Abstract
Metformin is quite an old drug, but it is optimal for the control of glycemia in Type 2 diabetes. It was reported, 15 years ago, that insulin resistance was abnormally high in most polycystic ovary syndrome (PCOS) patients. Starting from that moment, increasing numbers of studies were performed to demonstrate the efficacy of metformin in controlling and/or modulating several aspects of PCOS, which is the most common cause of menstrual irregularity, inesthetisms and infertility. Metformin induces higher glucose uptake, thus inducing a lower synthesis/secretion of insulin. Such an effect permits the possible restoration of the normal biological functions that are severely affected by the compensatory hyperinsulinemia reactive to the increased peripheral insulin resistance. These are the basis of the many positive effects of this drug, such as the restoration of menstrual cyclicity, ovulatory cycles and fertility, because abnormal insulin levels affect the hypothalamus-pituitary-ovarian function, as well as the use of glucose in peripheral tissues. Metformin improves the impairments typically observed in hyperinsulinemic PCOS patients, reducing the possible evolution towards metabolic syndrome and Type 2 diabetes; and when pregnancy occurs, it consistently reduces the risk of gestational diabetes, eclampsia and hypertension. PCOS seems to be the perfect physiopathological condition that might have higher benefits from metformin administration, obviously after Type 2 diabetes. This review focuses on the many aspects of PCOS and on the possible issues of this disease for which metformin might be a putative optimal treatment.
Keywords
Metformin is quite an old compound introduced into clinical use in Europe in 1957 and only in 1995 in the USA. Chemically, it is 1,1-dimethyl-biguanide hydrochloride, a biguanide currently used as an oral antihyperglycemic agent for diabetes mellitus. More or less 15 years ago it was reported for the first time that use of metformin in a group of obese patients with polycystic ovary syndrome (PCOS) was able to reduce insulin resistance [1]. Moreover, within 6 months, such treatment significantly reduced circulating androgen levels and body weight, together with the occurrence of more regular menstrual cyclicity and ovulation [1]. From then on, increasing evidence clearly demonstrated the importance of the use of metformin, not only in the presence of diabetes mellitus Type 2, but also in patients with PCOS and hyperinsulinemia, resolving several issues such as menstrual cyclicity, fertility, hormonal levels and metabolic syndrome (MS).
To better understand the clinical significance of the use of metformin, a brief review is needed to elucidate why the linkage between PCOS and this biguanide is so tight and so clinically relevant for women's health.
PCOS characteristics
Polycystic ovary syndrome is a very peculiar disease and is one of the most frequent endocrine disorders in women. This condition occurs in as many as 8–10% of women of reproductive age [2], with onset manifesting as early as puberty [3]. For many years there has been no agreement on the criteria on which to base the diagnosis of PCOS. This was probably a consequence of the heterogeneity of the syndrome, but also depended on the absence of clear pathogenetic mechanism(s) [4].
From the very beginning, diagnostic criteria proposed by the NIH for PCOS were the presence of hyperandrogenism and chronic anovulation with clear exclusion of related ovulatory or other androgen excess disorders (i.e., hyperprolactinemia, thyroid diseases, androgen-secreting tumors and adrenal dysfunction/hyperplasia) [5]. These criteria did not include the presence of polycystic ovaries at ultrasound examination because it was observed that polycystic ovaries could also be present in healthy eumenorrheic women [6].
A few years later, during the European Society of Human reproduction and Embryology (ESHRE)/American Society for reproductive Medicine (ASRM) conference, the diagnostic criteria were expanded and PCOS was considered as present when at least two of three features were diagnosed (Box 1): oligo or anovulation, clinical/biochemical hyperandrogenism and polycystic ovaries as assessed by ultrasound examination [6]. This evolution was relevant because it permitted the inclusion of women with PCOS who were excluded by previous NIH criteria [5]: those with polycystic ovaries affected by hyperandrogenism and ovulatory cycles, or chronic anovulation and normal androgen levels.
Polycystic ovary syndrome diagnostic features.
The European Society of Human reproduction and Embryology/American Society for reproductive Medicine conference stated that the diagnostic criteria were expanded and polycystic ovary syndrome was considered to be present when at least two of the three features below were present:
– Oligo or anovulation
– Clinical/biochemical hyperandrogenism
– Polycystic ovaries as assessed by ultrasound examination
More recently, the Androgen Excess and PCOS Society indicated that PCOS should always be considered an androgen excess disorder and concluded that PCOS was, above all, a disorder of androgen biosynthesis, utilization and/or metabolism in women [7].
Despite the diagnostic criteria, PCOS is still an unclear disease in terms of pathogenesis, and although the main clinical features related to the syndrome are menstrual irregularities, anovulation and clinical/biochemical signs of hyperandrogenism, PCOS patients frequently show other physiopathological characteristics.
As Palomba et al. recently stated, the reduced fertility rate that PCOS patients show cannot be ascribed to chronic anovulation alone [8], and other factors are probably responsible for the low fertility in these women, compromising oocyte quality, embryo evolution and/or implantation abnormalities [9]. Moreover, PCOS patients seem to be more exposed than the normal population to miscarriage risk, gestational diabetes, hypertension or preeclampsia [9].
Recently it has been made clear that both genetic and environmental factors may contribute to the onset of PCOS features [8,10]. Indeed, the prevalence of PCOS cases among patients’ first-degree relatives presents a statistically significant increase, as compared with several populations, which reveals a possible genetic predisposition of the syndrome [10]. In fact, several studies demonstrated that several genes are implicated in the pathogenesis of PCOS, and are categorized according to the pathophysiologic mechanism that each one interferes with (Table 1). On such genetic predisposition, environmental factors may play a key role, such as peculiar lifestyle, types of food, living conditions and also the impact during the intrauterine growth [10]. In fact, if intrauterine growth is characterized by an exposure to exaggerated androgen levels, this peculiar situation may lead to the development of hyperandrogenism and ovarian dysfunction later in life [10].
Genes involved in polycystic ovary syndrome.

Endocrine profile of PCOS patients
Polycystic ovary syndrome is characterized by higher plasma concentrations of ovarian and adrenal androgens, increased luteinizing hormone (LH) levels, high estrogen levels (especially estrone) due to extraglandular conversion from androgens, lower levels of sex hormone-binding globulin (SHBG) and higher levels of prolactin and insulin, the latter often in presence of overweight or obesity.
Although the pathogenesis of PCOS is still controversial [11–13], PCOS typically shows elevated LH and normal or relatively low follicular-stimulating hormone (FSH) secretion [14]. In fact, almost 50–60% of PCOS patients show a high LH:FSH ratio (>2.5) [11,12,15], an exaggerated LH response to gonadotrophin-releasing hormone (GnRH) stimulation test [11,12,16] and a higher frequency of LH pulsatile release from pituitary [11,12,14] that induces a higher stimulation on theca cells and an excess of androgen secretion as well as impaired follicular development [17,18].
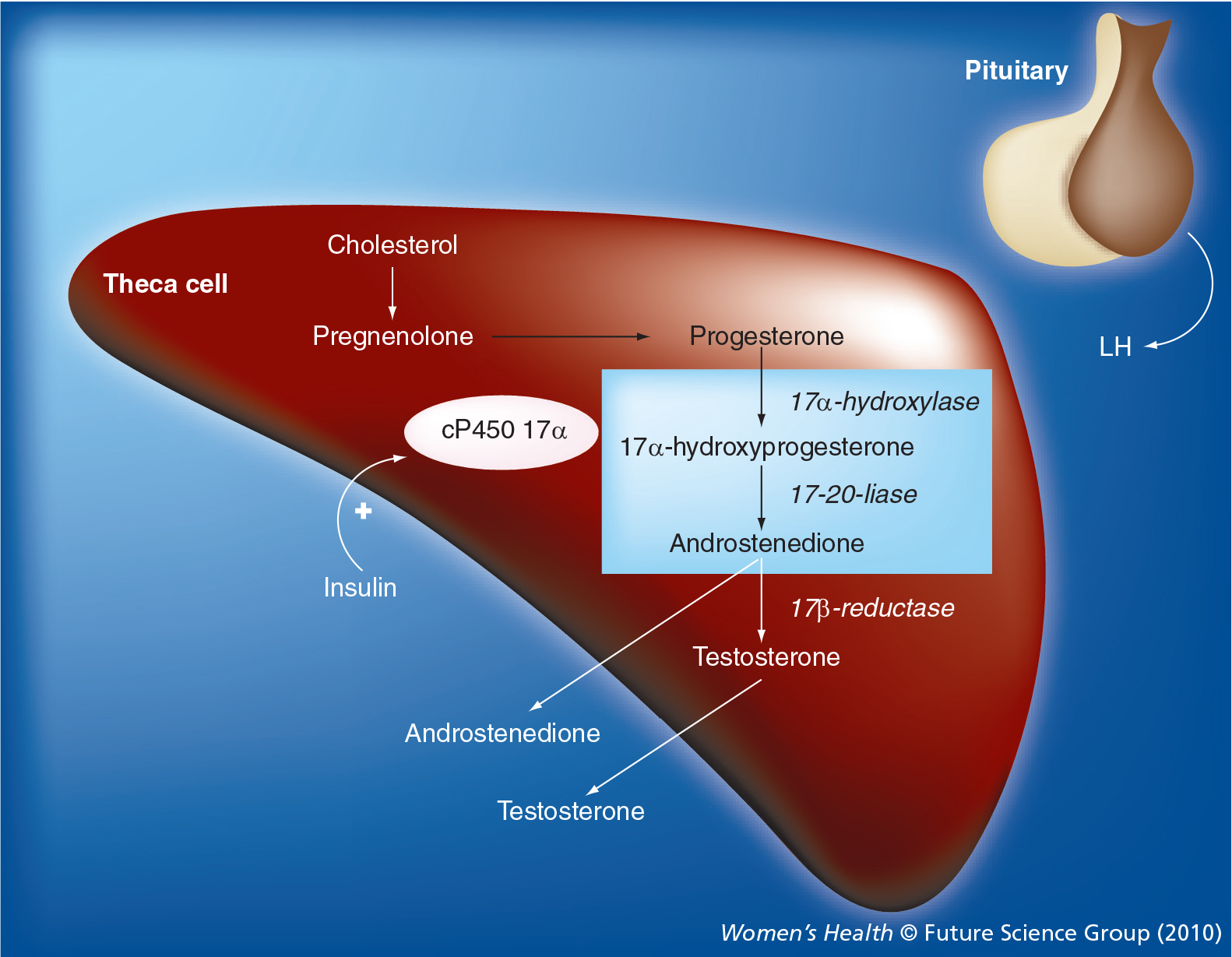
Hyperandrogenism is a key feature of the syndrome, although it is not constant [11]. It is mainly of ovarian origin with an adrenal contribution, since a certain percentage of PCOS patients might show a mild steroidogenetic defect in adrenal glands (such as for 21-hydroxylase) or just a higher adrenal hyperactivation due to stress [17]. Androstenedione and testosterone are the best markers of ovarian androgen secretion, while dehydroepiandrosterone sulfate (DHEAS) is the best marker of adrenal secretion. Most testosterone is derived from peripheral conversion of androstenedione and from direct ovarian production. In addition, the adrenal glands contribute in part to testosterone production in women [18], although in hyperandrogenic PCOS the main source of androgens is usually from the ovaries. Dysregulation of cytochrome p450c17, the androgen-forming enzyme in both the adrenal glands and the ovaries, is the central pathogenic mechanism underlying hyperandrogenism in PCOS (Figure 1) [13,19]. In the presence of 5α-reductase, testosterone is converted within the cell to the more potent androgen dihydrotestosterone. Excess or normal 5α-reductase activity in the skin determines the presence or absence of hirsutism [20]. Additionally, estrone plasma levels, a weak estrogen with biological activity 100-times less than estradiol, are increased as a result of peripheral conversion of androstenedione by aromatase activity – more active in PCOS than in healthy controls – while estradiol levels are normal or low because of the frequent anovulatory cycles. All this results in a chronic hyperestrogenic state with the reversal of the estrone:estradiol ratio that might predispose to endometrial proliferation and to a possible increased risk for endometrial cancer [19,21].
Normally, less than 3% of testosterone circulates as unbound in the serum. In fact, most circulating androgens are bound to SHBG, thus being biologically inactive. Any condition that decreases the levels of SHBG or other binding proteins can lead to a relative excess of free circulating androgens. In PCOS, hirsutism usually occurs with decreased SHBG levels and obesity [18,20].
Androgens may both directly and indirectly induce alterations in glucose metabolism, ultimately being an additional cause of abnormal insulin sensitivity. Androgens may directly inhibit peripheral and hepatic insulin action. In fact, testosterone could induce insulin resistance in women with PCOS acting on the post-binding signal, in particular by reducing the number and efficiency of glucose transport proteins, such as the type 4 glucose transporter (GLUT-4), especially in muscle and fat tissues [22]. In addition, it has also been reported that women with central obesity, typical of obese PCOS, have higher free androgen levels and exhibit significantly higher levels of insulin insensitivity compared with weight-matched controls [23]. Moreover, androgens and increased free fatty acids, commonly observed in central obesity, inhibit hepatic insulin excretion and insulin-stimulated glucose uptake in skeletal muscle, resulting in hyperinsulinemia and insulin resistance [24–26]. A great percentage of PCOS patients show overweight up to severe obesity, and typically any excess of weight can induce a reduction of peripheral tissues sensitivity to insulin, thus inducing the compensatory hyperinsulinism.
It is relevant to say that hyperinsulinemia may be central to the pathogenesis of the syndrome in many cases, because it can induce higher ovarian androgen production and anovulation [27,28], sustained also by the abnormal LH secretion, with a higher frequency of menstrual abnormalities than in normoinsulinemic women with PCOS (Figure 2) [29,30]. Insulin resistance and compensatory hyperinsulinemia are metabolic disturbances easily observable in at least 45–65% of PCOS patients, and frequently appear to be related to excessive serine phosphorylation of the insulin receptor [13,31].


Insulin actions on the main effectors of androgen production.
Metabolism & PCOS
In PCOS patients, there is an increased risk of developing Type 2 diabetes and coronary heart disease (CHD) [32–34]. Such risk has also been demonstrated to be higher in postmenopausal women, previously demonstrated to be PCOS during fertile life [35,36]. PCOS has been reported to have an increased risk of MS, which refers to a clustering within the same individual of hyperinsulinemia, mild-to-severe glucose intolerance, dislipidemia and hypertension, and an increased risk for cardiovascular disease (CVD) and diabetes [37–39]. All of them are CHD risk factors [39,40], and when observed in PCOS, a significantly higher odds ratio for the development of various cardiovascular risk factors occurs [36,41]. Moreover, PCOS patients have a significantly greater risk of MS when compared with controls [42]. In 2006, the International Diabetes Federation defined the features of the MS, and defined central obesity as present when the waist circumference is above 80 cm; in European women, this was considered as a necessary prerequisite risk factor for the diagnosis of MS [201]. However, it is of great relevance to point out that although the MS has been identified for more than 80 years, only in these last years has controversy about its definition emerged [37].
The risk factors for MS are:
Waist circumference is over 80 cm
Elevated triglycerides (≥1.7 mmol/l)
Reduced HDL (<1.29 mmol/l in women)
Specific treatment for lipid abnormalities
Elevated blood pressure (systolic ≥ 130 mmHb or diastolic ≥ 85 mmHg)
Specific treatment or precedent diagnosis of hypertension
Fasting plasma glucose at least 5.6 mmol/l
Previous diagnosis of Type 2 diabetes mellitus
The prevalence of MS in polycystic women is approximately 40–45% [43], and the main predictor factors are the elevated free serum testosterone and reduced serum SHBG level [44]. A significant inverse relationship between SHBG levels and the occurrence of MS in women with PCOS was also demonstrated recently [45]. The association of MS with PCOS appears to be particularly strong in those PCOS women who are young (<30 years) and overweight or obese (BMI > 27 kg/m2) [42].
Women with PCOS have lower HDL levels, higher LDL:HDL ratios and higher triglyceride levels than healthy eumenorrheic women [33]. All these are inductors of subclinical atherosclerosis as demonstrated by the increased thickness of the carotid intima media and by the higher endothelial dysfunction observed in PCOS patients [46,47], probably related to the insulin resistance and/or to the higher free testosterone plasma level [48,49].
Indeed, several studies reported an increased risk factor profile for cardiovascular disease in women with PCOS [50], in particular a decreased cardiac systolic flow velocity, diastolic dysfunction, increased vascular stiffness, low-grade chronic inflammation, increased oxidative stress, altered hemostasis including impaired fibrinolysis and increased tissue plasminogen activator antigen [50]. It is relevant to point out that very recent data demonstrated that postmenopausal women with a premenopausal history of irregular menses and hyperandrogenism (i.e., PCOS) have more angiographic coronary artery disease compared with non-PCOS women [35], thus providing evidence that identification of PCOS and its treatment may provide an opportunity to reduce CVD risk factors [35].
It is of great relevance the fact that women with PCOS have an increased risk for impaired glucose tolerance and Type 2 diabetes mellitus [51,52], with a tendency to an early development of glucose intolerance state [53,54]. In fact, the decrease of insulin sensitivity in PCOS women appears to be quite similar to that observed in patients with Type 2 diabetes mellitus and to be relatively independent from obesity, fat distribution and lean body mass [1]. On the other hand, there is strong evidence that obesity, particularly the abdominal phenotype, represents an important independent risk factor for glucose intolerance in PCOS women [52].
Rationale of metformin use in PCOS women
The logic for the use of insulin sensitizer drugs, such as metformin, to treat patients with PCOS is the fact that a large percentage of PCOS patients have been demonstrated to have insulin resistance and a compensatory hyperinsulinemia that negatively affect ovarian function in terms of steroid biosynthesis and follicular recruitment and maturation [8–9,55].
In fact, the presence of insulin resistance and the compensatory hyperinsulinemia are reported in at least 45–65% of PCOS patients [8,56], and this percentage is significantly higher than age- and BMI-matched healthy controls [57]. Interestingly, a higher basal secretion of insulin has been described in PCOS patients before as well as after weight loss, despite the demonstration of the perfect normalization of the response to the oral glucose tolerance test (OGTT) [58]. This observation suggests that in PCOS, independently from obesity, body fat distribution, androgen plasma levels and the reduction of insulin sensitivity might be related with an abnormal transduction of the signal induced by the insulinreceptor binding [53]. Obviously, when insulin resistance is present independently from obesity, whatever the weight gain that might occur, it certainly exaggerates insulin resistance and more severely alters the glucose metabolism and, later on, the hormonal profile.
It is important to remember that several studies, although not all [59], reported that insulin modulates LH secretion from pituitary cells in vitro [53] and in vivo (Figure 3), and that the reduction of hyperinsulinemia induces the significant decrease of LH plasma levels [51,60,61], although it is not clear whether decreased LH levels are due to the reduced insulin levels or secondary to the recovery of ovarian function (i.e., estrogen production) induced by decreased insulin plasma levels [61].
Other than ovarian function, insulin resistance affects several other organs and tissues. At the hepatic level, hyperinsulinemia decreases SHBG synthesis, thus increasing circulating free androgens, at the muscular level it alters the mitochondrial oxidative metabolism [62], and at ovarian levels hyperinsulinemia induces anovulation, follicular growth blockade and hyperandrogenism [8]. Excess insulin increases androgen concentrations blocking follicular maturation and increasing cytochrome P450c17a activity, a key enzyme in the synthesis of both ovarian and adrenal androgens [8,53]. This situation typically increases 17-hydroxyprogesterone (17OHP), androstenedione and testosterone plasma levels. The excess of intraovarian androgens negatively modulates follicular function and ovarian activity, thus inducing the typical stromal hypertrophy and maintaining ovarian atresia and anovulation [8,63].
When abnormal insulin sensitivity is diagnosed, the use of metformin might be suggested [8,64]. Metformin reduces hepatic glucose production from 9 to 30% and the action is exerted enhancing the suppression of gluconeogenesis and reducing the glucagon-induced gluconeogenesis, but specific effects on short and long-term gene expression effects cannot be excluded [8,65]. On peripheral tissues, such as muscle cells and adipocytes, metformin acts by increasing glucose uptake through the glucose transport system.
Patients with PCOS are insulin resistant and this condition typically decreases insulin's ability to stimulate glucose disposal into peripheral cells and decreases the glucose response (i.e., glucose uptake by the cells) to insulin [8]. In addition, metformin has been reported to act on insulin receptors, enhancing its response, thus inducing the reduction of both the synthesis of insulin and proinsulin in patients with Type 2 diabetes [65–67]. Other than the above-described effects on insulin, metformin positively acts on hormonal PCOS abnormalities through a direct and/or indirect action on steroidogenesis [8]. In fact, the recovery of normal ovulatory function is probably due to the direct modulation of metformin on the ovarian tissues and to the metformin-induced normalization of the ovarian steroidogenesis (lowering androgen production), thus determining the normal feedback on pituitary, lowering LH secretion and LH pulse characteristics [60,61]. Metformin improves steroidogenesis not only at the ovarian, but also at the adrenal level, since insulin plays specific modulatory roles on these two distinct endocrine glands that have the same enzymatic pathways [65,68]. In fact, it has been demonstrated that metformin administration ameliorates adrenal enzyme activities in PCOS patients [59].
It is important to remember that although many authors reported the significant reduction of the hyperandrogenic condition in PCOS patients, some others state that such an effect is less evident or totally lacking. A recent meta-analysis of the published studies demonstrated that the use of insulin sensitizers do not reduce hyperandrogenism better than oral contraceptives [69], but as recently reported, the typology of PCOS to be treated is of great relevance, since only when insulin sensitivity is abnormal does metformin show a greater efficacy on all the PCOS features, including hyperandrogenism [61]. Obviously, it cannot be excluded that other metabolically active hormones (e.g., leptin, resistin, adiponectin and gherelin) are positively activated by metformin administration and thus participate in the improvement of the reproductive function at the hypothalamus–pituitary–ovarian level [70].
In conclusion, data clearly show that metformin effectiveness on reproductive and on metabolic parameters is mainly exerted in association with a reduction of circulating insulin levels, thus supporting the hypothesis that a high insulin level is one of the main effectors/modulators of the clinical and endocrine dysfunctions of PCOS [8,12].
Metformin pharmacokinetics, side-effects & therapeutic regimens
Metformin has been shown to have a rather complex absorption [71]. It is adsorbed only in part at the gastrointestinal tract (20–30%), with the highest amount absorbed within 6 h from administration, and the amount absorbed is greater for low doses (500 mg) rather than for high doses, such as 1.5 g [71]. No binding of metformin with plasma proteins was reported [71]. The elimination is quite slow and this allows a relatively stable metformin disposal rate [71]. Since metformin is not metabolized, it is eliminated by the kidney with a mean half-life of 4–8 h [62].
The therapeutic dose of metformin cannot be standardized. Since its main indication is to treat diabetes, most of the dosages have been set according to the levels of glycemia achieved with the treatment, and the amount of metformin administered may vary from 500 to 1500 mg or more per day [8]. On the contrary, an extremely variable dosage has been used to treat PCOS, but up to now, no dose-finding study is available for PCOS, probably because there are various end points and goals for PCOS patients to reach, such as the recovery of menstrual cyclicity and of ovulation, loss of weight, reduction of hirsutism and skin defects. It is, however, important to point out that clinical studies clearly demonstrated that after longterm metformin treatment, drug suspension induces a quick reversion of the beneficial effects on peripheral insulin sensitivity [8,72]. In addition, because recent data showed that better clinical results were obtained in insulin-resistant rather than in non-insulin-resistant PCOS patients [61], a clear adjustment of the dose of metformin must be performed according to the BMI and insulin resistance. The treatment must be started with a low dose, administered few minutes before lunch and dinner (10–15 min before), because an empty stomach minimizes the possible drug-related side affects [8], and only after 3–7 days can it be increased, slowly, up to the most effective dosage for the patient, as reported by Nestler [73,74].
The known side-effects, although limited in incidence, are abdominal discomfort, constipation, diarrhea, flatulence, heartburn, indigestion, nausea and vomiting [8,75]. Metformin is a well-tolerated drug and the only relatively frequent side-effects are those related to gastrointestinal diseases (7.9–22.2%), such as diarrhea and/or nausea [75–77], but they usually disappear within a few days [8]. A recent report showed a good safety with an incidence of adverse events of 1% [78]. The rare side effects are uncommon when metformin is used for treating insulin resistance and compensatory hyperinsulinemia in PCOS patients [8,75], while they are more frequent in Type 2 diabetes, especially lactic acidosis [8,79]. In fact, almost all data on lactic acidosis pertained to Type 2 diabetes patients, and according to Palomba et al., it can be assumed that this complication is a rare drug-related side effect because the PCOS patients are generally young and without major medical conditions [8].
The length of metformin treatment in PCOS patients is difficult to standardize because various subjective variables may affect metformin efficacy. The real open question is whether metformin administration has to be considered as a symptomatic treatment or a real curative treatment [8]. The answer is ‘yes’, but the patient needs to help metformin modulation(s) on the metabolic side following all the advices concerning feeding and weight control. The experience of many researchers seems to suggest that metformin positively affects the metabolic problems of hyperinsulinemic, overweight/obese women together with a low caloric diet and a minimum of physical activity. Since metformin modulates insulin sensitivity, it works perfectly to restore insulin sensitivity within the normal ranges, and this usually happens when physical activity and loss of weight also induce the reduction of peripheral tissue insulin resistance together with metformin action [80]. However, it should be noted that previous data demonstrated that withdrawal of metformin treatment can be followed within 3 months by a reversal toward a pretreatment hyperinsulinemic state [72,81,82].
Recent data suggest that metformin is more effective in insulin-resistant PCOS patients, with normal BMI, probably because some constitutional abnormality might be at the basis of the insulin resistance. Nevertheless, overweight (but not necessarily obesity) remains a feature that represents a strong indicator of good results when coupled with insulin resistance [59,60], since a recent report showed equal response to metformin in those who were lean and obese [83]. However, it is interesting to point out that lifestyle changes have been demonstrated to be more effective in preventing diabetes risk and MS than treatment with metformin [84].
Previous data showed that after long-term treatment, metformin suspension induced a certain percentage of reversion of the beneficial effects on insulin sensitivity and on hyperandrogenism, although such a situation was not observed in all patients [72], and is probably related to the simultaneous worsening of both the ovarian function and the menstrual cyclicity [8].
In our opinion, the positive effects induced by metformin administration can be maintained when treatment is prolonged over the time (up to 12 months or more) and it is combined with a controlled diet and with moderate physical activity to aid metabolism and weight loss, especially when overweight or obesity is present. Indeed, Pasquali reported a greater weight loss when treatment is coupled with hypocaloric diet and physical activity [85].
Clinical features that predict metformin efficacy
It is quite clear that metformin may vary in the beneficial efficacy according to the clinical characteristics of the patient, and it is important to know whether these features are directly or indirectly affected by metformin action (Box 2). The more of these features present at the same time, the higher the chance of metformin treatment being effective [86].
BMI
Body weight, evaluated as BMI, is a fundamental characteristic. It is well-known that the higher the amount of fat, the higher the degree of insulin resistance and then of compensatory hyperinsulinism. Unfortunately, this is not a general rule; it is valid for overweight or obese patients only. In fact, several studies reported that a certain percentage of PCOS patients as well as a certain percentage of the normal population have constitutional insulin resistance concomitantly with a lean body mass [8,87,88], and when patients were treated independently from the BMI, all of them demonstrated a beneficial effect in insulin plasma levels, insulin sensitivity and in AUC for insulin under the OGTT [87]. Our data demonstrated a great efficacy of metformin administration in non-obese PCOS patients, mainly in those with abnormal insulin response to OGTT and/or fasting hyperinsulinism [61]. These data let us infer that the ability to be sensitive to insulin is probably a real constitutional aspect; every patient has their own and it is worsened when BMI increases. Indeed a recent meta-analysis demonstrated that in non-diabetic patients with hyperinsulinemia, the response to metformin administration was not different between lean and obese subjects, thus supporting the concept that BMI cannot be considered the only or most stringent predictor criteria [83]. As general advice, PCOS patients with high BMI are considered as optimal to undergo to metformin administration, probably using high dosages (1000–2000 mg/day) to achieve better results [89].
Potential predictors for metformin treatment.
Body weight
Insulin resistance/sensitivity
Androgen plasma levels
Waist-hip ratio
Genetic factors
Insulin sensitivity/resistance
This is probably the most important feature. Because metformin improves insulin sensitivity and induces the decrease of compensatory hyperinsulinemia, it is quite obvious that metformin has more indications for PCOS patients with high basal insulin levels and/or with abnormal response to the OGTT [61]. What is important to point out is that insulin resistance might be present independently from a normal or high BMI, and that only insulin-resistant individuals may benefit from metformin administration in terms of both insulin sensitivity and of insulin-induced metabolic and hormonal functions [8,61].
Androgen excess
In general, hyperandrogenism is a common feature of PCOS patients and cannot be considered a predictor of metformin efficacy. However, since ovarian dysfunction that leads to oligomenorrhea and anovulation is frequently observed in PCOS patients, and metformin administration is able to positively modulate hormonal control of ovarian function [61], it can be argued that the combination of menstrual irregularities, hyperandrogenism and anovulation can be positively affected by metformin administration. Various reports sustained that hyperinsulinism is a main cause of the ovarian function and hyperandrogenism, but it cannot be concluded that metformin can be used to cure hyperandrogenism per se [60,61]. Indeed, recent reviews reported that flutamide and other antiandrogens compounds are more effective than metformin at relieving hyperandrogenism [69].
Waist-to-hip ratio
The presence of overweight or obesity is certainly a risk factor for CVD in PCOS patients, and it is of great importance to state that a high waist-to-hip ratio (>0.8) specifically indicates a greater amount of fat at the abdominal level, which is the typical android distribution [90].
Genetic factors
For sure, environmental factors (such as lifestyle and feeding) deeply condition the occurrence of overweight or obesity as well as of compensatory hyperinsulinemia. However, this cannot be extended to all PCOS patients. Recent data clearly reported that there are several kinds of genetic situations that are the genetic causal triggers or might just be the starting triggers if combined with predisponent environmental factors [8,10]. It is important to observe that ethnic background is probably relevant as these genetic factors, since PCOS and/or mild-to-severe hyperandrogenism are more frequent in some populations than in others, as well as the incidence of metabolic diseases (i.e., diabetes Type 2), quite often mirroring a greater use of carbohydrates in the diet [90].
What metformin can ameliorate
From the clinical point of view it cannot be stated that metformin is the treatment for PCOS patients, but it is rather important to underline that metformin can be of great relevance when two or more of the aforementioned possible predictors are present (Box 1) [8]. From metformin being able to modulate glucose uptake and then insulin response, it is clear that the element that needs to be present is an abnormal insulin response to glucose load and/or hyperinsulinemia in fasting condition. Both indicate the presence of insulin resistance and consequently of a compensatory hyperinsulinism, which can be at the basis of the many hormonal dysregulation and dysfunctions that PCOS patients show. We will now review what metformin would be able to improve.
Effects on menstruation
Polycystic ovary syndrome patients quite frequently complain for menstrual irregularities and it has been reported that almost 80% of them have oligomenorrhea or amenorrhea and oligoovulation or anovulation [91,92]. Metformin administration was reported to restore normal menstrual cyclicity in a high percentage of the patients affected by oligomenorrhea or amenorrhea [59,93,94], but was less effective than oral contraceptives [95,96]. However, the action of metformin is rather complex because it is performed via the reduction of insulin and its negative modulation(s) exerted both on the ovarian microenvironment and on the hypothalamus–pituitary axis, while oral contraceptives improve menstrual cyclicity only through blockade of the reproductive axis [8]. Such an effect induces the consistent reduction of ovarian androgen production with no effect on the impairment induced by compensatory hyperinsulinism, if present.
Effects on fertility
The use of metformin seems to help when anovulatory PCOS patients attempt to get pregnant. Recent reviews and data clearly reported that metformin is not better than clomiphene citrate (CC) to induce ovulation [76,81,97,98], but considering the metformin mechanism of action, it might be beneficial in preparing the patients to a later better response to CC or to other drugs (i.e., gonadotropins) [8]. It is well-documented that the response of PCOS patients to CC or gonadotropin stimulations is different from that of healthy controls (with normal ovaries) [97], and in some cases is responsible for the ovarian hyperstimulation syndrome (OHSS) [99]. It is important to remember that metformin and CC have completely different mechanisms of action, and metformin may act on the reproductive axis as a secondary hormonal effect, mainly related to the improved (i.e., lowered) insulin plasma levels and insulin resistance [8,73,74]. In fact, while metformin acts on insulin pathways, CC acts directly on the reproductive axis. Clinical data suggested that hyperinsulinemia and insulin resistance could be responsible for the ovarian abnormal response occurring in OHSS [100]. Although no clear evidence has been provided, hyperinsulinemic PCOS patients undergoing metformin administration before and/or during ovarian stimulation for the search of spontaneous pregnancy or IVF might benefit from metformin administration. Recent papers and reviews sustain such hypothesis [8,9,101,102], although previous large trials report that metformin did not induce beneficial effects [97,98]. In addition, patients who have a condition of oligo or anovulation, also have endometrial vascularization, pattern and thickness abnormalities [53]. Endometrial pattern is a critical factor influencing the chance of pregnancy in spontaneous as well as in drug-induced (i.e., with CC or gonadotropins) cycles, and a tripleline pattern is associated with a significantly higher pregnancy rate [8,103]. PCOS patients undergoing metformin and with ovulatory cycles show a tripleline endometrial pattern at a rate not different from those observed in healthy controls [52]. Such an effect is probably due to different and concomitant actions: the positive modulation of the metformin-reduced insulin levels on glucose oxidation of endometrial tissues and the recovered optimal function of the hypothalamus–pituitary–ovarian function with a good balance of estradiol and progesterone secretions from granulosa cells and from corpus luteum [8,61].
Effects on hyperandrogenism
Under metformin treatment, a relative improvement might be recorded for hyperandrogenism and its clinical signs, such as acne and hirsutism. The presence of such esthetic disturbances is quite common from mild-to-severe grades in PCOS patients, but it also is highly related to other clinical aspects, such as overweight/obesity and lifestyle, as well as to genetic or ethnic predisposition. In general, hyperandrogenism and its signs are reduced by metformin administration because it reduces ovarian androgen production, ovarian P450c17a activity and free testosterone concentration [8], and within few months it also reduces the Ferriman–Gallway score [60,61].
Effects on body weight
Overweight and obesity are also positively modulated by metformin administration. Indeed weight reduction induced by diet and/or physical activity improves endocrine and metabolic functions, mainly helping the reduction of insulin resistance and compensatory hyperinsulinemia [85,104]. Metformin treatment and lifestyle intervention have been reported to be more effective in reducing body weight, BMI and visceral fat in obese subjects than lifestyle intervention alone [64,85]. This means that metformin cannot be considered a specific antiobesity drug because lifestyle changes and a balanced diet together with physical activity are fundamental to metformin cotreatment and might improve the chances of success [104,105]. However, it is of relevance to point out that metformin has been demonstrated to improve body weight control in obese PCOS patients by acting both directly on the CNS and indirectly via adiponectin modification [106], but in general it has a marginal effect on weight loss as monotherapy, as recently reviewed by Palomba et al., who stated that lifestyle modifications remain the cornerstone for weight loss in obese PCOS patients, although metformin cotreatment might improve the efficacy [8].
Effects on the risk of Type 2 diabetes & CVD
This issue has been partly discussed before, but it is relevant to underline that women with PCOS and insulin resistance have a higher risk for developing Type 2 diabetes [50,80]. The prevalences of insulin resistance and diabetes in PCOS patients, especially if they are obese, is approximately 30–40% and 5–10%, respectively, and this is three-to seven-fold greater than the normal population [80,104]. Since the incidence of cardiovascular diseases is quite high in diabetics, it has to be seriously considered that PCOS patients might also be exposed to a higher risk of CVD and/or its precursors such as hypertension [41,50], since they are at a higher risk for diabetes than the normal population. This fact is enforced by the demonstration that both hyperandrogenism and impaired peripheral insulin sensitivity may increase the risk of CVD [107,108], although prospective controlled data on CVD morbidity and mortality in PCOS patients has never been attently evaluated.
It is quite easy to understand that although metformin is not the panacea omnis malis, it represents a specific treatment to counteract the negative impacts of insulin resistance and obesity on well-being, especially for those women seeking to regain a regular weight, menstrual function and normal glucose metabolism together with a well-balanced lifestyle [8]. Recent studies clearly demonstrated that metformin has a cardioprotective effect on obese diabetic patients decreasing mortality and stroke end points [108,109]. Moreover, it has recently been reported in a murine model [110] and in humans [111] that metformin administration reduces cardiovascular end points in subjects at risk of developing Type 2 diabetes. Such observations seem to infer that metformin might be proposed for long-term therapy to reduce the risk for CVD in euglicemic subjects, such as PCOS patients [8].
Effects on women seeking pregnancy & on risk of miscarriage
All these aspects are of great importance and give clear relevance to the fact that PCOS patients have greater chances to benefit from metformin administration than the normal population, especially if their insulin resistance and glucose tolerance are impaired (see previous ‘Effects on fertility’ section). Such consideration is important, especially when PCOS patients are seeking pregnancy. In fact, being clear that metformin helps increase the chance of becoming pregnant both spontaneously or during induced ovulation/FIVET programs, some epidemiological data reported that PCOS patients have a risk of miscarriage, which has been computed as threefold greater than in normal controls [112,113], although other studies where PCOS patients and controls were matched for BMI did not demonstrate any increased risk of miscarriage [97,114]. Such different reports clearly underline the fact that a lot remains to fully understand PCOS.
Patients with PCOS are likely to develop gestational diabetes in 30–40% of cases [115,117], with a higher chance than normal pregnant women [116]. Various data also demonstrated that metformin use during pregnancy reduced the risk of gestational diabetes through the reduction of preconceptional BMI, fasting insulin levels and insulin resistance [117,118]. Various reports confirmed the efficacy of metformin treatment in PCOS pregnant women reducing significantly the risks of gestational diabetes [118–121] and of preeclampsia [122], although others did not show beneficial effects on miscarriage risk [97]. Although such data are of extremely great clinical relevance, not all of the reports studied large populations or were a placebo-controlled study. In any case, a recent meta-analysis confirmed that there is no difference in abortion risk in PCOS patients undergoing metformin treatment before pregnancy when compared with normal population [123,124]. However, the safety of metformin administration during pregnancy was attently evaluated, and no congenital abnormalities or adverse fetal outcomes were related to metformin [125,126]. Moreover, no negative effects on growth, motor and social development in infants were reported when metformin was administered to PCOS women during the first months of breast feeding [126–128].
Effects on mood & quality of life
Last but not least, one important aspect is the quality of life. PCOS patients have been demonstrated to perceive life significantly worse than healthy controls or other patients affected by other diseases [129]. In general, women are nearly twice as likely to experience major depressive disorders as men [130], but it is of great interest to report that several studies suggest quality of life, body age concerns and sexual dissatisfaction in PCOS [129,131,132], as well as a high degree of occurrence of depressive state [133]. Obesity, clinical signs of hyperandrogenism (acne, hirsutism and alopecia) and infertility seem to be the main elements on which such great psychosocial discomfort find basis [134,135], and when an adequate treatment improved all such features, quality of life and wellbeing improved again [136]. Interestingly, such a positive effect was also observed under metformin administration. Indeed, a significant improvement of quality of life in PCOS patients, especially for the psychological aspects, occurred and interesting correlations were observed between the clinical effects of metformin and the psychological improvement [137]. The reason fo such a positive effect on the impaired psychological trait of PCOS patients seems to be related to the metformin-induced effects on neurosteroid synthesis and secretion, especially allopregnanolone.
Among the components of the neurosteroid family, allopregnanolone, 3a-hydroxy-5a-pregnan-20-one (3a-5a-THP), is actually considered as the most important and clinically relevant [138–145]. It is a ring A-reduced pregnane derivative of progesterone and is synthesized in the gonads, adrenal cortex, CNS and by intraliver metabolism from progesterone [138–145]. Several experimental studies demonstrated the involvement of allopregnanolone in stress, mood and sexual behavior [146–148], and precise correlations have been suggested between brain allopregnanolone and reproductive function [149–151], probably mediated by its interaction with γ-aminobutyric acid A (GABAa) receptors or through an increase in GABAa receptor sensitivity to endogenous GABA [152–154].
Allopregnanolone plasma levels change throughout the menstrual cycle in humans [139,155,156], with levels higher during the luteal phase [138] as well as during pregnancy, especially during the third trimester [152], and lower during postmenopause [138]. Both the adrenal gland and the ovary are main sources of circulating allopregnanolone, as demonstrated by its increase both under GnRH and ACTH stimulating tests [157]. In eumenorrheic women, allopregnanolone has been demonstrated to be temporally coupled with LH pulses only during the luteal phase of the cycle, when progesterone production is higher [157], while such coupling has been observed with cortisol independently from the phase of the menstrual cycle [157]. Stress induces the increase of adrenal production of allopregnanolone both in patients with hypothalamic amenorrhea and in amenorrheic PCOS, since the ovarian production is blunted for the dysfunctional ovarian blockade [158,159], but when nonstressed oligomenorrheic PCOS patients were evaluated, they showed lower allopregnanolone plasma levels than healthy controls [157]. The high frequency of anovulatory cycles and the lack of adequate endogenous allopregnanolone production might be responsible for the aforementioned psychological state and depressive mood. Interestingly, when these PCOS patients were treated with metformin, allopregnanolone plasma levels increased [157]. At the basis of this effect there are the metformin-induced changes on insulin, on the local ovarian and adrenal environment and on the reproductive axis. The increase of allopregnanolone production and synthesis from its precursors and from endocrine tissues [157] might explain the efficacy of metformin treatment on PCOS quality of life [137].
In conclusion, the clinical use of metformin might be suggested in women with PCOS and abnormal insulin sensitivity. Metformin has been reported to induce positive modulation on peripheral tissues, improving metabolism and insulin sensitivity. The reproductive axis benefits the reduction of the insulin resistance since these facts restore a more efficacious recovery of the endocrine function of the ovary as well as of the hypothalamus-pituitary endocrine control. At the same time, metformin-induced insulin sensitivity counteracts the chance of being at risk of Type 2 diabetes and counteracts the negative effects of hyperinsulinism, hyperandrogenism and obesity on body health.
Future perspective
Examining the literature shows us about metformin use, it can be easily argued that this drug has a wide variety of effects, but some areas still have been relatively unexplored, such as the use of metformin in pregnant women. Controlled clinical trials should be promoted to evaluate this point, since pregnancy is one of the main goals of most of the PCOS patients undergoing to metformin administration and more data should be collected on the putative benefits and/or risks that the use of this drug might induce not only in the insulin-resistant mother but also in the fetus. Available data seem to be reassuring, but more must be done before metformin would be safely prescribed during pregnancy.
Another relatively unexplored issue is how much metformin use might be helpful or protective in CVD. Data from diabetic patients are certainly relevant and reassuring, but not all the patients under metformin administration are diabetics, and metformin is usually administered to improve insulin sensitivity in PCOS together with a change in lifestyle. Randomized controlled trials should be carried out to evaluate the long-term effects of metformin treatment on morbidity and mortality in PCOS patients, especially in those who are obese or with clear metabolic impairments.
Last but not least, the important field of scientific study is the putative role of metformin treatment on endometrium and on its biology. This is a rather important topic because the endometrium plays a crucial role in accepting the implantation of the pregnancy at its very beginning, and it should be evaluated how much metformin administration might be helpful in reducing/avoiding the miscarriage risk that PCOS patients have. In addition, randomized controlled trials should be performed to better understand how much metformin might decrease the premalignant endometrial diseases.
Acknowledgements
We are grateful to Dr Stefano Palomba, Department of Obstetrics and Gynecology, University Magna Graecia of Catanzaro, Catanzaro, Italy, to Professor Andrea R Genazzani and Professor Michele Luisi, Department of Obstetrics and Gynecology, University of Pisa, Italy, for the discussions and the advices given during the preparation of the present review.
Executive summary
Polycystic ovary syndrome (PCOS) is one of the most frequent endocrine disorders in women.
PCOS occurs in as many as 8–10% of women of reproductive age and in a higher percentage of oligo or amenorrheic patients.
Oligo or anovulation
Clinical/biochemical hyperandrogenism
Polycystic ovaries at ultrasound examination
PCOS patients frequently show hyperinsulinemia as a compensatory phenomenon related to the reduced insulin sensitivity of peripheral tissues. This condition induces a higher risk of developing Type 2 diabetes and coronary heart disease because PCOS patients have an increased risk of metabolic syndrome.
Better results have been observed in PCOS patients with hyperinsulinism, independently from BMI. In addition to metformin administration, obese/overweight PCOS with hyperinsulinism must also adopt lifestyle interventions to speed up the clinical improvement (i.e., body weight reduction).
The reduction of insulin plasma levels permits the improvement of ovarian function and the reduction of androgen synthesis/secretion, thus facilitating restoration of the ovarian cycle, ovulation and menstrual cyclity. Such effects may allow the patient to become pregnant.
Although increasing evidence demonstrates the relevance of metformin administration in PCOS patients, more studies must be performed to evaluate the potential benefits of metformin administration as a long-term treatment in hyperinsulinemic PCOS patients or during pregnancy.
Metformin is suggested in hyperinsulinemic PCOS patients.
Metformin reduces the risk of Type 2 diabetes, especially in overweight/obese PCOS patients.
Metformin improves reproductive axis function through the reduction of compensatory hyperinsulinism and endocrine impairments.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties. No writing assistance was utilized in the production of this manuscript.
