Abstract
Female sexual dysfunction is a common disorder that is present in over 40% of women in the USA. An evaluation of female sexual dysfunction differs greatly from male erectile dysfunction in that female sexual dysfunction is a very general term that encompasses a variety of sexual complaints in women, rather than a specific physiological disorder. No pharmacological treatment is presently approved for the treatment of the myriad of disorders that are involved in female sexual dysfunction. One of the more common disorders of female sexual dysfunction involves disturbances in objective or subjective sexual arousal. Sexual arousal in women has a central component and a peripheral, or vulvar, component. Disorders in vulvar blood flow and stimulation may lead to decreased genital and central arousal. A logical corollary of this statement is that an improvement in local blood flow by the use of a local or systemic vasodilator may lead to an increase in overall sexual arousal and an increase in sexual satisfaction. This article will explore the use of topical alprostadil as a local vasodilator to improve sexual arousal and sexual satisfaction in women with female sexual arousal disorder.
There has been increased interest in the past decade in evaluating the prevalence and treatment of female sexual dysfunction (FSD). FSD is a very general term encompassing a variety of sexual conditions that cause distress in women. It is often difficult to generalize a discussion of FSD in terms of diagnosis, evaluation and treatment due to the variety of conditions that are ‘lumped’ into the term FSD. Several studies have attempted to quantify the prevalence of FSD, which is present in 20–45% of women in the USA [1,2]. This prevalence would extrapolate to 30–40 million US women affected by the myriad disorders that represent FSD. Unfortunately, little new information has come to light regarding the efficacy and safety of methods used to treat FSD. Treatments have focused primarily on psychological therapy and counseling, with little basic research on the etiology of FSD. In many ways, research efforts directed at developing effective and safe pharmacological therapy for the treatment of FSD have been aided by the interest in and successful treatment of erectile dysfunction (ED) in men. Despite this increase in interest, no US FDA- or European Agency for the Evaluation of Medicinal Products (EMEA)-approved pharmacological therapy is available for the treatment of FSD. Basic science and clinical research efforts aimed at developing effective pharmacological therapies to treat FSD lag 10–15 years behind similar research efforts in male sexual dysfunction. This article will examine the progress of several attempts to develop effective pharmacological therapy for women with FSD, with specific emphasis on the use of topical alprostadil to treat female sexual arousal disorder (FSAD).
Classification of female sexual dysfunction
FSD is far more difficult to define than ED, as FSD represents a variety of disorders and therefore is subject to significant variability in terms of classification, evaluation and treatment (Box 1) [2,3]. As stated, a variety of disorders are classified in the
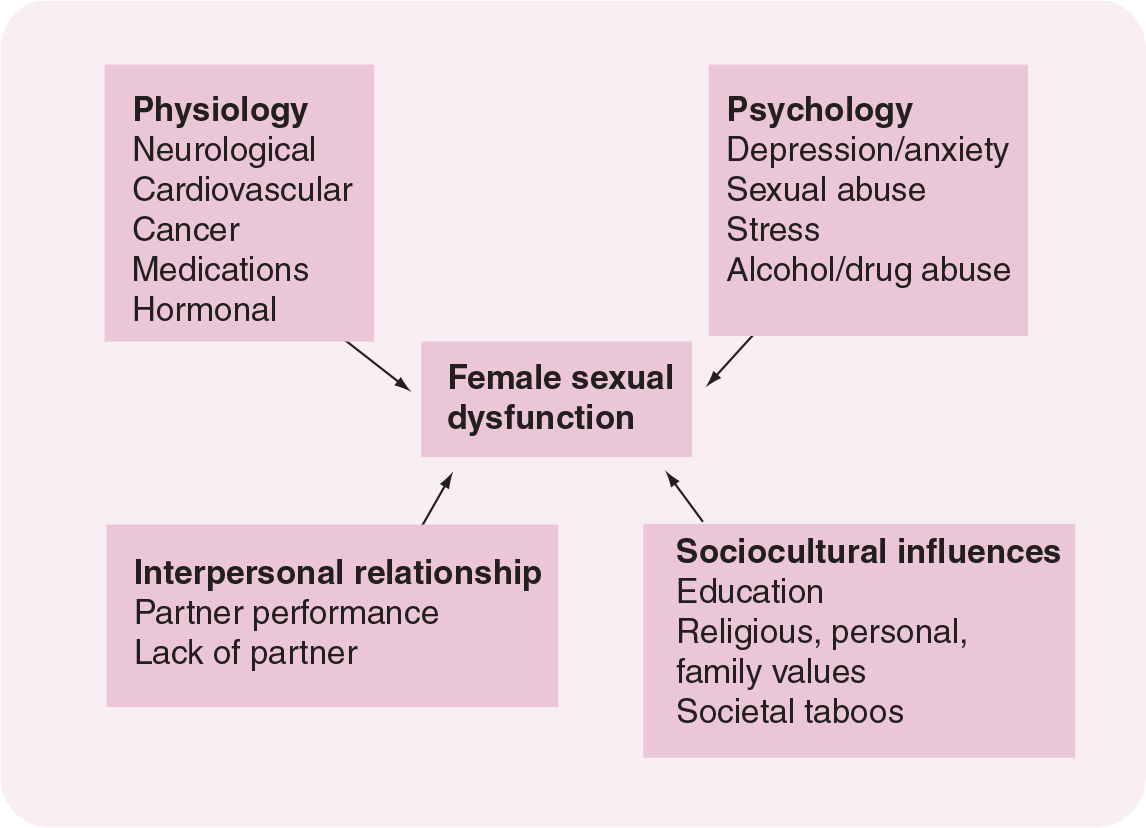
Multiple variables influencing female sexual function.
Classification of female sexual disorders.
Female sexual interest/desire disorder
Subjective sexual arousal disorder
Genital sexual arousal disorder
Combined sexual arousal disorder
Women's orgasmic disorder
Dyspareunia
Vaginismus
Sexual aversion
Persistent sexual desire disorder
From WHO 2nd International Consensus Conference On Men's And Women's Sexual Dysfunction. Paris (2003). Adapted from [3].
Female sexual arousal disorder
One of the more common disorders encompassing FSD involves sexual arousal problems in women. Arousal disorders occur in 19–30% of women aged 18–60 years (Table 1) and, interestingly, occur across different cultures [1,2,7–9]. In general, arousal disorders occur in women with an inadequate genital lubrication/swelling response or vaginal dryness during sexual response. There is also increasing agreement that sexual arousal requires cognitive as well as genital input. Disorders that specifically relate to inadequate genital arousal may involve a significant organic component (neurovascular) and may therefore be responsive to pharmacological therapy. The subepithelial region of the vaginal wall contains a dense network of capillaries that perfuse the vaginal epithelium [10–12]. In the nonaroused state, arterioles supplying the vaginal epithelium are constricted and AV anastomoses are unoccluded. This results in minimal blood flow, low surface partial pressure of oxygen in arterial blood (pO2), acidic pH and minimal fluid secretion. During sexual stimulation, there is a rapid increase in arterial inflow and a constriction of the AV anastomoses, leading to vasocongestion of the vaginal tissues, increased surface lubrication, increased pH and pO2, and an increase in vaginal sodium and chloride concentration toward that found in plasma [10–12]. Since dilatation of these vessels is central to the changes that occur during sexual arousal, vasodilators, both systemic and locally applied, may facilitate the arousal response and serve as an effective agent to treat FSAD.
Physiological effects of alprostadil
Alprostadil (prostaglandin [PG]E1) is a potent vasodilator that has effects not only on vascular smooth muscle but also as a local neuromodulator. It is an ubiquitous compound present in almost all tissues and is conserved evolutionarily in animals as diverse as Australian crickets and humans [13,14]. PGs are a diverse group of naturally occurring biologically active molecules that tend to be local mediators of blood flow and neuroregulation. Different receptor types mediate different biological activities. Table 2 outlines the variability of PG receptors as well as their biological activity. The female reproductive tract is exposed to high concentrations of PGE1 and other PGs present in semen. Pharmacologically, PGE2 is used for induction of cervical ripening at term labor and for the termination of pregnancy [15]. In addition, the female reproductive tract has been exposed to high levels of alprostadil from male partners using intraurethral alprostadil for the treatment of ED. Local and systemic toxicological studies in animals exposed to levels as high as 2000 μg/day via the intravaginal route demonstrate no local or systemic toxicity [16]. As expected, there are related increases in local blood flow and erythema but no related histological abnormalities. Long-term toxicology studies in dogs receiving alprostadil 1000 μg intravaginally did show nondose-related fibrotic liver changes in one study, that were preceded by elevated liver enzymes. This liver toxicity was believed to be a study anomaly and further studies have confirmed that the liver abnormalities did not represent a significant safety issue for adult women [16]. Reproductive toxicology studies in rabbits demonstrated no maternal toxicity or embryotoxicity, although there was a slight decrease in fetal weight at the highest dose [17,18].
Prostaglandin receptor subtypes and biological activity.

cAMP: Cyclic adenosine monophosphate; PG: Prostaglandin; PI: Inositol phospholipid.
Alprostadil for the treatment of erectile dysfunction
The modern era of effective pharmacotherapy for the treatment of ED began in 1982 with the introduction of intracavernosal injections (ICI) of papaverine [19,20]. Successful ICI programs ensued despite a lack of understanding of the anatomic, cellular and molecular pathways initiating and maintaining erection. ICI programs with papaverine yielded successful response rates as high as 75–90% [21–24]. Significant local complications with intracavernosal papaverine, including priapism and penile fibrosis, occurred in up to 30% of patients. Other compounds were studied in an attempt to duplicate the high efficacy of intracavernosal papaverine while decreasing the incidence of local side effects. Alprostadil, a synthetic form of PGE1, was used as intracavernosal monotherapy and in combination with papaverine with significant success. The FDA approved intracavernosal alprostadil for the treatment of ED in the USA in 1996.
Patient satisfaction with ICI programs was low. The incidence of local side effects and the complexity of ICI led to patient drop-out rates of 50%. To improve patient and clinician satisfaction alternate drug delivery methods were studied. Topical and intraurethral application of PG cream demonstrated moderate success. Large-scale Phase II and III studies using an intraurethral pellet of alprostadil began in 1991 [25]. These safety and efficacy trials were completed in 1995 and FDA approval was granted in the USA in December 1996.
Alprostadil delivered either by ICI or intraurethral suppository remained the leading treatment regimen for ED until the introduction of oral sildenafil in March 1998.
Pharmacodynamics of alprostadil in male cavernosal tissue
PGE1 is a naturally occurring lipid compound found in high concentrations (100–200 μg/ml) in seminal fluid. Alprostadil is the synthetic form of PGE1. The chemical name of alprostadil is prost-13-en-1-oic-acid, 11,15-dihydroxy-9-oxo-(11α,13E,15S)-(1
Pharmacokinetics in male corporal tissue
PGE1 is rapidly metabolized in the corpus cavernosum after ICI or intraurethral application by enzymatic oxidation. The principle metabolite, 15-keto PGE1, has little biological activity (1–2%). Systemic alprostadil delivered to the extrapenile circulation is rapidly metabolized on first pass through the lungs. Metabolites are principally excreted (88%) by the kidneys, with no evidence of tissue retention. The half-life of alprostadil is rapid, between 30 s and 10 min [29].
Mechanism of alprostadil
The mechanism by which alprostadil initiates cavernosal smooth muscle relaxation was elucidated in 1994 when the alternate adenylate cyclase pathway of cavernosal smooth muscle relaxation was described. Alprostadil binds directly to PG receptors on cavernosal smooth muscle cells. PGE1 initiates adenylate cyclase activity to convert ATP to cyclic adenosine monophosphate (cAMP), decreasing intracellular calcium ions in the cavernosal smooth muscle cells. Myofilament deactivation causes cavernosal smooth muscle relaxation. Once the cavernosal smooth muscle relaxes, blood flows into the cavernosal sinusoids, filling the corpora and compressing the emissary veins against the tunica albuginea. At the point where venous outflow is at a minimum and the sinusoids are filled, the penis becomes rigid. This mechanism may also induce vasodilatation in other vascular beds, including those present in the vaginal walls and clitoris.
Clinical studies using topical alprostadil as treatment for female sexual arousal disorder
Several studies have been completed using topical alprostadil to improve objective and subjective sexual arousal in women with FSAD. Preclinical studies have demonstrated that the application of alprostadil to the vulva significantly increases vulvar and clitoral blood flow. Visual inspection and vulvar Döppler measurements demonstrate a significant increase in blood flow in healthy women and women with FSAD after the application of alprostadil cream/gel or alprostadil in a liquid dispersant [30,31]. This increase in vulvar vasocongestion is highly desirable to stimulate the local reaction associated with sexual arousal. As a prelude to clinical studies looking to improve subjective arousal, alprostadil must demonstrate the ability to improve vulvar blood flow in order to initiate or improve sexual arousal at the level of the CNS. Since these preclinical studies have demonstrated that topical alprostadil does indeed improve vulvar blood flow, in-clinic and at-home studies were performed to further evaluate the role of topical alprostadil in the treatment of FSAD.
In-clinic trials in patients with female sexual arousal disorder
In a multicenter, randomized, placebo-controlled, crossover-design study to evaluate the effects of a topical alprostadil solution in the treatment of FSAD, healthy women aged 35–70 years who were either postmenopausal or posthysterectomy with oophorectomy and who had a diagnosis of FSAD based on history, physical exam and psychological screening were included [32]. The study also required that subjects had a normal pelvic exam within the previous 6 months.
A total of 110 individuals were screened. Of subjects admitted to the trial, 77 received alprostadil, 77 received placebo and 75 received both alprostadil and placebo.
Study design
Treatment for each study subject included one dose of alprostadil at the assigned dosage level and one dose of placebo administered by the subject after instruction in random order on separate clinic visits at least 72 h apart. Active and placebo doses were administered in the clinic by local application to the clitoris, the periurethral area, the vaginal introitus, the outer vagina and the internal surfaces of the labia minora.
Immediately following each dose of study medication, subjects viewed a 60-min video that contained, in order, 10 min of neutral video content, 20 min of visual sexual stimulation and 30 min of neutral video content.
Subjects selected videos from three categories (heterosexual, homosexual or bisexual) to allow for women of different sexual orientations to participate. Each category of videos contained two different videos (one for the first dosing visit and one for the second) that were balanced for content, and the study staff were blinded to the specific category of video stimulation selected by each study subject.
Immediately before dosing, and at 5, 15, 30, 45, 60, 90 and 120 min after dosing, subjects rated their genital wetness or lubrication, pelvic fullness, warmth and/or tingling, degree of pain, level of sexual arousal, satisfaction with level of sexual arousal, level of sexual satisfaction and overall level of physical comfort. Vital signs were assessed remotely, so as not to disturb subjects' sexual responses. Investigators also assessed the subject's genital erythema and edema immediately before dosing and at 60 and 120 min after dosing.
Alprostadil 100 and 400 μg doses were administered in volumes of 0.11 and 0.44 ml, applied to the external genitalia. Placebo doses consisted of a vehicle control solution administered at equal volumes to alprostadil doses.
Measures of treatment success
The sample was described with demographic variables, sexual function (Female Sexual Function Index [FSFI]) [33] and sexual distress (Female Sexual Distress Scale [FSDS]) [34] scales. The response to alprostadil was measured by observer ratings of genital vasocongestion, as evidenced by visually perceptible erythema and edema, and by subject ratings of sexual arousal, somatic sensations typically associated with sexual arousal and sexual satisfaction.
Observer ratings of genital erythema and edema were made immediately prior to each dose of study medication and at 60 and 120 min following each dose. These ratings were made using a four-point categorical rating scale.
Categorical subject ratings of genital wetness/lubrication, sensations of pelvic fullness, genital warmth/tingling, level of sexual arousal, satisfaction with sexual arousal and sexual satisfaction were assessed immediately before dosing and at 5, 15, 30, 45, 60, 90 and 120 min after each dose of study medication. These assessments were made using a five-point categorical scale. At these time points, subjects also rated levels of internal and external pain using similar categorical scales, and their overall physical comfort using a categorical scale with responses of ‘very comfortable’, ‘comfortable’, ‘neutral’, ‘uncomfortable’ and ‘very uncomfortable’. The use of categorical rating scales in research involving genital and subjective measures of sexual arousal in a controlled laboratory setting has been demonstrated to provide meaningful data regarding women's sexual response [35,36].
The safety of alprostadil was based on the incidence of adverse events and changes from screening to the end of the study in laboratory test values. Blood pressure and heart rate were measured, with the subject either seated or reclined, immediately before and at 5, 15, 30, 45, 60, 90 and 120 min after each dose of study medication.
Baseline arousal domain scores from the FSFI questionnaire confirmed that study subjects had a diminished ability to attain satisfactory arousal. FSFI questionnaire assessments also showed lower scores on study subjects' levels of desire and ability to achieve orgasm. These data therefore reinforce the importance of a careful medical history and psychological evaluation to rule out primary desire or orgasmic disorders. Distress levels were well balanced across the two dose groups evaluated in this study.
Both the 100 μg (82%) and 400 μg (79%) doses of alprostadil were associated with higher levels of local vascular engorgement when compared with placebo (18 and 21%, respectively), as evidenced by investigator observations of mild-to-moderate genital erythema. Although the alprostadil 100 μg dose did not differ significantly from placebo for any of the subjective evaluations, the alprostadil 400 μg dose resulted in significantly greater changes in genital warmth/tingling (p = 0.002), level of sexual arousal (p = 0.017), satisfaction with level of sexual arousal (p = 0.015) and level of sexual satisfaction (p = 0.021) than did placebo (Figure 2). These findings were based on the summed changes from baseline over the 120-min observation period. In addition, at the 30-min time point, just after the visual sexual stimulation ended, the 400 μg dose subjects reported ‘moderate’ or greater responses at 15–27% above placebo responses, with significant effects for pelvic fullness (p = 0.002), genital warmth and tingling (p = 0.006), and sexual satisfaction (p = 0.02). Thus, although this study was conducted entirely in the clinic and utilized visual sexual stimulation as opposed to subjects' at-home sexual activity, the 400 μg dose of alprostadil resulted in significant increases over placebo in both somatic sensations typically associated with sexual arousal and subject reports of sexual arousal and satisfaction.

Change from baseline of end points for in-clinic trial of alprostadil for the treatment of female sexual arousal disorder.
Adverse events commonly reported in this study were consistent with the known effects of alprostadil on blood vessels and sensory nerves, and included genital erythema, genital edema and genital burning. Moreover, genital burning was observed following placebo as well as alprostadil, indicating that some other ingredient in the topical product may be at least partially responsible for these effects. Importantly, overall comfort ratings with alprostadil did not differ significantly from placebo, and no subjects discontinued treatment due to adverse effects. Overall, alprostadil was well tolerated by this group of postmenopausal or hysterectomized women with FSAD.
The lack of significant effects on the wetness/lubrication assessment suggests that, in spite of this being criteria for the major diagnostic systems [37], it does not capture as much of the perceived change during sexual stimulation as do other variables. Subjective awareness of lubrication may be of lesser importance in terms of contributing to overall arousal than other objective genital responses such as warmth and erythema. This finding underscores the need to consider diagnostic revisions that address more facets of women's sexual experience [38].
The present study did not use vaginal photo-plethysmography [37,39,40] or other invasive vaginal vascular/physiological instrumentation to measure arousal response since the subjective assessments of physical genital sensations, arousal and satisfaction were more clinically relevant end points. The insertion of a vaginal probe to assess vaginal pulse amplitude may have actually interfered with the collection of these more important study measures. An examination of efficacy and partner response to the treatment in future studies would be important to clarify the extent to which topical alprostadil is effective and useful for sexual arousal complaints in women.
At-home trials using alprostadil for the treatment of female sexual arousal disorder
Phase II trials using alprostadil in a liquid dispersant have been performed in an at-home setting to improve sexual arousal in women with FSAD [41–43]. These studies are more relevant than in-clinic studies, as they represent an evaluation of the efficacy and safety of alprostadil in a more realistic home setting and measure the outcomes of sexual encounters using subject diaries as well as validated questionnaires. In addition, these double-blind, placebo-controlled studies were of long duration with multiple doses and dose ranges. A total of 269 naturally or surgically postmenopausal women with a diagnosis of FSAD (as ascertained by the principle investigator by interview and examination) were enrolled. After a run-in period, women were randomized to receive placebo, topical alprostadil 200, 400 or 800 μg over a 2-month period. Women were required to be sexually active and use a minimum of six doses. The primary study end point was the number of sexual encounters resulting in satisfactory arousal/excitement and/or orgasm. A statistically greater percentage of satisfactory events occurred in women receiving alprostadil over placebo for this end point (Figure 3). The most common adverse event was application-site burning, reported by 44–57% of patients. The majority of these cases were graded as ‘mild’ in intensity and lasted only minutes. There were no dose-related effects on vital signs, laboratory evaluations or physical exam findings. Less than 10% of patients discontinued treatment due to application-site discomfort. A similar study was performed in premenopausal women with FSAD. Premenopausal women aged 21–50 years with FSAD involved in a stable monogamous relationship were included [44]. Patients had mild-to-moderate FSD with FSDS scores of less than 60 and a Beck Depression Inventory score of less than 20. The study was a randomized, placebo-controlled design with crossover using placebo or topical alprostadil 400 μg. A total of 51 patients were randomized and, after the crossover was completed, a population of 25 patients who had received at least six doses of study drug were studied. Topical alprostadil was associated with a statistically significant increase in the number of satisfactory sexual events over placebo, as well as significant increases or trends in other diary items and questionnaire scores (Figure 4). Once again, there were no systemic side effects and the majority of application side effects (burning) were rated as mild and transient at the same frequency as demonstrated in the in-clinic studies (Table 3). Only one subject discontinued the study due to application-site discomfort. Larger, longer-term studies are needed to better assess the efficacy and safety of topical alprostadil in the at-home treatment of FSAD.
Adverse effects of topical alprostadil for the treatment of female sexual arousal disorder.

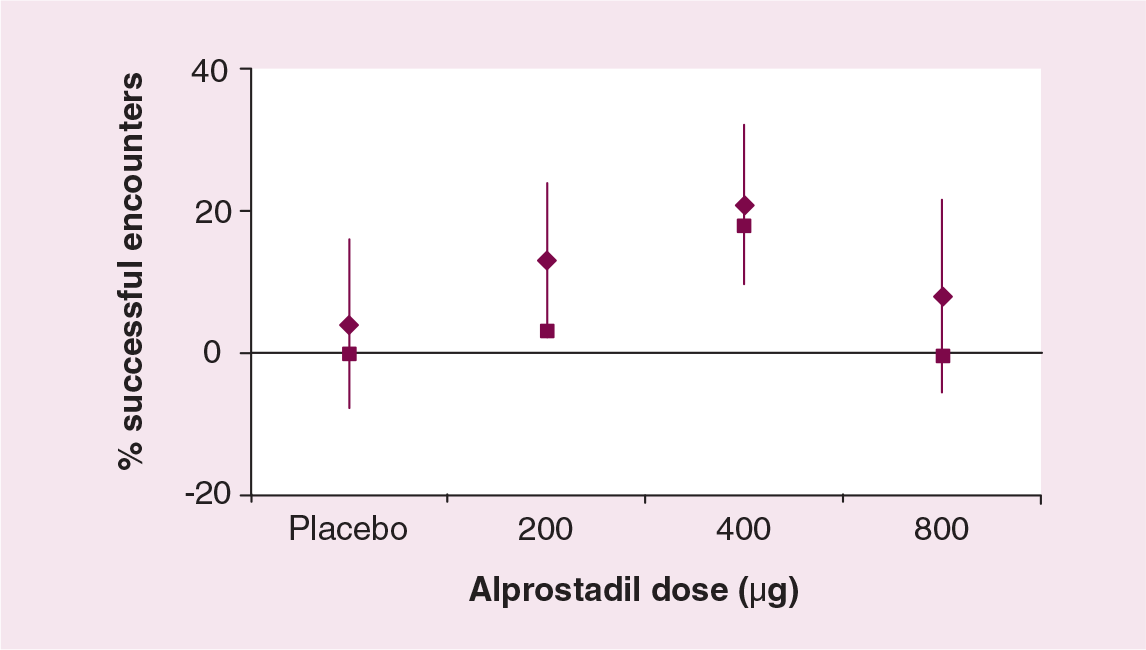
Percentage of encounters resulting in satisfactory arousal/excitement and/or orgasm after administration of topical alprostadil (change from baseline).
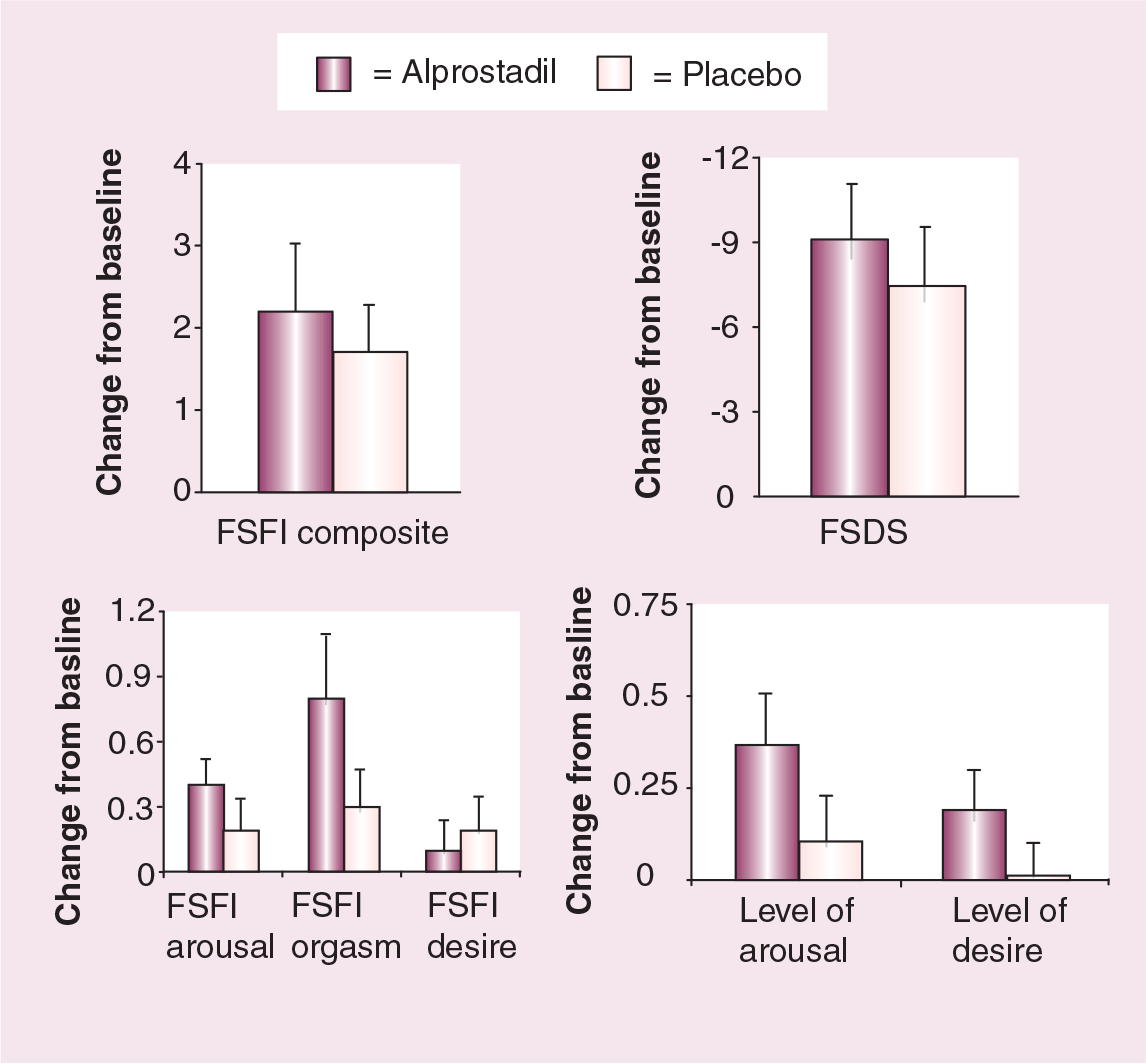
Improvement in validated sexual questionnaire scores/domains after administration of topical alprostadil.
Other formulations of alprostadil have been studied in an attempt to find an effective and safe pharmacological agent for the treatment of FSAD. Alprostadil cream has been studied in healthy women and women with FSAD in an at-home setting. In a large, double-blind, randomized, placebo-controlled, dose-ranging trial performed in China [45], 400 women with FSAD aged 21–65 years were randomly assigned to placebo, alprostadil 500, 700 or 900 μg for a ten-dose home treatment trial. An initial safety and efficacy treatment period was followed by a second evaluation period. The primary end point was improvement in subjective satisfactory sexual arousal during a sexual encounter assessed by validated questionnaire (Female Sexual Encounter Profile question 3). A total of 372 patients completed the study. At the end point of the second evaluation period, primary efficacy values were improved by 28.6, 41.9 (p = 0.053), 40.3 (p = 0.074) and 51.4% (p = 0.0002) in the placebo, alprostadil 500, 700 or 900 μg groups, respectively, over baseline (improved mean arousal success rate from baseline). Changes in baseline of subjects' FSFI and FSDS scores also demonstrated significant improvement or trends for alprostadil over placebo. No serious adverse events were reported and only five patients were withdrawn from the study due to adverse events.
Future perspective
Clearly, the ability of topical alprostadil to increase vaginal, vulvar and clitoral blood flow has been demonstrated in preclinical and clinical trials. Ongoing trials have also demonstrated that sexual satisfaction can be improved in women with FSAD by the application of topical alprostadil. Larger and more extensive trials are needed to fully evaluate the efficacy and safety of alprostadil for the treatment of FSD. Several issues remain before pharmacological intervention for the treatment of FSD is available.
As previously mentioned, isolated abnormalities of female sexual function seldom exist and the majority of women with FSD often have several confounding problems with sexual function. The principle reason that phosphodiesterase type 5 (PDE5) inhibitors, such as sildenafil, were unsuccessful in treating FSAD was that many women treated in the clinical trials had associated hypoactive sexual desire [46]. While PDE5 inhibition may have increased vulvar vasocongestion, it had no effect on the decreased sexual desire and was therefore ineffective at improving sexual satisfaction. After the realization that correct patient selection was the key to performing a useful clinical trial for the treatment of FSD, greater emphasis has been placed on correctly categorizing subjects and including women whose principle concern is sexual arousal disorder. Unfortunately, this selection process is very difficult and requires both an astute investigator and an extensive use of a combination of validated sexual questionnaires. Even if a drug is useful in treating an isolated female sexual disorder, the utility of this drug in a real-world situation where few patients have isolated sexual disorders is questionable.
Another issue complicating drug development is the question of the acceptance of a locally acting agent over a systemically administered drug. Local agents used to treat ED (e.g., intracavernosal/intraurethral alprostadil) have had very limited popularity and are almost solely reserved for use when oral agents fail. Issues of ease of administration, efficacy and local side effects have relegated locally acting agents to less than 5% of the total market for drugs used to treat ED. It remains to be seen if similar issues will arise with local agents designed to treat FSD.
Safety issues are at the forefront in the FDA's decision-making process for the approval of QoL drugs, including drugs to treat sexual dysfunction. In 2004, the FDA denied approval of a testosterone patch as treatment for decreased sexual desire in women. The principle concerns cited by the FDA were the unknown risks with regard to heart disease and breast cancer to women chronically exposed to exogenous testosterone administration [47]. Long-term studies with large numbers of women are needed to assess these safety concerns. A significant safety margin must exist in any drug designed to treat FSD in order for the drug to receive approval from the FDA. Perhaps locally metabolized compounds such as alprostadil will have an advantage due to the lack of systemic effects.
Executive summary
Female sexual dysfunction (FSD) is a common disorder that is present in over 40% of women in the USA, and no approved pharmacological therapy yet exists for the treatment of FSD.
Basic science and clinical research in the etiology and treatment of FSD lags behind male sexual dysfunction by at least a decade.
Clinical trials attempting to develop a treatment for FSD have significant hurdles including designation of successful treatment and institutional concerns regarding safety.
Female sexual arousal disorder (FSAD) may be improved by increasing genital blood flow by the use of systemic or locally applied vasodilators, which subsequently improve overall sexual satisfaction.
Prostaglandins are ubiquitous compounds that are synthesized locally, rapidly metabolized and are responsible for local vascular and neuroregulation.
Topical alprostadil (synthetic prostaglandin E1) has been approved for the treatment of erectile dysfunction in men and functions as a local vasodilator to improve penile blood flow by initiating cavernosal smooth muscle relaxation through the adenylate cyclase pathway.
Topical alprostadil can also improve vaginal blood flow, thus initiating an increase in vaginal lubrication, vaginal pH and vaginal partial pressure of oxygen in arterial blood.
Preclinical studies demonstrate that local application of alprostadil is safe in animal studies and will significantly increase vulvar blood flow when used in humans.
In-clinic studies have shown that 400 μg of alprostadil applied to the vulva significantly increases genital vasocongestion, warmth and tingling, leading to increased sexual arousal over placebo.
At-home studies demonstrate statistically significant increases in the number of satisfactory sexual encounters when 400–900 μg of alprostadil is compared with the administration of placebo in randomized, double-blind, placebo-controlled trials.
Trials have been performed in both pre- and postmenopausal women with similar results.
Several formulations of alprostadil including creams, gels and liquids have been used in clinical trials with similar results.
The most common side effects are local edema and burning.
Side effects are generally mild, transient and easily tolerated.
Systemic side effects are rare, with no changes in heart rate or blood pressure.
Large, long-term trials need to be performed to better determine the safety and efficacy of topical alprostadil for the treatment of FSAD.

