Abstract
Influential guidelines have supported the role of metformin in the management of polycystic ovary syndrome (PCOS) for a number of years. However, regulatory approvals for this therapeutic indication are still exceptional and exist only in a few countries, including for the originator, Glucophage®. PCOS is an insulin-resistant state, which drives hyperandrogenism and anovulatory infertility. The metabolic action of metformin involves amelioration of insulin resistance, which helps to resolve hormonal and metabolic disturbances and increases ovulation, pregnancy, and live birth rates relative to placebo. A combination of metformin with clomifene citrate (another widely used treatment for PCOS) is more effective than either alone and is a useful option in women with clomifene-resistant PCOS. Combining metformin with letrozole (1st-line agent for ovulation induction in women with PCOS and no other infertility risk factors) is not more effective than letrozole alone. Continuing metformin to the end of the 1st trimester at an effective dose (e.g. 1000–2000 mg/day) may help to reduce the rate of miscarriages. Metformin also has an adjunctive role in women with PCOS receiving assisted reproduction technology (ART) using the long gonadotrophin-releasing hormone agonist protocol, where it appears to increase the pregnancy rate and to reduce the risk of ovarian hyperstimulation syndrome. There is no role for metformin in women receiving short ART protocols. Where a successful pregnancy is achieved, metformin is generally safe for the mother and neonate. Further research is needed to define with greater precision the optimal dosage and times to initiate and discontinue metformin in women with PCOS who achieve pregnancy.
Plain language summary
Medical guidelines support prescribing metformin for some women with polycystic ovary syndrome (PCOS) although yet relatively few countries have an official clinical indication for this to support prescribing. The action of metformin involves improving the action of insulin, which helps to resolve hormonal and metabolic problems and increases the changes of successful ovulation, pregnancy and live births, especially when given together with clomifene citrate. Adding metformin to letrozole (another commonly used drug in this area) does not increase the effectiveness of letrozole alone. Continuing metformin to the end of the 1st trimester at an effective dose may help to reduce the rate of miscarriages. Metformin may also be useful for women with PCOS receiving one type of assisted reproduction technology (ART; long protocol), where it appears to increase the pregnancy rate and to reduce the risk of ovarian hyperstimulation syndrome. Where a successful pregnancy is achieved, metformin is generally safe for the mother and child. Further research is needed to define with greater precision the most effective dosage of metformin and when to start and stop treatment for women with PCOS who achieve pregnancy.
Introduction
Polycystic ovary syndrome (PCOS) is one of the most common hormonal conditions and the largest cause of anovulatory infertility affecting women of reproductive age. 1 As many as one woman in eight worldwide has PCOS, with up to 70% of cases undiagnosed. 2 Some ethnic groups bear a high burden of PCOS and its complications (described below). 2 PCOS is the underlying cause of anovulation in >90% of women attending infertility clinics. In addition, PCOS is associated with elevations of cardiometabolic risk factors, dysglycaemia, dyslipidaemia and obstructive sleep apnoea, with increased long-term risk of prediabetes, type 2 diabetes (T2D), gestational diabetes and cardiovascular disease.3 –7 PCOS also brings an approximately 3-fold elevation in the risk of developing metabolic dysfunction-associated fatty liver disease and associated long-term adverse clinical outcomes, including liver failure and cardiovascular events.8,9 Women with PCOS may display a range of distressing symptoms, including overweight/obesity, dysmenorrhoea, hyperandrogenism (which may include male pattern hair loss, acne, oily skin and hirsutism1,2), which promote issues with negative body image. 10
Table 1 summarises the key properties of three treatments that are used commonly in PCOS management: metformin, clomifene citrate and letrozole.11 –15 Increased insulin resistance within the pathology of PCOS (especially with comorbid obesity) contributes to clinical sequelae of PCOS (Figure 1).16,17 Briefly, insulin increases the frequency and amplitude of gonadotrophin-releasing hormone (GnRH) release, thus increasing the secretion of luteinizing hormone (LH) by the pituitary, with a consequent increase in androgen production by the ovaries. 16 In the ovaries, insulin acts together with LH and IGF-1 to increase the activity of cytochrome P450-17-alpha, a key enzyme in the biosynthesis of androgens. 16 In the adrenal glands, insulin resistance and hyperinsulinaemia may be associated with an increase in androgens due to the increased sensitivity of the adrenal glands to ACTH. 16 Thus, insulin resistance drives adverse hormonal and metabolic function in PCOS, suggesting a role for treatments that improve insulin signalling. 18 Metformin reduces blood glucose in T2D by enhancing the action of insulin, mainly through activation of the adenosine monophosphate activated protein kinase (AMPK) pathway and in key organs such as the liver, muscle and gut.11,19 The therapeutic use of metformin in women with PCOS has received support from major guidelines for use alone or alongside clomifene citrate in women with PCOS.20 –22
Overview of commonly used treatments for PCOS.

Information presented here is abstracted from the summaries of product characteristics of these drugs, available at https://www.medicines.org.uk/emc.
Abbreviation: PCOS, polycystic ovary syndrome.

Overview of the interactions between insulin resistance, hyperinsulinaemia and environmental factors in the pathogenesis of PCOS and its associated adverse impact on metabolism in muscle, fat and liver (left) and the hormonal mechanisms by which PCOS promotes infertility (right).
This article provides an update on the therapeutic use of metformin in women with PCOS. We describe the current evidence base for the effects of metformin on key aspects of the pathophysiology of PCOS and then focus on the effects of metformin on clinical fertility and pregnancy outcomes, and discuss its current role in the management of PCOS, alone or alongside other therapeutic agents.
Review search methodology
A literature search (EMBASE/PubMed) on 01 May 2023, focused on ‘metformin’, ‘PCOS’, ‘ovulation’ and ‘pregnancy’ (460 publications); other publications came from weekly literature alerts from EMBASE, PubMed and Google Scholar. About 900 references were screened for relevance and inclusion. Articles for citation here were ranked according to their design, with meta-analyses given the highest priority, followed by human randomised controlled trials and then observational studies in humans. These classifications also identify the grading of evidence presented below.
Metformin in PCOS – potential therapeutic mechanisms
Insulin resistance and accompanying hyperinsulinaemia drive the development of hyperandrogenism through reductions in the production of sex hormone-binding globulin (SHBG) in the liver and by inducing an imbalance of the hypothalamic-pituitary-ovary (HPO) and hypothalamic-pituitary-adrenal (HPA) axes (Figure 1). This occurs through actions on the brain, the pituitary, the ovary and the adrenal gland (associated with increased release of GnRH, LH, androgen hormones and adrenocorticotropic hormone (ACTH), respectively). 16 These adverse consequences of insulin resistance result in the reduced fertility associated with PCOS. Elevated levels of androgen hormones may also exacerbate insulin resistance by inhibiting the recruitment of GLUT4 glucose transporters by insulin: this impairs the ability of insulin to drive glucose uptake. 23 The insulin-resistant state also favours the development and maintenance of excess adiposity, with an increased risk of developing T2D and atherosclerotic cardiovascular disease. 24
Metformin reduces insulin resistance in women with PCOS, 21 opposing these adverse metabolic and hormonal phenomena (Figure 2), associated with an improvement in GnRH pulsatility, with a drop in LH and androgen production, in addition to an increase in sex hormone binding globulin (SHBG). 25 Reduction of hyperinsulinaemia during metformin treatment 21 improves the function of the HPO and HPA axes and opposes the increased production of androgen hormones characteristic of PCOS.25,26 Meanwhile, increased expression of SHBG and insulin-like growth factor binding protein-1 (IGFBP-1) during treatment with metformin reduces the circulating levels of free, active androgens and IGF-1, respectively.25,26 Normalisation of the LH level and the LH:FSH ratio in this way is perhaps the most prominent therapeutic action of metformin on fertility in women with PCOS. 25 Insulin resistance and hyperinsulinaemia are commonly observed features of obesity. However, clinical studies have concluded that metformin is effective in ameliorating the metabolic consequences of PCOS irrespective of obesity status, 27 although a larger effect of metformin has been observed in obese women28,29 or in non-obese women who demonstrate insulin resistance and hyperinsulinaemia. 30
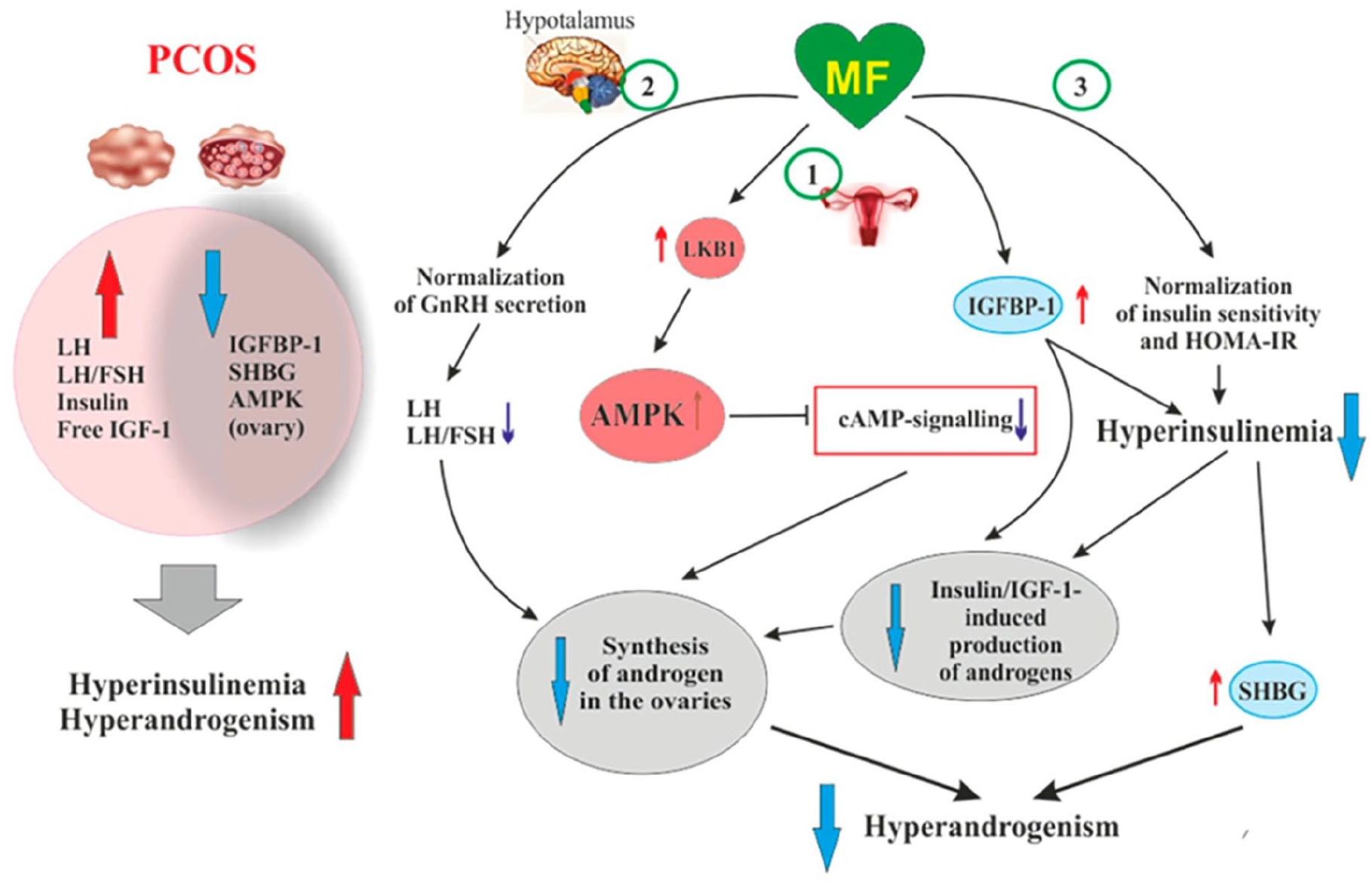
Potential cellular therapeutic mechanisms of MF in the setting of PCOS. Left: summary of metabolic abnormalities in the overlay in PCOS. Right: actions in the central nervous system (CNS) contribute to the amelioration of disturbances of the hypothalamic-ovarian axis, while increased signalling via the AMPK pathway and amelioration of insulin resistance opposes the development of hyperandrogenism, a key facet of the pathogenesis of PCOS.
The cellular actions of metformin arise largely via increased flux through the AMPK pathway (Figure 2), which also accounts for most of the actions of metformin on blood glucose in the setting of prediabetes and T2D. 31 A range of other cellular processes affected by metformin have been described and may also contribute, including reduction of systemic, low-grade inflammation. 26 This is consistent with observations that insulin resistance is an inflammatory state. 26 The effects of metformin on the clinical presentation of women with PCOS are described in the following sections. A summary of each subsection is provided in Table 2, which also provides a synthesis of the actions of metformin.
Summary of effects of metformin in women with PCOS.

ART: assisted reproduction technologies; GnRH: gonadotrophin-releasing hormone; HOMA-IR: homeostasis model assessment for insulin resistance; PCOS, polycystic ovary syndrome. See text for references.
Clinical actions of metformin in women with PCOS
Hormone levels
Treatment of women with PCOS with metformin (compared with non-metformin-treated control subjects) has been shown to reduce the level of total testosterone and androstenedione in meta-analyses published in 202321,32 and in a 10-year observational study in a population of 159 overweight/obese women with PCOS. 33 Metformin also reduced the free androgen index (FAI), a diagnostic measure of hyperandrogenism, 34 in the meta-analysis described above 28 and in a randomised, 6-month, placebo-controlled trial (the PPCOS study). 35 Clomifene citrate (with or without metformin) was more effective than metformin alone in reducing FAI in this study. 35 However, total testosterone was reduced significantly only in metformin-containing groups. 31 Meta-analyses demonstrated reduced insulin resistance (HOMA-IR) and reduced fasting insulin in women with PCOS women treated with metformin, consistent with the hypothesised mechanisms described above.36,37
Menstrual function
Irregular menstruation or amenorrhoea are classic symptoms of PCOS, associated with insulin resistance, hyperandrogenism and anovulation.38,39 Indeed, many women with PCOS never achieve normal cyclicity.38,39 Cochrane meta-analyses have shown that metformin increases menstrual frequency versus placebo, while metformin was inferior compared with combined oral contraceptives.40 –43 These hormonal treatments may, however, adversely affect some cardiovascular disease risk factors and have been associated with increased risk of insulin resistance. Metformin was as effective as other medications used in diabetes for normalising menstrual function in women with PCOS in a network meta-analysis. 44 Observational data also support a role for the use of metformin for normalising cyclicity in PCOS during treatment periods of up to 10 years.33,45 –48 One observational study was conducted in women with PCOS who did not demonstrate insulin resistance. 47 This suggested that improvements in menstrual function on metformin may not depend completely on an insulin-sensitising effect of metformin.
Ovulation
Metformin versus no metformin or clomifene citrate
The meta-analysis embedded within the 2023 guidelines for PCOS management reported a strong trend towards increased ovulation rate with metformin versus placebo, with no difference for metformin versus clomifene citrate (Figure 3). 21 Metformin plus clomifene citrate was more effective than clomifene citrate alone, but not metformin alone in this analysis (Figure 3).

Comparisons of effects on important fertility outcomes between metformin-containing regimens and selected other common treatments for PCOS from a meta-analysis commissioned for 2023 internal guidelines for the management of PCOS.
A Cochrane review from 2012 that included 44 clinical trials found that metformin improved ovulation rates in women with PCOS when given alone (odds ratio (OR) versus no metformin 1.81 (95% CI: 1.13–2.93)) or with clomifene citrate (OR versus clomifene citrate alone 1.74 (95% CI: 1.50–2.00)). 41 Later (2017 42 and 2019 43 ) Cochrane meta-analyses also demonstrated a significant effect of metformin on ovulation rates per woman: ORs were 2.55 (95% CI: 1.81–3.59) and 2.64 (95% CI: 1.85–3.75) for metformin versus no metformin, respectively, and 1.57 (95% CI: 1.28–1.92) and 1.65 (95% CI: 1.35–2.03) for metformin plus citrate versus clomifene citrate, respectively. The 2019 Cochrane analysis found no influence of obesity status on ovulation rates, although the 2017 analysis suggested a larger effect of metformin in non-obese women.42,43
A network meta-analysis of 54 studies (2017) compared the effects of several first-line therapies for PCOS on ovulation rates and other parameters. 48 Metformin increased ovulation rate versus placebo (OR 4.06 (2.02–8.19)) in the network analysis, although not in an accompanying pairwise meta-analysis (OR 3.63 (95% CI: 0.45–29.35)). Metformin alone was less effective than clomifene citrate alone (OR 0.57 (95% CI: 0.36, 0.92)). Clomifene citrate plus metformin was superior to metformin alone (OR 2.50 (95% CI: 1.43–4.36)) and similarly effective versus clomifene citrate alone (OR 1.43 (95% CI: 0.92–2.22)) in the network meta-analysis. A further meta-analysis that restricted analysis to randomised, controlled trials with a low risk of bias also found that ovulation rates were lower for metformin alone versus clomifene citrate alone in women with PCOS. 21
Metformin was less effective than clomifene citrate for induction of ovulation in the randomised PPCOS Study (6-month evaluation of metformin, clomifene citrate and both in combination in 626 infertile women with PCOS); the mean absolute difference in ovulation rate was 20.0% (95% CI: 9.1–30.9). Similarly, metformin plus clomifene citrate was more effective than metformin alone (mean absolute difference 31.4% (95% CI: 24.7–38.0)) and clomifene citrate alone (mean absolute difference 11.4% (95% CI: 4.2–18.4)). 35 No comparison between metformin and a non-clomifene control group was available from this study. Another randomised, placebo-controlled trial evaluated the effect of 3 months of metformin in 200 women of reproductive age with PCOS. 49 Ovulation occurred in 86% of the metformin group and in 20% of the placebo group (p = 0.003).
Metformin (± clomifene citrate) versus letrozole
Letrozole is recognised as a 1st-line agent for ovulation induction in women with PCOS and no other infertility risk factors. The meta-analysis within the 2023 international guideline reported that combinations of metformin plus letrozole were more effective on ovulation than metformin plus clomifene citrate or clomifene citrate alone (Figure 3). However, adding metformin to letrozole did not improve the ovulation rate versus letrozole alone, as the 95% CI for this comparison crossed unity (Figure 3). 21 Metformin was less effective for inducing ovulation than letrozole in the network meta-analysis described above (OR 0.29 (95% CI: 0.15–0.51)), although the efficacy of letrozole and metformin plus clomifene citrate were similar (OR 0.71 (95% CI: 0.41–1.26)). 48
The rate of ovulation with letrozole monotherapy (82%) was about twice that seen with metformin plus clomifene citrate (43%) in a randomised trial in 202 women with clomifene-resistant PCOS. 50 Another randomised trial, in 150 infertile women with PCOS who did not ovulate in response to letrozole, involved retreatment with this agent after 3 months of metformin treatment: the proportion with normal ovarian function tended to be higher in women with (66%) versus without (34%) metformin treatment. 51 A post hoc analysis of data from a randomised trial showed that concurrent treatment with metformin did not alter the dose of clomifene citrate necessary to induce ovulation. 52 Finally, the combination of letrozole plus metformin was more effective than metformin alone (88.9% versus 69.4%) in inducing ovulation in an observational study of 72 infertile women with PCOS. 53
Clinical pregnancy rates
Metformin versus no metformin or clomifene citrate
The three Cochrane reviews in this area found consistently that metformin approximately doubled the pregnancy rate in women with PCOS with ORs of 1.93–2.31 (all were statistically significant) when compared with placebo or no treatment.41 –43 Metformin was more effective than clomifene citrate in non-obese women (OR 1.56 (95% CI: 1.06–2.29)), while the opposite was observed in the obese subgroup (OR 0.34 (95% CI: 0.21–0.55)). 43 The meta-analysis within the 2023 PCOS guideline reported a similar benefit for metformin versus placebo (Figure 3), 21 as did the network meta-analysis (versus no metformin as the control group; OR 3.59 (95% CI: 1.97–6.56)). 48 Another meta-analysis from 2011 found similar pregnancy rates with metformin or clomifene citrate alone in women with BMI ⩽32 kg/m2. 54 Metformin plus clomifene citrate was more effective than clomifene citrate alone for increasing clinical pregnancy rates in the 2019 Cochrane analysis (OR 1.62 (95% CI: 1.32–1.99)), and in the analysis within the 2023 PCOS guideline (Figure 3), 21 consistent with the data on ovulation rates presented above. 44 The latter analysis found no significant differences between metformin and clomifene citrate, or between clomifene citrate plus metformin and metformin (Figure 3). 21
Clomifene citrate plus metformin was more effective than either clomifene citrate alone (OR 1.71 (95% CI: 1.28–2.27)) or metformin alone (OR 1.64 (95% CI: 1.12–2.40)) for increasing pregnancy rates in the network meta-analysis. 44 This was true only for metformin plus clomifene citrate versus clomifene citrate in the meta-analysis within the 2023 PCOS guideline. 21 Two additional recent (2023) meta-analyses of 27 trials 55 and 12 trials 56 also reported a significant increase in the pregnancy rate with metformin. The second of these (reported in abstract form at the time of writing) found that the clinical pregnancy rate was higher if women with PCOS took metformin to the end of the first trimester, rather than discontinuing immediately after a positive pregnancy test. 56
A randomised trial in 320 women with PCOS involved treatment with metformin for 12 weeks followed by the addition of another fertility treatment if required. 57 Metformin-based treatment increased pregnancy rates significantly (hazard ratio (HR) 1.60 (95% CI: 1.13–2.27)); after stratification for BMI with a significant effect seen only in the obese subgroup, in contrast to the findings of the Cochrane analysis described above. It should be noted that women with obesity received a higher metformin dose (2000 mg/day) than their non-obese counterparts (1500 mg/day). A small randomised trial reported that neither clomifene citrate nor metformin impacted pregnancy rates markedly and that the pregnancy rate was low in obese women whatever strategy was used. 58
The PPCOS study reported a higher pregnancy rate in women randomised to metformin plus clomifene citrate versus metformin alone (mean absolute difference 22.4% (95% CI: 15.0–29.8)) but not versus clomifene citrate alone (mean absolute difference 7.2% (95% CI: −1.3 to 15.7)); consistent with these data, clomifene was more effective on the pregnancy rate than metformin alone (mean absolute difference 15.2% (95% CI: 8.3–22.1)). 35 Another randomised, placebo-controlled comparison of metformin with clomifene citrate in 92 non-obese women with PCOS found that the pregnancy rate on metformin was higher during the 6th monthly cycle (33%) than in the 1st cycle (7%), the opposite was true for clomifene citrate (13% versus 0%). 59 A follow-up randomised trial from the same group in which metformin was maintained for 6 months irrespective of whether patients ovulated demonstrated equivalent effects of metformin and clomifene citrate on the pregnancy rate, leading the authors to recommend that metformin treatment should be maintained for this time in women with PCOS. 60
Metformin (± clomifene citrate) versus letrozole
Metformin plus letrozole was more effective for increasing clinical pregnancy rates than clomifene citrate with or without metformin, but not versus letrozole alone in the meta-analysis within the 2023 PCOS guideline (Figure 3). 21 The efficacy of metformin plus clomifene citrate was not significantly different from that of letrozole alone (Figure 3). 21 In the network meta-analysis, metformin was less effective on pregnancy rates than letrozole (0.62 (95% CI: 0.41–0.92), with no difference between letrozole and metformin plus clomifene citrate (OR 1.01 (0.70–1.46). 48 A randomised trial demonstrated higher pregnancy rates for letrozole (36%) versus metformin plus clomifene citrate (10%, p < 0.001), 50 although pregnancy rates were similar among a population of 292 anovulatory women with PCOS who received letrozole or clomifene citrate plus metformin within another 3-month randomised trial. 61 Another randomised trial in 100 women with PCOS showed that adding metformin to letrozole increased the pregnancy rate versus letrozole alone, 62 although the meta-analysis did not support a difference in the clinical pregnancy rate between metformin plus letrozole and letrozole alone (Figure 3). 21 Similarly, the pregnancy rate was higher (p = 0.029) on metformin plus letrozole (86%) versus metformin alone (64%) in an observational study of 72 women with PCOS. 53
Live births and early pregnancy loss
Metformin versus no metformin or clomifene citrate
The 2017 42 and 2019 43 Cochrane reviews reported an increased live birth rate with metformin versus no metformin (OR 1.59 (95% CI: 1.00–2.51) for both), as did the meta-analysis within the 2023 PCOS guideline (Figure 3). 21 The earlier Cochrane review 42 reported a comparable OR (1.80 (95% CI: 0.52–6.16)) which did not achieve statistical significance. Additionally, the 2019 Cochrane analysis found no effect of obesity status on the live birth rate. 43 Additionally, metformin plus clomifene citrate did not improve the live birth or miscarriage rate versus clomifene citrate alone in the 2019 Cochrane analysis or the meta-analysis within the 2023 PCOS guideline (Figure 3).21,43 The 2017 network meta-analysis found no significant effect of metformin compared with placebo/no treatment or clomifene citrate on live birth or miscarriage rates; there were also no significant differences for metformin plus clomifene citrate versus placebo (OR 4.66 (95% CI: 0.59–36.68)) metformin alone (OR 1.45 (95% CI: 0.88–2.41)) or clomifene citrate alone (OR 1.62 (95% CI: 0.91–2.91)). 48
Metformin did not influence miscarriage rates relative to the other treatments studied in these analyses, other than a marginally significantly increased rate for metformin versus clomifene citrate in the analysis within the 2023 PCOS guideline (Figure 3). 21 Other data from women with PCOS suggest reduced risk of miscarriage with metformin versus placebo/no treatment, including another recent meta-analysis (17 randomised trials, 2022; RR 0.40 (95% CI: 0.20–0.78) in favour of metformin) 63 and in a randomised trial in 76 women who did or did not receive metformin until the end of the first trimester (10.8% versus 42.2%, respectively, p < 0.05). 64 Meta-analyses from 2023 (12 studies) 56 and 2015 (11 studies 65 and 5 studies 66 ) reported reduced rates of miscarriage in women with PCOS who continued metformin after confirmation of the pregnancy. Observational data reported a higher rate of miscarriage (36.3%) among 80 women with PCOS who discontinued metformin early in the pregnancy, compared with 11.6% (p < 0.00001) among 120 women who maintained metformin 1000–2000 mg/day throughout the pregnancy. 67
The results of individual randomised comparisons of metformin and clomifene citrate on the live birth rate have been conflicting. The PPCOS study reported live birth rates of 23% on clomifene citrate and 7% on metformin, 35 while an earlier randomised trial reported live births for 56% of pregnancies on clomifene citrate and 84% on metformin. 59
Metformin (with or without clomifene citrate) versus letrozole
Metformin alone was inferior to letrozole for the live birth rate in the network meta-analysis (OR 0.54 (95% CI: 0.29–0.98)), while there was no significant difference between the effects of clomifene citrate and metformin (OR 0.98 (95% CI: 0.37–2.57)). 48 No comparisons involving letrozole demonstrated a significant effect on miscarriages in the meta-analysis within the 2023 PCOS guideline (Figure 3). 21 There were no significant differences in live birth rates or miscarriages in the randomised trial in 268 women with PCOS for comparisons between letrozole and clomifene citrate ± metformin, or between letrozole and letrozole plus metformin. 61
Body weight
Excess adiposity is a key driver of increased insulin resistance; more than half of women with PCOS are obese and difficulty losing weight is a major source of concern and distress for these women, along with irregular cyclicity and infertility.68,69 The meta-analysis conducted for the 2023 guideline for the management of PCOS concluded that metformin reduced BMI and waist-hip ratio, compared with placebo.21,23 A model-based meta-analysis of 28 randomised trials predicted a maximum effect of metformin on BMI of −5.88% (monotherapy) and −11.8% (combination therapy), with a dosage range of metformin 1000–2000 mg/day. 70 Another meta-analysis of 33 studies in populations with PCOS found that the reduction of BMI on metformin was larger versus combined oral contraceptives with or without antiandrogen drugs. 71 Metformin was also associated with weight loss in the PPCOS study (where it was more effective than clomifene citrate) 31 and in observational studies.33,46,72
Psychological well-being and quality of life
PCOS is associated with a heavy burden of debilitating symptoms and infertility that promote a substantial level of psychological distress for many women.69,73 A recent systematic review (33 randomised or observational studies, 2023) found that the burden of reduced health-related quality of life (HRQoL) measured using validated instruments in women with PCOS rivaled or exceeded that associated with heart disease, diabetes, or breast cancer. 73 This review also found that most treatments for PCOS improved HRQoL. 73 PCOS has been associated significantly in observational studies with increased risk of serious adverse neuropsychological outcomes such as major depression, 74 bipolar disorder 75 or schizophrenia 76 ; in each case, treatment with metformin was associated with reduced risk of developing the condition.
Metformin in women with PCOS undergoing assisted reproduction technologies (ART)
The use of metformin during ART is relatively common, as shown by an international web-based survey of 44 countries in which 10.4% of 5260 subjects reported that they took metformin during ART. The delivery of ART commonly employs one of two protocols. Briefly, the ‘short protocol’ occurs entirely within one cycle and may involve FSH administration from day 2 of the menstrual cycle, sometimes followed by administration of a GnRH antagonist, followed by an injection of human chorionic gonadotrophin (hCG) when the ovarian follicles are suitably developed, with oocyte collection occurring 1.5 days later. 77 The ‘long protocol’ typically involves GnRH agonist treatment from the mid-luteal phase of the preceding cycle to suppress endogenous ovarian function with a period of FSH administration leading up to one week before the stimulating hCG injection is given in the following cycle. The long protocol is more commonly used, although the short protocol may carry less risk of ovarian hyperstimulation syndrome (OHSS) and is more suitable for selected women. There is no firm evidence for superiority in general of either protocol over the other for supporting a subsequent pregnancy. 78
In women receiving a long-GnRH agonist protocol, metformin increased the clinical pregnancy rate (RR 1.32 (95% CI: 1.08–1.63)), but did not increase live birth rates (RR 1.30 (95% CI: 0.94–1.79)), according to a Cochrane meta-analysis of six randomised trials from 2020. 79 Only two randomised trials involving administration of metformin to women using the short-GnRh antagonist protocol for ART were available for meta-analysis by the Cochrane Collaboration in 2020; metformin exerted no significant effect on the pregnancy rate (relative risk (RR) 1.38 (95% CI: 0.21–9.14)). 79 Another recent meta-analysis (35 randomised trials, 2022) which did not stratify patients for the type of ART protocol received reported similar findings to the Cochrane analysis for women receiving the long ART protocol (increased pregnancy rate, no effect on live birth rate and a reduction in the rate of OHSS). 80 A randomised comparison of metformin versus placebo, which involved treatment for at least 12 weeks with metformin 2000 mg/day did report an increase in the live birth rate (48.6% versus 32.0%; p = 0.04), in addition to an increased pregnancy rate (50.0% versus 33.3%; p = 0.04). 81
Only one randomised trial (in 153 women with infertility related to PCOS) evaluated metformin in women receiving the short protocol; the live birth rate was significantly lower in the metformin versus placebo groups (RR 0.48 (95% CI: 0.29–0.79)). 77 Although this study involved a very short period of administration of metformin (from 7 days before a natural menstrual period or the first day of an induced period to the day before oocyte collection), there is no clinical evidence base to support the therapeutic use of metformin in women receiving ART via the short protocol.
Metformin did not significantly influence the miscarriage rate versus placebo in the Cochrane analysis after pooling eight randomised trials irrespective of the ART protocol used (RR 0.86 (95% CI: 0.56–1.32)). 79 The rate of OHSS was reduced significantly with metformin versus control (11 randomised trials; RR 0.46 (95% CI: 0.29–0.72)). 79 Another meta-analysis published in 2022 with 40 randomised trials involving women at high risk of OHSS with or without PCOS, for which the main outcome was moderate-to-severe OHSS, reported only a trend to benefit for metformin versus placebo (RR 0.4 (95% CI: 0.12–1.23). 82
Safety of metformin in relation to its use in women with PCOS
Importance of evaluating the safety of metformin during pregnancy
It is inevitable that some pregnancies will be exposed to metformin, as some women with PCOS take metformin with the intention of increasing their chances of becoming pregnant. Moreover, some evidence discussed above suggests that the optimal administration of metformin in a woman with PCOS may involve treatment at least until the end of the 1st trimester of pregnancy. Establishing the safety of metformin during pregnancy is thus crucial in relation to the potential use of metformin within the management of PCOS.
Safety of metformin in women with PCOS who become pregnant
Pregnancy loss and preterm delivery
Meta-analytic or randomised trial evidence from studies in populations with PCOS, described above, showed that treatment with metformin did not affect the rate of miscarriage/early pregnancy loss, especially when treatment with metformin was not stopped at the time of discovery of the pregnancy.43,56,63,65,67 The meta-analysis conducted for the 2023 management guideline reported an OR of 0.48 (95% CI: 0.14–1.62) for miscarriage for metformin versus placebo, as per above. 21 There was a benefit for CC alone versus metformin alone (OR 2.44 (1.03–5.82)), but again no difference for metformin plus versus metformin plus CC (OR 0.63 (0.30–1.31)). 21
Treatment with metformin reduced the risk of preterm delivery according to meta-analyses of randomised trials from 2022 (RR 0.42 (95% CI: 0.25–0.71)) 59 and 2016 (OR 0.38 (95% CI: 0.16–0.90)). 65 The PREGMET2 study involved randomisation of 487 pregnant women with PCOS to metformin or placebo. 83 The primary endpoint (a composite of late miscarriage (13–22 weeks) and preterm delivery (24–36 weeks)) was not met (OR 0·50 (95% CI: 0.22–1.08), p = 0.08). The effect of metformin became significant after post hoc pooling of these data with earlier data from this group (N = 790 women; OR 0·43 (95% CI: 0.23–0.79), p = 0.004), although further subgroup analyses revealed a significant effect only in obese women. A large case-control study from Taiwan (N = 9735) confirmed the increased risk of preterm delivery in women with PCOS and reported that this risk was reduced by about 10% with metformin treatment. 84 Another database study from Sweden, analysed all 1,016,805 singleton births in that country between 2006 and 2016. 85 Again, the higher risk of preterm delivery in women with PCOS was observed; metformin essentially abolished this excess risk, which became similar to that seen in women without PCOS.
Preeclampsia and gestational hypertension
Metformin did not influence the risk of preeclampsia or gestational hypertension in a meta-analysis of studies in women with PCOS. 65 A randomised study described an increased risk of pre-eclampsia after stopping metformin at confirmed conception versus continuing metformin through the pregnancy (RR 2.65 (95% CI: 1.95–3.59)). 86 The large database study from Sweden found that the risk of preeclampsia increased in women with versus without PCOS who were not taking metformin; treatment with metformin effectively abolished the excess risk of preeclampsia associated with PCOS. 85 Treatment with metformin also reduced the risk of preeclampsia in women without PCOS. 85
Women with PCOS and gestational diabetes (GDM)
Meta-analyses have found a reduced risk of GDM in women with PCOS receiving versus not receiving metformin,63,65 although the evidence base is currently confined to analyses of observational studies and not randomised trials. 65 Another meta-analysis suggested a stronger effect of metformin on the prevention of GDM in women with versus without confirmed PCOS, and when metformin was started before conception versus during pregnancy. 87 The large database study from Sweden found a surprising 3.9-fold increased risk of GDM associated with any use of metformin during pregnancy, 83 which probably arose from confounding by reverse causality, as metformin is used commonly 88 and increasingly 89 within the management of GDM. Observational data suggest that continuing metformin through the pregnancy may reduce the risk of GDM in the setting of PCOS.90,91 Metformin reduced the risk of subsequent T2D in women with impaired glucose tolerance and a prior history of GDM in the randomised Diabetes Prevention Program. 92
Maternal pregnancy outcomes
Metformin has been shown to limit weight gain in mothers with PCOS during pregnancy, with no evidence of adverse effects. 83 In Sweden, treatment with metformin appeared to negate the excess risk of Caesarean section associated with PCOS, as it did for other adverse outcomes summarised above. 85 There is no evidence for other significant safety concerns associated with the use of metformin during pregnancy in populations without PCOS. Interestingly, reduced gestational weight gain and a reduction in the risk of Caesarean section have also been noted in the general population of pregnant women receiving versus not receiving metformin.93,94
Neonatal outcomes
An increased head circumference has been observed in neonates born to mothers with PCOS who received versus did not receive metformin, with no evidence of differences in other anthropometric indices.83,95,96 Observational data suggested that offspring born to metformin-treated mothers with PCOS were not at increased risk of low birth weight/small for gestational age (or preterm birth), and no increased risk of macrosomia, large for gestational age (LGA) or 5-min Apgar score <7, highlighting that metformin can be considered safe throughout the complete gestation. 85
Safety of metformin in populations of pregnant women not defined by the presence of PCOS
The Metformin In women with T2D in pregnancY (MiTy) study involved randomisation of 502 insulin-treated pregnant women (6 to <23 weeks’ gestation) with T2D to additional metformin 2000 mg/day or placebo. 97 There was no significant difference in the primary composite of foetal and neonatal outcomes (40% in either group, p = 0.86). Women randomised to metformin versus placebo achieved better glycaemic control at 34 weeks’ gestation (average difference mean glucose –0·2 mmol/L) despite needing less insulin (difference –0·4 U, p < 0·0001), gained less weight (difference –1·8 kg, p < 0·0001) and had fewer caesarean births (53% versus 63%, p = 0·031). Metformin-exposed neonates were smaller on average (difference –218 g, p = 0·002), and 13% in the metformin group and 7% in the placebo group were small for gestational age (p = 0·026). Neonatal outcomes were secondary endpoints in the EMERGE trial, which randomised 510 women with GDM to metformin or placebo initiated during or before the 29th week of the pregnancy. 98 Neonates in metformin-treated mothers were less likely to weigh >4 kg at birth (7.6% versus 14.8%, p = 0.02)) or to be >90th percentile for birth weight (6.5% versus 14.9%, p = 0.003), and had smaller crown-heel length on average (51.0 cm versus 51.7 cm, p = 0.02), all versus placebo-treated mothers. There were no significant differences between groups for birth weight <2.5 kg (6.1% versus 3.4%, p = 0.12), neonatal intensive care admissions (15.6% versus 12.5%, p = 0.37), respiratory distress requiring respiratory support (9.2% versus 6.8%, p = 0.41), major congenital anomalies (3.8% versus 2.7%, p = 0.62), neonatal hypoglycaemia (13.7% versus 12.9%, p = 0.88), or jaundice requiring phototherapy (one case occurred, in the metformin group). Maternal weight gain was also lower on metformin versus placebo (0.8 kg versus 2.0 kg, p = 0.03), with no significant differences between groups for other maternal outcomes. The Metformin in Gestational Diabetes (MiG) study involved randomisation of 751 women with GDM to insulin or metformin (with supplemental insulin as required); 93% of the metformin group continued treatment until delivery. 99 The incidence of the primary composite foetal/neonatal outcome was 32% in each group. Secondary endpoints (neonatal anthropometric measurements, maternal glycaemic control, maternal hypertensive complications, and postpartum glucose tolerance) also did not differ between groups.
Follow-up of the MiG study (MiG-TOFU) revealed that children born to mothers who received metformin were larger on average at 9 years of age compared with children of women previously randomised to insulin. 100 However, there were no differences in total or regional body fat percentages, indices of glycaemia, insulin, insulin resistance, lipids, liver transaminases, leptin or adiponectin. In contrast, follow-up of all children born in the MiTY study demonstrated no significant differences in anthropometric indices at 2 years of age. 101
A post hoc study of the Finnish observation study CLUE (Consequences for life of children with in-utero exposure to metformin in Finland – a register-based cohort study) found that children exposed to metformin in utero did not have an increased risk of small or large for gestational age birth compared with children born to mothers with a GDM diagnosis but not treated medically.102,103 A meta-analysis of 21 randomised trials (2022) broadly confirmed these findings relating to maternal weight gain and birth weight, for pregnancies complicated by GDM or T2D. 104 Additionally, treatment with metformin versus insulin was associated with less gestational hypertension (RR 0.63 (95% CI: 0.48–0.82)), maternal hypoglycaemia (RR 0.33 (95% CI: 0.15–0.73)), neonatal hypoglycaemia (RR 0.56 (95% CI: 0.49–0.64)) and neonatal intensive care unit admission (RR 0.73 (95% CI: 0.64–0.83)).
General tolerability and safety of metformin
The principal side effects of metformin occur in the gastrointestinal tract (mainly diarrhoea and abdominal pain).11,12 These tend to occur early during treatment and can be minimised by cautious titration of the metformin dose (‘start low, go slow’). 105 The prolonged-release formulation (metformin XR or SR, depending on the country) is as effective as the immediate-release formulation but is better tolerated 106 : a switch to the XR/SR formulation has allowed some previously metformin-intolerant people with T2D to resume treatment with metformin. 107 In addition, long-term treatment with metformin in people with T2D has been associated with vitamin B12 deficiency which can be corrected with a B12 supplement. 108
Metformin-associated lactic acidosis (MaLA) is a very rare but life-threatening adverse event, with an estimated incidence of 3–9 cases/100,000 patient-years of exposure to metformin. 109 Severe renal dysfunction (GFR <30 mL/min/1.73 m2) conveys an increased risk of MaLA.110,111 Indeed, this severity of renal dysfunction is an absolute contraindication for the therapeutic use of metformin, along with other situations that may decrease renal function acutely (e.g. severe infection, shock, administration of contrast media) or limited tissue perfusion (e.g. acute cardiac insufficiency). While everyone who receives metformin should be screened for contraindications, these are less likely to occur in younger populations such as women of childbearing age.
What the guideline says
This section summarises guidance relating to the therapeutic use of metformin from the 2023 international guideline for the management of PCOS. 21 Recommendations on the use of metformin alone focus mainly on the control of menses and cardiometabolic factors. Letrozole is the 1st-line pharmacologic intervention to improve ovulatory function in women with PCOS who want to become pregnant and have no other risk factors for infertility, according to this guidance. The use of metformin in combination with clomifene citrate to improve ovulation and pregnancy rates is emphasised, consistent with the evidence summarised above. Box 1 summarises the principal recommendations relating to the use of metformin for women with PCOS in this guideline.
Key points relating to the use of metformin from the 2023 international guideline for the management of polycystic ovary syndrome (PCOS).
Metformin monotherapy may be appropriate for:
• Adults with PCOS and BMI ⩾25 kg/m2 for anthropometric and metabolic outcomesa
• Adolescents with or at risk of PCOS for cycle regulationa,b
• Considered for adults with PCOS and BMI <25 kg/m2 b,c
• Consider COCP before metformin for control of hirsutism in women with PCOS and irregular mensesd
• Consider metformin before COCP for control of irregular menses where COCP treatment is not possible (other interventions will be needed to address hirsutism)a,d
• Consider metformin before COCP for control of cardiometabolic risk factorsa
• Metformin plus COCP may be most suitable for women with PCOS and BMI >30 kg/m2 with additional prediabetes or diabetes risk factorsd
• If metformin alone is used for a woman with PCOS and anovulatory infertility but no other risk factors for infertility, women should be counseled that more effective approaches to treat anovulation are availablea
Combined use of metformin and other drugs:
• Metformin plus COCP is not more effective than either alone for adults with PCOS and BMI <30 kg/m2 a
• Prescribe clomifene citrate before metformin to improve ovulation and chance of a successful pregnancy for women with anovulatory PCOS and no other risk factors for infertility (consider both agents together rather than either alone for these women)a
• Counsel women on the risk of multiple pregnancies when prescribing clomifene citrate (± metformin)d
• Note that the monitoring requirements for clomifene citrate plus metformin are the same as for clomifene citrate aloned
• Consider gonadotrophin before clomifene citrate plus metformin for clomifene-resistant women with anovulatory PCOS and no other risk factors for infertility (laparoscopic ovarian surgery is a less effective option for these women)a
Metformin and assisted reproduction technology (ART) in women with PCOS
• Metformin may be used as adjunctive therapy before and/or after ovarian stimulation during long protocol ART to reduce the risk of OHSS and miscarriagea
• For long protocol ART, start metformin at the start of GnRH treatment and continue to a successful pregnancy test or continue beyond this if indicatedd
• Titrate metformin carefully (to optimise gastrointestinal tolerability) to a dose of 1000–2500 mg/day
Other recommendations relating to metformin:
• Consider metformin where appropriate to reduce the risk of preterm delivery and to limit excess gestational weight gain, in pregnant women with PCOSa
• Counsel women that the consequences of metformin exposure on long-term offspring health remain unclear and there is a suggestion of increased childhood weight, although causality is not certaind
• Consider metformin for ameliorating the adverse psychological impact of PCOS (also consider hair removal, lifestyle interventions, COCP)d
• Consider metformin before inositol for hirsutism and central adiposityd
• Where a therapeutic indication for the use of metformin or other drugs PCOS is not available, healthcare professionals should counsel women on the therapeutic properties and risk: benefit considerations relating to the chosen therapyd
• Brief women on the gastrointestinal side-effects of metformin and the fact that the overall healthcare burden (including cost, monitoring and need for clinic visits) are lower with metformin than some other optionsd
• Discuss age and other risk factors for fertility before prescribing metformind
aEvidence-based recommendation. bSupported by limited evidence. cConsensus recommendation (expert opinion on drug actions). dPractice point (expert opinion on delivery of care). Recommendations have been paraphrased for conciseness: refer to the full guideline for further details. COCP: combined oral contraceptive pill. OHSS: ovarian hyperstimulation syndrome. Abstracted from information presented in reference 21.
It is important to stress that all women with PCOS should be encouraged to undertake lifestyle improvements (improved diet and more physical activity), which is similar to recommendations for the management of other conditions involving increased cardiometabolic risk, such as T2D or cardiovascular disease. 112 The 2023 guideline notes that lifestyle intervention and metformin have similar efficacy in the management of PCOS, although it should be remembered that the ability and willingness to undertake long-term lifestyle interventions varies greatly between individuals. Other recommendations involve optimising the tolerability of metformin, which is discussed above. Also, women should be counselled on the lack of evidence that metformin prevents the onset of GDM in pregnant women with PCOS, and that treatment with metformin does not prevent late miscarriages (after 12–22 weeks), hypertension/preeclampsia, and macrosomia (the extent of the evidence base for effects on these parameters is also discussed above).
Discussion
Influential guidelines support a role for metformin in the management of PCOS. The latest guideline, with input from experts in 71 countries, confirmed and expended the place of metformin in PCOS care. 21 Paradoxically, relatively few national and international product labels provide support and recognition for the use of metformin in women with PCOS. Providing an indication is important for facilitating access to treatments for patients, depending on their local healthcare and reimbursement systems.
Many clinical studies have been performed in this area over decades, and consideration of the evidence base for pharmacologic interventions in women with PCOS relies heavily on meta-analyses of studies with differing populations and methodologies. Accordingly, the optimal deployment of metformin to optimise fertility in women with PCOS is still controversial. Letrozole is probably more effective than metformin in increasing ovulation and pregnancy rates when given as monotherapy, but further studies are needed to establish whether combinations of metformin + letrozole are more effective than letrozole alone, as suggested by the meta-analyses described above. Giving a treatment that increases the antihyperglycaemic response to insulin is a rational strategy for women with severe insulin resistance and marked hyperinsulinaemia. In addition, the observation in a randomised trial, described above, that a longer period of treatment with metformin was associated with markedly increased pregnancy rates, while the opposite occurs with clomifene citrate, 59 raises important questions about trials that involve repeated challenges with clomifene citrate during background treatment with metformin. First, the remaining trial participants who do not become pregnant during the early cycles of clomifene citrate may be enriched with women who are clomifene resistant. Also, the dose of metformin requires careful titration to minimise the occurrence of gastrointestinal side effects, so that optimal efficacy with this agent will not be achieved early in such trials. It appears that a sufficient intensity and duration of treatment with metformin (e.g. at least 6 months (or up to 12 weeks gestation) even when ovulation has occurred before this) may optimise the response to metformin in women with PCOS. 60 Continued treatment with metformin appeared to reduce the rate of miscarriage in meta-analyses and randomised trials.56,65 –67,82
Limitations
The main limitation of our review is its narrative structure (it is not a formal systematic review). However, we have provided details of our search methodology, and how we selected articles for inclusion here. The large size of this field, and the lack of randomised, controlled trials in this area, meant that we necessarily had to concentrate to a large extent on meta-analyses, which tend to carry forward any faults within their included data.
Conclusions
Metformin involves amelioration of insulin resistance, which helps to resolve hormonal and metabolic disturbances and increases ovulation, pregnancy and live birth rates, including in women with PCOS. A combination of metformin with clomifene citrate is a useful option in women with clomifene-resistant PCOS, although adding metformin to letrozole does not appear to bring additional efficacy for fertility outcomes. Continuing metformin to the end of the 1st trimester at an effective dose (e.g. 1000–2000 mg/day) may help to reduce the rate of miscarriages. However, the optimal protocol for starting, maintaining and withdrawing metformin after the establishment of a pregnancy has yet to be established. Metformin also has an adjunctive role in women with PCOS receiving assisted reproduction technology (ART) using the long (but not the short) gonadotrophin-releasing hormone agonist protocol. Metformin is generally safe for the mother and neonate.
