Abstract
Increasingly effective adjuvant therapies mean that the prognosis for postmenopausal women with breast cancer has never been better. Weight problems are common among breast cancer patients and worsen due to the impact of diagnosis and treatment. Recent studies have linked excess weight with the risk of recurrence of breast cancer among premenopausal women. While general obesity (body mass index) does not appear to influence the already much improved prognosis for postmenopausal women, there is some evidence that limiting central obesity and improving insulin resistance may improve survival. The focus of attention for postmenopausal breast cancer survivors is also shifting to consider the mortality and morbidity from other weight-related cancers and noncancer causes, such as cardiovascular disease, making weight control a potentially important adjunct to endocrine therapy. This paper outlines the rationale and optimal design for effective weight management strategies among postmenopausal breast cancer patients receiving endocrine therapy.
Excess adiposity is a well-known factor in the development of breast cancer after the menopause and has also been historically found to influence prognosis [1,2]. Earlier detection and more effective adjuvant treatments mean the likelihood of recurrence and death from postmenopausal breast cancer has decreased significantly. The focus of attention for postmenopausal breast cancer survivors is now shifting to consider mortality and morbidity from other cancers and noncancer causes such as cardiovascular (CV) disease, many of which are linked to adiposity. A significant number of women have excess weight and weight/diet-related comorbidities at the time of diagnosis [3]. The diagnosis and treatment of breast cancer may lead to gains in adiposity and increase the likelihood and severity of these weight-related comorbidities, which may be amenable to preventive treatment. The American Cancer Society has recommended diet, exercise and weight control for breast cancer survivors [4]. This recommendation is not widely accepted by other experts and is not currently incorporated into clinical practice. This paper outlines the rationale for effective weight management strategies among postmenopausal breast cancer patients receiving endocrine therapy for early (operable) breast cancer that is estrogen receptor (ER)-positive.
Early breast cancer in postmenopausal women
Trends in incidence & mortality
Breast cancer is the most common malignancy among women in Western countries, and typically accounts for 25–30% of all female cancers. A total of 80% of women diagnosed with breast cancer are aged 50 or over, and it is women of this age group who are experiencing the greatest increase in breast cancer rates (Fig 1a). The majority of breast cancer (80–95%) among postmenopausal women is localized at the time of diagnosis [5], while nearly three quarters is estrogen responsive. With earlier detection and advances in adjuvant treatment, the prospects of postmenopausal women surviving breast cancer have never been better (Fig 1b). Between 1988 and 2003, breast cancer mortality in the UK fell by 34% for women aged 50–64, 32% for women aged 65–69, and 12% for women over 70 years of age [6]. The most recent and undoubtedly conservative estimates indicate that there are over 2 million women living with a history of breast cancer in the USA [201] and approximately 172,000 in the UK [6]. The long-term health and well being of older cancer patients is thus an increasingly important consideration for healthcare professionals.

Age-specific incidence rates of female breast cancer (A) and breast cancer mortality rates (B) of women in the UK 1975–2001 [6].
Currently recommended treatment for postmenopausal women with early breast cancer
The majority of breast cancer patients have some form of breast surgery, followed by a short course of radiotherapy for those women who have had conservation surgery. Adjuvant endocrine therapy is used in all node-positive and most node-negative women with ER-positive tumors to block the cancer-promoting effect of estrogen. A 5-year course of the selective estrogen receptor modulator (SERM), tamoxifen, was the standard treatment [7]. There is, however, an increasing body of evidence to support the benefits of aromatase inhibitors over tamoxifen with respect to disease-free survival, contralateral breast cancer and decreased risk of vascular events and endometrial cancer [8]. Anastrozole (Arimidex®, AstraZeneca) may be substituted for tamoxifen wherever tamoxifen is contraindicated or not tolerated [7,9]. The clear benefits of switching to any of the aromatase inhibitors, such as exemestane (Aromasin®, Pharmacia), letrozole (Femar®, Novartis) and anastrazole, after an initial period of tamoxifen treatment have recently been demonstrated in the Intergroup Exemestane Study (IES) [10], the MA-17 study [11], the Italian Tamoxifen Arimidex (ITA) study [12] and the Austrian Breast and Colorectal Cancer Study Group (ABCSG) 8/ARimidex–NOlvadex (ARNO) 95 trials [13].
Comorbidities among breast cancer patients
Many postmenopausal breast cancer patients have comorbidities at the time of diagnosis, such as hypertension (25–50%), coronary heart disease (15–27%), vascular disease (3–5%), non-insulin-dependent diabetes mellitus (NIDDM) (5–10%), arthritis (12–25%) and chronic obstructive pulmonary disease (7–12%) [14]. Weight gain after diagnosis and subsequent treatment of breast cancer may exacerbate existing comorbidities or increase the likelihood of additional ones developing.
The stage of breast cancer is the strongest prognositic indicator of survival for breast cancer patients of any age. Comorbidities are, however, a major cause of mortality among women diagnosed with localized breast cancer [14], particularly among older women [3]. A recent Surveillence Epidemiology and End Results program (SEER) report, based on 174,000 postmenopausal women with localized disease, found cumulative probability of death from breast cancer to be between 0.14 and 0.28 over 28 years from diagnosis [3]. The major competing causes of death were CV disease and other cancers. CV disease accounted for 32.3% of deaths in women aged 50–59, 40.5% in women aged 60–69 and 52% in women older than 70 years of age (Table 1).
Percentage of death from causes other than breast cancer among black and white postmenopausal women 1973–2000.
From the Surveillance Epidemiology and End Results (SEER) program, adapted from Schairer et al. (2004) [3].
Weight issues
Prevalence of overweight & obesity among breast cancer patients
There have been dramatic upward trends in the prevalence of overweight (body mass index [BMI] 25–29.9 kg/m2) and obesity (BMI > 30 kg/m2) within both Western and developing countries. In the UK, rates of obesity have particularly increased among postmenopausal women (Figure 2a). Modest increases in central obesity have also been seen among postmenopausal women. Between 1993 and 2002, waist:hip ratio increased from 0.82 to 0.84 (Figure 2b) [202]. Given that excess weight is a risk factor for breast cancer, it is not surprising that the prevalence of overweight and obesity in breast cancer patients has also increased significantly. A total of 63% of postmenopausal early breast cancer patients recruited to the Arimidex or Tamoxifen Alone or in Combination (ATAC) trial between 1996 and 2000 were overweight or obese (39% overweight and 24% obese) [ATAC trialists' group Pers. Comm.], compared with 50% of women recruited to the National Surgical Adjuvant Project for Breast and Bowel cancers (NSABP)-14 trial from 1982–1988 (35% overweight and 15% obese) [15]. These are likely to be conservative estimates, since women recruited into clinical trials are known to be more health conscious than the general population.

The prevalence of overweight and obesity (A) and waist:hip ratio (B) among postmenopausal women in the UK 1993–2001 [143].
Changes in weight & body composition in women receiving adjuvant endocrine therapy
It is estimated that 60% of women gain weight after the diagnosis of breast cancer. On average, modest gains occur in postmenopausal breast cancer patients (approximately 2 kg), which are greater than those seen in the healthy population, although less than those seen with adjuvant chemotherapy [16,17]. There is a perception among patients that endocrine therapies lead to weight gain, yet gains in weight with adjuvant tamoxifen and anastrazole are comparable to gains seen in breast cancer patients not receiving treatment [17–20]. Weight gain is widely believed to reflect the normal effect of inactivity among women recently diagnosed with breast cancer who are recovering from breast surgery and undergoing radiotherapy [20].
Weight change is a poor predictor of the amount of fat, lean body mass and the distribution of fat, particularly among postmenopausal women [21]. Prospective measurements of changes in weight and body composition over the first year of adjuvant anastrozole or tamoxifen among 23 of the authors' postmenopausal early breast cancer patients showed a modest weight gain (mean: 2.6 kg, standard error [SE]: 0.8 kg) but comparatively greater gains in total fat (mean: 4.2 kg, SE: 0.8 kg), particularly central fat (i.e., waist circumference: 4.4 cm, SE: 1.3 cm; and abdominal skin fold: 11 cm, SE: 1.4 mm), and a greater loss of fat free mass (FFM) (mean: −1.6 kg, SE: 0.5 kg), which exceed those previously reported in healthy women (Figure 3) [22]. Gains in body fat and waist circumference will be perceived by women themselves, as they will be conscious of fat redistribution to the abdomen. This may explain the large numbers of women (approximately 40%) who complain of weight gain during adjuvant endocrine therapy [23,24] when seemingly only modest changes in weight occur.
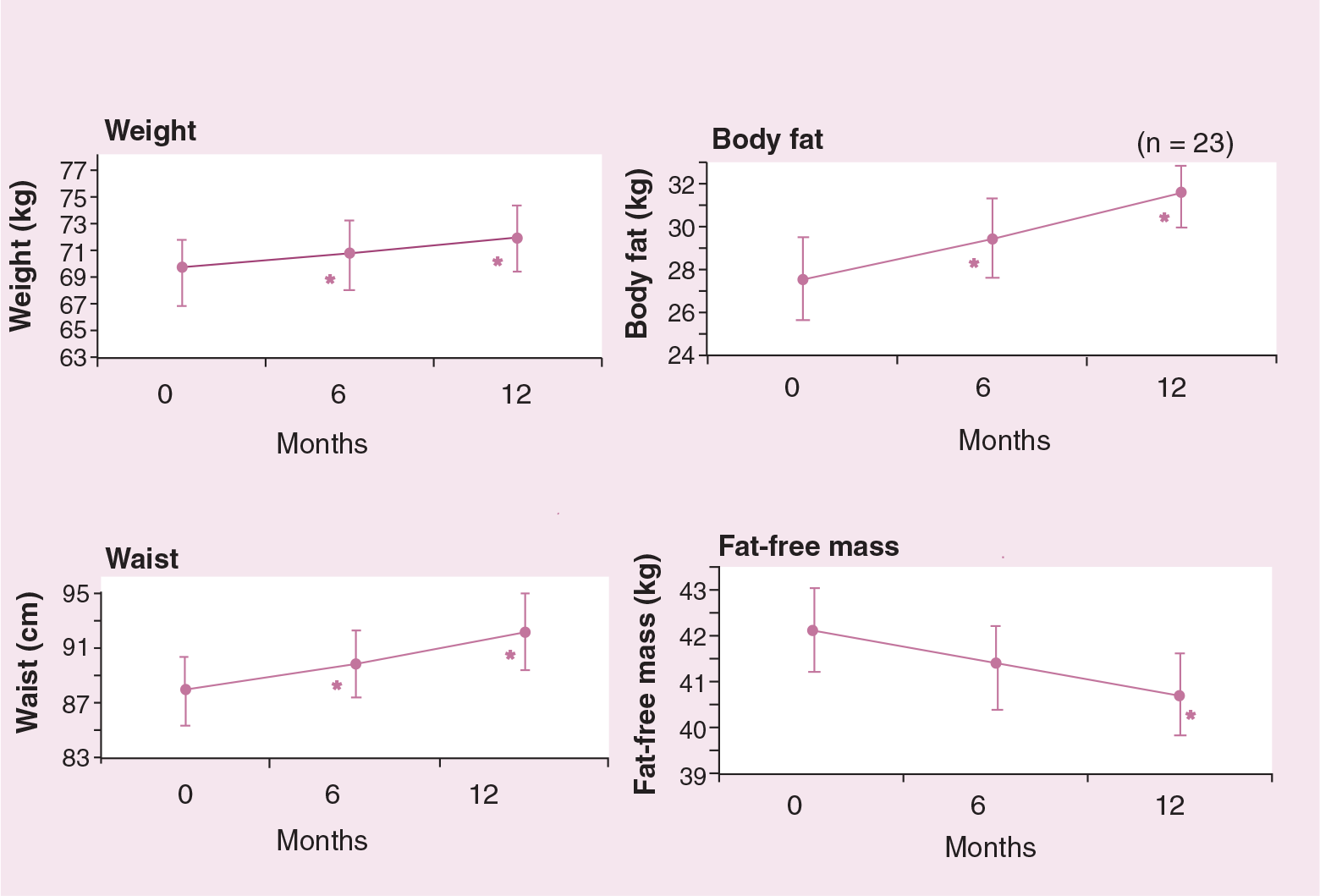
Changes in weight and body composition over 1 year in postmenopausal women receiving adjuvant endocrine therapy (n = 23) [22].
Estrogen and insulin-like growth factor (IGF)-1 influence the amount and distribution of fat [25] and FFM [26,27], as well as energy balance and body weight [28,29]. It is possible that some of the changes in body composition among women receiving adjuvant therapy may specifically relate to the hormonal effects of stopping hormone replacement therapy at the time of diagnosis [30], and the additional effects of lower estrogen levels (with anastrozole) or estrogen agonist/antagonist actions (with tamoxifen). An earlier prospective, randomized tamoxifen study among healthy women, however, reported minimal weight change, but significant increases in central fat among both patients receiving tamoxifen and control patients [31].
The effect of excess weight on disease-free survival
The controversy of whether excess weight or weight gain after diagnosis has an adverse effect on the prognosis of breast cancer has been investigated over the past 20–30 years. Much of the increased risk is thought to be linked to later detection and increased nodal involvement among heavier women. However, a critical review published in 1990 concluded that body size had a modest independent adverse prognostic effect among postmenopausal women, particularly among women with limited node-negative disease [32].
Obesity adversely affects the prognosis of both ER-positive and negative tumors [33,34]. This may be mediated by the adverse hormonal and secretory effects of adipose tissue. For example, it is known that adiposity is linked to increased estradiol, testosterone, insulin, IGF-1, leptin and inflammatory markers such as tumor necrosis factor-α, interleukin-6, plasminogen activator inhibitor 1 and decreased production of adiponectin [35]. A significant proportion of the adverse effects in ER-positive tumors is thought to be linked to higher levels of bioavailable estradiol (as a result of increased aromatization within adipocytes and lower circulating concentration levels of sex hormone binding globulin [SHBG]) among heavier breast cancer patients [36]. Predictably, the adverse effects of weight and weight gain on breast cancer prognosis have not been demonstrated in more recent studies of postmenopausal women, many of whom were receiving adjuvant tamoxifen therapy (Table 2) [15,37–42].
Weight at diagnosis and weight change and disease-free survival among postmenopausal breast cancer patients.
C: Chemotherapy; CI: Confidence interval; ER: Estrogen receptor; HR: Hazard ratio; IBCSG: International Breast Cancer Study Group; NSABP: National Surgical Adjuvant Breast and Bowel Project; RCT: Randomized controlled trial; RR: Relative risk; T&C: Tamoxifen and chemotherapy.
Obese = BMI > 30 kg/m2.
The recently reported Women's Intervention Nutrition Study (WINS) assessed the effect of a low-fat dietary intake (33.3 ± 16.7 g/day compared with 51.3 ± 24.4 g in the control group) on disease-free survival among 2437 postmenopausal women with early-stage resected breast cancer over 5 years. Dietary fat reduction improved the relapse-free survival in the overall population (hazard ratio [HR]: 0.76; 95% confidence interval [CI]: 0.6–0.98; p = 0.034). The benefits were greatest among the relatively small group of ER-negative women receiving chemotherapy (HR: 0.58; 95% CI: 0.37–0.91; p = 0.018) but were somewhat weaker and did not achieve statistical significance among ER-positive women receiving tamoxifen (HR: 0.85; 95% CI: 0.63–1.14; p = 0.277). How much of the improved survival was attributable to modest weight loss (average of 2 kg in the study group), or to the fat restriction and increased fruit and vegetable consumption in the intervention group, is not known [43].
Weight & contralateral breast cancer
Women diagnosed with breast cancer are at a higher risk of developing breast cancer in the other (or contralateral) breast. For postmenopausal women the risk is estimated to be approximately 0.6% each year and is significantly reduced by tamoxifen (50%) and the aromatase inhibitor anastrazole (70–80%) [8]. BMI is an independent risk factor for the development of contralateral breast cancer and appears to be a major factor in the higher rates of contralateral breast cancer among African, compared with white, American women in the recent NSABP-14 adjuvant tamoxifen trial. In this study, the 10-year cumulative incidence of contralateral breast cancer was 5.6% in obese women compared with 3.3% in underweight or normal-weight women (BMI: 18.5–24.9 kg/m2; HR: 1.58; CI: 1.1–2.5) [44].
Central obesity insulin resistance & breast cancer prognosis
While general obesity does not appear to affect breast cancer prognosis among ER-positive women receiving tamoxifen, there is some evidence that women with central obesity (and higher levels of insulin and increased insulin resistance) may be more likely to relapse than their thinner, more insulin-sensitive counterparts. Borugian and colleagues reported the relative risk of breast cancer mortality for each 0.1 unit increase in waist:hip ratio to be 1.4 (95% CI: 0.9–2.1) [45]. Hyruniuk and colleagues found that diabetic women receiving tamoxifen had a HR of breast cancer mortality of 2.4 compared with nondiabetics receiving tamoxifen (p < 0.006) [46]. In addition, Goodwin and colleagues linked higher fasting insulin concentrations to both decreased disease-free survival and overall breast cancer survival among women receiving adjuvant tamoxifen therapy. HR for women with the highest compared with the lowest insulin concentrations were 2.1 (CI: 1.3–3.4) and 3.1 (CI: 1.7–5.9), respectively [47].
Does excess weight modify the efficacy of endocrine therapy?
The possibility that endocrine therapy may exert a greater protective effect among heavier subjects due to a greater relative reduction in estradiol [48], or that the dose of aromatase inhibitor/tamoxifen may be inadequate among heavier women, are both unlikely. Within the NSABP-14 trial, tamoxifen reduced the risk of recurrence by 40% compared with placebo within normal, overweight and obese women [15]. Similarly, a recent analysis within the ATAC trial showed that the efficacy of anastrozole was not modified by body weight [ATAC Trialist's Group Pers. Comm.]. Weight does not appear to influence the metabolism of tamoxifen [49] or the declines in estradiol and estrone with aromatase inhibitors [50].
Effect of weight on comorbidities
While BMI and weight gain do not appear to exert adverse effects on recurrence, they have a negative impact on survival due to non-breast cancer causes [15]. Within the NSABP-14 adjuvant tamoxifen trial, obese women were nearly 50% more likely to die from non-breast cancer causes than normal weight women (HR: 1.49; 95% CI: 1.15–1.92). Being overweight also appeared to have a detrimental effect on survival but this did not reach statistical significance (HR: 1.19; 95% CI: 0.94–1.52). Secondary cancers are major competing causes of death among breast cancer patients [3]. There was some indication that heavier women were at increased risk of endometrial cancer in this study. The cumulative incidence of endometrial cancer 10 years after breast surgery was 0.009 (0.004–0.013) for underweight/normal weight women compared with 0.012 (0.005–0.019) for overweight women and 0.016 (0.007–0.026) for obese women [15]. The increased risk of endometrial cancer in heavier women is known to occur, particularly among women receiving tamoxifen [51]. The HR of other second primary cancers (which were seen over 37 different sites) was also increased in the NSABP-14 study among overweight (HR: 1.29; 95% CI: 0.95–1.76) and obese (HR: 1.62; 95% CI: 1.16–2.24) women compared with normal/underweight women [15].
CV disease is another major competing cause of death for breast cancer patients [3]. In the noncancer setting, the risk of CV disease is significantly increased even within normal ranges of BMI and modest adult weight gains. Typically, risk increases linearly above a BMI of 21 kg/m2 [52]. Among women, each unit increase in BMI is linked to a 14% increase in risk of CV disease, while each kg of weight gain is linked to a 3.1% increase [53].
The decreased estrogen and increased androgenicity produced by endocrine therapies may have an adverse effect on lipid profiles and their attendant CV risk [54]. Tamoxifen has been shown to have beneficial effects on some, but not all, surrogate markers of CV disease risk among postmenopausal early breast cancer patients. Specifically, tamoxifen reduces total and low-density lipoprotein (LDL) cholesterol, increases apolipoprotein A1 concentrations [55], decreases C-reactive protein (CRP) and fibrinogen (particularly among heavier women) [56] and lowers carotid intima–media thickness [57] but increases triglyceride concentrations [58]. Although tamoxifen does not appear to prevent accumulation of central fat, it has been shown to limit the adverse effects of central adiposity on LDL cholesterol and fibrinogen [31]. However, these favorable effects of tamoxifen on surrogate markers of CV disease do not translate into an actual reduction in CV events; a recent pooled analysis reported the relative risk for myocardial infarction for tamoxifen to be 0.90 (95% CI: 0.66–1.23) [59]. Adjuvant tamoxifen is associated with a 1.68 (95% CI: 1.22–2.32) risk of stroke and a 2.46 (95% CI: 1.24–4.91) risk of pulmonary emboli [59]. The risk of thromboembolic events is particularly increased among heavier patients [44].
Data are beginning to accumulate on the effect of aromatase inhibitors on CV disease. Interestingly, the different aromatase inhibitors have a varying effect on lipid profiles. Short-term studies in early breast cancer patients have linked anastrozole to increased levels of high-density lipoprotein (HDL) cholesterol and decreased triglyceride, but have also linked it to either no effect on [60] or increased levels of total and LDL cholesterol [61]. Exemestane is linked to nonsignificant reductions in total LDL and HDL cholesterol [62]. Recent data from the MA17 trial showed no appreciable effect of letrozole on total LDL or HDL cholesterol, triglyceride or lipoprotein over 3 years in women who have completed 5 years of tamoxifen [63]. Whether these small alterations in lipid profiles translate into differences in CV risk in the adjuvant setting are not known. Anastrazole has a comparable effect to tamoxifen on ischemic CV disease [64], but the risk of thromboembolic and cerebrovascular disease is lower [8]. Preliminary data from the Breast International Group (BIG) 1–98 adjuvant letrozole and the IES exemestane switch trial have linked these aromatase inhibitors to a slight excess of coronary events and deaths compared with tamoxifen.
Heavier women receiving tamoxifen are more likely to develop nonalcoholic steatohepatitis, which is characterized by fatty liver with inflammatory changes and fibrosis not related to alcohol intake [65]. The clinical significance of these changes is not clear, as they readily reverse within 1–2 years after cessation of tamoxifen in most cases [66] and do not display histologic, biochemical or clinical signs of progression to cirrhosis after 10 years of follow-up.
Obesity and weight gain have well-documented adverse effects on physical function, bodily pain and vitality among breast cancer patients [67], as is the case in the general population [68]. Excess body weight and fat are related to disability and limitations in activities of daily living and are better predictors of disability in the next 3 years than amount of muscle mass among women aged 65 and older [69].
Lymphedema, an undesirable complication following axillary clearance or radiotherapy to the axilla, is linked to obesity [70]. Increased risk is most likely due to postoperative infections, coupled with the increased likelihood of technical problems during axillary surgery among heavier women. There is little work on the effects of weight loss on existing lymphedema. A recent study undertaken at The Royal Marsden Hospital, London, UK, compared the effects of a dietitian-led weight reduction intervention with no intervention in overweight women with lymphedema in a randomized, controlled study. Arm volume, as measured by circumference measurements, was significantly reduced in the weight reduction group [C Shaw, Pers. Comm. 2004].
Lifestyle changes after diagnosis of breast cancer
Gains in body fat occur when energy intake exceeds energy expenditure. There are few direct measurements of energy intake among women receiving endocrine therapy, although adoption of disinhibited eating behavior and loss of normal, restrained eating patterns is thought to be a factor in weight gain after diagnosis of breast cancer [71]. Although many breast cancer patients (40%) report changing their diet in an effort to maintain health and prevent recurrence [72], these changes are often not well-informed, evidence-based choices. Common practices include the removal of dairy products [203] and commencing herbal or nutritional supplements [73]. Objective dietary measurements among breast cancer patients suggest dietary change may actually be considerably more modest than reported [74], and that the adoption of healthier diets is least likely in those with unhealthy diets at the time of diagnosis and among the overweight and obese [75,76].
Postmenopausal breast cancer patients become less active after diagnosis. Although reductions are not as great as those among women receiving adjuvant chemotherapy, postmenopausal women aged 50–59 and over 60 report significant declines in their overall level of physical activity (−9.5 to −11.6%, respectively), specifically for exercise of moderate (−6 to −26%) and vigorous intensity (−29 to −40%) [77]. A recent survey found that 43% of postmenopausal breast cancer patients did not exercise routinely; furthermore, 48% of these stated they had no plans to increase their level of physical activity [78]. Greater declines in exercise occur in heavier subjects, which suggests that these patients should be specifically targeted to increase or maintain levels of exercise [77].
Optimum weight-control programs
Weight-loss studies
There have been surprisingly few studies of weight reduction in breast cancer patients. The first demonstration that weight loss was feasible among breast cancer survivors was a randomized, calorie-restricted weight-loss intervention among postmenopausal breast cancer patients in 1993 [79]. Plans for a larger follow-up study were abandoned on the assumption that any potential benefits of weight loss on survival would be negated with the widespread introduction of adjuvant tamoxifen therapy. Current studies have focused on exercise alone [80] or fat-reduced high fruit and vegetable diets rather than weight reduction. The WINS study has been discussed previously [43]. The Women Healthy Eating and Living (WHEL) study includes 1000 postmenopausal and 2000 premenopausal women and is similarly testing the effect of a low fat (15–20% energy), high fruit (three fruit servings/day) and vegetable (five vegetable servings/day), high fiber (30 g/day) diet on disease-free survival. Results of this study are expected in 2007. Minimal weight change between the intervention and control group means the effect of weight control or weight loss is unlikely to be evaluated from this study [81].
Treatment goals
Weight-loss programs should focus on attaining a weight loss of more than 5% of body weight among currently overweight or obese women, or maintaining current weight for healthy-weight women. Whether weight loss will improve overall breast cancer survival among women receiving adjuvant endocrine therapy is not known. The authors' recent observational study, which linked weight loss of greater than 5% after the menopause to a 25% reduction in the development of primary breast cancer [82], may suggest reduced rates of contralateral breast cancer.
In the noncancer setting, modest weight loss (> 5%) in overweight subjects has been linked to a 60% reduction in the risk of NIDDM [83,84] and a 36% reduction in hypertension [85], as well as significant reductions in symptomatic osteoarthritis [86] and respiratory disease. A recent overview of randomized, controlled trials linked a 10% weight loss to a 0.25 mol/l reduction in cholesterol and a 6.1 mm/hg reduction in systolic blood pressure. These changes were estimated to reduce the risk of CV disease by 10–12% [87]. Intentional weight loss of approximately 10% is known to reduce the risk of colorectal (9%) and endometrial cancer (4%) [88].
Diet & exercise strategies for weight loss & weight maintenance
Dietary advice should aim to optimize weight loss/weight control as well as reducing the risk of recurrence of breast cancer and promoting CV and bone heath. Weight loss is difficult to achieve and maintain. Low-fat diets have long been considered the optimum approach for weight loss, but it is becoming increasingly clear that these may be no better and may even be less effective than other dietary approaches [89]. Among breast cancer survivors, low-fat dietary interventions (15–20% of energy) have been linked to either modest (2 kg) [43] or no weight loss [90]. Moreover, in the latter study, the low-fat intervention brought about, albeit small, adverse declines in HDL cholesterol (3%) and increases in triglycerides (13%) [91].
The benefits of a low-fat diet for improving disease-free survival among women receiving adjuvant endocrine therapy requires further consideration. Despite encouraging results overall, the benefits of a low-fat diet among women receiving adjuvant endocrine therapy were not clearly demonstrated in the recent WINS study [43]. Increasing evidence that breast cancer risk has positive links with saturated and n-6 polyunsaturated fat and inverse relationships with monounsaturated (oleic acid) and n-3 fat [92] suggest that the survival benefits in the WINS study may have been specifically linked to reductions in saturated and n-6 fat rather than total fat itself. Replacing saturated and n-6 fats in the diet with monounsaturated or n-3 fats may prove to be beneficial for the breast cancer survivor but this area requires further study.
While we await the results of such studies it may be prudent to advise a Mediterranean, energy-restricted diet (35% energy from fat: 15% monounsaturated fat, 8% polyunsaturated fat, 7% saturated fat and high fiber), rich in olive oil, fish, poultry, fruit, vegetables, legumes and nuts, for weight loss among postmenopausal breast cancer survivors. This diet is superior to a low-fat diet in limiting calorie intake on account of the higher fat and protein content [93] and lower glycemic load [94]. This diet also has metabolic benefits compared with a low-fat weight-reducing diet for risk of CV disease [95,96], as well as potential anti-angiogenic, anti-inflammatory [97] and immunocompetent effects [98].
Fruit and vegetables may be expected to have anticancer effects. Specifically, carotenoids have retinoid effects on cellular differentiation and apoptosis [99] and brassicas have a favorable effect on estrogen metabolism among postmenopausal women via cytochrome P450 induction [100]. Although the beneficial effects of fruit and vegetable intake on breast cancer prognosis have not been resolved [101], fruit and vegetables are recommended for breast cancer survivors for their cardioprotective effects [102].
Fiber is an important, cardioprotective component of a successful weight-control diet [103,104]. Increasing fiber intake from 22 to 29 g/day led to significant declines in serum concentrations of total and free estradiol among postmenopausal women receiving tamoxifen [105]. The relevance of these hormonal changes to prognosis among breast cancer survivors on endocrine therapy is not clear.
Alcohol has been linked to the risk of postmenopausal breast cancer [106]. However, a recent overview reported that none of the eight studies examining the effect of alcohol on prognosis linked alcohol with the survival of women diagnosed with breast cancer [101]. Alcohol restriction is, however, prudent for women seeking to control their weight, due to its intrinsic calorie content and appetite-stimulating effects. The well-documented cardioprotective effects of a modest alcohol intake for postmenopausal women suggest that up to 2 units of alcohol/day can be included as part of a calorie-controlled diet [107].
Exercise alone does not appear to control weight among postmenopausal breast cancer survivors [80]; however, it is clearly a vital component of energy-restricted weight-control programs in this population [108]. Exercise exerts independent benefits on disease-free survival [109], quality of life (QOL), CV fitness [110] and osteoporosis [111]. A recent randomized study failed to demonstrate beneficial effects of exercise on insulin sensitivity among postmenopausal breast cancer survivors [112].
Delivery of weight-management programs
Multidisciplinary teams are recommended for weight management [84], but these may not be feasible in the breast cancer setting. Dietitians are not historically involved in the nutritional management of breast cancer patients, while physiotherapy input is mainly limited to postoperative arm rehabilitation. Physician involvement would undoubtedly lend credence and most likely adherence to the weight-loss advice [113]. Oncology practitioners have not, however, traditionally considered weight management a priority and are not inclined to raise the issue with patients [114]. Nurse-led weight-management programs have been shown to be cost-effective in the CV setting. However, a recent UK survey highlighted that one in five breast-care nurses do not currently have enough time to provide adequate information and basic psychological support to patients [204]. Many do not feel they have adequate skills to promote lifestyle modification and weight control [22]. Nurse-led interventions would only become feasible with additional breast-care nurse staffing and training.
The level of support required to achieve and maintain weight loss varies widely between patients. In the noncancer setting, sufficiently motivated individuals (approximately 9%) may opt to lose weight on their own after discussing the options with their healthcare practitioner [115]. In the noncancer setting, there has been a tendency to promote group therapy above individualized counseling for weight loss. Groups were believed to provide better weight-loss outcomes and a cost-effective way of addressing patient numbers. It is clear, however, that individuals have preferences regarding the group or individual approach, and that matching treatment to preferences provides better results [116]. Obese breast cancer survivors receiving group-based interventions appear to require additional individual counseling to meet their physical and psychological needs [117,118]. In a study by Djuric and colleagues, weight loss over 12 months in a commercial slimming group was −2.7 ± 2.1 kg compared with 8.0 ± 1.9 kg in an individually counseled group and −9.5 ± 2.7 kg in a group attending the slimming group with additional individual counseling [118]. Telephone-based counseling has also been shown to be efficacious in changing dietary and exercise behavior among breast cancer survivors in a research setting [119,120].
Promoting a home-based exercise program within the clinic may encourage previous exercisers to resume activity. Previous nonexercisers, however, require structured exercise programs and additional support to increase their level of activity [121]. A recent needs-assessment study suggested that breast cancer patients wished to exercise in specific breast cancer groups [122].
The timing of interventions needs careful consideration in order to optimize their impact. Attempts to deliver lifestyle advice too soon after diagnosis may worsen the feeling of guilt and the psychological impact of diagnosis. Disruption to daily activities at this time may be a barrier to changing diet and exercise behavior. Equally, the diagnosis of breast cancer is known to be an opportune time when women may be more responsive to making beneficial lifestyle changes [123]; a total of 70% of postmenopausal breast cancer survivors indicated a preference to receive lifestyle advice in the 6 months immediately after diagnosis [78].
Pharmacotherapy for weight management
The agents orlistat (Xenical®, Roche) and sibutramine (Meridia®/Reductil®, Abbott) are currently available as part of a weight-management program for overweight and obese patients in the noncancer setting. Neither agent has been linked to the development of breast cancer, but the different pharmacologic properties, contraindications and side effects of these agents require consideration for patients with breast cancer (Table 3).
Eligibility for initiation and continuation of orlistat and sibutramine for weight management.
Diabetes, hyperlipidemia, hypertension, coronary heart disease, stroke
Diabetes, hyperlipidemia. Sibutramine is contraindicated if BP >145/90 mm hg
BMI: Body mass index; BP: Blood pressure.
Adapted from National Obesity Forum UK [205].
Orlistat is a specific inhibitor of gastric and pancreatic lipases. The inactivated enzymes are thus unavailable to hydrolyze dietary fat, allowing 30% of dietary fat to pass through the gut unabsorbed. Orlistat has been associated with an additional weight loss of 3.2 kg (CI: 4.15–2.37) compared with standard dietary advice [124]. Patients follow a hypocaloric diet containing less than 30% of energy from fat, since high-fat diets lead to steatorrhea, flatus and fecal urgency. Treatment with orlistat may potentially impair the absorption of fat-soluble vitamins (A, D, E and K). Although the vast majority of patients receiving up to 4 years of treatment with orlistat in clinical studies had vitamin A, D, E and K and beta-carotene levels within the normal range, diets rich in fruit and vegetables should be encouraged.
Sibutramine inhibits the uptake of serotonin and norepinephrine in the brain. This has been linked with additional weight loss of 3.4 kg (95% CI: 4.45–2.35) compared with standard dietary advice. Weight loss is achieved through early satiety, with a reported 20% reduction in food intake, while sympathetically mediated thermogenesis maintains the basal metabolic rate, which normally falls as weight is lost. Sibutramine is contraindicated in patients with a history of psychiatric illness, depression and eating disorder, and in those with hypertension. The prevalence of psychiatric disorders [125] and hypertension [14] among obese breast cancer survivors may preclude the use of sibutramine for a significant number of patients.
Evaluating the effectiveness of weight-management interventions
Assessment of body weight & body composition in breast cancer patients
Body fat is best assessed using imaging techniques such as dual energy x-ray absorpiometry (DXA) for total body fat and magnetic resonance imaging (MRI), or computerized topography (CT), at the L3 L4 level for visceral and subcutaneous central fat stores. These techniques are expensive and not routinely available for the majority of patients, hence simpler methods such as bioelectrical impedance are increasingly used to assess body composition in weight-management settings. Impedance is the resistance of the body to the flow of an alternating current. This technique is based on the fact that lean tissues have a high water and electrolyte content and are good conductors, whereas fat is a poor conductor. Impedance measurements are thus related to the volume of the conductor, which relates to the amount of body water, FFM and fat mass.
A limitation of impedance among women receiving adjuvant endocrine therapy is that impedance is relatively insensitive to changes in central fat stores [126]. Changes in waist circumference should therefore be monitored. Waist circumference is an accepted proxy measurement of central fat (particularly visceral fat) stores [127]. Waist measurement is a useful marker of the risk of CV disease, NIDDM, insulin sensitivity and breast cancer prognosis [45,128]. For non-Asian women, a waist circumference greater than 32 inches (80 cm) is considered to place them at an increased risk of insulin resistance and the metabolic syndrome, while a waist circumference of greater than 35 inches (88 cm) places women at a substantially increased risk. For women of South Asian descent, a waist circumference greater than 32 inches (80 cm) is associated with a substantially increased risk [128].
Biomarkers
Biomarkers of efficacy of weight-control interventions are essential for both the clinician and patients, for whom they can serve as key motivators. Key markers include fasting serum concentrations of insulin or insulin-sensitivity (using the homeostasis model based on fasting insulin and glucose concentrations) [129], serum testosterone concentration and markers of inflammation such as high sensitivity (hs)CRP. Higher levels of fasting insulin have been linked to decreased disease-free survival and overall survival, independent of the effects of weight among women receiving adjuvant tamoxifen therapy [47], and are linked to an increased risk of NIDDM and CV disease. Bio-available testosterone is an independent predictor of coronary heart disease [130], NIDDM [131] and dyslipidemia [132] among postmenopausal women. A small case–control study linked higher levels of testosterone to subsequent risk of breast cancer recurrence among postmenopausal women, both receiving and not receiving tamoxifen. Although the CIs are wide, women with testosterone levels of 0.5–0.93 ng/l were 7.2-times (95% CI: 2.4–21.4) more likely to experience recurrence compared with women with testosterone of 0.16–0.33 ng/l. Interestingly, risk was reduced among women who lowered their levels of testosterone during a 12-month low glycemic index (GI), low saturated fat, high fiber, high phytoestrogen, weight-losing diet (HR: 0.15; 95% CI: 0.03–0.71) [133]. Inflammatory markers such as hsCRP have been linked to future risk of CV disease [134], and most likely to the development of breast cancer [135], although the relationship to the risk of other cancers, such as colorectal cancer, is not entirely consistent in women [136].
Quality of life
The impact of weight-loss interventions on QOL should be assessed. The diagnosis and treatment of breast cancer is a major life event that can exert a significant psychological impact on women of all ages [137]. The most common long-term psychological effects include mood disorders, anxiety, fatigue and problems with sexual functioning and body image [138]. Weight gain has a negative impact on QOL outside the cancer setting. The effect of excess weight on QOL among older breast cancer patients has not been studied, although weight gain has been cited as a problem among 40% of these patients [138].
Cardiovascular disease risk
The precise relationship between lipid profiles, blood pressure and future CV disease in breast cancer patients receiving endocrine treatments is not known and requires further study. It is, however, prudent to assess established risk factors for cardiovascular disease (i.e., systolic blood pressure, total and HDL cholesterol) around the time of diagnosis and on a regular basis throughout the weight-loss program. These parameters will provide an ongoing estimate of 10-year CV disease risk to guide management [139]. Statins should be commenced in women with a 10-year CV risk of 20% or greater and LDL cholesterol greater than 100 mg/dl, and antihypertensives commenced among women with blood pressure greater than 140/90 mmhg. Aspirin therapy should also be used in women at high risk of CV disease [139].
Conclusion
General and central adiposity are common among newly diagnosed postmenopausal breast cancer patients and worsen due to the impact of diagnosis and treatment. Increasingly effective adjuvant therapies mean that the prognosis for postmenopausal women with breast cancer has never been better. Available evidence suggests that weight control is unlikely to improve this further, although the specific benefits of limiting central obesity and insulin resistance may yet prove to be beneficial.
Weight control is, however, a potentially important adjunct to adjuvant endocrine therapy to minimize comorbidities such as heart disease, NIDDM, other cancers, stroke and lymphedema and to improve well-being among breast cancer patients. Weight management is not a routine part of cancer care, in marked contrast to cardiac rehabilitation where it is an integral component of management.
Weight loss is notoriously difficult to achieve and maintain. Combined diet and exercise weight-control interventions are recommended. Evidence that older women may be more likely to adhere to long-term energy restriction [140] and the motivational effect of cancer diagnosis may increase the success of interventions in this group [141]. The optimum timing, mode of delivery and degree of support requires further study to ensure that efficacious patient-orientated programs are developed. Such programs need to be evaluated in terms of their effect on body fat and central adiposity as well as weight, and, most importantly, their impact on biomarkers of future risk of CV events and NIDDM. Cost–benefit analyses of delivering such interventions will be required to justify the inevitable increased staffing needs.
Future perspective
Despite the expansion of breast cancer prevention strategies among high-risk women, few would disagree that the number of overweight/obese postmenopausal women diagnosed with early breast cancer is likely to increase over the next 5–10 years. Current trends predict that the population of older women [142], in particular obese older women [202], at risk of breast cancer is set to increase. The recent extension of the UK national screening program to include women aged 65–70 years means that the number of older women diagnosed with localized breast cancer and receiving adjuvant endocrine treatment will increase.
Widespread use of aromatase inhibitors either as a first- or second-line treatment after a period of tamoxifen is likely to reduce further mortality from breast cancer and make the management of comorbidities and improved QOL increasingly important. The possible benefits of a low-fat diet (20% energy from fat) demonstrated in the recent WINS study needs to be considered in the context of widespread use of aromatase inhibitors. Low-fat diets are not optimal to limit CV disease compared with Mediterranean higher-monounsaturated fatty acid diets [95,96], and may not be appropriate among patients receiving aromatase inhibitors in whom dietary approaches minimizing CV morbidity (rather than breast cancer mortality) may become a priority. The relationship between CV markers and subsequent risk of CV disease among women receiving adjuvant endocrine treatment will be a research priority.
Ever-increasing numbers of older, heavier breast cancer patients means that efficacious and cost-effective weight-control interventions need to be developed. Ongoing studies of particular interest are being conducted in the USA. Demark-Wahnefried and colleagues are studying the effect of a personally tailored telephone counseling program on diet and exercise behaviors among older breast and prostate cancer survivors (Leading the way in Exercise And Diet [LEAD] study) [143]. This group are also planning the Reach-out to ENhancE Wellness (RENEW) study, which will assess the effect of a portion-controlled healthy diet and exercise intervention via print materials and telephone counseling among 640 overweight long-term breast, prostate and colorectal cancer survivors. The authors are currently developing a nurseled diet and exercise weight-control information package for postmenopausal breast cancer patients. The authors are planning to assess how much nursing reinforcement is required to maximize the efficacy of this package in the context of a randomized, controlled trial of package versus package plus reinforcement.
Executive summary
Rates of breast cancer are increasing, particularly among postmenopausal women.
Advances in adjuvant treatment have significantly improved prognosis; current estimates predict 83% of women aged 50–59 in the UK have a 5-year disease-free survival.
The beneficial effects of aromatase inhibitors (anastrazole, letrozole and exemestane) either as a primary adjuvant therapy or after a course of adjuvant tamoxifen, predict their increasing usage in early breast cancer patients.
Comorbidities are common among breast cancer patients. At the time of diagnosis, 25–50% have hypertension, 15–27% have coronary heart disease and 5–12% have non-insulin dependent diabetes mellitis (NIDDM).
Cardiovascular (CV) disease and other cancers are the major competing causes of death among postmenopausal breast cancer patients.
Minimizing CV disease will be a priority among women receiving aromatase inhibitors.
A total of 63% of early breast cancer patients are overweight or obese at the time of diagnosis.
Modest weight gain (mean ± standard error (SE): 2.6 ± 0.8 kg) but greater gains in body fat (4.2 ± 0.8 kg) and waist circumference (4 ± 1.3 cm) occur in the year after diagnosis.
Generalized obesity (body mass index) does not influence disease-free survival among women receiving adjuvant endocrine therapy. Central obesity may, however, have an adverse effect on prognosis; a 0.1 increase in waisthip ratio is linked to a relative risk of breast cancer mortality of 1.4 (95% CI: 0.9–2.1).
Obese breast cancer patients are more likely to develop contralateral breast cancer; over 10 years, 5.6% of obese women compared with 3.3% normal-weight women developed contralateral breast cancer.
Obese breast cancer patients are more likely to die from other causes, such as CV disease and other cancers; hazard ratio for death from other causes in obese compared with normal-weight women is 1.49 (95% CI: 1.15–1.92).
Weight control may reduce lymphedema and mobility and improve quality of life.
On average, activity levels reduce after diagnosis (−11%).
Many women make poorly informed dietary changes that are not evidence based, i.e., dairy-free diets and nutritional supplements.
Weight loss of greater than 5% of body weight has been linked to significant reductions in primary breast cancer (25%), diabetes (60%) and hypertension (36%) in the noncancer setting.
The benefits of low-fat diets among women receiving adjuvant endocrine therapy were not clearly demonstrated in the recent Women's Intervention Nutrition Study (WINS). The benefits of replacing saturated and n-6 fats in the diet with monounsaturated fatty acids and n-3 fats for breast cancer patients requires further study.
An energy-restricted Mediterranean diet and exercise program is recommended to promote weight-loss/maintenance and to minimize the risk of CV disease, diabetes and osteoporosis.
The timing and mode of delivery of weight-control programs and the benefits of anti-obesity drugs require further study in this population.
Weight-control interventions should be evaluated in terms of their effects on body fat, central fat, markers of breast cancer and CV disease risk (i.e., insulin sensitivity, testosterone and inflammatory markers) and quality of life, as well as body weight.
The relationship between lipid profiles and CV risk among women receiving adjuvant endocrine therapy is not known and requires further study.
