9]. However, so far, no single gene defect has been identified and, therefore, PCOS is more likely to be a polygenetic disorder. Experimental animal models have demonstrated that prenatal exposure to androgens is associated with many of the features typical of PCOS, such as abnormal luteinizing hormone secretion, hyperandrogenism, anovulation, abdominal adiposity and insulin resistance in adult life [10]. However, the clinical relevance of this hypothesis remains to be elucidated. Furthermore, it has been hypothesized that insulin resistance is a socioecological adaptation to behavioral changes in reproductive and lifestyle habits [11]. Overweight and obesity are of particular importance for the development of PCOS [12].
A major concern for women with PCOS is decreased fertility, which is aggravated by obesity and metabolic aberrations. This review will focus on the association between PCOS and obesity and the mechanisms by which obesity Influences reproductive function and pregnancy outcome in women with PCOS.
Hyperandrogenism & insulin resistance
Hyperandrogenism and insulin resistance are the endocrine cornerstones in the pathogenesis of PCOS, which could explain the various symptoms of the disorder. There is evidence of a primary abnormality of the theca cells whereby they produce increased amounts of androgens [13,14]. The ovarian hyperandrogenism is augmented by disordered feedback control of pulsatile gonadotropin-releasing hormone secretion, leading to elevated luteinizing hormone secretion and a relative follicle-stimulating hormone deficiency, which will favor androgen synthesis [15]. Increased production of androgens impairs follicular development, resulting in a large number of small follicles and an increased amount of stroma due to enhanced follicle atresia [16]. The clinical consequences of increased production of androgens from the ovary are hirsutism, the typical polycystic ovarian morphology (

4D ultrasound of a typical polycystic ovary.
It has been demonstrated that patients with PCOS also have an increased occurrence of insulin resistance, which is independent of obesity [17]. There is support that many patients with PCOS have a genetic post-receptor defect in the insulin signaling pathway that causes insulin resistance and secondary hyperinsulinemia. The molecular mechanisms for this defect may involve serine phosphorylation of the insulin receptor, which inhibits insulin signaling [18–20]. Interestingly, the insulin resistance associated with PCOS appears to be tissue selective and has been reported in muscle, adipose tissue and the liver, but not in the ovary [19,21]. Insulin may act directly or synergistically with luteinizing hormone to enhance androgen production from the ovarian theca cells [22]. Insulin also inhibits hepatic synthesis of sex hormone-binding globulin (SHBG), and thereby increases free or bio-available testosterone levels [23]. Furthermore, insulin decreases hepatic and ovarian synthesis of insulin-like binding protein-1, which promotes IGF-I actions, such as stimulation of ovarian androgen production [22]. Therefore, hyperinsulinemia contributes to hyperandrogenism and ovarian dysfunction in women with PCOS. Furthermore, insulin resistance is associated with metabolic disorders that predispose an individual to Type 2 diabetes.
Inversely, testosterone may induce insulin resistance through several mechanisms. It has been suggested that testosterone facilitates catecholamine-stimulated lipolysis in visceral fat tissue, thus exposing the liver to a high flux of free fatty acids, which could result in hepatic insulin resistance [24]. Testosterone may also cause insulin resistance by inducing decreased capillary density in peripheral muscle tissue [25]. Furthermore, it has been demonstrated that short-term testosterone treatment in postmenopausal women induces a change in the expression of lipolytic signaling proteins in abdominal subcutaneous adipose tissue in relation to lipolytic activity and insulin resistance [26,27]. It has not been elucidated whether hyperandrogenism or reduced insulin sensitivity is the primary factor in PCOS or if the combination of both factors is necessary for the development of the syndrome.
Obesity & appetite regulation in PCOS
Accumulation of abdominal/visceral fat is a common clinical finding in patients with PCOS, even if they are of normal weight [12,28]. Hyperandrogenism in these women might contribute to abdominal fat deposition as demonstrated by long-term testosterone treatment in female-to-male transsexuals [29]. Abdominal fat predisposes an individual to insulin resistance, dyslipidemia and hypertension; in other words, the metabolic syndrome, leading to increased long-term risk of diabetes mellitus and cardiovascular disease [1,12]. Among women with PCOS, 40% have impaired glucose tolerance and 10% develop Type 2 diabetes by their fourth decade [30,31]. The reported prevalence of obesity in women with PCOS has varied greatly depending on population and ethnicity, but it could be approximately 50% [2–3,12]. Recently, it was demonstrated that the prevalence of obesity among women with PCOS in the USA had increased from 51% in 1987 to 74% in 2002, paralleling the corresponding increase of obesity in the general population [32].
Weight gain often precedes the development of PCOS and, conversely, weight loss is associated with resolution of symptoms [12]. Weight gain may induce a vicious circle of endocrine/metabolic abnormalities and aggravating clinical symptoms (
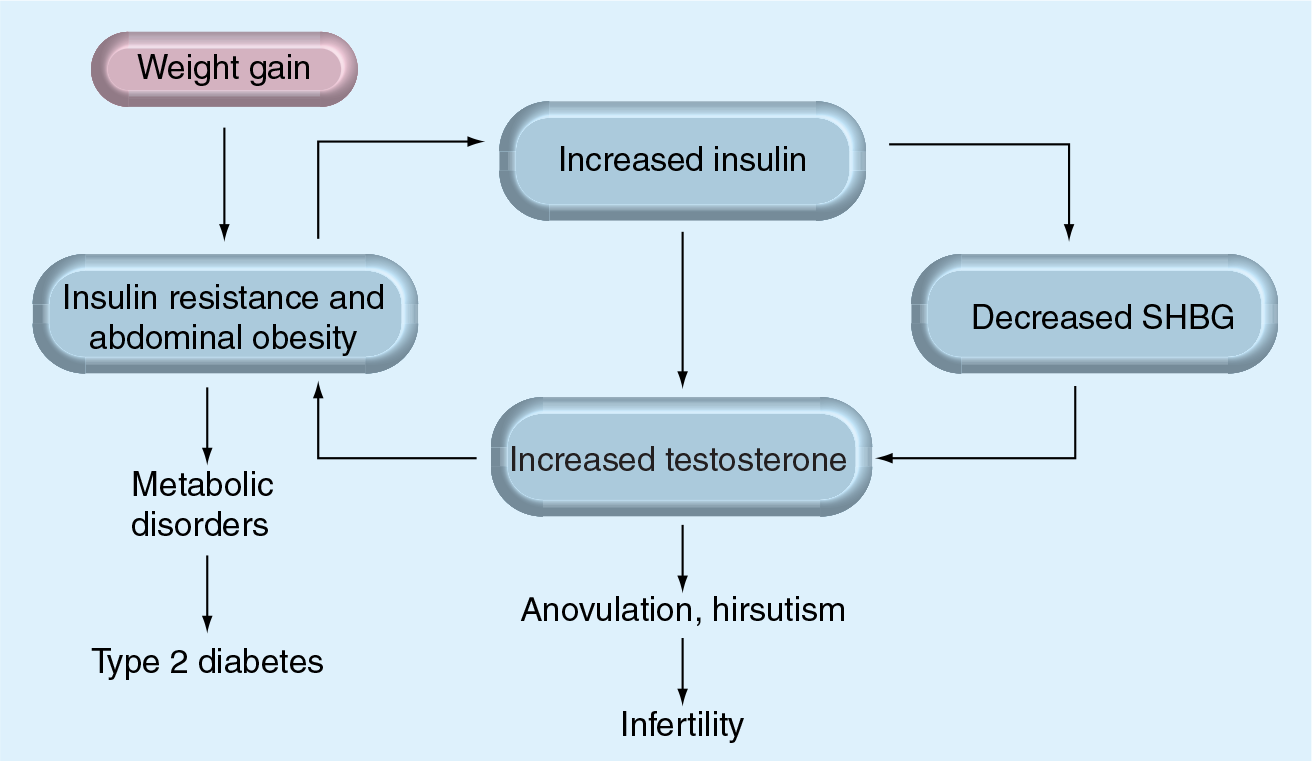
Weight gain induces a vicious circle of endocrine/metabolic abnormalities and aggravating clinical symptoms.
Many women with PCOS suffer from a craving for sweets, a reduced feeling of satiety and a tendency toward binge-eating [33]. We have demonstrated that women with PCOS have reduced meal-related secretion of the gastrointestinal ‘satiety peptide’, cholecystokinin, compared with BMI-matched controls [34]. Furthermore, patients with PCOS display dysregulation of ghrelin, a hormone secreted from the gastric mucosa, which stimulates hunger and food intake [35]. Thus, there is evidence of disturbed appetite regulation in patients with PCOS, which, together with the characteristic endocrine/metabolic abnormalities, may explain why these women have to struggle to maintain normal bodyweight. Increased appetite in patients with PCOS may be part of an anabolic constitution that predisposes an individual to obesity and insulin resistance. It seems likely that this constitution, probably of genetic nature, provides evolutionary advantages in times of nutrition shortages. Furthermore, we have demonstrated that the constitution of women with PCOS may be an advantage for physical performance [36]. However, in times of unlimited food supplies it may, together with a sedentary lifestyle, lead to the development of obesity and reproductive failure.
Impact of obesity on fertility in women with PCOS
Impaired reproductive function in women with PCOS is usually explained by anovulation. PCOS is considered to be the most common cause of anovulatory infertility, explaining up to 70% of the cases [37]. However, implantation failure and a tendency towards higher incidence of miscarriage [38], as well as recurrent miscarriage [39], indicate that endometrial factors may also be of importance for reproductive dysfunction in PCOS. There is increasing evidence that the endocrine and metabolic abnormalities, such as unopposed estrogen, hyperandrogenism and hyperinsulinemia in PCOS, could have adverse effects on the endometrium and contribute to infertility in these patients [40]. Furthermore, ovarian hyperandrogenism has been associated with reduced oocyte and embryo quality [41].
Obesity, particularly the abdominal type, has a major impact on reproductive function in women with PCOS. Obesity is related to more severe hirsutism, more frequent anovulatory cycles and increased frequency of infertility in women with PCOS [42–44]. Therefore, obese patients have an increased need for assisted reproduction treatment. There is also evidence of a higher incidence of blunted responsiveness to pharmacological treatments that induce ovulation and a reduced pregnancy rate in obese PCOS patients [45–47]. Furthermore, obesity is associated with lower embryo quality and a decreased rate of implantation by IVF treatment [48–50]. In a recent study, Jungheim et al. demonstrated that morbid obesity (BMI ≥40 kg/m2) in women with PCOS was related to significantly lower pregnancy rates after IVF compared with patients who are not morbidly obese (odds ratio [OR]: 0.44; 95% CI: 0.22–0.87) [51]. Live birth rates also tended to be lower in the group of patients with morbid obesity; however, the difference did not reach statistical significance.
The negative impact of obesity on fertility in patients with PCOS is probably related to aggravated endocrine and metabolic disturbances (
Several studies have demonstrated that obese patients with PCOS have a greater risk of miscarriage than normal-weight patients [55,56]. There is support that the increased miscarriage rate associated with PCOS could be attributed to obesity per se [57]. In a recent meta-analysis of 16 studies, it was concluded that overweight and obesity increase the general risk of miscarriage [58]. The mechanism by which obesity contributes to the risk of miscarriage is not fully understood. However, it seems plausible that the endocrine disturbances characteristic of both obesity and PCOS, including hyperinsulinemia and hyperandrogenism, could have a negative influence on endometrial and ovarian function. Furthermore, hyperinsulinemia is associated with elevated plasminogen activator inhibitor-1 concentrations, which have been related to an increased risk of miscarriage in patients with PCOS [59,60]. It is suggested that hypofibrinolysis, induced by plasminogen activator inhibitor-1, may cause a defective hemostatic response leading to placental thrombosis and subsequent fetal loss [59,60].
Obesity & pregnancy outcome in PCOS
Polycystic ovary syndrome is considered to have significant implications for pregnancy outcomes. In a large meta-analysis involving 15 studies with 720 PCOS patients and 4505 weight-matched controls, it was demonstrated that women with PCOS have a significantly higher risk of developing gestational diabetes (OR: 2.94; 95% CI: 1.70–5.08), pregnancy-induced hypertension (OR: 3.67; 95% CI: 1.98–6.81) and pre-eclampsia (OR: 3.47; 95% CI: 1.95–6.17) [61]. Furthermore, these women have an increased risk of neonatal complications, such as preterm birth (OR: 1.75; 95% CI: 1.16–2.62), neonatal intensive care (OR: 2.31; 95% CI: 1.25–4.26) and mortality (OR: 3.07; 95% CI: 1.03–9.21) [61]. However, it remains unclear whether PCOS itself is responsible for the increased pregnancy risks or if the associations could be explained by obesity. Furthermore, it is not known how PCOS and obesity in combination influence pregnancy outcomes. It is also noteworthy that most of the patients with PCOS included in these studies had conceived after ovulation induction or IVF, whereas the controls, in majority, were spontaneously pregnant [61].
Impact of obesity on reproduction in women with polycystic ovary syndrome.
Increased frequency of anovulatory cycles and infertility compared with normal-weight patients [41–43].
Higher incidence of blunted responsiveness to pharmacological treatments to induce ovulation and reduced pregnancy rate [44–46].
Lower embryo quality and decreased rate of implantation by IVF treatment [47–50].
Higher risk of gestational diabetes [66].
Since insulin resistance is a common feature of PCOS, it would be expected that women with PCOS run an increased risk of developing gestational diabetes. In a retrospective case–control study, it was demonstrated that women with a history of gestational diabetes indeed demonstrated a higher prevalence of PCOS compared with women with uncomplicated pregnancies [62]. Conversely, several studies have demonstrated an increased risk of gestational diabetes in women with PCOS [63–66]. There are studies suggesting that the higher incidence of gestational diabetes in women with PCOS could be explained by a higher occurrence of obesity in these women [67]. However, a meta-analysis has demonstrated that the increased risk of developing gestational diabetes in women with PCOS compared with controls remained after exclusion of studies in which a higher BMI among women with PCOS was reported [61]. On the other hand, obesity appears to constitute an additional risk for gestational diabetes in women with PCOS.
Pregnancy-induced hypertension and pre-eclampsia also appear to be associated with PCOS according to a meta-analysis of eight studies [61]. The underlying mechanisms for this association have not been elucidated. However, they may be related to metabolic disorders in women with PCOS since insulin resistance in early pregnancy is associated with an increased risk of pre-eclampsia [68]. Higher risk of pregnancy-induced hypertension and pre-eclampsia suggest that women with PCOS may run an increased risk of placental insufficiency, which in turn could lead to neonatal complications.
There is some evidence of increased incidence of preterm delivery in patients with PCOS according to a meta-analysis of eight studies [61]. Preterm birth may partly explain the higher incidence of admission to a neonatal intensive care unit and perinatal mortality in the children born to women with PCOS [66,69–72]. Reported causes of perinatal mortality were lethal malformations, cervical insufficiency, sepsis and placental abruption [66,69–72]. Although women with PCOS are often obese and have insulin resistance prior to pregnancy as well as a higher rate of gestational diabetes, there is no evidence of an increased incidence of macrosomia in the infants born to these women [65–66,73]. Furthermore, there is no increased risk of small-for-gestational-age infants to women with PCOS [65–66,73].
Strategies to improve reproduction in obese women with PCOS
The first choice of treatment for overweight and obese patients with PCOS is a change of lifestyle aimed at weight loss. Diet and exercise resulting in weight loss comprise the most successful strategy to improve reproductive and metabolic parameters of PCOS. Several studies have demonstrated that even a 5–10% loss in bodyweight can restore menstrual cyclicity and ovulation [74–79]. The effects of different types of diets on the endocrine and metabolic parameters in PCOS have been compared in smaller, randomized studies [75,79,80]. It has been suggested that a diet of increased protein intake at the expense of carbohydrates could be more favorable than other diets with regard to the improvement of insulin sensitivity, weight reduction and maintenance of weight loss in patients with PCOS [75]. However, a recent, large, randomized study in overweight subjects reported similar weight loss for diets with different compositions of fat, protein and carbohydrates after 2 years of treatment [81]. Exercise as the only intervention has also been demonstrated to improve fertility, insulin sensitivity and cardiopulmonary functional capacity in women with PCOS [77,82]. Preliminary data from our 4-month interventional trial have demonstrated that diet and exercise are equally effective at improving fertility in spite of significantly greater weight loss by diet than by exercise [Nybacka et al. Unpublished Data]. Although there is no doubt that weight loss is clearly beneficial in overweight women with PCOS, a great clinical problem is posed by many patients failing to lose weight by lifestyle changes or quickly regaining weight after an initial weight loss. Structured lifestyle management with long-term support may improve patients' compliance.
It has been proposed that women with PCOS benefit from metformin therapy [83]. Metformin is a biguanide that inhibits the production of hepatic glucose, thereby decreasing insulin secretion, and enhances insulin sensitivity in peripheral tissues [83]. The effect of metformin on bodyweight and fat distribution in patients with PCOS is unclear since some studies have demonstrated weight loss and reduction in waist circumference while others have not [83]. However, metformin appears to improve the efficacy of lifestyle modifications resulting in weight loss and reduction in visceral fat [76,84]. There is evidence of a beneficial effect of metformin on the menstrual pattern and hormone levels in patients with PCOS [84,85]. Interestingly, metformin seems to be more effective in nonobese patients than in obese patients [86]. A meta-analysis demonstrated that metformin is more efficient than placebo in terms of inducing ovulation in women with PCOS (OR: 3.88; 95% CI: 2.25–6.69) [87]. However, a large, randomized trial demonstrated that ovulation induction by clomiphene is clearly superior to metformin in achieving live births in patients with PCOS who have not previously been treated [88]. However, in clomiphene-resistant patients, metformin increases the ovulation frequency when added to clomiphene (OR: 4.41; 95% CI: 2.37–8.22) [87].
Metformin treatment has also been used in patients with PCOS during pregnancy. A placebo-controlled study in 40 patients demonstrated reduced pregnancy-related complications, such as preterm delivery and pre-eclampsia, in the metformin group when compared with the placebo [89]. In other prospective but uncontrolled studies, metformin has been demonstrated to reduce the frequency of miscarriages and gestational diabetes in women with PCOS [90,91]. Recently, a placebo-controlled study involving 29 patients with PCOS confirmed the finding of a reduced abortion risk [92]. A meta-analysis of eight studies did not find any evidence for adverse pregnancy outcome in PCOS women undergoing treatment with metformin [93]. However, data that confirm the beneficial effects of metformin from large-scale randomized studies are needed before the treatment could be recommended for patients with PCOS during pregnancy.
There are few studies evaluating the effects of antiobesity drugs in PCOS. Orlistat is a potent inhibitor of gastric and pancreatic lipase, inhibiting the digestion of dietary triglycerides and decreasing the absorption of lipids. Orlistat reduces weight and is as beneficial as metformin in reducing testosterone levels [94]. Sibutramin acts by inhibiting the reuptake of serotonin and norepinephrine, thereby increasing satiety, thus resulting in weight reduction of 5–10% in obese patients [95]. This drug potentiates the effect of lifestyle modification resulting in weight loss and improvements on metabolic and cardiovascular risk factors in PCOS patients [96–98].
The modest and short-lasting effects of lifestyle interventions and antiobesity drugs in the treatment of obesity have led to an increasing interest in bariatric surgery. A recent meta-analysis of more than 22,000 procedures of bariatric surgery found an average excess weight loss of 61% (BMI decrease: −14.2), which was associated with significant improvements in metabolic and cardiovascular symptoms [99]. A large prospective study has confirmed that the weight loss and metabolic benefits were maintained 10 years after the surgical procedure [100]. Bariatric surgery has been demonstrated to be highly effective in treating patients with PCOS with morbid obesity resulting in sustained weight loss, resolution of PCOS symptoms and improved fertility [101,102]. It has therefore been suggested that bariatric surgery should be considered as first-line treatment in patients with PCOS with severe obesity.
In conclusion, encouragement of lifestyle changes should always be the first-line strategy in the management of obese patients to improve fertility (

Lifestyle modification should be the first-line strategy in the management of obese patients to improve fertility.
Future perspective
According to the WHO, obesity has become a global epidemic with at least 300 million adults afflicted worldwide. The increasing incidence of child obesity is of particular concern. The health consequences of obesity are well known and include Type 2 diabetes, cardiovascular disease and certain forms of cancer. Obesity per se is also associated with reduced fertility. In an American study, Yildiz et al. demonstrated that the obesity rate among women with PCOS rose considerably during the last decades, whereas the prevalence of PCOS was only minimally increased [32]. These results may imply that genetic factors play a more important role than environmental factors for the development of PCOS. However, it still appears likely that clinical symptoms of PCOS may worsen and increase in frequency in parallel with the increasing incidence of obesity in the general population.
Executive summary
Hyperandrogenism and insulin resistance are related conditions that compose the endocrine cornerstones in the pathogenesis of polycystic ovary syndrome (PCOS).
Increased ovarian production of androgens is responsible for hirsutism, polycystic ovarian morphology and anovulation, whereas insulin resistance is associated with metabolic complications.
Insulin resistance leading to secondary hyperinsulinemia stimulates ovarian androgen production and inhibits the synthesis of sex hormone-binding globulin, which results in increased levels of free testosterone.
Testosterone may induce insulin resistance by various mechanisms in the liver, muscle and subcutaneous adipose tissue.
Abdominal fat accumulation is a characteristic feature of PCOS and predisposes an individual to insulin resistance, the metabolic syndrome and increased long-term risk of Type 2 diabetes.
The prevalence of obesity in women with PCOS varies with population and ethnicity, but has been estimated to be approximately 50%.
Increased weight and obesity exacerbate all symptoms of PCOS, whereas weight loss is associated with resolution of symptoms.
There is evidence of disturbed appetite regulation in PCOS, which may partly explain why many patients with PCOS struggle to not gain weight.
Impaired reproductive function in women with PCOS is primarily explained by ovarian dysfunction, but there are also indications of changed endometrial receptivity and decreased oocyte and embryo quality.
Obesity in women with PCOS is associated with increased frequency of menstrual disturbances and infertility and an increased need for assisted reproduction treatment.
There is also evidence of a higher incidence of blunted responsiveness to pharmacological treatments that induce ovulation and a reduced pregnancy rate in obese PCOS patients.
Obese patients with PCOS have a greater risk of miscarriage than normal-weight patients.
Women with PCOS have a significantly higher risk of developing gestational diabetes, and obesity appears to constitute an additional risk.
PCOS is also associated with pregnancy-induced hypertension and pre-eclampsia, which may be related to metabolic disorders.
There is some evidence of increased incidence of preterm delivery, higher incidence of admission to a neonatal intensive care unit and perinatal mortality in the children born to women with PCOS.
There is no evidence of increased incidence of macrosomia or small-for-gestational-age infants born to women with PCOS.
Diet and exercise resulting in weight loss comprise the most successful strategy to improve reproductive and metabolic parameters of PCOS. Even a moderate weight loss of 5–10% can restore menstrual cyclicity and ovulation.
There is evidence of a beneficial effect of metformin on the menstrual pattern and in inducing ovulation in women with PCOS.
Clomiphene citrate is more effective than metformin for the induction of ovulation and pregnancy. However, in clomiphene-resistant patients with PCOS, metformin in combination with clomiphene increases the ovulation frequency.
Antiobesity drugs may potentiate the effect of lifestyle modification, resulting in weight loss, as well as improving metabolic and cardiovascular risk factors in patients with PCOS.
In patients with severe obesity, bariatric surgery appears to be the most effective therapy to lose weight and to improve fertility.
Clinical symptoms of PCOS may worsen and increase in frequency in parallel with the increasing incidence of obesity in the general population.
An ethical dilemma is that more and more patients are becoming obese and fail to lose weight to achieve recommended levels of BMI of less than 35 before fertility treatment.
For the future it is of great importance to develop strategies for the prevention of overweight and obesity in order to improve reproductive and metabolic health.
The most important challenge is to develop programs of lifestyle modification that are effective in the long run, since many fail to result in sustained weight reduction.
Since obesity is related to difficulties in conceiving and increased risks during pregnancy and delivery, obese women are advised to lose weight prior to pregnancy. However, in spite of clinical support, many women with PCOS fail to lose weight to achieve the recommended BMI of less than 35 prior to fertility treatment [103,104]. Should these patients not be offered ovulation induction therapy and other kinds of assisted reproductive technologies? This is an ethical dilemma that will become more and more evident for clinicians while obesity escalates in the population.
For the future, it is of great importance to develop strategies for the prevention of overweight and obesity in order to stop the growing obesity trend. Women diagnosed with PCOS should be informed about the role of weight gain in the aggravation of clinical symptoms and the significance of weight loss for their potential resolution. Lifestyle consolation should be offered to all patients who need it. An important challenge is to develop effective programs of lifestyle modification in order to improve reproductive and metabolic health in women with PCOS. Both diet and exercise programs appear to be efficient in the short run but often fail to result in sustained weight reduction. Adjuvant pharmacologic treatments, including metformin and antiobesity drugs, may enhance the effect of lifestyle intervention alone. However, the effect of pharmacologic treatments on weight loss is limited. For severely obese patients, bariatric surgery appears to be the most effective treatment.
Footnotes
Désirée Lie, MD, MSEd, Clinical Professor, Family Medicine, University of California, Orange; Director, Division of Faculty Development, UCI Medical Center, Orange, California. Disclosure: Désirée Lie, MD, MSEd, has disclosed no relevant financial relationships.
Polycystic ovary syndrome,obesity & reproductive implications
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions and earn continuing medical education (CME) credit, please go to http://www.medscape.com/cme/futuremedicine. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.com. If you are not registered on Medscape.com, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,
