Abstract
For the last 15 years, there has been a vigorous ongoing debate as to whether or not all conservatively treated patients with ductal carcinoma in situ (DOS) require radiation therapy following excision or whether selected patients with DOS can be treated by excision alone. At just about all breast cancer symposia where DOS is discussed, experts are assigned to debate the pros and cons of radiation therapy after excision. The debate is often heated. This article outlines numerous reasons to consider excision alone in the treatment of selected DOS patients.
Ductal carcinoma in situ (DCIS) of the breast is a heterogeneous group of lesions with diverse malignant potential and a range of treatment options. This article will focus on the issue of whether or not to irradiate every patient who selects breast conservation.
Ductal carcinoma in situ is the most rapidly increasing subgroup in the breast cancer family of disease with more than 62,000 new cases estimated to be diagnosed in the USA during 2008 (26% of all new cases of breast cancer) [1]. Most new cases (>90%) are nonpalpable and discovered mammographically.
There are three forms of local treatment for patients with DCIS: mastectomy, excision plus radiation therapy or excision alone. The nature of the excision can vary widely from a small focused excision to an extensive oncoplastic quadrant resection and to any of these excisions, radiation therapy may or may not be added. This latter issue – whether or not to irradiate – has been the focus of much debate during the last 20 years.
For most patients with DCIS, there will be no single correct treatment. There will generally be a choice. The choices, although seemingly simple, are not. As the choices increase and become more complicated, frustration increases for both the patient and her physician [2,3].
Counseling the patient with biopsy-proven ductal carcinoma in situ
It is never easy to tell a patient that she has breast cancer. But is DCIS really cancer? From a biologic point of view, DCIS is unequivocally cancer. But when we think of cancer, we generally think of a disease that, if untreated, runs an inexorable course toward death. That is certainly not the case with DCIS [4]. We must emphasize to the patient that she has a borderline cancerous lesion, a preinvasive lesion, which at this time is not a threat to her life. In our series of 1363 patients with DCIS through April 2008, the raw mortality rate is 0.5%. Numerous other DCIS series confirm an extremely low mortality rate [5–10].
Patients often ask why is there any mortality rate at all if DCIS is truly a noninvasive lesion? If DCIS recurs as an invasive lesion and the patient dies from metastatic breast cancer, the source of the metastases is clear. But what about the patient who undergoes mastectomy and, sometime later, develops metastatic disease or a patient who is treated with breast preservation who never develops a local invasive recurrence, but still dies of metastatic breast cancer? These latter patients probably had an invasive focus with established metastases at the time of their original treatment, but the invasive focus was never found during routine histopathologic evaluation. No matter how carefully and thoroughly a specimen is examined, it is still a sampling process and a 1–2 mm focus of invasion can be missed.
One of the most frequent concerns expressed by patients once a diagnosis of cancer has been made is the fear that the cancer has spread. We are able to assure patients with DCIS that no invasion was observed microscopically and the likelihood of systemic spread is minimal.
The patient needs to be educated that the term breast cancer encompasses a wide range of lesions of varying degrees of aggressiveness and lethal potential. The patient with DCIS needs to be reassured that she has a minimal lesion and that she is probably going to need some additional treatment, which may include surgery, radiation therapy, an anti-estrogen or some combination of these. She needs reassurance that she will not need chemotherapy, that her hair will not fall out and that it is highly unlikely that she will die from this lesion. She will, of course, need careful clinical follow-up.
End points for patients with DCIS
When evaluating the results of treatment for patients with breast cancer, a variety of end points must be considered. Important end points include local recurrence, both invasive and DCIS, regional recurrence, such as the axilla, distant recurrence, breast cancer-specific survival, overall survival and quality of life. The relative importance of each end point varies depending on whether the patient has DCIS or invasive breast cancer
When treating invasive cancer, the most important end points are distant recurrence and breast cancer-specific survival; in other words, living with or dying from breast cancer. For invasive breast cancer, a variety of different systemic treatments have been demonstrated to significantly improve survival. These include a wide range of chemotherapeutic regimens and endocrine treatments.
Variations in local treatment were thought not to affect survival [11,12]. However, they do effect local recurrence. Recently, literature has demonstrated that for every four local recurrences prevented, one breast cancer death is prevented [13].
Ductal carcinoma in situ is similar to invasive breast cancer in that variations in local treatment affect local recurrence, but no study to date has demonstrated a significant difference in distant disease-free or breast cancer-specific survival, regardless of any treatment (systemic or local), and no study is likely to show a difference since there are so few breast cancer deaths in patients with pure DCIS. The most important outcome measure, breast cancer-specific survival, is essentially the same regardless of which local or systemic treatment is given. Consequently, local recurrence has become the most commonly used end point when evaluating treatment for patients with DCIS.
A meta-analysis of four randomized DCIS trials comparing excision plus radiation therapy versus excision alone was published in 2007. It contained 3665 patients. Radiation therapy increased local control by a statistically significant 60%, but overall survival was slightly worse in the radiation therapy group with a relative risk of 1.08 [9]. These data are dissimilar to those of the Early Breast Cancer Trialist' Collaborative Group, perhaps owing to much shorter follow-up [13]. Half of the recurrences in the DCIS meta-analysis were noninvasive and could not possibly affect survival. Of the remaining invasive recurrences, 80–90% are generally cured by early detection and treatment. This should result in a slight trend toward a lower survival for the excision-alone group, but exactly the opposite was observed: a nonsignificant trend toward a better survival. The authors of the meta-analysis believe that with longer follow-up, the higher local recurrence rate for excision alone will probably result in a lower overall survival. However, for the time being, that has not happened and a detrimental effect secondary to radiation therapy must be considered a possibility.
Local recurrences are clearly important to prevent in patients treated with DCIS. They are demoralizing. They often lead to mastectomy and, theoretically, if invasive, they upstage the patient and are a threat to life. However, protecting DCIS patients from local recurrence at all costs must be balanced against the potential detrimental effects of the treatments given.
Following treatment for DCIS, 40–50% of all local recurrences are invasive. Approximately 10–20% of DCIS patients who develop local invasive recurrences develop distant metastases and die from breast cancer [14,15]. In the long-term, this could translate into a mortality rate of approximately 0–0.5% for patients treated with mastectomy, 1–2% for conservatively treated patients who receive radiation therapy (if there is no mortality associated with radiation therapy) and 2–3% for patients treated with excision alone. In order to save their breasts, many patients are willing to accept this theoretic, and as of now unproven, small absolute risk associated with breast-conservation therapy.
Treatment options
Mastectomy
Mastectomy is, by far, the most effective treatment available for DCIS if our goal is simply to prevent local recurrence. Most mastectomy series reveal local recurrence rates of approximately 1% with mortality rates close to zero [16]. In my own series, we have had only one breast cancer death among 467 patients treated with mastectomy (0.2%).
However, mastectomy is an aggressive form of treatment for patients with DCIS. It clearly provides a local recurrence benefit but only a theoretical survival benefit. Therefore, it is often difficult to justify mastectomy, particularly for otherwise healthy women with screen-detected
DCIS, during an era of increasing utilization of breast conservation for invasive breast carcinoma. Mastectomy is indicated in cases of true multicentricity (multiquadrant disease) and when a unicentric DCIS lesion is too large to excise with clear margins and an acceptable cosmetic result.
Genetic positivity to one of the breast cancer-associated genes (BRCA1 and BRCA2) is not an absolute contraindication to breast preservation, but many patients who are genetically positive and who develop DCIS seriously consider bilateral mastectomy and bilateral oophorectomy.
Breast conservation
Surveillance Epidemiology and End Results (SEER) data reveal that 74% of patients with DCIS are treated with breast conservation. While breast conservation is now widely accepted as the treatment of choice for DCIS, not all patients are good candidates. Certainly, there are patients with DCIS whose local recurrence rate with breast preservation is so high that mastectomy is clearly a more appropriate treatment. However, the majority of women with currently diagnosed DCIS are candidates for breast conservation. Clinical trials have demonstrated that local excision and radiation therapy in patients with negative margins can provide excellent rates of local control [5,8–10,17–20]. However, even radiation therapy may be overly aggressive since many cases of DCIS may not recur or progress to invasive carcinoma when treated by excision alone [4,21–25].
Prospective, randomized trials: the gold standard
All of the prospective, randomized DCIS trials have demonstrated a significant reduction in local recurrence for patients treated with radiation therapy compared with excision alone, but no trial has reported a survival benefit, regardless of treatment [5,8, 9,17–20,26,27].
Only one trial has compared mastectomy with breast conservation for patients with DCIS and the data were only incidentally accrued. The National Surgical Adjuvant Breast Project (NSABP) performed protocol B-06, a prospective, randomized trial for patients with invasive breast cancer [28,29]. There were three treatment arms: total mastectomy, excision of the tumor plus radiation therapy and excision alone. Axillary nodes were removed regardless of the treatment assignment.
During central slide review, a subgroup of 78 patients was confirmed to have pure DCIS without any evidence of invasion [28]. After 83 months of follow-up, the percentage of patients with local recurrences were as follows: 0% for mastectomy, 7% for excision plus radiation therapy and 43% for excision alone [30]. In spite of these large differences in the rate of local recurrence for each different treatment, there was no difference in breast cancer-specific survival among the three treatment groups.
Contrary to the lack of trials comparing mastectomy with breast conservation, four prospective, randomized trials comparing excision plus radiation therapy with excision alone for patients with DCIS have been published: the NSABP (protocol B-17) [17], the European Organization for Research and Treatment of Cancer (EORTC), protocol 10853 [20], the UK, Australia, New Zealand DCIS Trial (UK Trial) [8] and the Swedish Trial [10].
The results of NSABP B-17 were updated in 1995 [27], 1998 [19], 1999 [18] and 2001 [5]. In this study, more than 800 patients with DCIS excised with clear surgical margins were randomized into two groups: excision alone versus excision plus radiation therapy. The main end point of the study was local recurrence, invasive or noninvasive (DCIS). The definition of a clear margin was nontransection of the DCIS. In other words, only a fat or fibrous cell needed to be present between DCIS and the inked margin to call the margin clear. Many margins, of course, were much wider.
After 12 years of follow-up, there was a statistically significant, 50% decrease in local recurrence of both DCIS and invasive breast cancer in patients treated with radiation therapy. The overall local recurrence rate for patients treated by excision alone was 32% at 12 years. For patients treated with excision plus breast irradiation, it was 16%, a recurrence rate that is considered to be the current gold standard for conservatively treated patients with DCIS [5]. There was no difference in distant disease-free or overall survival in either arm. These updated data led the NSABP to confirm their 1993 position and to continue to recommend postoperative radiation therapy for all patients with DCIS who chose to save their breasts. This recommendation was clearly based primarily on the decreased local recurrence rate for those treated with radiation therapy and secondarily on the potential survival advantage it might confer.
The early results of B-17, in favor of radiation therapy for patients with DCIS, led the NSABP to perform protocol B-24 [18]. In this trial, more than 1800 patients with DCIS were treated with excision and radiation therapy, and then randomized to receive either tamoxifen or placebo. After 7 years of follow-up, 11% of patients treated with placebo had recurred locally, whereas only 8% of those treated with tamoxifen had recurred [5]. The difference, while small, was statistically significant for invasive local recurrence but not for noninvasive (DCIS) recurrence. Data presented at the 2002 San Antonio Breast Cancer Symposium suggested that the ipsilateral benefit was observed only in estrogen receptor-positive patients [31]. Again, there was no difference in distant disease-free or overall survival in either arm of the B-24 trial.
The EORTC results were published in 2000 [20,26]. This study was essentially identical to B-17 in design and margin definition. More than 1000 patients were included. The data were updated in 2006 [32]. After 10 years of follow-up, 15% of patients treated with excision plus radiation therapy had recurred locally compared with 26% of patients treated with excision alone, results similar to those obtained by the NSABP at the same time point in their trial. As in the B-17 trial, there was no difference in distant disease-free or overall survival in either arm of the EORTC trial. In the initial report, there was a statistically significant increase in contralateral breast cancer in patients who were randomized to receive radiation therapy. This was not maintained when the data were updated.
The UK, Australia, New Zealand DCIS Trial (UK Trial) was published in 2003 [8]. This trial, which involved more than 1600 patients, performed a two-by-two study in which patients could be randomized into two separate trials within a trial. The patients and their doctors chose whether to be randomized in one or both studies. After excision with clear margins (same nontransection definition as the NSABP), patients were randomized to receive radiotherapy (yes or no) and/or to tamoxifen versus placebo. This yielded four subgroups: excision alone, excision plus radiation therapy, excision plus tamoxifen and excision plus radiation therapy plus tamoxifen. Those who received radiation therapy obtained a statistically significant decrease in ipsilateral breast tumor recurrence similar in magnitude to the ones demonstrated by the NSABP and EORTC. In contrast to the findings of the NSABP, there was no significant benefit from tamoxifen. As with the NSABP and the EORTC, there was no benefit in terms of survival in any arm of the UK DCIS study.
The Swedish DCIS Trial randomized 1046 patients into two groups: excision alone versus excision plus radiation therapy. Microscopically clear margins were not mandatory. A total of 22% of patients had microscopically unknown or involved margins. Radiation therapy resulted in a 67% reduction in local recurrence rate with a median follow-up of 5.2 years. There were no differences in distant metastases or deaths [10].
Overall, all these trials reach the same conclusions. They show that radiation therapy decreases local recurrence by a relative 50% and they all demonstrate no survival benefit, regardless of treatment. The only difference is that the NSABP B-24 trial demonstrates a significant decrease in local recurrence attributable to tamoxifen while the UK Trial does not.
With the prospective DCIS trials demonstrating a highly significant benefit in favor of the radiation therapy arms, you might wonder how I could have possibly advocated for excision alone in selected patients. The key words are ‘selected patient’.
Limitations of the prospective, randomized trials
The randomized trials were designed to answer a single broad question: does radiation therapy decrease local recurrence? They have accomplished that goal. All have clearly shown that, overall, radiation therapy decreases local recurrence, but they cannot identify in which subgroups the benefit is so small, that the patients can be safely treated with excision alone?
Many of the parameters considered important in predicting local recurrence (tumor size, margin width, nuclear grade and so on) were not routinely collected prospectively during the randomized DCIS trials. In addition, the trials did not specifically require the marking of margins or the measurement of margin width. The exact measurement of margin width was present in only 5% of the EORTC pathology reports [26]. The NSABP did not require size measurements and many of their pathologic data were determined by retrospective slide review. In the initial NSABP report, more than 40% of patients had no size measurement [17]. Unfortunately, if margins were not inked and tissues not completely sampled and sequentially submitted, then these predictive data can never be determined by retrospective review.
The relative reduction in local recurrence appears to be the same in all four trials – approximately 50% for any given subgroup at any point in time. What does this relative reduction mean? If the absolute local recurrence rate is 30% at 10 years for a given subgroup of patients treated with excision alone, radiation therapy will reduce this rate by approximately 50%, leaving a group of patients with a 15% local recurrence rate at 10 years. Radiation therapy appears to be indicated for a subgroup with such a high local recurrence rate. However, consider a more favorable subgroup, a group of patients with a 6–8% absolute recurrence rate at 10 years. These patients receive only a 3–4% absolute benefit. We must irradiate 100 women in order to observe a 3–4% decrease in local recurrence. Here, we must ask whether the benefits are worth the risks and costs involved and we should make every attempt possible to identify low-risk subgroups.
11 reasons to consider excision alone
There are 11 lines of reasoning that suggest that excision alone may be an acceptable treatment for selected patients with DCIS:
Excision alone is already common in spite of the randomized data, which suggest that all conservatively treated patients benefit from radiation therapy. The 2003 SEER data reflect that excision alone is being used as complete treatment for DCIS in 35% of all DCIS patients. American doctors and patients have embraced the concept of excision alone.
Anatomic: evaluation of mastectomy specimens using the serial subgross tissue processing technique reveals that most DCIS is unicentric (involves a single breast segment and is radial in its distribution) [33–38]. This means that in many cases, it is possible to excise the entire lesion with a segment or quadrant resection. Since DCIS, by definition, is not invasive and has not metastasized, it can be thought of in Halstedian terms. Complete excision should cure the patient without any additional therapy. Holland et al. and Faverly et al. have shown that if 10 mm margins are achieved in all directions, the likelihood of residual DCIS is less than 10% [37,38].
Biologic: some DCIS is simply not aggressive, for example, small well-excised low-grade lesions bordering on atypical ductal hyperplasia. Lesions such as these carry a low potential for development into an invasive lesion, approximately 1% per year at most [4,21,39–42]. This is only slightly more than lobular carcinoma in situ, a lesion that is routinely treated with careful clinical follow-up.
Pathology errors: the differences between atypical ductal hyperplasia and low-grade DCIS may be subtle. It is not uncommon for atypical ductal hyperplasia to be called DCIS. Such patients treated with radiation therapy are indeed ‘cured of their DCI’.
Prospective, randomized data: the prospective, randomized DCIS trials demonstrate no difference in breast cancer-specific survival or overall survival, regardless of treatment after excision with or without breast irradiation [5,8–10,20]. If this is true, why not strive for the least aggressive treatment?
Radiotherapy may do harm: numerous studies have demonstrated that radiation therapy for breast cancer may increase mortality from both lung cancer and cardiovascular disease [43–47]. Current radiotherapy techniques, which make use of computerized tomography planning, make every attempt to spare the heart and lungs from radiation exposure but long-term data are not available. If there is no proof that breast irradiation for patients with DCIS improves survival and there is proof that radiation therapy may cause harm, it makes perfect sense to spare patients from this potentially dangerous treatment whenever possible.
Radiation therapy is expensive, time consuming and is accompanied by significant side effects in a small percentage of patients (e.g., cardiac and pulmonary) [48]. Radiation fibrosis continues to occur but it is less common with current techniques than it was during the 1980s. Radiation fibrosis changes the texture of the breast and skin, makes mammographic follow-up more difficult and may result in delayed diagnosis if there is a local recurrence.
Some series demonstrate that there are more invasive recurrences in irradiated patients than in non-irradiated patients. In our own series, 34% of excision-only patients who recurred, recurred with invasive disease whereas 53%% of irradiated patients who recurred, recurred with invasive disease (p < 0.01). This is true in the series of Schwartz et al. [25] and Wong et al. [49]. In our series, the median time to recur after excision alone was 23 months while after excision and irradiation, it was 58 months (p < 0.01). This delay in the diagnosis of recurrence may contribute to the increased rate of local invasive recurrence in irradiated patients.
If radiation therapy is given for the initial DCIS, it cannot be given again, at a later time, if there is a small invasive recurrence. In general, in favorable patients, I prefer to withhold radiation therapy initially and only give it to the few that ultimately recur with invasive disease. The use of radiation therapy with its accompanying skin and vascular changes make skin-sparing mastectomy, if needed in the future, more difficult to perform.
Using commonly available histopathologic parameters, we can do better than the gold standard for local recurrence established by the prospective, randomized trials. The gold standard for irradiated patients is a 16% local recurrence rate at 12 years. This was established by the NSABP B-17 trial [5,17–19]. Using tools such as the University of Southern California (USC)/Van Nuys Prognostic Index (VNPI), it is possible to select patients with low scores ranging from 4 to 6. These patients recur at a rate of 10% or less at 12 years without radiation therapy.
Finally, within the 2008 National Comprehensive Cancer Network (NCCN) guidelines, excision without radiation therapy (excision alone) has been added as an acceptable treatment for selected patients with a low risk of recurrence [101]. Excision alone is now accepted and mainstream for favorable patients with DCIS.
Distant disease & death
When a patient with DCIS, who was previously treated by any modality, develops a local invasive recurrence, followed by distant disease and death owing to breast cancer, this step-wise Halstedian progression makes sense. The patient has been upstaged by her local invasive recurrence. The invasive recurrence becomes the source of the metastatic disease and death is now a possibility.
By contrast, when a previously treated patient with DCIS develops distant disease and there has been no invasive local recurrence, a completely different sequence of events must be postulated. This sequence implies that invasive disease was present within the original lesion but was never discovered and was already metastatic at the time of the original diagnosis. The best way to avoid missing an invasive cancer is with complete sequential tissue processing at the time the original lesion is treated. Nevertheless, even the most extensive evaluation may miss a tiny focus of invasion.
If, during histopathologic evaluation, even the tiniest invasive component is found, this patient can no longer be classified as having DCIS. She has invasive breast cancer and she needs to be treated as such. She will need sentinel node biopsy, radiation therapy, if treated conservatively, and appropriate medical oncologic consultation and aftercare.
Predicting local recurrence in conservatively treated patients with DCIS
There is now sufficient, readily available information that can aid clinicians in differentiating patients who significantly benefit from radiation therapy after excision from those who do not. These same data can highlight patients who are better served by mastectomy because recurrence rates with breast conservation are unacceptably high, even with the addition of radiation therapy.
Our research [50–54] and the research of others [22–24,27,41,55–59] have demonstrated that various combinations of nuclear grade, the presence of comedo-type necrosis, tumor size, margin width and age are all important factors that can be used to predict the probability of local recurrence in conservatively treated patients with DCIS.
University of Southern California/Van Nuys Prognostic Index
A score, ranging from 1 for lesions with the best prognosis to 3 for lesions with the worst prognosis, is given for each of the four prognostic predictors: tumor size, margin width, pathologic classification (based on nuclear grade and necrosis) and age (
University of Southern California/Van Nuys Prognostic Index scoring system.
A total of 1–3 points are awarded for each of four different predictors of local breast recurrence (size, margins, pathologic classification and age). Scores for each of the predictors are totaled to yield a Van Nuys Prognostic Index score ranging from a low of 4 to a high of 12.
USC/VNPI = pathologic classification score + margin score + size score + age score
Scores range from 4 to 12. The patients least likely to recur after conservative therapy had a score of 4 (small, low grade, well-excised lesions in older women). The patients most likely to recur had a score of 12 (large, poorly excised, high-grade lesions in younger women). The probability of recurrence increased as the USC/VNPI score increased.
Updated results using the USC/VNPI
All through April 2008, our group treated 1363 patients with pure DCIS. A total of 467 patients were treated with mastectomy and are not included in any analysis that uses local recurrence as the end point. In total, 896 patients were treated with breast conservation: 562 by excision alone and 334 by excision plus radiation therapy. The average follow-up for all patients was 87 months: 85 months for mastectomy, 111 months for excision plus radiation therapy and 76 months for excision alone.
There were 161 local failures, 71 of which (44%) were invasive. The probability of local failure was reduced, overall, by 60% if radiation therapy was given, a result almost identical with the prospective, randomized trials. The local recurrence-free survival is shown by treatment in

Probability of local recurrence-free survival by treatment for 1363 patients with ductal carcinoma in situ.
Seven patients (2.5%) treated with radiation therapy developed local recurrences and distant metastases, six of whom have died from breast cancer. One patient (0.2%) treated with excision alone developed local invasive recurrence and metastatic disease and died from breast cancer. Two patients with mastectomy developed distant disease after developing local invasive recurrences. One has died from breast cancer. There is no statistical difference in breast cancer-specific survival (
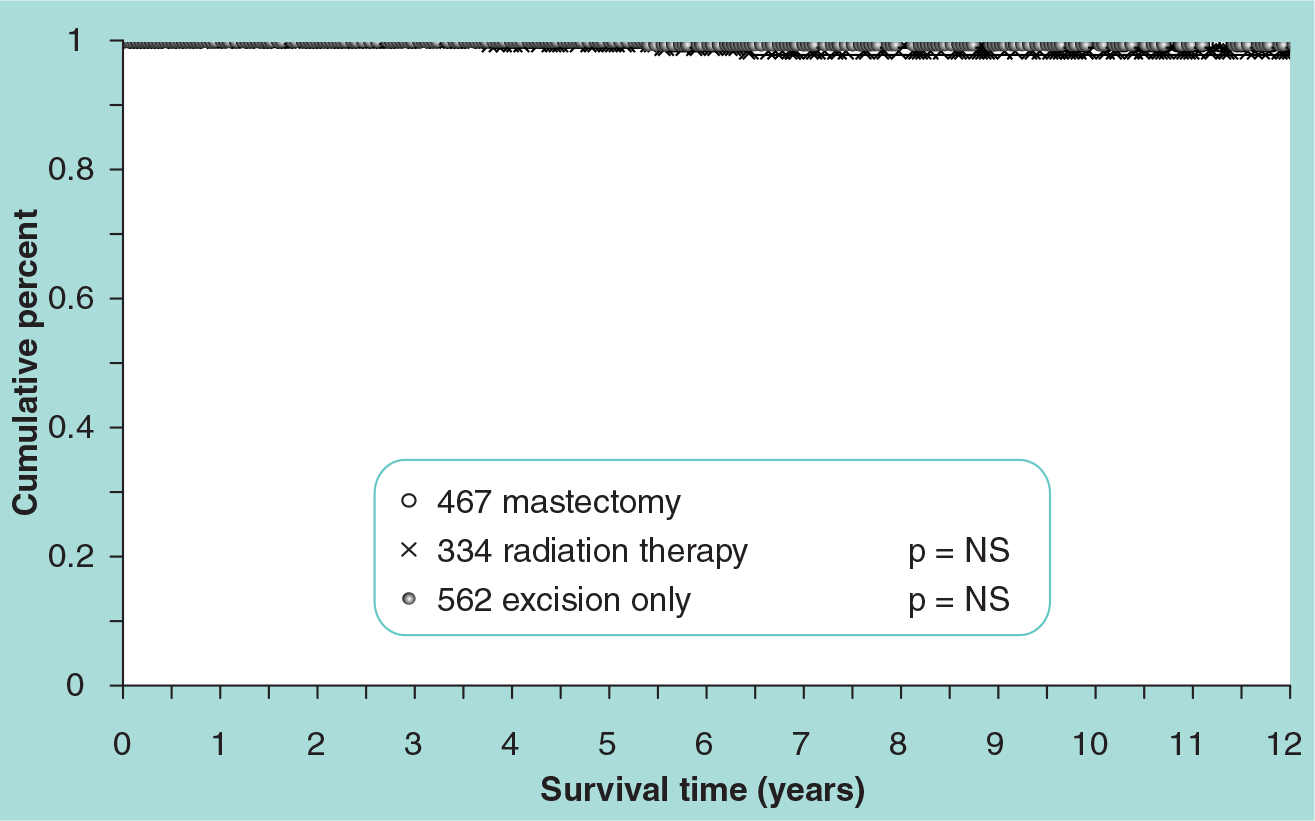
Probability of breast cancer-specific survival by treatment for 1363 patients with ductal carcinoma in situ.

Probability of overall survival by treatment for 1363 patients with ductal carcinoma in situ.
A total of 71 additional patients have died from other causes without evidence of recurrent breast cancer. The 12-year actuarial overall survival, including deaths from all causes, is 90%. It is virtually the same for all three treatment groups and for all three USC/VNPI groups (

Probability of breast cancer-specific survival for 896 breast-conservation patients grouped by modified USC/VNPI score (4, 5 or 6 vs 7 and 8; 9 vs 10, 11 or 12).
The local recurrence-free survival for all 896 breast-conservation patients is shown by tumor size in
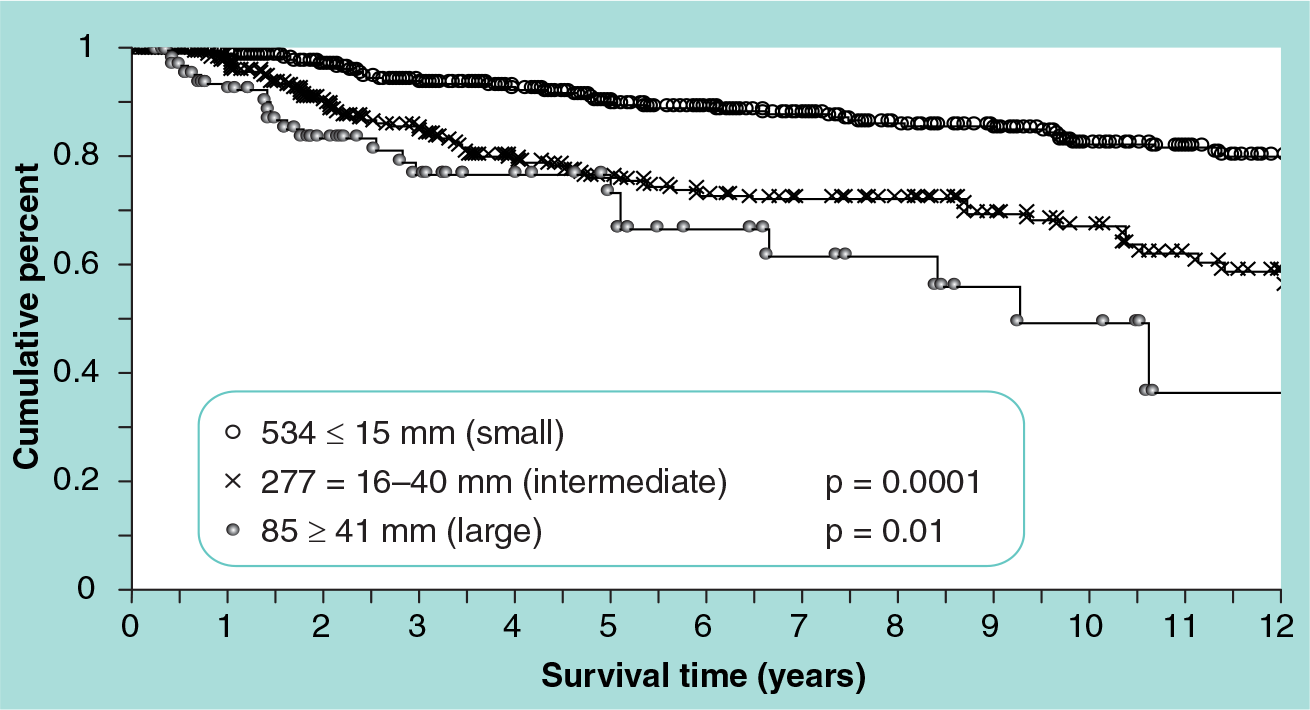
Probability of local recurrence-free survival by tumor size for 896 breast-conservation patients.

Probability of local recurrence-free survival by margin width for 896 breast-conservation patients.

Probability of local recurrence-free survival for 896 breast-conservation patients using Van Nuys ductal carcinoma in situ pathologic classification.
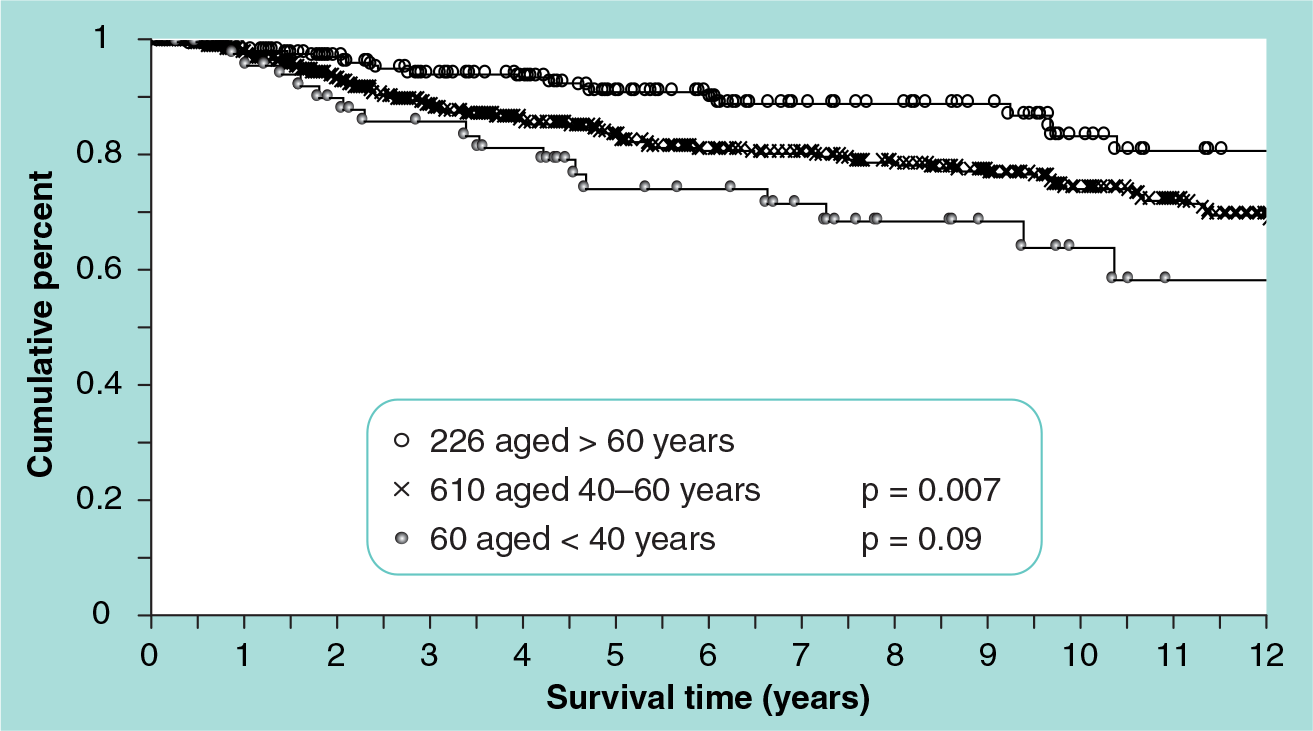
Probability of local recurrence-free survival by age group for 896 breast-conservation patients.

Probability of local recurrence-free survival for 896 breast-conservation patients grouped by USC/VNPIndex score (4, 5 or 6 vs 7 or 8; 9 vs 10, 11 or 12).
Patients with USC/VNPI scores of 4, 5 or 6 do not show a local recurrence-free survival benefit from breast irradiation(p = not significant;
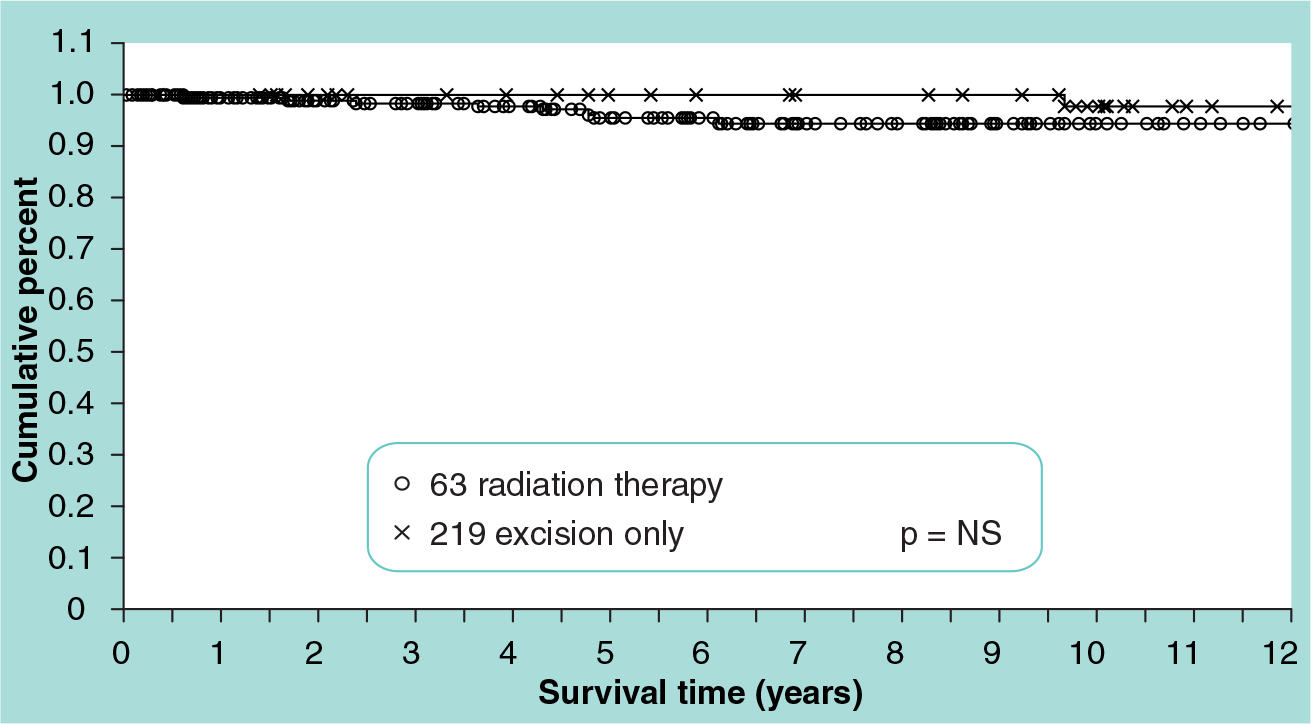
Probability of local recurrence-free survival by treatment for 282 breast-conservation patients with University of Southern California/Van Nuys Prognostic Index scores of 4, 5 or 6.
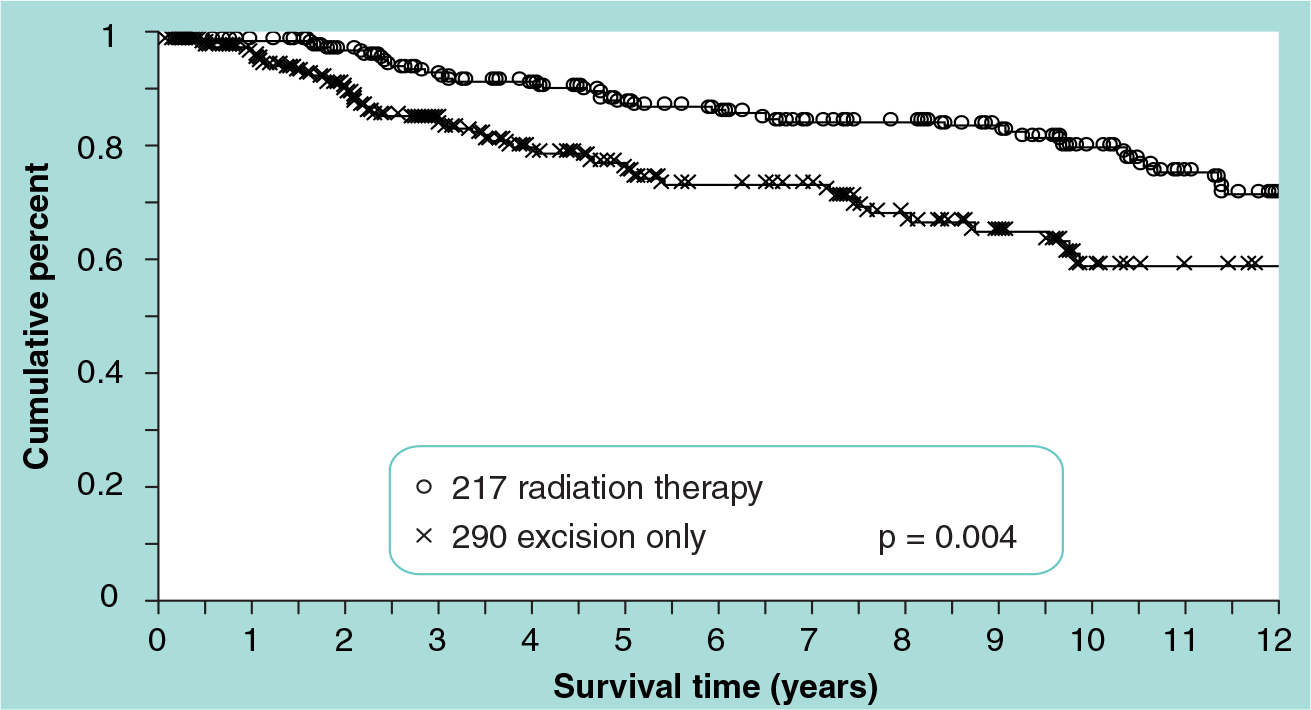
Probability of local recurrence-free survival by treatment for 507 breast-conservation patients with Univeristy of Southern California/Van Nuys Prognostic Index scores of 7, 8 or 9.

Probability of local recurrence-free survival by treatment for 106 breast-conservation patients with modified University of Southern California/Van Nuys Prognostic Index scores of 10, 11 or 12.
Current treatment trends
In the current era of evidence-based medicine, it is reasonable to interpret the prospective, randomized data as support for the tenet that all conservatively treated patients with DCIS should receive postexcisional radiation therapy. However, in spite of these data, the number of patients with DCIS being treated with excision alone continues to increase [60]. With the inclusion of excision alone as an acceptable alternative within the NCCN guidelines in 2008 [49], the percentage of patients being treated by excision alone is likely to increase in coming years.
The USC/VNPI is offered as an aid to the complex treatment decision-making process. It can be used as a starting point to suggest reasonable treatment options supported by local recurrence data. The USC/VNPI divides patients with DCIS into three groups with differing probabilities of local recurrence after breast conserving surgery. Although there is an apparent treatment choice for each group (
Conclusion
Ductal carcinoma in situ is now relatively common, and its frequency is increasing. This is largely as a result of better and more frequent mammography in a greater proportion of the female population.
Not all microscopic DCIS will progress to clinical cancer; however, if a patient has DCIS and is not treated, she is more likely to develop an ipsilateral invasive breast cancer compared with a woman without DCIS.
The comedo subtype of DCIS is more aggressive and malignant in its histologic appearance and is more likely to be associated with subsequent invasive cancer than the noncomedo subtypes. Comedo DCIS is more likely to have a high S-phase, overexpress HER2/neu and demonstrate increased thymidine labeling as compared with noncomedo DCIS. In addition, comedo DCIS treated conservatively is more likely to recur locally than noncomedo DCIS. However, separation of DCIS into two groups by architecture is an oversimplification and does not reflect the biologic potential of the lesion as well as separation by nuclear grade and comedo-type necrosis.
Most DCIS detected today will be non-palpable. It will be detected by mammographic calcifications. It is not uncommon for DCIS to be larger than expected by mammography, to involve more than a quadrant of the breast and to be unicentric in its distribution.
Preoperative evaluation should include film-screen mammography with compression magnification and ultrasonography. MRI is becoming increasingly more popular and I use it on every patient diagnosed with any form of breast cancer. The surgeon and the radiologist should plan the excision procedure carefully. The first attempt at excision is the best chance to get a complete excision with a good cosmetic result.
Treatment guidelines, USC/VNPI
4–6: excision only
7–9: excision and radiation
10–12: mastectomy
USC: University of Southern California; VNPI: Van Nuys Prognostic Index.
Re-excisions often yield poor cosmetic results and the overall plan should be to avoid them whenever possible. In light of this, the initial breast biopsy should be an image-guided needle biopsy.
After the establishment of the diagnosis, the patient should be counseled. If she is motivated for breast conservation, the surgeon and radiologist should plan the procedure carefully, using multiple wires to map out the extent of the lesion.
When considering the entire population of patients with DCIS without subset analyses, the prospective, randomized trials have demonstrated that postexcisional radiation therapy can reduce the relative risk of local recurrence by approximately 50% for conservatively treated patients. However, in some low-risk DCIS patients, the costs may outweigh the potential benefits. In spite of a relative 50% reduction in the probability of local recurrence, the absolute reduction may only be a few percent. While local recurrence is extremely important, breast cancer-specific survival is the most important end point for all patients with breast cancer, including patients with DCIS, and no DCIS trial has ever shown a survival benefit for radiation therapy when compared with excision alone. Moreover, radiation therapy is not without financial and physical cost. As a result, in recent years, an increasing number of selected patients with DCIS have been treated with excision alone.
The USC/VNPI uses five independent predictors to predict the probability of local recurrence after conservative treatment for DCIS; these include tumor size, margin width, nuclear grade, age and the presence or absence of comedo necrosis. In combination, they can be used as an aid to identify subgroups of patients with extremely low probabilities of local recurrence after excision alone, for example, patients who score 4, 5 or 6 using the USC/VNPI. If size cannot be accurately determined, margin width by itself can be used as a surrogate for the USC/VNPI, although it is not quite as good.
Oncoplastic surgery combines sound surgical oncologic principles with plastic surgical techniques. Coordination of the two surgical disciplines may help to avoid poor cosmetic results after wide excision and may increase the number of women who can be treated with breast-conserving surgery by allowing larger breast excisions with more acceptable cosmetic results. Oncoplastic surgery requires cooperation and coordination of surgical oncology, radiology and pathology.
Executive summary
Ductal carcinoma in situ (DCIS) is relatively common, representing 26% of all newly diagnosed breast cancers in the USA, and its frequency is increasing.
Most DCIS detected today will be nonpalpable and detected by mammographic calcifications.
DCIS is not a systemic disease.
Local treatment and preoperative planning are important.
DCIS is generally a segmental disease.
Selected patients can be treated with excision alone and with little risk.
The most important cause of local recurrence is inadequate excision.
Margin width is the most important predictor of local failure and it is surgeon controlled.
Radiation therapy improves local control in all subgroups, but the absolute improvement in some subgroups is very small.
There is no benefit in survival, regardless of treatment.
Radiation therapy is not a substitute for inadequate surgery or poor pathology.
Oncoplastic resection is a therapeutic procedure and not a breast biopsy, and is performed on patients with a proven diagnosis of breast cancer. New oncoplastic techniques that allow for more extensive excisions can be used to achieve both acceptable cosmesis and widely clear margins, alleviating the need for radiation therapy in many cases of DCIS.
The decision to use excision alone as treatment for DCIS should only be made if complete and sequential tissue processing has been used and the patient has been fully informed and has participated in the treatment decision-making process.
Excision alone is now an accepted alternative treatment for low-risk DCIS patients within the 2008 NCCN guidelines.
Future perspective
Our knowledge of DCIS genetics and molecular biology is increasing at a remarkably rapid rate. Future studies are likely to identify markers that will allow us to differentiate DCIS with an aggressive potential from DCIS that is merely a microscopic finding. Once we can identify DCIS that will soon develop the potential to invade and metastasize, the treatment selection process will become much simpler. As molecular medicine improves, we will probably be able to identify the earliest changes that lead to breast cancer and hopefully reverse or prevent them.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
