Abstract
Raloxifene hydrochloride is a selective estrogen receptor modulator that has antiestrogenic effects on breast and endometrial tissue and estrogenic effects on bone, lipid metabolism and blood clotting. Tamoxifen is the prototypical selective estrogen receptor modulator and reduces the risk of both in situ and invasive breast cancers by half when compared with placebo. The limitations on the use of tamoxifen for breast cancer risk reduction relate to its well-known, but rare, side effects. A number of clinical trials have established the benefit of raloxifene on osteoporosis and fracture. Raloxifene significantly improves serum lipids and serum markers of cardiovascular disease risk, but has no significant effect on the risk of primary coronary events. In several osteoporosis trials and the Raloxifene Use for The Heart (RUTH) trial, raloxifene decreased the risk of estrogen receptor-positive breast cancer by 44–90%. In the Study of Tamoxifen And Raloxifene (STAR) trial, the effect of raloxifene on invasive breast cancer was equivalent to that of tamoxifen, with more favorable effects on uterine malignancy and clotting events. Symptomatic side effects are acceptable. In total, the available data indicate that raloxifene represents an acceptable alternative to tamoxifen for the reduction of the risk of postmenopausal breast cancer in high-risk women. The potential market for a compound shown to reduce the risk of breast cancer in postmenopausal women who are at increased risk for breast cancer is more than 10 million women in the USA alone.
Keywords
The development of antiestrogens, now known as selective estrogen receptor modulators (SERMs), was a decades-long process that began in the 1960s and has been thoroughly reviewed [1]. It will be summarized briefly here. In the 1980s, few side effects were noted with the SERM tamoxifen compared with cytotoxic chemotherapy, so tamoxifen became the standard of care for women with estrogen receptor (ER)-positive breast cancer. The widespread use of tamoxifen in node-negative breast cancer patients increased interest in testing the worth of tamoxifen as a potential chemopreventive in high-risk women. Tamoxifen prevented mammary carcinogenesis in animals, reduced the occurrence of contralateral primary breast cancers and had few major side effects.
Both tamoxifen and raloxifene (originally known as keoxifene) maintained bone density in ovariectomized rats at doses that would prevent the development of carcinogen-induced rat mammary tumors. A difference in the target site-specific actions of tamoxifen in the mouse uterus or human breast tumor transplanted into immunodeficient mice resulted in the observation that tamoxifen enhanced the growth of human ER-positive endometrial carcinomas but blocked the growth of breast tumors transplanted into the same immunodeficient animal. These data translated to the clinic with a small but significant increase in the incidence of endometrial cancer in postmenopausal women. Raloxifene was noted to have fewer estrogen-like actions in the rodent uterus and on human endometrial cancer.
SERMs interact either with membrane-bound ER as part of the cell surface receptor signal transduction phosphorylation cascade or with the nuclear ERs. The shape of the SERM determines optimal antiestrogenic folding of the ER complex. Two ERs (ER-α and ER-β) produce opposing biological actions based on the shape or the phosphorylation state of the SERM–ER complex, in addition to interactions with either a coactivator protein, to induce a response, or a corepressor protein, to block a response. In addition, based on conformational shape, there may be either destruction of the SERM–ER complex by proteosomes or accumulation. Finally, the SERM–ER complex may activate the promoter region of genes directly at the DNA level or via activator protein (AP)-1 sites.
During the past decade, there has been an enhanced understanding of the complex interactions within tumors and estrogen target tissues that modulate the actions of the SERM–receptor complex. Although there was originally believed to be only one ER, it is now clear that at least two ERs (ER-α and ER-β) can modify estrogen or SERM action. The two receptors are distributed differently within the body and may modify ligand interactions by homo- or hetero-dimerization, as well as through possible interactions with other proteins, to modulate signal transduction. SERM structure is important in causing distinct ER folding that affects interactions with other proteins such as coactivators or corepressors. For both raloxifene and tamoxifen, minor differences in SERM ligand interaction with specific amino acids produce different intrinsic estrogen actions. The tamoxifen–ER complex is more estrogen-like in vitro, reflecting more estrogen-like action in the uterus. By contrast, the raloxifene–ER complex is much less estrogen-like and has fewer estrogen-like properties in the uterus.
Tamoxifen
Tamoxifen is the prototypical SERM, and the clinical data regarding tamoxifen serve as the comparison for clinical evaluations of raloxifene both for its efficacy in reducing the risk of breast cancer and for its safety. In the Breast Cancer Prevention Trial (BCPT), a randomized comparison of tamoxifen and placebo in women at increased risk for breast cancer [2,3], tamoxifen reduced the risk of invasive breast cancer by 49% (two-sided p < 0.00001), with a cumulative incidence through 69 months of follow-up of 43.4 versus 22.0/1000 women in the placebo and tamoxifen groups, respectively. The decreased risk was greatest among women aged 60 years or older, and risk was also reduced in women with a history of lobular carcinoma in situ (LCIS) (56%) or atypical hyperplasia (86%). Tamoxifen reduced the risk of noninvasive breast cancer by 50% (p < 0.002). Tamoxifen administration reduced fractures in the hip, radius (Colles') and spine. The rate of endometrial cancer was increased in the tamoxifen group, predominantly in women aged 50 years or older. The relative risk (RR) of women aged 49 years or younger was 1.21 (95% confidence interval [CI]: 0.41–3.60), whereas it was 4.01 (95% CI: 1.70–10.90) in women aged 50 years or older.
The limitations in the use of tamoxifen for breast cancer risk reduction relate to its well-known, but rare, side effects [4]. In the BCPT, the increase in incidence of invasive endometrial cancer after tamoxifen administration was observed early in the follow-up period. During 66 months of follow-up, the cumulative incidence was 5.4/1000 women in the placebo group and 13.0/1000 women in the tamoxifen group. The rates of stroke, pulmonary embolism and deep-vein thrombosis (DVT) were elevated in the tamoxifen group; these events occurred more frequently in women aged 50 years or older. Women who experienced both a stroke and a transient ischemic attack, or both a pulmonary embolism and DVT, were categorized according to the most severe event, that is, stroke or pulmonary embolism, respectively. While not statistically significant, the incidence of stroke increased from 24 events in the placebo group to 38 events in the tamoxifen group (i.e., from 0.92/1000 participants/year in the former group to 1.45/1000 participants/year in the latter group). Among women aged 50 years or older, 20 strokes occurred in those who received placebo and 35 in those who received tamoxifen (RR: 1.75; 95% CI: 0.98–3.20).
Pulmonary emboli were observed in almost three-times as many women in the tamoxifen group as in the placebo group (RR: 3.01; 95% CI: 1.15–9.27). When the incidence of pulmonary embolism was related to the age of participants there was an increase in those events in postmenopausal women who received tamoxifen (RR: 3.19; 95% CI: 1.12–11.15). More women who received tamoxifen developed DVT than did women who received placebo. The average annual rates per 1000 women were 1.34 in the tamoxifen group versus 0.84 in the placebo group (RR: 1.60; 95% CI: 0.91–2.86). The excess risk appeared to be greater among women aged 50 years or older.
Clinical trials of raloxifene for osteoporosis & heart disease end points
During the past decade, a number of clinical trials have been conducted to assess the benefit of raloxifene on osteoporosis and fracture. After the publication of the results of the BCPT, these osteoporosis trials reported data related to the incidence of invasive breast cancer among women receiving raloxifene compared with those receiving placebo. The results are summarized in Table 1. A trial in women who were at increased risk of coronary heart disease (CHD) also reported breast cancer end points [13]. The design of each of these trials will be described separately and the breast cancer outcomes will be reviewed. The toxicities associated with raloxifene in each of the trials will also be reviewed. Finally, the data from the Study of Tamoxifen And Raloxifene (STAR) will be reviewed in great detail as they comprise most of the prospective data related specifically to the reduction of breast cancer risk.
Prospective, randomized, placebo-controlled clinical studies of raloxifene with invasive breast cancer as an end point.
CHD: Coronary heart disease; CORE: Continuing Outcomes Relevant to Evista® trial; MORE: Multiple Outcomes of Raloxifene Evaluation trial; RUTH: Raloxifene Use for The Heart trial; STAR: Study of Tamoxifen And Raloxifene trial.
Multiple Outcomes of Raloxifene Evaluation (MORE) trial
The Multiple Outcomes of Raloxifene Evaluation (MORE) trial was conducted to determine primarily whether women receiving raloxifene would have a lower risk of fracture; invasive breast cancer was included as a secondary end point [5–10]. It was a multicenter, randomized, double-blind trial, in which women taking raloxifene or placebo were followed for a median of 40 months, from 1994–1998, at 180 clinical centers composed of community settings and medical practices in 25 countries, mainly in the USA and Europe. Participants were a total of 7705 postmenopausal women with osteoporosis who were younger than 81 years of age (mean age: 66.5 years). They were randomly assigned raloxifene 120 or 60 mg/day or placebo.
Continuing Outcomes Relevant to Evista® (CORE) trial
The Continuing Outcomes Relevant to Evista (CORE) trial was conducted to examine the effect of 4 additional years of raloxifene therapy on the incidence of invasive breast cancer in women in MORE who agreed to continue in CORE [11–12]. There were 3510 women who had been randomly assigned to receive raloxifene (either 60 or 120 mg/day) in MORE who were assigned to receive raloxifene 60 mg/day in CORE. Women who had been assigned to receive placebo in MORE continued on placebo in CORE (n = 1703).
Raloxifene Use for The Heart (RUTH) trial
Raloxifene therapy has been associated with improvement in the levels of serum lipoprotein cholesterol, fibrinogen and homocysteine. The favorable effect of raloxifene on markers of cardiovascular risk, coupled with evidence from observational studies that treatment with estrogen was associated with a reduced risk of CHD in postmenopausal women, led to the design of the Raloxifene Use for The Heart (RUTH) trial to determine the effect of raloxifene on clinical coronary events [13]. In the trial 10,101 postmenopausal women (mean age: 67.5 years) with CHD or multiple risk factors for CHD were randomly assigned to either 60 mg/day raloxifene or placebo and were followed for a median of 5.6 years. The two primary outcomes were coronary events (i.e., death from coronary causes, myocardial infarction or hospitalization for an acute coronary syndrome) and invasive breast cancer.
Study of Tamoxifen And Raloxifene (STAR) trial
To compare the relative effects and safety of raloxifene and tamoxifen on the risk of developing invasive breast cancer and other disease outcomes, the National Surgical Adjuvant Breast and Bowel Project conducted the STAR trial, a prospective, double-blind, randomized clinical trial that began in July 1999 in nearly 200 clinical centers throughout the USA [14,15]. Patients were 19,747 postmenopausal women, with a mean age of 58.5 years, with increased 5-year breast cancer risk (mean risk: 4.03 ± 2.17%) as estimated by the Gail model. Participants were randomly assigned to receive either tamoxifen 20 mg/day or raloxifene 60 mg/day over 5 years. Outcomes of interest were incidence of invasive breast cancer, uterine cancer, noninvasive breast cancer, bone fractures and thromboembolic events. The trial was designed to assess statistical equivalence of the two therapies and was powered to report data when 327 cases of invasive breast cancer occurred.
Overview of the market
The potential market for a compound shown to reduce the risk of invasive breast cancer in postmenopausal women who are at risk for breast cancer is large [16]. Of the more than 65 million women aged 35–79 years without reported breast cancer in the USA in 2000, more than 10 million women (15.5%) would be eligible for tamoxifen chemoprevention using the eligibility criteria of the BCPT. In the USA, there are more than 33 million postmenopausal women, and 9 million of these women are eligible for risk reduction with a SERM. Of the 50 million white US women aged 35–79 years, more than 2 million, or approximately 5%, would have a positive benefit:risk index for SERM-based chemoprevention. An estimate of the net benefit among nonwhite women is difficult due to the lack of these women in the clinical trials. Efforts are ongoing to increase participation of nonwhite women in risk reduction clinical trials. Among the postmenopausal women, 1.2 million women would derive net benefit from tamoxifen. Among white women in the USA, more than 28,000 breast cancers would be prevented or deferred if those women who have a positive net benefit index took tamoxifen over the next 5 years. A similar number of women in Europe could also derive benefit from chemoprevention of breast cancer with a SERM. Estimates for the rest of the world are not available. Approximately 500,000 women worldwide are currently receiving raloxifene for the management of osteoporosis.
Introduction to raloxifene
Chemistry
Raloxifene hydrochloride is a SERM that has antiestrogenic effects on breast and endometrial tissue and estrogenic effects on bone, lipid metabolism and blood clotting [17–20]. It is a benzothiophene ([6-hydroxy-2-(4-hydroxyphenyl)-benzothiophen-3-yl]-[4-(2-[1-piperidyl]ethoxy) phenyl]methanone) with characteristics similar to but distinct from the triphenlyethylene SERMs, such as tamoxifen. Its chemical structure is shown in

Estrogens, tamoxifen and raloxifene.
Pharmacodynamics
In vitro, raloxifene binds to both the α and β subtypes of the ER (ER-α and ER-β, respectively) [21]. Compared with 17β-estradiol, raloxifene has a relative binding affinity of 46% at the human ER-α and 26% at the rat ER-β. In functional transactivation studies, raloxifene antagonizes 17β-estradiol-induced estrogen response element (ERE)-mediated activity via ER-α and ER-β. By contrast, raloxifene acts as an agonist at ER-α and ER-β in a response element transactivation system. The conformation of ER when occupied by 17β-estradiol is different from its conformation when occupied by raloxifene [22]. As noted earlier, these distinct ER–ligand conformations appear to recruit different coactivators and corepressors onto the promoter of a target gene by differential protein–protein interactions at the receptor surface. Raloxifene acts as a partial agonist in bone but does not stimulate endometrial proliferation in postmenopausal women, presumably due to some combination of differential expression of transcription factors in the two tissues and the effects of this SERM on ER conformation.
Pharmacokinetics & metabolism
Raloxifene is rapidly absorbed after oral administration [17,18]. Absolute raloxifene bioavailability is 2%; approximately 60% of an oral dose is absorbed and enterohepatic cycling occurs. Although not considered clinically significant, absorption may increase when raloxifene is given with a high-fat meal, as shown by a 28% increase in peak serum concentration (Cmax) and a 16% increase in the area under the plasma concentration–time curve (AUC). For 30–150 mg single doses of raloxifene, the apparent volume of distribution is 2348 1/kg and is not dose-dependent. After multiple doses, Cmax and AUC values of raloxifene are 1.36 μg/l/mg/kg and 24.2 μg * h/l/mg/kg.
Raloxifene and its glucuronide conjugates are highly bound to plasma proteins albumin and α1-acid glycoprotein, but not to sex hormone-binding globulin. First-pass metabolism to the glucuronide conjugate is extensive; 1% unconjugated raloxifene appears in the plasma as a result of interconversion between conjugated and unconjugated forms. Excretion is predominantly via the fecal route and occurs within 5 days of administration; approximately 5% of the administered dose is excreted in urine as glucuronide conjugates. Its elimination half-life is 32.5 h (range: 15.8–86.6) with multiple dosing [23].
Coadministration of warfarin with raloxifene produces small but significant reductions in clearance and volume of distribution of the warfarin enantiomers. A 60% reduction in the absorption and enterohepatic recirculation of raloxifene has been reported after a single dose of the anion exchange resin cholestyramine.
Clinical efficacy
Effects on osteoporosis
A meta-analysis of all randomized, double-blind, placebo-controlled trials has been published and was performed to determine whether the reduction in the risk for vertebral fracture reported with raloxifene was consistent among studies, and to define more accurately the point estimate of the odds ratio (OR) [24]. Three osteoporosis prevention studies [25], two arms of the MORE trial [5] and two additional treatment studies [26,27], in which fracture data were available from prospectively scheduled spinal radiographs, were included in the analysis. The effects of raloxifene 60 and 120 mg/day pooled with 150 mg/day were analyzed using an intention-to-treat analysis to minimize biases. There was no significant heterogeneity among the studies included in the meta-analysis. OR estimates for the reduction of fracture risk were 0.60 (95% CI: 0.49–0.74) for raloxifene 60 mg/day and 0.51 (95% CI: 0.41–0.64) for raloxifene 120 and 150 mg. These data indicate that raloxifene consistently reduces the risk of vertebral fracture in postmenopausal women. On the other hand, it does not reduce the incidence of nonvertebral fractures, particularly those in the hip.
A post hoc analysis of the MORE osteoporosis treatment trial assessed the risk:benefit profile of raloxifene in 7705 postmenopausal women [28]. A global index of potential benefits and risks was defined using criteria from the Women's Health Initiative (WHI) as the first occurrence of CHD, stroke, pulmonary embolism, invasive breast cancer, endometrial cancer, colorectal cancer, hip fracture or death owing to other causes. To provide results based on analyses similar to those used for the global index, as originally applied in the WHI trial, events reported after a patient discontinued from the trial were censored from the analysis. Results were similar for each dose group compared with placebo. This major disease outcomes global index resulted in annual rates of 1.39% and 1.83% in the raloxifene and placebo groups, respectively (hazard ratio [HR]: 0.75; 95% CI: 0.62–0.92), compatible with a favorable risk:benefit profile for raloxifene in treating postmenopausal osteoporosis.
In the RUTH trial, raloxifene reduced the risk of clinical vertebral fractures (HR: 0.65; 95% CI: 0.47–0.89; absolute risk reduction: 1.3/1000) [13].
Effects on cardiovascular events
The effect of raloxifene on the incidence of cardiovascular adverse events in postmenopausal women has been reported in the MORE and CORE trials [29,30]. Of the 7705 participants originally enrolled in MORE, 4011 were enrolled in CORE. The 8-year incidence of serious cardiovascular adverse events did not differ significantly between the raloxifene (5.5%) and placebo (4.7%) groups (HR: 1.1; 95% CI: 0.86–1.56). Similar results were obtained when coronary (HR: 1.22; 95% CI: 0.82–1.83) or cerebrovascular (HR: 1.19; 95% CI: 0.78–1.84) events were analyzed separately, and when cardiovascular events were analyzed in the 459 MORE–CORE participants who were at increased risk of cardiovascular events using previously established criteria (HR: 1.03; 95% CI: 0.58–1.82). Thus, there was no evidence of either a beneficial or harmful effect of raloxifene on the incidence of cardiovascular events overall, or coronary or cerebrovascular events, in postmenopausal osteoporotic women who were at relatively low risk of cardiovascular events.
Consistent with these results, raloxifene does not appear to cause adverse effects on lipids and lipoproteins. A post hoc analysis reported the effects of raloxifene on lipids and lipoproteins in 2659 women with either normal (≤150 mg/dl) or high (>150 mg/dl) triglyceride levels from a substudy of the MORE trial [31]. In both triglyceride subgroups, raloxifene significantly improved low-density lipoprotein cholesterol, total cholesterol, non-high-density lipoprotein cholesterol (non-HDL-C), apolipoprotein B, apolipoprotein A-I and fibrinogen compared with placebo (p < 0.05). After raloxifene treatment, women with high triglycerides experienced an equal or greater reduction in cholesterol, lipoprotein parameters and ratios of total cholesterol:HDL-C and non-HDL-C:HDL-C than were observed in women with normal triglycerides. Mean levels of low-density lipoprotein cholesterol were reduced by 16.5% and apolipoprotein B by 15.8% in women with high triglycerides, and by 12.7% and 11.3%, respectively, in women with normal triglycerides. These findings substantiate that raloxifene improves concentrations of both cholesterol and β-lipoprotein [32]. The subgroup of women with high triglycerides, who have elevated cardiovascular risk, appear to derive at least equal, if not greater, overall effect on lipid and lipoprotein lowering with raloxifene. These are important safety considerations when evaluating raloxifene for a potential breast cancer risk reduction indication.
RUTH trial
In light of the data just reviewed, it is not unexpected that, compared with placebo, raloxifene had no significant effect on the risk of primary coronary events (533 vs 553 events; HR: 0.95; 95% CI: 0.84–1.07) in the RUTH trial [13]. There was no significant difference between the raloxifene and placebo group in the incidence of the primary outcome of death from coronary causes, nonfatal myocardial infarction or hospitalization for an acute coronary syndrome, or for death from coronary causes, nonfatal myocardial infarction or hospitalization for an acute coronary syndrome individually. The effect of treatment on the primary outcome of coronary events did not differ significantly among women with established CHD or in women who were at increased risk for CHD. There is no evidence, therefore, for a beneficial effect of raloxifene on CHD events in older postmenopausal women. Its effect on younger postmenopausal women has not been reported.
Effects on invasive breast cancer
MORE trial
In the MORE trial, 13 cases of breast cancer were confirmed among the 5129 women assigned to raloxifene versus 27 among the 2576 women assigned to placebo (RR: 0.24; 95% CI: 0.13–0.44; p < 0.001) [6–9]. To prevent one case of breast cancer, 126 women would need to be treated in this older population of low-to-average-risk women. Raloxifene decreased the risk of ER-positive breast cancer by 90% (RR: 0.10; 95% CI: 0.04–0.24), but not ER-negative invasive breast cancer. Similar to tamoxifen in the BCPT, raloxifene increased the risk of venous thromboembolic disease (RR: 3.1; 95% CI: 1.5–6.2), but did not increase the risk of endometrial cancer (RR: 0.8; 95% CI: 0.2–2.7).
In the MORE trial, prespecified subgroups were defined by age (≥65 vs <65 years), age at menopause (≥49 vs <49 years), body mass index (≥25 vs <25 kg/m2), family history of breast cancer, serum estradiol level (5–10 vs <5, >10 vs <5 pmol/l), prior estrogen therapy, bone mass at baseline entry into the MORE trial, and 5-year predicted risk of breast cancer (assessed using the modified Gail model). In the placebo group, older age, higher estradiol level and a family history of breast cancer were associated with an increased breast cancer risk (p < 0.05). Raloxifene therapy was associated with a reduced breast cancer risk in women at lower and higher breast cancer risk. The therapy–family history interaction was significant (p = 0.04), indicating a greater benefit in those women reporting a history of breast cancer in their first-degree relatives.
CORE trial
During the 4 years of the CORE trial, there were 61 cases of breast cancer (30 in the placebo group and 31 in the raloxifene group) that were confirmed by adjudication [11–12]. Of the 61 breast cancer cases, 52 cases (28 in the placebo group and 24 in the raloxifene group) were classified as invasive breast cancer. Women in the raloxifene group had a 59% reduction in the incidence of invasive breast cancer compared with women in the placebo group (2.1 vs 5.2 cases/1000 woman-years; HR: 0.41; 95% CI: 0.24–0.71). The ER status was determined for 46 of the 52 cases of invasive breast cancer; 36 cases (78%) were ER-positive. Women in the raloxifene group had a 66% reduction in the incidence of invasive ER-positive breast cancers compared with women in the placebo group (1.3 vs 3.9 cases/1000 woman-years; HR: 0.34; 95% CI: 0.18–0.66). By contrast, the incidence of invasive ER-negative breast cancer in women who received raloxifene was not different from that in women who received placebo. The overall incidence of breast cancer, regardless of invasiveness, was reduced by 50% in the raloxifene group compared with the placebo group (2.7 vs 5.5 cases/1000 woman-years; HR: 0.50; 95% CI: 0.30–0.82)
For the 7705 MORE participants, the total number of reported breast cancers confirmed by adjudication from randomization in MORE to the end of their participation in either MORE or CORE was 121 (56 cancers in the raloxifene group and 65 cancers in the placebo group). During these 8 years, 40 invasive breast cancers were reported in the raloxifene group (1.4 cases/1000 woman-years) and 58 invasive breast cancers were reported in the placebo group (4.2 cases/1000 woman-years). These data are shown in Figure 2.
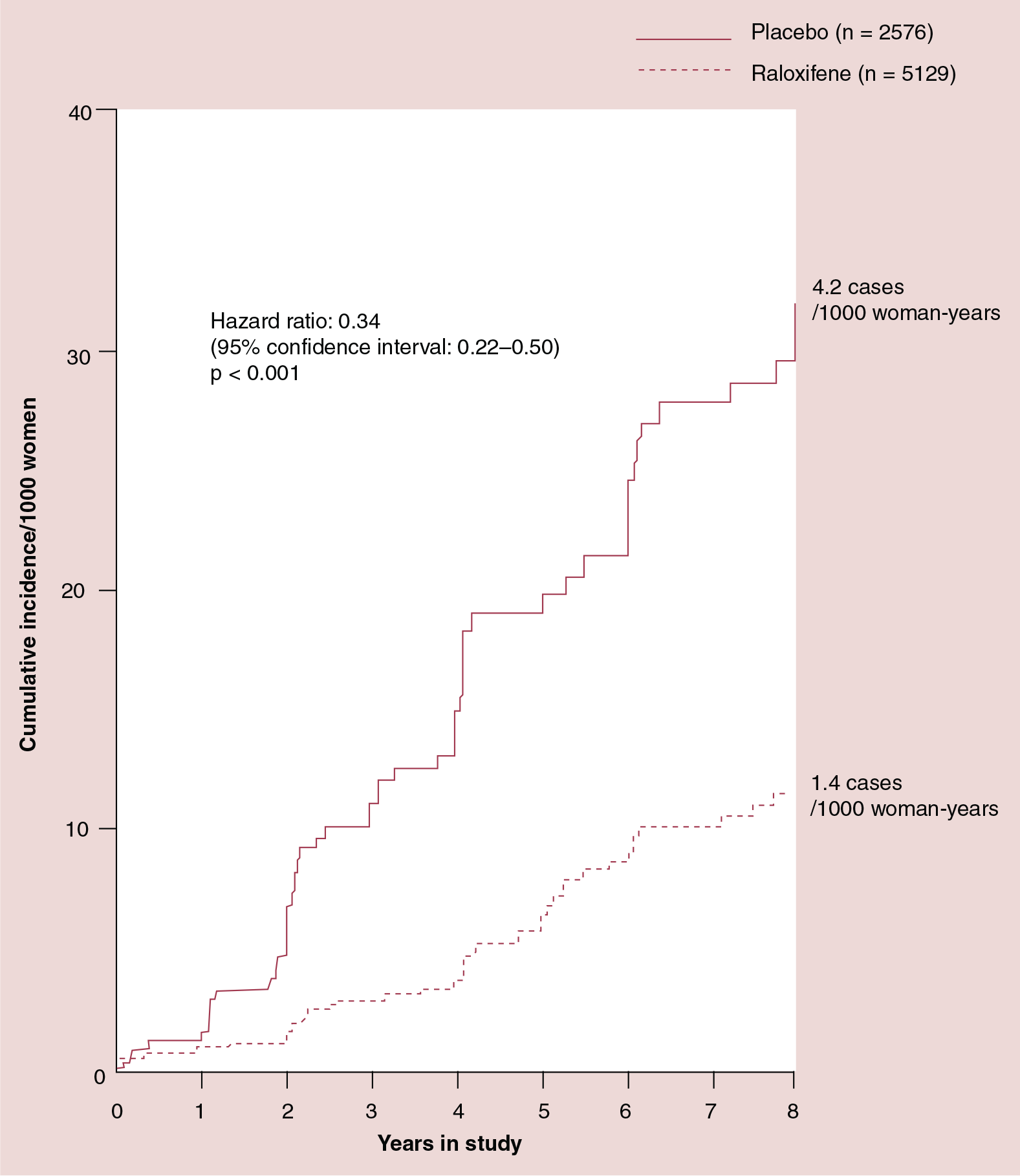
Breast cancer risk reduction attributable to raloxifene compared with placebo in the MORE/CORE trials.
Therefore, the raloxifene group had a 66% reduction in the incidence of invasive breast cancer compared with the placebo group (HR: 0.34, 95% CI: 0.22–0.50). ER status was determined for 88 cases and 75% of these were ER-positive. During these 8 years, the raloxifene group had a 76% reduction in the incidence of invasive ER-positive breast cancer compared with the placebo group (0.8 vs 3.2 cases/1000 woman-years; HR: 0.24; 95% CI: 0.15–0.40). There was no difference in the incidence rates of invasive ER-negative breast cancer between the raloxifene group and the placebo group. There was no statistically significant difference in the incidence of noninvasive breast cancers reported in the two treatment groups (16 cases for the raloxifene group vs 7 cases for the placebo group; HR: 1.12; 95% CI: 0.46–2.73; p = 0.80). During the 8 years of the MORE and CORE trials, the overall incidence of breast cancer, regardless of invasiveness, was reduced by 58% in the raloxifene group compared with the placebo group (HR: 0.42; 95% CI: 0.29–0.60; p = 0.001).
RUTH trial
In the RUTH trial, raloxifene reduced the risk of invasive breast cancer in lower-risk older women by 44% (HR: 0.56; 95% CI: 0.38–0.83) [13]. Similar to the findings in the MORE and CORE trials, raloxifene primarily reduced the incidence of ER-positive invasive breast cancer: the absolute risk reduction per 1000 women treated with raloxifene for 1 year was 1.2 cases of ER-positive invasive breast cancer.
STAR trial
After a median of 3.2 years of therapy in the STAR trial, there were 163 cases of invasive breast cancer in women assigned to tamoxifen and 168 in those assigned to raloxifene (incidence: 4.30/1000 vs 4.41/1000; RR: 1.02; 95% CI: 0.82–1.28) [14]. The cumulative incidence through 72 months for the two treatment groups was 25.1 and 24.8/1000 for the tamoxifen and raloxifene groups, respectively (p = 0.83). When the treatment groups were compared by baseline categories of age, history of LCIS, history of atypical hyperplasia, Gail model 5-year predicted risk of breast cancer [33,34] and the number of relatives with a history of breast cancer, the pattern of no differential effect by treatment assignment remained consistent. There were no differences between the treatment groups in regard to distributions by tumor size, nodal status or ER level.
Breast cancer risk reduction in studies with raloxifene.
CORE: Continuing Outcomes Relevant to Evista® trial; MORE: Multiple Outcomes of Raloxifene Evaluation trial; RUTH: Raloxifene Use for The Heart trial; STAR: Study of Tamoxifen And Raloxifene trial.

Cumulative coronary heart disease events (A) and invasive breast cancers (B) in the RUTH trial.
There were fewer cases of noninvasive breast cancer in the tamoxifen group (57 cases) than in the raloxifene group (80 cases) (incidence: 1.51 vs 2.11/1000; RR: 1.40; 95% CI: 0.98–2.00). Cumulative incidence over 6 years was 8.1/1000 in the tamoxifen group and 11.6 in the raloxifene group. Approximately 36% of the cases were LCIS and 54% were ductal carcinoma in situ (DCIS), with the balance being mixed types. The pattern of fewer cases among the tamoxifen group was evident for both LCIS and DCIS.
Safety and tolerability
MORE trial
In the MORE trial, raloxifene increased the risk of venous thromboembolic disease (RR: 3.1; 95% CI: 1.5–6.2), but did not increase the risk of endometrial cancer (RR: 0.8; 95% CI: 0.2–2.7).
CORE trial
During the CORE trial, the RR of thromboembolism in the raloxifene group (2.9 events/1000 woman-years) compared with those in the placebo group (1.3 events/1000 woman-years) was 2.17 (95% CI: 0.83–5.70) for the CORE enrollees and 3.11 (95% CI: 0.92–10.44) for the 3200 CORE enrollees who resumed taking study medication during the CORE trial. During the 8 years of the MORE and CORE trials, the incidence rate for venous thromboembolic events was 2.2 and 1.3 events/1000 woman-years for the raloxifene and placebo groups, respectively. During the CORE trial, no cases of pulmonary embolism were reported in the placebo group, compared with nine cases reported in the raloxifene group (p = 0.066). During the 8 years from randomization in MORE until the end of the CORE trial, two women assigned to placebo (0.16%) and 17 women assigned to raloxifene (0.62%) developed a pulmonary embolism (p = 0.048). One death due to pulmonary embolism was reported in the raloxifene group during the 4 years of the CORE trial.
RUTH trial
In the RUTH trial, four common adverse events (an acute coronary syndrome, anxiety, constipation and osteoporosis) were reported more frequently in the placebo group than in the raloxifene group and seven symptoms or adverse events (i.e., arthritis, cholelithiasis, dyspepsia, hot flushes, intermittent claudication, muscle spasm and peripheral edema) were reported more frequently in the raloxifene group than in the placebo group. Hot flushes, leg cramps, peripheral edema and gallbladder disease were all statistically significantly more common in women assigned to raloxifene than to placebo. The rates of cholecystectomy did not differ significantly between the treatment groups (p = 0.25). The incidences of endometrial cancer and all cancers other than breast cancer did not differ significantly between treatment groups.
Although there was no significant difference in the rates of death from any cause or total stroke according to group assignment, raloxifene was associated with an increased risk of fatal stroke (59 vs 39 events; HR: 1.49; 95% CI: 1.00–2.24; absolute risk increase: 0.7/1000 woman-years) and venous thromboembolism (HR: 1.44; 95% CI: 1.06–1.95; absolute risk increase: 1.2/1000 woman-years).
The RUTH trial did not incorporate a global index that weighed risks and benefits as was reported in the BCPT [4], but comparison of the absolute reductions and increases in individual end points allows assessment of the net risk:benefit balance [35]. The rate of invasive breast cancer was reduced by 44%, which translates to 1.2 fewer cancers/1000 women treated with raloxifene/year among low-risk, older postmenopausal women. The rate of clinical vertebral fractures was 3.7/1000 women/year, and the absolute reduction was 1.3 fractures/1000 women-years. These two benefits came at a cost of 1.2 more cases of venous thromboembolism and 0.7 excess fatal strokes/1000 women-years. For women in the RUTH cohort, which was comprised of individuals with or at increased risk for CHD, the moderate benefits of raloxifene for breast cancer risk reduction appeared to some observers not to justify the risks [35].
STAR trial
In the STAR trial, patient-reported symptoms were collected from all participants using a 36-item symptom checklist [15]. Quality of life was measured with the Medical Outcomes Study Short-Form Health Survey (SF-36) [36–39], the Center for Epidemiologic Studies – Depression (CES-D) [40] and the Medical Outcomes Study Sexual Activity Questionnaire [41] in a substudy of 1983 participants with a median follow-up of 5.4 years (range: 4.6–6.0 years). Questionnaires were administered before treatment, every 6 months for 60 months and at 72 months. Primary quality-of-life end points were the SF-36 physical (PCS) and mental (MCS) component summaries.
Among women in the quality-of-life analysis in STAR, mean PCS, MCS and CES-D scores worsened modestly throughout the study, with no significant difference between the tamoxifen and raloxifene groups. Sexual function was slightly better for participants assigned to tamoxifen. Of the women in the symptom assessment analyses, those in the raloxifene group reported greater mean symptom severity over 60 months of assessments than the women in the tamoxifen group for musculoskeletal problems, dyspareunia and weight gain. Women in the tamoxifen group reported greater mean symptom severity for gynecological problems, vasomotor symptoms, leg cramps and bladder control symptoms. However, no significant differences existed between the tamoxifen and raloxifene groups in patient-reported outcomes for physical health, mental health and depression, although the tamoxifen group reported better sexual function.
There were 36 cases of uterine cancer with tamoxifen and 23 with raloxifene (RR: 0.62; 95% CI: 0.35–1.08). No differences were found for other invasive cancer sites, ischemic heart disease events or stroke.
Thromboembolic events (i.e., pulmonary embolism and DVT) occurred less often in the raloxifene group (RR: 0.70; 95% CI: 0.54–0.91). The absolute rate of venous thromboembolism was significantly lower among women assigned to raloxifene (2.6/1000) than among those assigned to tamoxifen (3.7/1000). The cumulative incidence of serious clotting events at 6 years was 21.0/1000 for the tamoxifen group and 16.0/1000 for the raloxifene group. Pulmonary embolism and DVT occurred in 54 versus 35 women (RR: 0.64; 95% CI: 0.41–1.00) and in 87 versus 65 women (RR: 0.74; 95% CI: 0.53–1.03) assigned to tamoxifen and raloxifene, respectively.
There were 53 stroke events associated with tamoxifen in the STAR trial and 51 stroke events among the women receiving raloxifene.
The number of osteoporotic fractures in the groups was similar. There were fewer cataracts (RR: 0.79; 95% CI: 0.68–0.92) and cataract surgeries (RR: 0.82; 95% CI: 0.68–0.99) in the women receiving raloxifene. There was no difference in the total number of deaths or in causes of death.
Regulatory affairs
Raloxifene is approved by both the EU and the US FDA for the prevention and treatment of osteoporosis in postmenopausal women. Based on the extensive clinical experience reported to date, approval is being sought both in the USA and Europe for the use of raloxifene to reduce the risk of invasive breast cancer in high-risk, postmenopausal women. These women would include those with high quantitative risks for breast cancer, those with LCIS or atypical ductal or lobular hyperplasia, and those with a history of breast cancer in a first-degree female relative (mother, sister or daughter). Approval is expected in 2007 for this indication. As there are no clinical data in premenopausal women regarding either the safety or efficacy of raloxifene, its use in this population is not recommended and no evaluations of raloxifene in premenopausal women are planned.
Conclusion
The findings from the MORE and CORE trials suggest that raloxifene therapy is associated with a reduced risk of invasive breast cancer in postmenopausal women irrespective of the presence or absence of risk factors, but that its effects are greater in women with a family history of breast cancer. The cumulative results from all of the trials considered together indicate that both absolute benefits and absolute risks will vary depending on the risk profiles of women receiving treatment. Raloxifene does not significantly affect the risk of cardiac outcome events despite its known favorable effects on serum lipids. In addition, in the older population of women in RUTH who were at low-to-average risk of breast cancer, the benefits of raloxifene in reducing the risks of invasive breast cancer and vertebral fracture were reduced by the increased risks of venous thromboembolism and fatal stroke. The small difference (0.6/1000 woman-years) in the rate of noninvasive breast cancer reported in the STAR trial when comparing tamoxifen with raloxifene will have a small impact on the estimate of the net benefit of raloxifene for reducing the risk of breast cancer given that noninvasive breast cancer is not a life-threatening event and should not diminish the perceived utility of raloxifene for this indication.
Raloxifene is a unique SERM with distinct activity and toxicity profiles. Extensive experience from prospective investigations has established its safety and efficacy in the management of postmenopausal osteoporosis. Although it has no apparent beneficial effect on CHD, it has no adverse effect either. Three prospective clinical trials have established its benefit in reducing the risk of invasive breast cancer, and it offers safety advantages when compared with tamoxifen in postmenopausal women who are at increased risk for breast cancer. Symptomatic side effects are acceptable as reported in the large, prospectively blinded clinical populations summarized here and followed during years of raloxifene administration. The risk of other cancers, fractures, ischemic heart disease and stroke are similar for both raloxifene and tamoxifen. Raloxifene thus offers an alternative to tamoxifen for the reduction of breast cancer risk in high-risk postmenopausal women with a superior risk:benefit profile based upon the benefits and risks reviewed here.
Future perspective
SERMs will retain their role for the management of postmenopausal osteoporosis and in the reduction of the risk of postmenopausal breast cancer until ongoing trials of aromatase inhibitors establish them as more effective, safer agents in postmenopausal women who are at increased risk of breast cancer.
Information resources
American Society of Clinical Oncology offers news and meeting reports, technology assessments, education and training, practice resources and updates on legislative activities for cancer professionals.
National Cancer Institute of the USA provides information for both health professionals and the public. Information includes cancer topics and statistics, clinical trials, funding opportunities, and advisory boards and groups.
American Cancer Society has comprehensive information for both the public and professionals including cancer facts and figures, statistics, research programs and funding opportunities, treatment decision tools, information regarding clinical trials, a bookstore and an international program.
National Comprehensive Cancer Network is an alliance of 19 of the world's leading cancer centers and provides an authoritative resource for both healthcare professionals and clinicians; information includes Clinical Practice Guidelines in Oncology™ and a compendium of drugs and biologicals. They provide treatment guidelines for patients and resource lines for physicians, including information regarding clinical trials.
Eli Lilly and Company provides summaries of completed clinical studies and/or references to publications or scientific presentations by product.
Center Watch is a clinical trials listing service providing information regarding new drugs for both health professionals and the public; there are clinical trial results, a drug directory and a bookstore for both patients and professionals.
Effects on the skeleton
Three prevention studies, two arms of the Multiple Outcomes of Raloxifene Evaluation (MORE) trial and three additional treatment studies, showed that the effects of raloxifene 60 mg/day and 120 mg/day pooled with 150 mg/day reduce the risk of veterbral fracture by 40% for raloxifene 60 mg/day and 49% for raloxifene 120 and 150 mg/day. In the Study of Tamoxifen And Raloxifene (STAR) trial, the effect of raloxifene on the risk of fracture was comparable to that of tamoxifen. These data indicate that raloxifene consistently reduces the risk of vertebral fracture in postmenopausal women.
Raloxifene does not appear to cause adverse effects on lipids and lipoproteins. In the Continuing Outcomes Relevant to Evista® (CORE) trial, the incidence of serious cardiovascular adverse events during 8 years was compared between treatment groups, and the 8-year incidence of serious cardiovascular adverse events did not differ significantly between the raloxifene and placebo groups.
Although fatal strokes were increased among raloxifene users in the Raloxifene Use for The Heart (RUTH) trial, no difference in stroke deaths was seen in the STAR trial.
Compared with placebo in postmenopausal women at average risk of breast cancer, raloxifene reduced the risk of invasive breast cancer by 44–76%.
Among younger postmenopausal women who were at increased risk of breast cancer, raloxifene was as effective as tamoxifen in reducing the risk of invasive breast cancer.
Raloxifene appeared to be less effective than tamoxifen in reducing the risk of in situ breast cancer, although the results were not statistically significant.
In high-risk, younger postmenopausal women, raloxifene appears to offer net benefit when weighing reduction of the risk of breast cancer and the prevention of fractures with the risk of stroke, venous thromboembolic events, uterine events and symptomatic side effects.
In the MORE and CORE trials, there was no evidence of an increased risk of invasive uterine malignancy when comparing women receiving raloxifene with those receiving placebo.
In the STAR trial, there were statistically significant reductions in the incidence of both invasive uterine malignancy and hysterectomy when comparing women taking raloxifene with those taking tamoxifen.
In STAR, the risk of pulmonary embolism and deep venous thrombosis was 30% less among the women in the raloxifene group compared with those in the tamoxifen group.
Although there was no significant difference in the rates of death from any cause or total stroke according to group assignment, raloxifene was associated with an increased risk of both fatal stroke and venous thromboembolism in the RUTH trial.
In the STAR trial, the incidence of stroke with raloxifene was identical to the risk with tamoxifen, although there were more fatal strokes with raloxifene, as was also seen in the RUTH trial.
Compared with placebo, raloxifene causes an increase in hot flashes, leg cramps, peripheral edema and gallbladder disease.
Compared with tamoxifen, there are more musculoskeletal symptoms, dyspareunia and weight gain with raloxifene, although the differences are not large. Vaginal bleeding is not increased by raloxifene when compared with placebo.
