Abstract
Trials with tamoxifen have clearly shown that estrogen receptor-positive breast cancer risk can be reduced at a late stage in the natural history with prophylactic agents. Approximately half of the cases were prevented. The current challenge is to achieve this or better efficacy, and to reduce side effects. A recent trial found that the selective estrogen receptor modulator raloxifene has similar efficacy to tamoxifen but fewer side effects. Long-term Phase III trials are currently underway studying two aromatase inhibitors. Results from other studies suggest that these agents have a better side-effect profile and may prevent 70–80% of receptor-positive breast cancers. New agents are needed for receptor-negative breast cancer and several possibilities are currently under investigation.
Keywords
Prospects for the prevention of breast cancer have never been greater. We are beginning to find the lifestyle factors that can reduce the chance of breast cancer in women at average risk, and targeted chemoprevention for high-risk women is developing on a number of fronts.
Targeting individuals at increased risk has been highly effective for cardiovascular disease. Over the last 50 years, cardiac deaths have decreased by more than 50% in the USA and death from strokes has been reduced by more than two-thirds (Figure 1). Much of this can be attributed to the identification of high-risk individuals by measuring blood pressure and cholesterol levels, and offering them targeted preventive treatment. This has not yet been done for cancer, but breast cancer is leading the way, and we now have some important risk factors and biomarkers with a high population attributable risk. A key requirement for a biomarker is that it responds to treatment in a way that quantitatively predicts the extent of risk reduction for an individual. At present, we only have candidate biomarkers for a few cancers, notably breast cancer and prostate cancer. Mammographic density is the most promising biomarker for breast cancer, and more than 40 studies that date back to the original work by Wolfe have shown an increased risk for women with radiographically dense breasts [1]. Since then other researchers have shown that quantification of the proportional area of the breast that is covered by mammographic dense tissue is the best measure available. We can expect further improvements in the measurement of density through the use of computerized assessments, volume measurement and the identification of other radiological features, such as diffuse disease versus nodular pattern and structured densities. However, even using current techniques, breast density is a common, readily measurable factor that indicates an appreciable increase in risk in both premenopausal and postmenopausal women [2]. Although much remains to be learned about how changes in density affect risk, the fact that breast density is reduced by tamoxifen [3] and increased by hormone replacement therapy [4] suggests that we might be able to predict the effect on risk from modification of breast density.
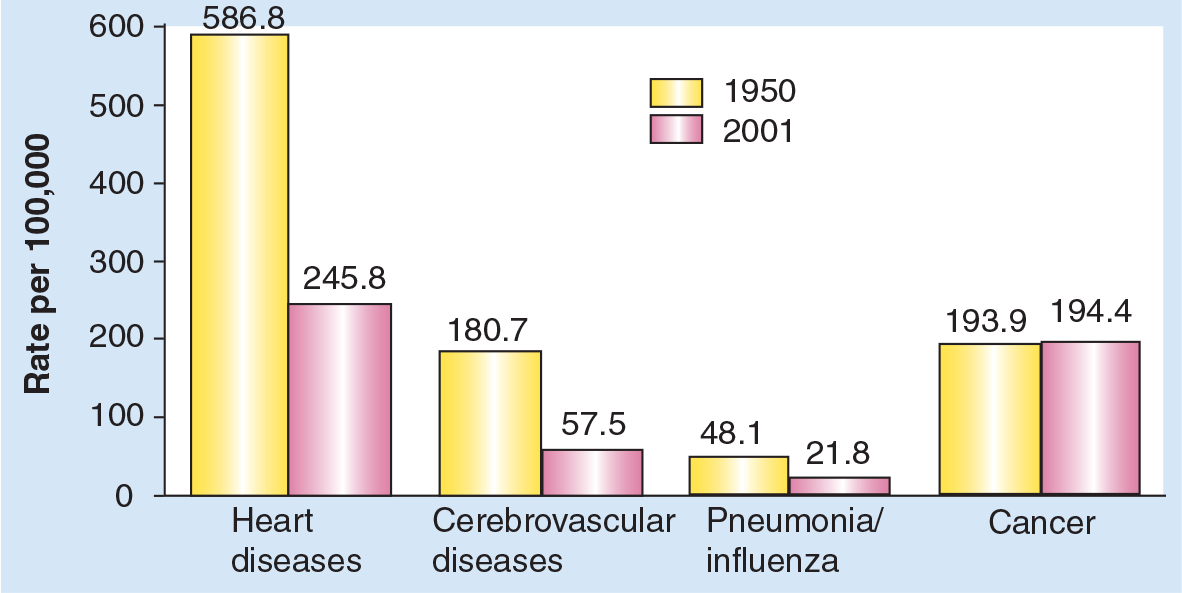
Change in the US death rates* by cause, 1950 & 2001.
Chemoprevention agents
Tamoxifen
Tamoxifen was first shown to prevent new contralateral tumors in women with breast cancer in 1985 [5]. This, together with supporting animal studies by Jordan and colleagues [6], led to the proposal to use tamoxifen in the primary prevention of high-risk women [7]. Four prevention trials have now been completed and substantial evidence is available on new isolated contralateral tumors in the adjuvant trials (Table 1). Overall, these trials have shown that breast cancer is partially preventable at a relatively late stage by hormonal maneuvres. The combined results of these trials indicated that approximately half of estrogen receptor- (ER)-positive tumors can be prevented with 5 years of prophylactic tamoxifen (Figure 2a), but this agent has no impact on ER-negative women (Figure 2b) [8]. Overall, this amounts to a 38% reduction in the risk of breast cancer.
Breast cancer prevention trials using tamoxifen.
ER+: Estrogen receptor positive; IBIS-1: International Breast Cancer Intervention study-1; NSABP: National surgical adjuvant breast and bowel project.
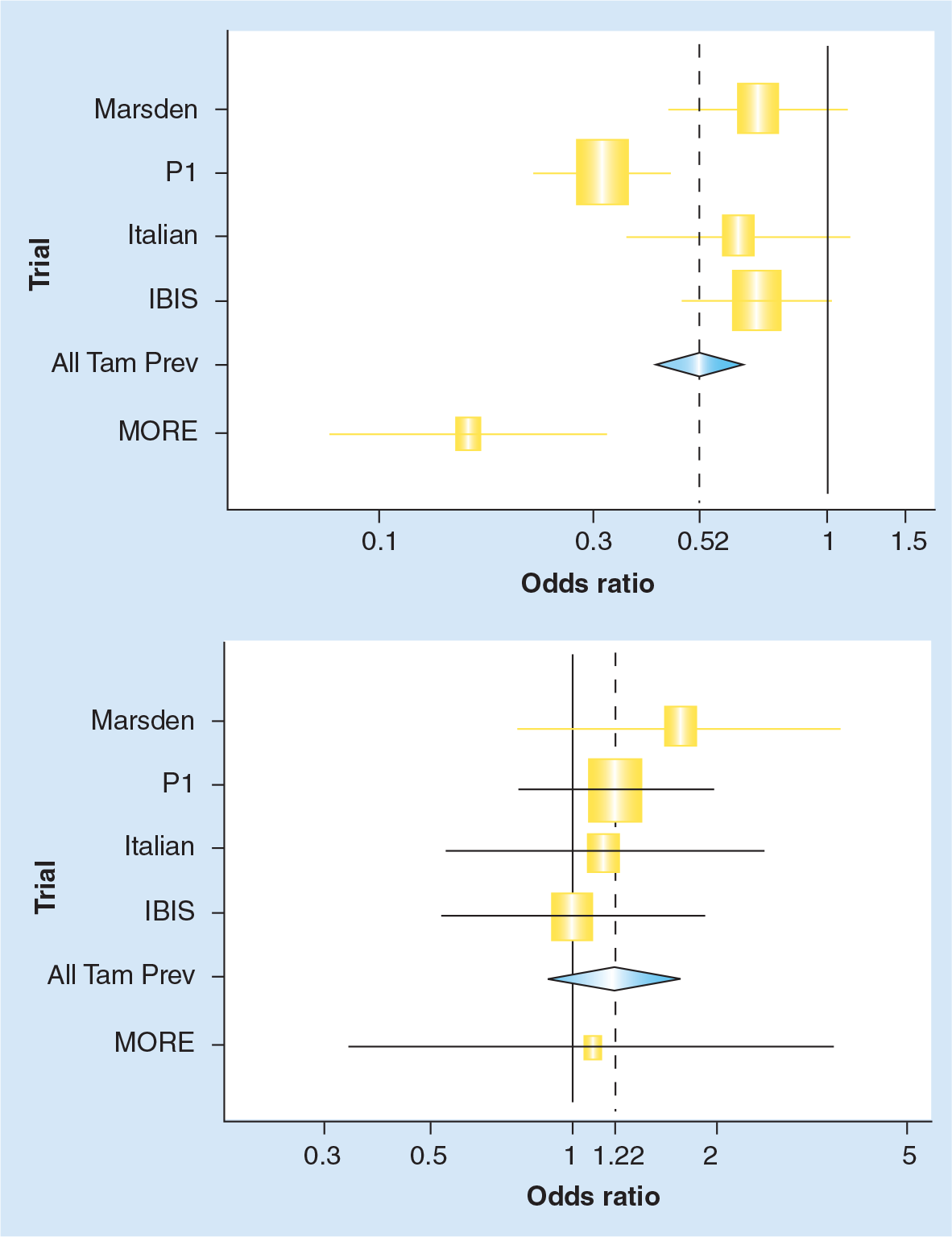
Overview of tamoxifen prevention trials: estrogen receptor-positive and -negative invasive breast cancer.
On the other hand, there were two major side effects of tamoxifen: increases in endometrial cancer and venous thromboembolic events. The former is increased by approximately 2.5-fold, whereas the latter is approximately doubled. In simple terms, giving 5 years of tamoxifen to 1000 women aged 50 years at double the population risk would lead to 11 fewer breast cancers, six additional deep vein thromboses and three extra endometrial cancers in the first 5 years of follow-up (Table 2). Given that breast cancer is the most serious of these events, the balance appears reasonably favorable. However, a key question will be the extent to which benefits and side effects extend beyond the 5-year treatment period, and the data for this are still scarce. When used as treatment, 5 years of tamoxifen shows a substantial effect on the reduction of recurrences in years 6–10, and if this holds for prevention, it will greatly improve the overall benefit:risk ratio. This is likely to be crucial for the widespread acceptance of tamoxifen as a preventive agent, as the current risk:benefit ratio, although positive for many women, is not overwhelmingly so, and another increment in the benefits without further toxicity could make a major difference.
Predicted 5-year outcome in 1000 women at high risk of breast cancer followed for 5 years.
Raloxifene
Two separate follow-up periods of a trial initially known as the Multiple Outcomes of Raloxifene Evaluation (MORE) and subsequently as the Continuing Outcomes Relevant to Evista™ (CORE) trial have reported on the reduction of breast cancer in osteoporotic women. The original intent of this trial was to reduce bone fracture rates [30]. Following 4 years of treatment, a 65% reduction in all breast cancer was found in the MORE segment. This led to another 4 years of blinded treatment in the CORE study, where breast cancer was the primary end point. Results here were also very favorable with a 50% reduction in breast cancer. Raloxifene appears to be associated with a similar level of thromboembolic complications as tamoxifen, but does not stimulate the endometrium, so that there is no excess of endometrial cancers or other gynecological problems.
The Raloxifene Use for The Heart (RUTH) study [9], whose primary aim was to study the impact of raloxifene on cardiovascular end points in 10,101 women at increased risk of cardiovascular events [9], has also reported on its effect on breast cancer (Table 3). This report found no benefit for cardiovascular disease, but a 50% reduction in breast cancer incidence which is very much in line with the MORE/CORE result. Also, the Study of Tamoxifen And Raloxifene (STAR) trial comparing raloxifene directly with tamoxifen in over 19,000 women at high risk of breast cancer has also announced its initial results. In this study, raloxifene also led to identical reductions in invasive breast cancer incidence as with tamoxifen, but with fewer gynecological and thromboembolic events. Taken together, these data indicate that raloxifene has similar preventive efficacy as tamoxifen, but with an improved side-effect profile. This is somewhat disappointing in view of the anticipated greater impact on breast cancer from MORE/CORE, but still indicates that this drug has a role to play for postmenopausal women at increased risk. Its role in pre- and perimenopausal women has not been investigated, although this may ultimately be the setting in which it has the greatest value.
Prevention trials using raloxifene.
CHD: Coronary heart disease; CORE: Continued outcomes relevant to evista; MORE: Multiple Outcomes of Raloxifene Evaluation; RUTH: Raloxifene Use for The Heart; STAR: Study of Tamoxifen And Raloxifene
Aromatase inhibitors
Efficacy
Most of what we know about the potential use of aromatase inhibitors (AIs) in prevention derives from adjuvant studies in women with early breast cancer, where the development of isolated contralateral tumors as a first event is a model for the prevention of new tumors in healthy women. This has proved a reliable source for estimating the qualitative effects of tamoxifen in prevention, both in terms of major side effects and in terms of efficacy. This approach has generally been more reliable than animal models or observational epidemiological studies, although randomized intervention studies in the prevention setting have been essential for fully quantifying effectiveness and balancing risks and benefits.
To date, seven different adjuvant trials have reported on the use of three different AIs for postmenopausal women with breast cancer [10–15]. In these trials, adjuvant AIs have been found to be effective in three clinical settings. In the first setting, the AI was compared with tamoxifen as initial adjuvant hormonal therapy in patients with resected operable breast cancer. In the Anastrozole, Tamoxifen, Alone or in Combination (ATAC) trial, 5 years of anastrozole significantly improved disease-free survival when compared with 5 years of tamoxifen [10,16,17]. Similar results have also been reported with letrozole in the Breast International Group (BIG)1-98 study [18]. In the second setting, an AI was compared with tamoxifen in patients who had already received 2–3 years of adjuvant tamoxifen. In the Intergroup Exemestane Study (IES) trial, 2–3 years of exemestane improved disease-free survival compared with tamoxifen in patients who had already completed 2–3 years of tamoxifen therapy [12,13]. Similar or longer benefits have been seen in trials of the same design using anastrozole in Austria, Germany and Italy [12,15]. In the third setting, the AI was evaluated as extended adjuvant hormonal therapy following completion of 5 years of adjuvant tamoxifen. The MA-17 trial compared 5 years of letrozole with 5 years of placebo following completion of 5 years of adjuvant tamoxifen, and demonstrated significant improvement in disease-free survival in favor of the group which received the AI [14,17]. A benefit of similar magnitude has also been reported in an Austrian trial [15]. Based on the results from the above trials, AIs are currently being increasingly utilized in all three of these clinical settings.
In all of the above trials evaluating adjuvant AIs, a consistent reduction in the rates of contralateral breast cancer has been observed in the group receiving the AI (Figure 3). For example, in the ATAC trial, the number of contralateral breast cancers was reduced from 59 in the tamoxifen arm to 35 in women receiving anastrozole, a 42% reduction (95% confidence interval [CI]: 12–62%; p = 0.01). A larger reduction of 53% (95% CI: 27–71%; p = 0.001) was seen in the hormone receptor-positive patients [10]. Tamoxifen is known to reduce the incidence of contralateral tumors by 46% in women with mostly ER-positive primary tumors, suggesting that the overall reduction of receptor-positive breast cancer associated with anastrozole may be around 70–80%. Information on the receptor status of the second cancers in this trial is not yet available, but one would expect the preventive effect to be restricted to ER-positive contralateral tumors, and to be greater for this group than for new breast tumors overall.

Contralateral tumors in adjuvant aromatase inhibitor trials.
Side effects
The profound estrogen depletion associated with AIs produces a new state of human existence, and this is bound to have other effects beyond those related to breast carcinogenesis. These effects can only be reliably studied in prevention trials where a placebo is employed. There are suggestions from adjuvant trials that AIs may also reduce endometrial cancer and cerebrovascular events to below baseline rates, but full evaluation is difficult as there is no untreated comparison group. Bone loss and increased fracture rates appear to be the most serious side effects, and methods for combating them will be essential if these drugs are to be used prophylactically. Due to the importance of side effects in the prevention setting, details of the available toxicity data from current adjuvant AI trials are summarized in the following sections.
Anastrozole
Data from the ATAC trial have been published for 36, 47 and 68 months of median follow-up. They are very similar, and only the latest data are discussed here (Table 4) [10]. Anastrozole was better tolerated than tamoxifen with approximately 25% fewer drop-outs due to side effects and a surprising 20% relative reduction in hot flushes (at any time or severity). Tamoxifen-induced side effects such as venous thromboembolic events, vaginal bleeding and discharge, other gynecological symptoms and endometrial cancer did not occur with anastrozole. However, further follow-up is needed to be certain of this. Cerebrovascular events, including strokes and transient ischemic attacks, were reduced by 30%.
ATAC: pre-defined adverse events*.
Adverse events on treatment or within 14 days of discontinuation.
Excludes patients with prior hysterectomy and includes on- and off-therapy adverse events.
Fractures occurring at anytime prior to recurrence (includes patients no longer receiving treatment). Reproduced with permission from [10].
There were two side effects that were clearly increased by anastrozole. One was the occurrence of musculoskeletal events – primary arthralgias that were increased by approximately one third, from 30 to 36%. Most of these were mild and did not lead to a discontinuation of treatment. They are probably best explained by being due to a ‘second menopause’ induced by lower levels of estrogen, and appear to be similar to the aches and pains associated with the normal menopause.
More important is the bone loss associated with estrogen deprivation. In a subset of 308 patients in the bone substudy, there was a median bone mineral loss over 2 years of 4.1% in the lumbar spine and 3.9% in the hip with anastrozole compared with small increases in tamoxifen [19]. With the median follow-up of 68 months, this has translated into an increase of fractures from 7.7–11.0%, or a 50% relative increase.
Letrozole (Breast International Group 1–98 trial)
The side-effect profile is shown in Table 5 and was qualitatively similar to that seen for anastrozole [8]. Arthritis, arthralgia and myalgia were all significantly increased. Hot flushes were also increased compared with placebo and vaginal bleeding was decreased. Fracture rates were also significantly increased by 44% (228 vs 162; p = 0.0006). Follow-up time for this trial was short, and it seems likely that significant increases in bone loss and fracture will occur with longer treatment. At this stage it is not possible to compare the absolute effects on bone of letrozole with anastrozole, but it is clear that both will lead to bone loss and increased fracture rates and the relative effects appear to be similar (Table 6). Further data on the issue will be important for use in prevention studies.
BIG 1–98: targeted adverse events.
Serial cholesterol levels are being reviewed.
CVA: Cerebrovascular accident; TIA: Thrombosis-inducing activity
Adapted from [18].
ATAC vs BIG 1–98: bone fractures.
ATAC: Anastrozole, Tamoxifen, Alone or in Combination; BIG: Breast International Group. Adapted from [10].
Exemestane (intergroup exemestane study)
Data on general side effects of exemestane compared with tamoxifen in the IES are shown in Table 7 [13]. Again, similarities with other AIs emerge. There were fewer thromboembolic events, gynecological symptoms and cramps with exemestane, but more cases of arthralgia and osteoporosis (7.4 vs 5.7%; p = 0.05). Curiously, diarrhea was reported more often (4.3 vs 2.3%; p <0.001), which has not been seen with the other AIs.
Intergroup exemestane study: significant differences in adverse event incidence.
From [13].
Early studies in animals suggested that the steroidal structure of exemestane might lead to bone preservation and even enhancement [20]. Preliminary data from the IES trial do not support this possibility and suggests a picture similar to the other AIs. Again, further data are required be able to compare the effect on bone of exemestane with that of the other AIs. Major cardiac events may be increased in this trial and with letrozole in the BIG I-98 trial, but no excess has been seen with anastrozole based on data from the ATAC trial with longer follow up (Table 8). These concerns with exemestane and letrozole are based on limited early data, and further follow-up is needed to determine if they are real.
Deaths: ATAC and BIG 1–98 trials.
ATAC: Anastrozole, Tamoxifen, Alone or in Combination; BIG 1–98: Breast International Group 1–98; moFU: Months of follow up. From [10].
Prevention trials
Two primary prevention trials using AIs are currently in progress. One uses anastrozole while the other uses exemestane.
International Breast Cancer Intervention-II Trial
The International Breast cancer Intervention Study (IBIS)-II Trial began in February 2003 and is comparing anastrozole with placebo in 6000 postmenopausal women at increased risk of breast cancer. This study is still, at present, open to recruitment. Entry criteria are similar to IBIS-I, except that women with mammographic density covering at least 50% of the breast are also eligible.
Mammary Prevention trial 3 (MAP.3)
Another prevention trial with AIs is currently underway using exemestane. This trial, sponsored by the National Cancer Institute of Canada Clinical Trials Group, compares exemestane for 5 years vs placebo in 3000 postmenopausal women at increased risk. Risk factors needed for eligibility include one of the following: a Gail Score above 1.66, aged over 60 years, prior atypical ductal or lobular hyperplasia, or ductal carcinoma in situ (DCIS) treated with mastectomy. The sample size calculations are based on an expected risk reduction of 65% with exemestane versus placebo.
New agents
Several lines of investigation for improved agents are underway. The most developed is the search for selective estrogen receptor modulators (SERMs) that have an even more favorable profile that raloxifene, which still has thromboembolic concerns and leads to vasomotor symptoms. However, its lack of gynecological symptoms has stimulated the search for a perfect SERM that would be antiestrogenic for the breast, endometrium and lipid and clotting profiles, but have estrogenic effects on bones and brain (vasomotor symptoms). Two compounds are in stage II human testing, arzoxifene and larzoxifene, and several more are in early development.
ER-negative tumors remain a challenge for prevention, and new targets will be needed to prevent these tumors. There is interest in epidermal growth factor receptor (EGFR) blockers (gefitinib), agents targeting human epidermal growth factor 2 (HER2), such as trastuzumab, and joint blockers (lapatinib). Nonsteroidal anti-inflammatory drugs (NSAIDs) [21,22], cyclooxygenase-2 (COX-2) inhibitors [23,24] and statins [25–27] may also protect against both receptor-positive and receptor-negative tumors, but only results from observational studies are available at the moment, and the results still have some inconsistencies.
Conclusions & future perspective
The ability to prevent receptor-positive breast cancer is well-established, and the challenge now is to reduce side effects and find agents with more favorable benefit:risk ratios. This will be critical if chemoprevention is to become widely used. For tamoxifen we still need more data on the carryover effects after treatment is completed. If benefits continue, but side effects do not, then this would make it an even more attractive agent. Raloxifene appears to have similar efficacy and a better side-effect profile in terms of both gynecological and thromboembolic events. There is no long term data on carryover effects for this agent, but the better toxicity suggests that it may be preferable for many postmenopausal women. Raloxifene has not been used in premenopausal women, where tamoxifen is currently the only proven agent. The AIs look very promising for postmenopausal women, but this is still based on the reduced incidence of contalateral tumors in women being treated for breast cancer in the adjuvant setting. Completion of the AI-based prevention trials is a high priority. A summary of the available evidence for tamoxifen, raloxifene and the AIs is shown in Figure 5. These are based on different levels of evidence as discussed above. Side-effect profiles differ from drug to drug and will be critical in determining which drugs to use, both overall and for individual patients. Good biomarkers will greatly accelerate our ability to evaluate new agents, and breast density is currently the most attractive candidate. However, its ability to predict the degree of risk reduction still needs to be validated. The prevention of ER-neg-ative breast cancer remains an unmet challenge; however, work on new approaches offers an approach for the treatment of these cancers as well. I would expect that once new agents targeted for other non-ER-receptor based pathways (epidermal growth factor 1 [EGF1], HER2 and insulin-like growth factor [IGF]) are developed with lower toxicity profiles for the treatment of ER-negative cancers, these will begin to be explored for prevention, especially in premenopausal women where their relative incidence is higher.

International breast cancer intervention study II: prevention stratum.
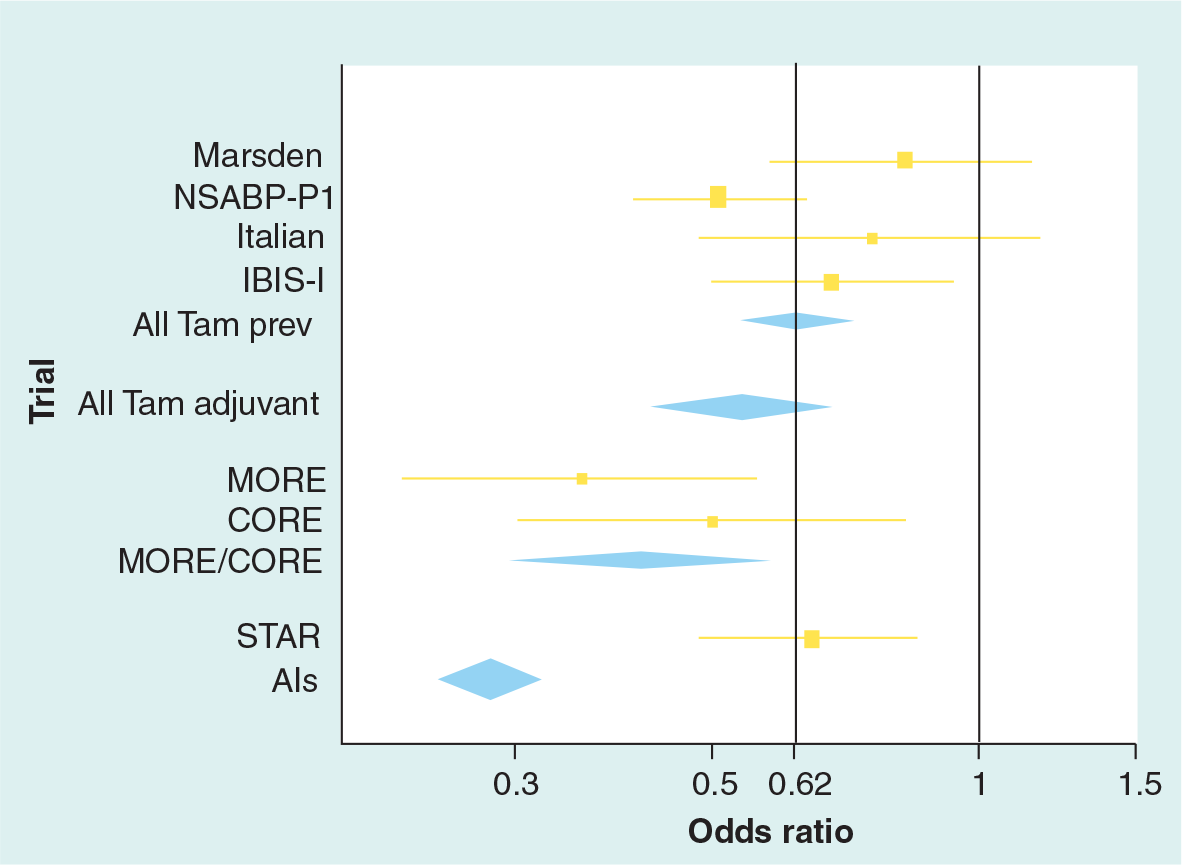
Overview: all breast cancers (including ductal carcinoma in situ except adjuvant).
Executive summary
Prospects for the prevention of breast cancer have never been greater.
The prevention trials using tamoxifen have clearly established that this agent can reduce estrogen receptor (ER) -positive breast cancer incidence by approximately 50%.
Recent data for postmenopausal women shows that raloxifene has a similar effect, but with fewer gynecological and thromboembolical side effects.
The risk:benefit ratio for these drugs will strongly depend on the carryover protection against breast cancer in the 5 years after ending treatment, and more data will soon be available on this.
Neither of these agents have any impact on ER-negative breast cancer.
Data on new contralateral tumors in adjuvant trials suggests that the aromatase inhibitors may be able to reduce incidence of ER-positive tumors by 75%, although no impact on ER-negative tumors is anticipated
These agents are only effective for postmenopausal women.
High priority should be given to completing the ongoing aromatase inhibitor prevention trials (IBIS-II and MAP3).
New approaches to prevention are needed for premenopausal women and women with ER-negative tumors.
Footnotes
The author's research is partly supported by AstraZeneca and Aventis, and he acts as a statistical consultant for AstraZeneca and is on advisory boards for Lilly and Pfizer.
