Abstract
P53 protein has an intrinsic role in modulating the cellular response against DNA radioinduced damages and has been pointed out as an indirect indicator of individual radiosensitivity. The rate of cell proliferation is also a parameter that has been related to tissue sensitivity to radiation. However, this feature is yet understudied. In this context, the aim of this work was to employ Flow Cytometry (FC) for simultaneously assessing of p53 protein expression levels together with cellular proliferation rate of irradiated human lymphocytes. From in vitro irradiated human blood samples, mononuclear cells were isolated and labeled with Carboxylfluorescein Diacetate Succinimidyl Ester (CFSE) prior to phytohaemagglutinin (PHA) stimulation in culture for 96 hours. Cells were also labeled with anti-p53 monoclonal antibody PE-conjugated in order to analyze either proliferation rate or p53 expression levels by FC. It was verified a reduction in the proliferation rate of irradiated lymphocytes and, in parallel, a rise in the p53 expression levels, similar for quiescent and proliferating lymphocytes. The results emphasize the importance of the use of CFSE-stained lymphocytes in assays associated to proliferation rate and the use of this methodology in several studies, such as for evaluating individual radiosensitivity.
1. INTRODUCTION
The role of some molecules in the repair of radio-induced DNA damage has been studied and such molecules have been proposed as bioindicators of individual radiosensitivity. The achievement of this goal would be especially important in cancer radiotherapy effectiveness, since individuals differ widely in their responses to ionizing radiation exposures (Borgmann et al. 2002; Schnarr et at. 2007; Pinto et al. 2010). The characteristic of cancer implies that the target volume to be irradiated is generally outlined beyond the tumor boundaries, leading to side effects of radiotherapy that are issues of damages to normal (healthy) tissues. Therefore, normal connective tissue tolerance has an important role in the protocol definition as well as for the effectiveness of radiotherapy (Barnett et al. 2009). In this context, for example, bioindicators of individual radiosensitivity would act as prognostic parameter in cancer radiotherapy.
Due to its intrinsic role in modulating the cellular response against DNA damages, p53 protein levels have being pointed out as an indirect indicator of individual radiosensitivity (Dahm-Daphi 2000; Mayer et al. 2002; Komuro et al. 2003; Cavalcanti et al. 2008; Lu and El-Deiry 2009; Cavalcanti et al. 2011). The most advanced investigations concerning this protein expression and its correlation with radiosensitivity use blood-based assays with stimulated cells, mainly lymphocytes. As human lymphocytes are typically in resting phase (G0), they can reenter the cell cycle if appropriately stimulated to mitosis. However, culture conditions, cell cycle position and differentiation stages are factors that can influence the lymphocyte radiosensitivity in terms of cell death. For example, on actively proliferating cells, apoptosis may occur during the interphase right before mitosis or after some rounds of cell divisions (Carloni et al. 2001). Despite of this fact, little information has shed light on how mitogen stimulation and proliferation are affected and influences p53 protein expression levels in human irradiated lymphocytes.
One of the technologies used to quantify bioindicators, flow cytometry (FC) has been widely applied in investigation of antigens located on the cell surface, cytoplasm or nucleus, in a large number of cells in short time (Andrzej et al. 1998; Nolan and Yang, 2007). The FC has being also used to assess cell proliferation status or capacity, by labeling with intracellular fluorescent dyes. Among the commercially available dyes, Carboxylfluorescein Diacetate Succinimidyl Ester (CFSE) stands out as one of the most used and versatile in terms of homogeneity of lymphocyte staining, membrane stability, optimal concentration and cost-efficiency (Banks et al. 2011). The CFSE staining approach can be associated with fluorochrome-labeled monoclonal antibodies, enabling simultaneous analysis of protein expression level and cell proliferation (Parish 1999; Tario et al. 2007; Parish et al. 2009).
In this scenario, this work was designed to use FC in rapid simultaneous determination of p53 protein expression and the proliferation rate of circulating human lymphocytes irradiated in vitro.
2. MATERIAL AND METHODS
2.1 Studied population and ethical aspects
Blood samples from 5 volunteers (25.6 ± 3.9 years old, coded as A, B, C, D and E) were studied, being 3 females and 2 males. All individuals signed a written informed consent before blood collection, following recommendations of ethics committee from Federal University of Pernambuco (project 297/09).
2.2 Sampling and irradiation
From each donor, a total of 20 mL of peripheral blood was collected by venipuncture in sodium-heparin tubes (Vacuette, USA) and divided into two 10 mL aliquots. One aliquot was irradiated with 4 Gy (Cobalt Irradiator, dose-rate of 4.312 Gy/h) and the remaining aliquot was used as control (non-irradiated).
2.3 Isolation of peripheral blood mononuclear cells
After irradiation, blood was diluted (1:1) with phosphate-buffered saline (PBS, pH 7.2) and deposited onto the Ficoll-Paque PLUS density (1.077 g/mL) gradient (GE Healthcare, USA). After centrifugation, peripheral blood mononuclear cells (PBMC) were individually removed and washed twice with PBS.
2.4 CFSE labeling
4 × 106 cells in 1 mL of PBS were ressuspended with PBS containing 0.25 μM of CFSE (Invitrogen, USA) and incubated at 37 °C for 10 min. CFSE concentration was carefully titrated in order to establish the best concentration to avoid inhibition of cell proliferation or even cell death. After incubation, the labeling was quenched with 1 mL of ice cold (4 °C) RPMI 1640 containing 2 mM of L-glutamine, 50 mg/L of gentamicin sulfate and 2 mg/L of amphotericin B, supplemented with 10% fetal bovine serum (both Cultilab reagents, Brazil). The cells were pelleted and washed once with PBS followed by resuspension with 1 mL of RPMI supplemented at density of 2 × 106 cells/mL.
2.5 Cell culture
Cells were plated in 96-well bottom plates (TPP, Switzerland) at density of 2 × 105cells/well with 10 μg/mL phytohemagglutinin (PHA). From each subject, one part of cells was cultivated without stimulation in order to set-up the flow cytometry. Plates were incubated in a humidified atmosphere of 5% CO2 at 37 °C for 96 hours.
2.6 Cell labeling with antibodies
Following 96 hours of culture, viability of CFSE labeled cells was assessed by the trypan blue dye exclusion method, which stains non-viable cells. Then, cells were harvested, washed once with RMPI supplemented, resuspended with 1% paraformaldehyde and incubated for 20 min. After incubation, cells were washed once with RMPI supplemented, centrifuged for 5 min at 300 × g and, then, cells were ressuspended with permeabilizing solution (50% FACS lysing solution 1X [BD Pharmingen, USA], 50% paraformaldehyde 4% in PBS) and incubated for 10 min at room temperature. The fixation and permeabilization methods employed in this work have been previously established and tested as published elsewhere (Cavalcanti et al. 2008, Cavalcanti et al. 2011). After this, cells were washed twice with PBS 0.5% Tween-20 solution (Merck, Germany). Cell pellet was resuspended with 200 μL of PBS 0.5% Tween20 solution and stained with monoclonal antibodies fluorochrome-conjugated, for 20 min at room temperature. Non-irradiated cells were incubated with mouse IgG1 phycoerithrin (PE)-conjugated (Clone MOPC-21 -BD Biosciences, USA), the isotype control antibody. For detection of p53, irradiated and non-irradiated cells were incubated with anti-p53 antibody PE-conjugated (Clone G59-12 - BD Biosciences, USA). After labeling, cells were washed twice with PBS 0.5% Tween-20 solution and fixed with 500 μL of 1% paraformaldehyde in PBS.
2.7 Flow cytometry analysis
The analyses were performed on a FACScalibur flow cytometer (Becton Dickinson Company, USA) equipped with an argon laser (wavelength 488 nm). Fluorescence of 50,000 lymphogated events, based on scatter parameters of size and granulosity was acquired. Data were analyzed and treated with FlowJo 7.6 (Tree Star Inc., USA). Non stimulated cells were used during analysis for setting quadrant parameters, and the levels of p53 protein were subtracted from the value of isotype control (less than 1%).
2.8 Statistical analysis
The results were presented as mean and standard deviation. Differences between means were evaluated by Student's t-test, and they were considered statistically significant when p < 0.05.
3. RESULTS
For each subject studied, control of labeling and proliferation was done with non-stimulated cells. Figure 1 shows dot-plots from lymphocytes non-stimulated with mitogen, non-irradiated (A) and irradiated with 4 Gy (B).

Dot-plots obtained during analysis of lymphocytes non-stimulated with mitogen, non-irradiated (A) and irradiated with 4 Gy (B). The upper quadrants allows the visualization of p53 expression (left, negative; right, positive) and the lower quadrants, the pattern of proliferation (left, proliferating; right, quiescent).
As expected, non-stimulated lymphocytes kept in culture were not able to proliferate and showed high levels of CFSE identified for its high fluorescence.
Figure 2 presents dot-plots obtained during analysis of non-irradiated (0 Gy) and irradiated (4 Gy) mitogen-stimulated samples from five subjects (coded as A, B, C, D and E). In each graphic, lower right quadrant displays quiescent lymphocytes (CFSE high fluorescence) non-positive for p53; upper right quadrant displays quiescent cells positive for p53 expression; lower left quadrant displays proliferating lymphocytes (CFSE low fluorescence) non-positive for p53; upper left quadrant displays proliferating cells positive for p53 expression.
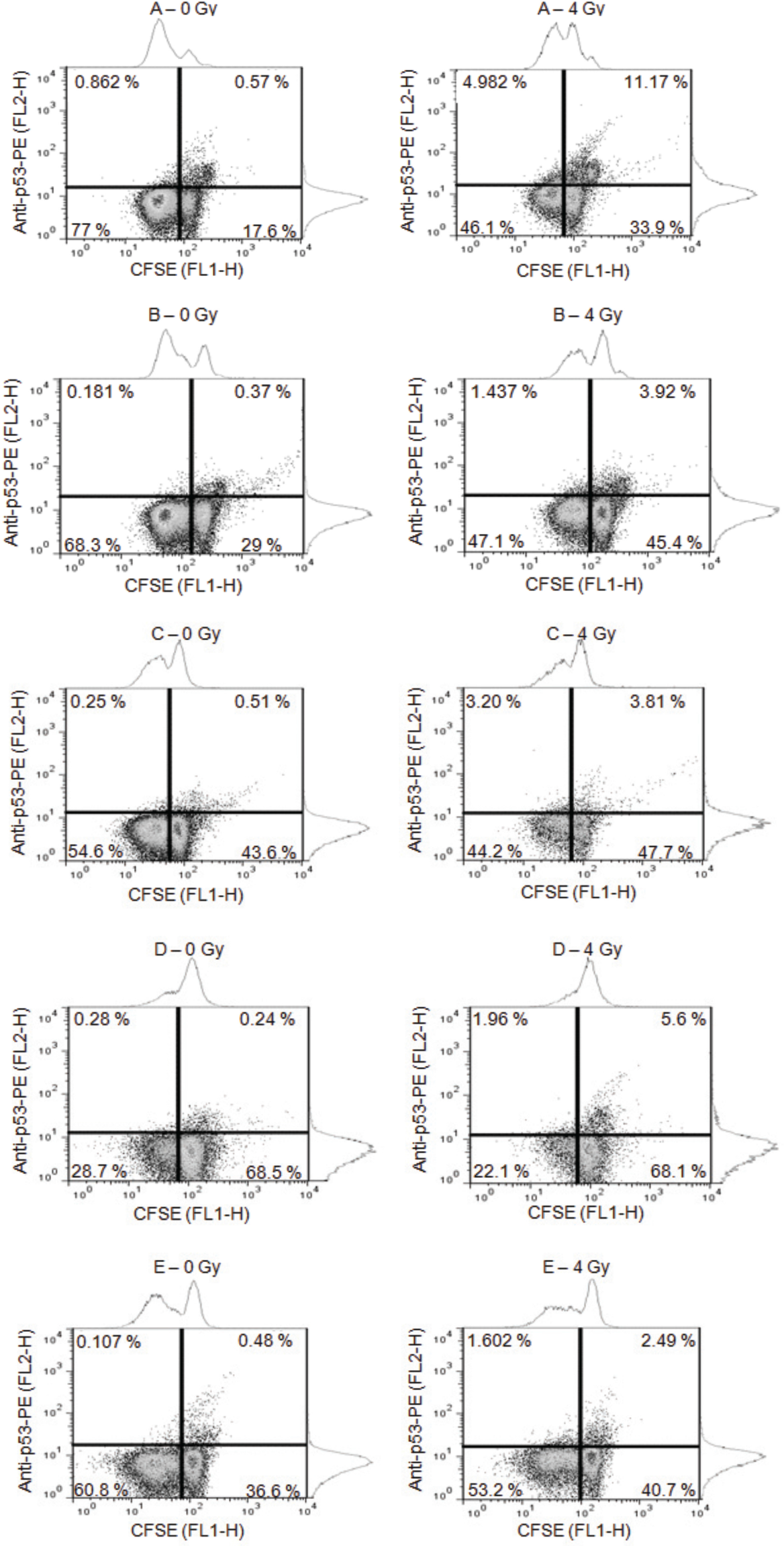
Dot-plots obtained during the analysis of non-irradiated (0 Gy) and irradiated (4 Gy) proliferating samples from subjects A, B, C, D and E. Lower right quadrant displays quiescent lymphocytes (CFSE high fluorescence) non-positive for p53 and upper right quadrant displays quiescent cells positive for p53 expression; lower left quadrant displays proliferating lymphocytes (CFSE low fluorescence) non-positive for p53 and upper left quadrant displays proliferating cells positive for p53 expression.
The exposure of blood samples to 4 Gy resulted in an increase of quiescent lymphocytes, from (39.06 ± 19.07)% to (47.16 ± 12.83)%, and the consequent decreasing of proliferating lymphocytes, from (57.88 ± 18.33)% to (42.54 ± 11.91)%. However, this radioinduced effects was statistically significant only for proliferating cells (p < 0.05).
About p53 protein expression, Table 1 presents expression levels values for quiescent and proliferating lymphocytes from unexposed and 4 Gy-exposed peripheral blood samples of each subject.
p53 expression levels in all samples analyzed before (control non-irradiated) and after in vitro irradiation.

Regarding this data, for each group (control or irradiated lymphocytes) there is no statistical difference in p53 expression between quiescent and proliferating cells. However, it is observed a significantly increase of this protein expression levels after irradiation (p < 0.05), either for quiescent or proliferating lymphocytes.
4. DISCUSSION
In this research, lymphocytes were chosen due to the ease of obtaining and handling such cells in laboratory and as a result of the well-established method of labeling them with CFSE, especially for studies of immunology. Although PHA is widely employed on researches aimed to investigate molecular parameters in stimulation of lymphocytes, this mitogen is mainly effective in the stimulation and activation of T cells, acting differently for B lymphocytes that are negligibly stimulated (Vilasová et al. 2008; Moghbeli-Nejad et al. 2012). Henceforth, all results presented in this report are related to T lymphocytes (O'Donovan et al. 1995).
This research employs the same CFSE concentration successfully used in similar investigation (Brenchley and Douek 2004; Seggewiss et al. 2005). However, in proliferation rate studies, there is a discussion whether CFSE concentration can keep cells alive or not. Some authors highlight that high concentrations of CFSE can inhibit cell proliferation (the cellular event to which this dye is made to detect) (Quah et al. 2007) or even kill the cells. Lašt'ovička et al. (2009) have described a range of concentrations in which CFSE can damage lymphocytes enough to kill them. To avoid this drawback, in this study CFSE concentration was carefully titrated with a wide range of concentrations to achieve the best concentration able to keep cells alive and normally proliferating, as confirmed by the use of trypan blue dye method. The choice of the trypan blue dye method was a practical decision due to either the low costs or its efficiency in the evaluation of cell viability.
CFSE is a very versatile dye which permits to monitor in vivo and in vitro lymphocyte division (Lyons 1999; Parish 1999; Lyons 2000; Veiga-Fernandes et al. 2000). At the molecular level, CFSE is non-fluorescent with ability to penetrate in the cell and couples to cellular proteins. Once in the cell, esterases remove acetate groups of the molecule that from this point becomes highly fluorescent. CFSE has the advantage of labeling cells regardless of their stage in the cell cycle (Asquith et al. 2006) and its fluorescence intensity is progressively halving after each cell division (Quah et al. 2007). Actually, flow cytometric evaluation allows the detection and quantification of several different molecules produced by a single cell by using markers with different light emission properties.
However, the analyses employing probes with similar CFSE light emission are not straightforward (Tario et al. 2007). The use of CFSE dye for evaluating cell proliferation is cost-efficient in comparison with other techniques, such as the gold standard tritiated thymidine (3H-TdR). 3H-TdR is a radioactive molecule that labels the DNA of proliferating cells, but due to its cost and need of dedicated lab facilities and special procedures, this methodology has been progressively replaced (Evrard et al. 2010).
Our results showed an increase in the level of quiescent lymphocytes and a decrease of proliferating lymphocytes, which might indicate a direct consequence of the apoptotic phenomenon. One of the effects of cellular irradiation is cell cycle arrest, a phenomenon commonly seen as a time-lapse to evoke proteins involved in the repair of damages or even to induce cell death (Wood and Chen, 2008, Landsverk et al. 2011).
In normal conditions it is not expected an increase in the levels of p53 protein due to its short average lifetime. On the other hand, after cell irradiation an increase of this protein concentration occurs, which is accompanied by an increase in its mean life span (Fuchs et al. 1998; McVean et al. 2000; Fei and El-Deiry 2003; Cavalcanti et al. 2008). As expected, the results of this study showed an increase of p53 expression at different levels for each subject. However, it was not expected that p53 could be expressed for both quiescent and actively proliferating cells. P53 is a central molecule in the response to radioinduced damages on the DNA, which are followed by activation of a complex process that acts to arrest the cell cycle and allows the identification and repair of damages (Stehg 2012). The role of p53 in the response to DNA damages in lymphocytes actively proliferating has yet to be well determined. These results about the diminishment of lymphocyte proliferation in irradiated samples are indicative of a relationship between p53 expression and cell death. Nevertheless, the exactly mechanism by which p53 and survivin interact to induce cellular apoptosis is not well established.
The results of this research are suggestive that the levels of p53 expressed by proliferating lymphocytes are related with either the cellular apoptotic process to eliminate cells carrying chromosomal damages induced by IR, or the prolonged time-culture and its consequences to the cell survival. Hence, in order to correlate the expression of p53 and the radiosensitivity of tissues, one may postulate that in case the studied subjects are accidentally exposed to IR, at the same conditions, all of them will respond similarly to the irradiation, with exception of perhaps the subject A.
Observing the Table 1, the levels of p53 expression of subject A after irradiation are considerably higher when compared to the others. Moreover, the levels of p53 expressed by the quiescent population irradiated (11.17%) were approximately two fold higher than their proliferating counterparts (4.98%). These high p53 expression levels of the subject A would indicate that this individual will probably overreact to the radiation exposure differently, when compared to the others.
Researchers have shown that some tissues from humans and mice expressing high levels of p53 have in common an elevated sensitivity to IR (MacCallum et al. 1996; Dainiak 2002). Cavalcanti et al. (2011) emphasized a dose-dependent elevation of p53 levels in human lymphocytes. Even being of extreme importance, p53 is not the only factor which might contribute to the cellular fate after exposure to IR.
The assessment of proliferative status of cells from other tissues can also be done in combination with the staining for other intracellular or extracellular proteins. In this sense, we recommended assays with CFSE-labeled lymphocytes to identify differences on cell proliferation rate and p53 protein expression. By this methodology, it was identified a rise of p53 expression levels in irradiated lymphocytes, either quiescent or actively proliferating, and a reduction of the proliferation rates of irradiated lymphocytes. This latter is likely a consequence of activation of a cell death program, eliminating cells by apoptosis even when they are under mitotic stimulation. This approach was completely original in the field of p53 studies since, as far as we know, there is no data in the literature contrasting, at the same time, the expression of p53 and the proliferation status by the use of CFSE dye in human irradiated lymphocytes. The results presented in this report suggest the use of this methodology for more accurate evaluation of protein dose-response from irradiated cells. Naturally, to demonstrate the usefulness of this methodology, further studies with a greater number of subjects are needed in order to support the findings.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank the Centro de Pesquisas Aggeu Magalhães (Fiocruz-CPqAM-Brazil) for providing laboratory facilities; Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq-Brazil) for financial support. Also, we warmly acknowledge Maria Carolina Accioly Brelaz de Castro for technical contributions. The authors declare that there are no conflicts of interest.
