Abstract
Present communication reports the effects of environmentally available, low doses of tetra chloro di benzo-p-dioxin (2,3,7,8 TCDD) to lysosomal enzymes in mice liver. The study tests the hypothesis, in vivo exposure of low dose TCDD provokes dose and duration dependent toxic effects to key lysosomal enzymes and thereby causes cellular apoptotic changes. Three groups of female Swiss albino mice were subjected to two doses of TCDD (0.004 mg/kg bw/d, 0.04 mg/kg bw/d) for 2, 4 and 6 days of exposure durations. The results indicated significant exposure duration dependent effects of TCDD in mice liver cells. The results suggested that TCDD possibly induced an increase in intracellular ions or ROS which in turn altered different physiological activities by affecting different metabolic pathway of the liver cells. The altered functions of key lysosomal enzymes by TCDD may also evoke the process of cellular apoptosis.
INTRODUCTION
2,3,7,8 TCDD is a toxic compound of Dioxin family which produces a variety of toxic effects in animals and even in human. The histopathological changes produced by TCDD differ quantitatively and qualitatively in different species and to some extent in acute and chronic administration (Poland and Knutson, 1987). Xenobiotics like pesticides, organic solvents and many other chemicals are concerned with public health and environmental pollution. TCDD was reported to cause serious effects such as liver damage, chlorachne, oedema formation in animals (Higginbotham et al. 1968). The chlorinated dioxin commonly occurs in food chain and can still cause long term health effects (Van den Berg et al. 2000). Dioxins are known to produce hypoglycemia which can alter Krebs cycle and dysregulated fatty acid oxidation, enhances reactive oxygen species (ROS) which can produce intracellular toxic load. The production of the oxidative stress occurs through exposure of organochlorine compounds and metabolism by different oxido-reductase cycle (Livingstone et al. 1990). Lysosome functions as the cell waste disposal system and commonly used for digestion of macromolecules produced by various chemical reactions with the help of hydrolases enzymes it contains (Jigyasi and Kundu, 2013a). It has been reported earlier that after TCDD exposure, changes in multiple endocrine and growth factor system have been occurred which was tissue specific, sex and age dependent (Birnbaum and Tuomisto, 2000). TCDD is known to effect different cellular activities and growth factor signal transduction by affecting complex chain of reactions into the cell (Jigyasi and Kundu, 2013b). TCDD causes mitochondrial toxicity characterized by ROS production, lipid peroxidation, decreased ATP level and altered electron transport chain (ETC) enzyme activity and expression (Forgacs et al. 2010). TCDD also reported to disrupt glycolysis and gluconeogenesis by altering relevant enzyme activity (Lu et al. 2010). Organochlorine pollutants are endocrine disruptors following to disrupt several metabolic pathways by inducing β- oxidation of fatty acid and amino acid (Lin et al. 2011). Going through the literature it was observed that the study on the effects of TCDD to lysosomal enzymes is rare. The present study was undertaken to examine a hypotheses, in vivo exposure of environmentally available low concentration of TCDD provokes dose and duration dependent toxic effects to lysosomal enzymes and may cause cellular apoptotic changes by affecting lysosomal enzyme activity in mice liver cells.
MATERIALS AND METHODS
A total of three groups of adult female Swiss albino mice, around 3 months of age and weighing 30 ± 5 g, were used for the study. The animals were fed with commercially available rodent diet and water ad libitum, and kept in the animal house facilities under hygienic condition as per CPCSEA India, guidelines. Humidity, temperature was controlled (25 ± 2°C) and diurnal cycle of 14:10 h was maintained. All experiments were conducted according to norms approved by CPCSEA, India. The Dioxin, 2,3,7,8 TCDD, in its purest form, was obtained from Sigma Aldrich Chemicals Pvt. Ltd. (CAS No. 1746-01-6). All other chemicals used for this study were of analytical grade and procured from reputed chemical companies. A total of 81 inbred female Swiss albino mice of the same age and weight group were taken for experimental studies. The selection of doses were based on the (a) TCDD residues available in the environment and possible human exposure through oral route from different environmental sources (b) evaluation of toxicity studies and Minimum Risk Dose (MRD) for extrapolating from animal model to human for TCDD administered through oral route. A separate control group was maintained that received equivalent amount of corn oil which was used as vehicle. Prior to the experiment, a test was carried out to validate the findings between treated with corn oil control and untreated control animals. There was no significant difference between treated and untreated control groups. Groups of mice were exposed different doses of TCDD (0.004 mg/kg bw/d, 0.04 mg/kg bw/d) for 2, 4 and 6 days of exposure durations. After exposure to TCDD, the liver tissue was pooled from at least three animals for each dose group and suspended in chilled Sucrose- EDTA-Imidazole (SEI) buffer (pH 7.1) to remove excess blood and other membranous substances. Known amount of tissue was randomly sampled from the pooled tissue of all animals and homogenized in chilled phosphate buffer (pH 7.0) to obtain a 10% (w/v) homogenate. Enzyme extract preparation for purified lysosomal enzymes was carried out by the method of Beaufay (1972). Homogenate was centrifuged at 2000 rpm for 8 min at 4 −C. the obtained supernatant was re-suspended in phosphate buffer and centrifuged at 11,000 rpm for 40 min to get lysosomal fraction. The resultant sediment was re-suspended in phosphate buffer with 0.1% Triton X 100 to obtain a supernatant of lysosomal fraction. The activity of Acid Phosphatase, α-Galactosidase, β-Glactosidase and β-Glucuronidase were estimated using this lysosomal fraction. The enzyme assay was done as per the method of Tettamanti and Masserini (1984). Protein concentration of the tissue homogenate was determined by the Lowry et al. (1951), using bovine serum albumin as the standard. The obtained data were subjected to different statistical analyses for their cumulative acceptability and hypotheses testing. All statistical analyses were done as per Sokal and Rohlf (1969).
RESULTS AND DISCUSSION
Results of the present study showed noticeable alterations in the lysosomal enzymatic activity in all the doses and exposure durations. The specific activity of acid phosphatase showed inhibitory trend in higher dose and higher exposure duration followed by an insignificant stimulation in 4 days of exposure duration (Fig. 1a). Similarly, the activity of α-galactosidase showed inhibitory trend in higher exposure duration in higher dose, whilst, there was also insignificant stimulation was observed in the lower dose of TCDD exposure (Fig. 1b). In case of β-galactosidase, the specific activity showed little stimulation in higher exposure duration in higher dose, but very less inhibition was observed after 4 days of exposure durations (Fig. 1c). The specific activity of β-glucuronidase showed inhibitory trend in all exposure durations in all doses of TCDD (Fig. 1d).

Graphs showing dose and duration dependent alterations in the specific activity (Pi released /mg protein/hr) after in vivo TCDD intoxication. Error bars represents SD and ‘*’ sign represents the significant variations at P = 0.05 level in the specific activity of mice.
Dioxin and dioxin like polychlorinated biphenyls (PCBs) have adverse physiological effects, all which require binding to the aryl hydrocarbon receptor (AhR) and the route of exposure has little influence effects rather than the hepatic sequestration (Okey, 2007; Nguyen and Bradfield, 2008). They also reported that most of the intracellular receptor, after binding to the ligand, can induce necrosis or cell apoptosis by altering the enzymatic activity of lysosomes. The receptor AhR, after binding with TCDD into the cytoplasm, release to the chaperons proteins where it can associates with dioxin responsive elements (Whitlock et al. 1996). PCBs and dioxins are lipophilic and mainly accumulated in adipose tissue (Jigyasi and Kundu, 2013c, d). In mammals, highly chlorinated dioxin congeners are reported to have slow metabolism and tend to increase their potential to disrupt the signal pathway (Whitlock 1999). Dioxins also known to alter the endocrine pathway which is strongly associated with metabolic fat (Jigyasi and Kundu, 2013 a, b). The toxicity of dioxin and dioxin like PCBs varies from species to species and depends on the accumulation of organochlorine compounds into the lipid droplets (Michele et al. 2013). TCDD is a widespread persistent organic pollutant in the environment. In most human exposure cases, intake of TCDD occurs via food and remains constant in blood and adipose tissue because of its low degradability. TCDD is generally remains in equilibrium in the lipid compartments and is sequestered in adipose tissue. The inhibition of phosphatase and glycosidases enzymes may induce accumulation of macromolecules or other byproduct into the cell which are responsible for interruption of different cycles relevant to carbohydrate metabolic pathway.
In the present study, the statistical analyses employed showed significant variations in the specific activity of all selected lysosomal enzymes (Table 1, 2, 3). The results of two-factor ANOVA showed the exposure duration dependent significant variations in all estimated enzymes. Highly significant variation was observed in the activity of acid phosphatase enzyme (F = 39.61, p = 0.05) (Table 1). It was evident from the present study that TCDD significantly impaired the lysosomal activity. The observed results revealed that both the doses of lipophilic TCDD could alter the lysosomal enzymatic stabilization. Results of the't’ test showed significant variations in the activity of all enzymes estimated. A highly significant variation was observed in the activity of β-glucuronidase (Table 2). The results of one-way ANOVA between individual exposure duration within each group also showed similar results. However, highly significant variations were observed in the activity of α- galactosidase after the intoxication of higher dose group (Table 3).
Results of Two-factor ANOVA between control and toxicated groups. The results of Two-factor ANOVA showed significant time dependent toxic effects of TCDD.

Significance at P = 0.05 (F crit of dF = 3,8) = 3.63
Significance at P = 0.05 (F crit of dF = 8,35) = 2.59
Results of t-test between control and individual exposure duration with in each dose group in liver tissue. The statistical analysis showed that all the selected enzymes are significantly altered in all exposure durations.
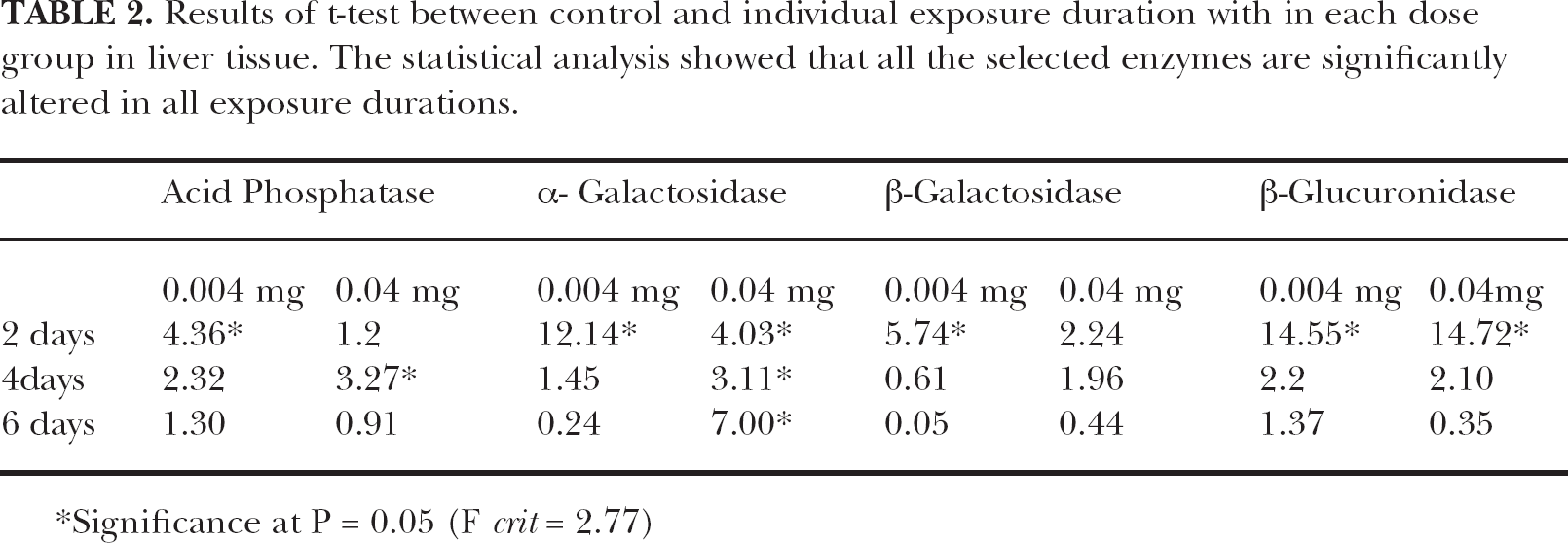
Significance at P = 0.05 (F crit = 2.77)
Results of Single Factor ANOVA between individual exposure duration within each dose group. Results showed the significant variations in the selected enzymes in each group.
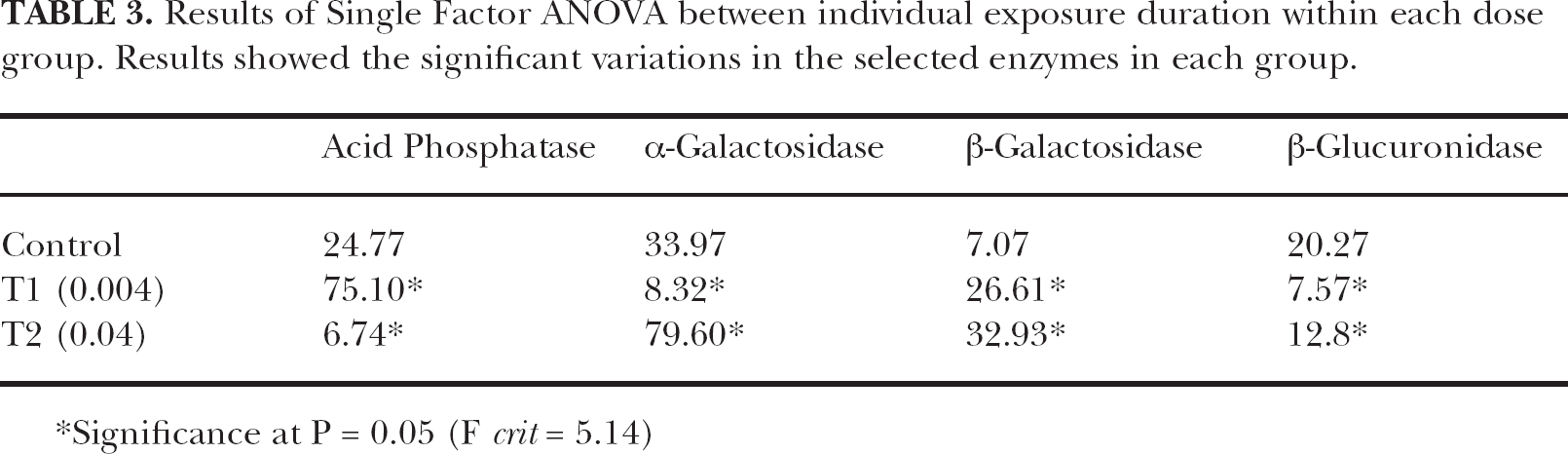
Significance at P = 0.05 (F crit = 5.14)
The cytotoxic effects of organochlorine compounds are known to alter the morphology and functionality of lysosome and accumulate its byproduct for digestion of waste products (Moore, 1991 a, b). It was also reported that when this content exceeds the capacity of lysosome, intra-cellular damage and leakage takes place causing severe cellular damage (Deckers et al. 1980; Viarengo et al. 1981; Moore et al. 1984). Therefore, the lysosomal stability is an extremely sensitive stress index related to a variety of organic and inorganic xenobiotics (Camus et al. 2000). Cytotoxicity can be initiated by changes in the lysosomal volume and thus, related to non-specific manifestations of sublethal injury due to pollutant exposure. The lysosomel activity is considered as general stress marker for bio-monitoring analyses (Lowe et al. 1981; Marigomez et al. 1989; Cajaraville et al. 2000). It was earlier reported that TCDD increases intracellular ions by producing oxidative stress into the cell (Jigyasi and Kundu, 2013a). The organochlorine compounds have the ability to affect the various lysosomal enzymes directly and alter the specific activity which causes apoptotic process followed by the destabilization of lysosomal enzymes in kidney and intestine cells of mice (Bhuva et al. 2014). TCDD was reported to disturb cell homeostasis, caused cell swelling and cell rupture (Pathak and Kundu, 2013 a, b). Similarly, intracellular increase in ions can elevate the prerequisite CPLA2, which is responsible for plasma and lysosomal destabilization (Mukherjee et al. 1997). Further, it was reported that intracellular ions are able to increase intra-lysosomal pH, probably by interacting with membrane proton pump of the cell (Viarengo et al. 2000). These effects together can produce lysophospholipids and another elevating factor or ROS, which can initiate to destruction of cellular activity (Sapirstein et al. 1996).
CONCLUSION
The overall results of the present study showed that even very low concentration of TCDD can alter lysosomal enzymatic activity. This was due to one of the major possibilities that intracellular ion homeostasis was altered by TCDD molecules which were prerequisite for cellular destruction or ROS which are produced by toxic stressors. Organochlorine compounds produce ROS which could also play important role to alter the different physiological activity. Destabilization of lysosomal enzymatic activity might have initiated the accumulation of intracellular macromolecules which leading cellular damage or cell leakage, the prerequisite process of cell apoptosis or cell death.
Footnotes
ACKNOWLEDGEMENT
Authors are thankful to UGC, Govt. of India, New Delhi for supporting this study through its DSA /CAS programme. The senior author is thankful to UGC, New Delhi for Meritorious Scholarship.
