Abstract
Central thalamus regulates forebrain arousal, influencing activity in distributed neural networks that give rise to organized actions during alert, wakeful states. Central thalamus has been implicated in working memory by the effects of lesions and microinjected drugs in this part of the brain. Lesions and drugs that inhibit neural activity have been found to impair working memory. Drugs that increase activity have been found to enhance and impair memory depending on the dose tested. Electrical deep brain stimulation (DBS) similarly enhances working memory at low stimulating currents and impairs it at higher currents. These effects are time dependent. They were observed when DBS was applied during the memory delay (retention) or choice response (retrieval) but not earlier in trials during the sample (acquisition) phase. The effects of microinjected drugs and DBS are consistent with the Yerkes-Dodson law, which describes an inverted-U relationship between arousal and behavioral performance. Alternatively these results may reflect desensitization associated with higher levels of stimulation, spread of drugs or current to adjacent structures, or activation of less sensitive neurons or receptors at higher DBS currents or drug doses.
INTRODUCTION
Impairments of working memory and other aspects of prefrontal executive function are among the most debilitating effects of neuropsychiatric disorders for which there are no effective cures. Currently available clinical treatments focus on drugs that act systemically on monoaminergic or cholinergic systems to enhance prefrontal cortical activity. Cholinergic and monoaminergic neurotransmitters are normally released in response to state-related changes in stress, affect, and arousal, and have distinct but widespread effects on cortical and subcortical functions (Parikh and Sarter, 2009; Robbins and Arnsten, 2009). This paper will focus on an alternative approach to activating prefrontal cortex by stimulating central thalamus with either microinjected drugs or electrical deep brain stimulation (DBS).
Central thalamic nuclei offer important advantages as a means to manipulate frontal cortical function although they present a number of challenges for translational research aimed at developing effective clinical treatments. Central thalamus includes the midline and intralaminar nuclei and paralaminar areas of adjacent nuclei (Figure 1). These nuclei contain dense concentrations of matrix neurons that project to superficial layers of cortex and are hypothesized to synchronize activity in neural networks that underlie cognitive functions (Jones, 2009). They receive common visceral and arousal-related inputs from the brainstem including the deep mesencephalic reticular nucleus; brainstem noradrenergic, serotonergic, and cholinergic nuclei; and histaminergic and orexinergic neurons in hypothalamus (Steriade et al., 1997; Bayer et al., 2002; Krout et al., 2002). Central thalamic neurons exhibit both activated “up” states, which correspond to alert wakefulness and “down” states, which correspond to sleep and drowsy inattentiveness (Glenn and Steriade, 1982; Kinomura et al., 1996; Paus, 2000). These neurons also exhibit more momentary responses to external stimuli that trigger behavioral actions (Matsumoto et al., 2001; Wyder et al., 2003).

Organization of central thalamus. Photomicrographs of coronal sections through anterior (A), middle (B), and posterior (C) levels of rat thalamus stained with cresyl violet (approximately 7 mm, 6 mm, and 5 mm anterior to the interaural line in a flat skull position). Locations are marked for the rostral intralaminar nuclei, including centrolateral (CL), paracentral (PC), and central medial (CM); caudal intralaminar nuclei, including the parafascicular (PF) nucleus; dorsal midline nuclei, including paraventricular (PV) and paratenial (PT); and ventral midline nuclei, including rhomboid (Rh), reuniens (Re), and ventral reuniens (VRe). Other labels indicate the locations of the third ventricle (3V), stria medularis of thalamus (sm), fasciculus retroflexus (fr), mammillothalamic tract (mt), habenula (Hb), and the mediodorsal (MD) and interanteromedial (IAM) nuclei.
Central thalamic nuclei have distinct afferent and efferent connections that appear organized to influence specific aspects of arousal and awareness. Individual nuclei project preferentially to functionally distinct areas of frontal cortex and to anatomically related targets in the basal ganglia and limbic system. Thus they appear organized to influence large-scale neural networks that underlie the capacity for intentional responding and consciousness (Van der Werf et al., 2002; Vertes, 2006; Schiff, 2008). In the rat, the dorsal (paraventricular and paratenial) and ventral (reuniens and rhomboid) midline nuclei (Figure 1) project heavily to areas of ventral midline prefrontal cortex (Hoover and Vertes, 2007; Vertes and Hoover, 2008), an area important for supervisory attentional control, behavioral flexibility, and sensory working memory (Kesner, 2000; Dalley et al., 2004). Rostral intralaminar nuclei (central lateral, paracentral, central medial in Figure 1) project to dorsal prefrontal cortex and adjacent secondary motor areas (Berendse and Groenewegen, 1991; Hoover and Vertes, 2007) which are important for motor planning, temporal coding, and motor working memory (Kesner, 2000; Bailey and Mair, 2004, 2007). Caudal intralaminar nuclei (lateral parafascicular in Figure 1) innervate primary sensorimotor cortex, an area important for skilled movements and sensory-guided responding (Bailey and Mair, 2004; Alaverdashvili and Whishaw, 2008).
AROUSAL AND COGNITIVE PERFORMANCE
Stress and anxiety have powerful effects on cognitive performance. Low levels of arousal result in drowsiness, partial awareness, and inefficient thinking. Moderate levels are required for alert and attentive states, optimal for efficient, quick reactions and organized responding during complex tasks. High levels of stress and anxiety can lead to fear and rage, restricted awareness, and confused, disorganized behavior. The relationship between arousal and cognition is governed by two principles derived from a relatively simple experiment conducted by Yerkes and Dodson in 1908. Yerkes and Dodson varied levels of electric shock used to condition mice to perform three different brightness discriminations. In the easiest discrimination, where the brightness difference was greatest, mice performed progressively better, requiring fewer trials to learn the discrimination as the intensity of the shock increased “from the threshold of stimulation to the point of harmful intensity.” When discriminations were made more difficult by reducing the brightness difference, mice required more trials in general to learn discriminations and exhibited curvilinear (inverted-U) relationships between shock intensity and performance. Yerkes and Dodson argued that: i. for all but the easiest discrimination problems performance improves as stimulation increases above threshold to an optimal level and then deteriorates at higher levels of stimulation; and ii. the optimal level of stimulation “depends upon the difficultness of the habit” with more difficult tasks having lower levels of optimal arousal.
Over the century since Yerkes and Dodson, we have developed a more sophisticated understanding of neural networks that allow organisms to decide between response alternatives and their interactions with brain mechanisms that regulate attention and arousal. Successful decision-making requires cognitive processing of relevant information, estimation of relationships between actions and their potential consequences, and executive functions to optimize decision-making performance. Convergent evidence suggests that decision making depends on parallel pathways that connect frontal cortex with striatum, pallidum, and thalamus (Mair et al., 2002; Bailey and Mair, 2004; Balleine et al., 2007; Lee et al., 2007; Shiffett et al., 2010). Sensorimotor areas of frontal cortex and dorsolateral striatum/ putamen play a critical role in stimulus-guided actions that are relatively automatic or habitual. Prefrontal cortex and dorsomedial striatum/ caudate play a critical role in flexible, goal-directed actions guided by working memory by representing information in the brain in absence of external sensory information (Goldman-Rakic, 1996; Arnsten, 2009).
The activity of cerebral cortex is modulated by neurotransmitter systems that are differentially affected by behavioral state conditions related to stress, mood, attention, and arousal. Many of these systems function in both phasic and tonic modes, reflecting short latency responses to behaviorally relevant stimuli along with more gradual state-related changes in arousal and awareness. Although different neurotransmitters have distinct effects on cognition they affect diverse cortical and subcortical targets and thus diverse functions (Robbins and Roberts, 2007; Parikh and Sarter, 2009; Robbins and Arnsten, 2009). There are number of excellent recent reviews of these neurochemical systems, their influence on prefrontal cortical function, and their utility for treating conditions that affect prefrontal function such as ADHD, bipolar disorder, schizophrenia, and dementia (Aston-Jones and Cohen, 2005; Robbins and Roberts, 2007; Arnsten, 2009; Parikh and Sarter, 2009; Robbins and Arnsten, 2009).
The catecholamines norepinephrine and dopamine are probably the best understood of the neuromodulatory systems that affect prefrontal function. Stimulants that increase the release of catecholamines in prefrontal cortex have long been the mainstay for treating ADHD. Although it was once thought that stimulants had a paradoxical calming effect on individuals with ADHD, it is now clear that low doses of stimulants enhance prefrontal functions in normal and ADHD individuals and that higher doses tend to disrupt these functions. Studies of rodent and primate species have provided evidence of comparable inverted-U relationships between catecholamine activity and prefrontal function. The beneficial effects of stimulants in ADHD are thought to reflect a normalization of function in prefrontal cortico-striatal circuits that are impaired in these disorders (Shafritz et al., 2004; Arnsten, 2006). In monkeys, drugs blocking either noradrenergic α2a or dopaminergic D1 receptors impair working memory (Sawaguchi and Goldman-Rakic, 1991; Li and Mei, 1994) and disrupt coding properties of prefrontal neurons during brief memory delays (Williams and Goldman-Rakic, 1995; Li et al., 1999). Moderate levels of catecholamines selectively activate α2a and D1 receptors and enhance working memory performance and neural coding in prefrontal cortex (Vijayraghavan et al., 2007; Wang et al. 2007). Excessive release of catecholamines, observed with high levels of stress, activate lower affinity α1 and β adrenoreceptors and produce excessive levels of D1 activation. Excessive α1 stimulation disrupts prefrontal activity through its effects on phosphatidylinositol-protein kinase C, while excessive D1 activation disrupts prefrontal activity through its effects on cyclic AMP (Hains and Arntsen, 2008).
THALAMIC CONTROL OF AROUSAL AND AWARENESS
Interactions between thalamus and cortex play a critical role in regulating arousal and awareness. It has long been recognized that cortical states of arousal are controlled by neurons in the brainstem reticular formation (Moruzzi and Magoun, 1949) and that central thalamic nuclei play an important role in what was once viewed as a diffuse, nonspecific reticulo-thalamo-cortical activating system (Lindsley, 1951). Thalamic neurons switch between two modes of action related to behavioral state: a depolarized tonic firing mode associated with awake attentive states and a burst firing mode produced by sustained periods of hyperpolarization associated with drowsy inattentiveness and sleep (Llinas and Steriade, 2006). The tonic mode is associated with subthreshold high frequency oscillations in the gamma range (30 to 60 Hz) and the burst mode with low frequency (0.5 to 1 Hz) oscillations, both of which are synchronized across wide regions of cortex and thalamus through diffuse corticothalamic and thalamocortical projections (Jones, 2009). These state-related modes of thalamic activity are influenced by projections originating in cortex and by inputs from brainstem arousal systems, including neurons releasing acetylcholine and norepinephrine. Recent evidence suggests that manipulations of thalamic activity by intrathalamic application of drugs acting on noradrenergic or cholinergic receptors can shift the activity of cortex directly between desynchronized activated states associated with arousal and synchronized deactivated states indicative of drowsiness (Hirata and Castro-Alamancos, 2010).
Studies of neural activity in awake, behaving organisms have confirmed the importance of central thalamus in regulating arousal and awareness. Functional imaging of human subjects has revealed activation of central thalamus and midbrain reticular formation during wakefulness that is increased as demands on attention are increased (Kinomura et al., 1996; Paus, 2000). By contrast, central thalamic activity is diminished when subjects are rendered unconscious by general anesthesia (Fiset et al., 1999; White and Alkire, 2003). This relationship has been confirmed by evidence that the effects of anesthesia are reversed in animal models when central thalamus is activated by microinjection of nicotine or an antibody to a voltage-gated potassium channel directly into central thalamus (Alkire et al., 2007, 2009).
Central thalamic neurons show phasic as well as tonic patterns of activation. Analyses of fMRI have demonstrated momentary shifts in central thalamic activity associated with EEG signs of cortical arousal (Nagai et al., 2004). Single unit recordings in monkeys have also revealed momentary responses of central thalamic neurons to behaviorally significant events. Thus neurons in sensorimotor areas of posterior intralaminar nuclei exhibit temporally discrete responses to visual, auditory, or somatosensory stimuli (Matsumoto et al., 2001), while prefrontal-related areas in rostral intralaminar nuclei exhibit stimulus, response, and delay-period activity in a delayed saccade task (Wyder et al., 2003). These results provide evidence of momentary fluctuations in central thalamic activity related to salient sensory or behavioral events superimposed on tonic levels of activity associated with behavioral state conditions.
DISCRETE CENTRAL THALAMIC LESIONS DISRUPT SPECIFIC FRONTAL CORTICAL FUNCTIONS
The importance of the rostral intralaminar nuclei in working memory was first appreciated through clinical case studies of amnesia and the pyrithiamine-induced thiamine deficiency (PTD) model of amnesia (Mair, 1994). The PTD model produces lesions that target the intralaminar nuclei (Figure 2A) and produce a pattern of impaired working memory and spared rule learning consistent with amnesia. The importance of the intralaminar nuclei was confirmed by evidence that memory is spared in rats that lack intralaminar lesions after recovery from PTD treatment (Mair et al., 1988, 1991), as well as in rats where lesions are prevented by pharmacological blockade of NMDA receptors (Robinson and Mair, 1992). Other studies have demonstrated comparable patterns of impairment following lesions damaging the intralaminar nuclei, but not following lesions of other areas damaged by PTD treatment (Mair and Lacourse, 1992; Burk and Mair, 1998).

Central thalamic lesions. Photomicrographs of lesions in coronal plane and stained with cresyl violet. Lesions include damage produced by pyrithiamine-induced thiamine deficiency (A) and NMDA microinjected into the rostral intralaminar nuclei (B), ventral midline nuclei (C), and caudal intralaminar nuclei (D).
The effects of central thalamic lesions must be analyzed with care, given the close proximity of nuclei and fiber tracks and the likelihood that a lesion producing substantial damage in one nucleus will produce unintended damage to adjacent structures (Figure 2). Two approaches have been taken to examine the specificity of lesion effects. The first is to compare the effects of lesions aimed at different areas of central thalamus predicted to have distinct effects on cognitive function. The second is to compare the effects of lesions aimed at discrete nuclei with lesions in areas of cortex, basal ganglia, or the limbic system innervated by those nuclei. The first approach has revealed discrete effects of lesions in different parts of central thalamus. We recently compared effects of lesions in the rostral intralaminar (central lateral, paracentral), ventral midline (reuniens, rhomboid), and caudal intralaminar (parafascicular) nuclei (Figure 2 B, C, and D). We found evidence of a double dissociation in which lesions of the caudal intralaminar site impaired visually guided responding while sparing spatial memory, whereas lesions in ventral midline produced the opposite pattern of impairment (Hembrook and Mair, 2010). The specificity of these deficits were confirmed by: evidence that lesions damaging other parts of central thalamus did not exacerbate impairments observed with discrete lesions restricted to these sites; and evidence from other studies that the effects of the ventral midline and caudal intralaminar lesions are similar to deficits observed with lesions in the terminal fields innervated by these nuclei.
The second approach has provided evidence that intralaminar lesions affect functions mediated by the areas of cortex and striatum they innervate (Mair et al., 2002; Bailey and Mair, 2004, 2006, 2007). Delayed match-to-position (DMTP) is a task that requires the capacity for motor working memory and response flexibility thought to depend on dorsal and ventral parts of medial prefrontal cortex (Kesner, 2000). Rostral intralaminar lesions (Figure 2B) produce a delay-independent pattern of DMTP impairment in which comparably severe impairments are observed across the memory delays tested (Bailey and Mair, 2005). Similar delay independent patterns of impairment have been observed for lesions damaging anatomically-related areas in prefrontal cortex (Mair et al., 1998), ventral and dorsomedial striatum (Burk and Mair, 2001), and ventral pallidum (Zhang et al., 2005). The importance of circuitry linking rostral intralaminar nuclei with these areas was confirmed in a study in which the rostral intralaminar nuclei were reversibly inactivated in rats by microinjections of lidocaine into thalamus in either the same hemisphere or the hemisphere opposite a lesion of ventral striatum (Porter et al., 2001). Unilateral inactivation produced significantly greater impairment when administered on the side opposite the lesion. This crossed lesion effect provides evidence of a common circuit involving ventral striatum and the rostral intralaminar nuclei underlying this impairment.
CENTRAL THALAMIC REGULATION OF PREFRONTAL FUNCTION: PHARMACOLOGICAL STUDIES
Central thalamic activity is influenced by multiple neurotransmitter systems that provide potential targets for pharmacotherapy. The inhibitory neurotransmitter GABA is released by extrathalamic projections arising in ventral pallidum and pars reticulata of substantia nigra, projections from the thalamic reticular nucleus, and local interneurons. Working memory has been shown to be impaired by microinjection of either the GABAA agonist muscimol or the GABAB agonist baclofen in central thalamus (Romanidies et al., 1999; Mair and Hembrook, 2008). Comparable effects have been observed with pharmacological stimulation of ventral pallidal neurons that release GABA in central thalamus (Kalivas et al., 2001; Zhang et al., 2005).
Central thalamic activity is also influenced by many of the same neuromodulators that regulate frontal cortical activity (Asanuma, 1997). Cholinergic innervation of thalamus arises from the mesopontine nuclei and plays a critical role in the control of natural states of sleep and waking (Asanuma, 1997; Steriade, 2004). Acetylcholine depolarizes thalamocortical neurons and thus blocks low frequency rhythms characteristic of slow-wave sleep and generates fast rhythms characteristic of alert states. Stimulation of mesopontine cholinergic nuclei produces similar effects (Steriade, 2004). There is relatively little known about the influence of mesopontine-thalamic cholinergic projections on cognitive function. Non-selective, conventional lesions of the mesopontine cholinergic nuclei impair performance of tasks measuring attention (Inglis et al., 2001) and memory (Keating and Winn, 2002) functions of prefrontal cortex. Pharmacological blockade of nicotinic cholinergic receptors in the rostral intralaminar nuclei has been found to affect reaction time and accuracy of visually guided responding (Newman and Mair, 2007). Central thalamus is uniquely targeted by orexins, hypothalamic neuropeptides that promote alertness and arousal, particularly after lengthy periods of wakefulness (Sakurai, 2007). The orexins act directly on the cell bodies and axon terminals of central thalamic neurons that project to prefrontal cortex (Lambe et al., 2007). While orexins have been shown to have powerful wakefulness-promoting effects there is relatively little known about their effects on cognition (Deutch and Bubser, 2007).
To examine the influence of central thalamus on prefrontal function, Mair and Hembrook (2008) trained rats to perform the DMTP task and then microinjected drugs into the rostral intralaminar nuclei. To test the utility of treating prefrontal impairment with these drugs, results were compared for rats with lesions in medial prefrontal areas known to affect DMTP performance. Inhibiting central thalamic activity with the GABAA agonist muscimol produced impairment that increased as a function of dose (Figure 3). This impairment was delay-independent, consistent with results for lesions damaging the rostral intralaminar nuclei or anatomically related areas in dorsal prefrontal cortex, ventral striatum, or ventral pallidum (Mair et al., 1998; Burk and Mair, 2001; Bailey and Mair, 2005). Injecting muscimol in a nearby site as an anatomical control eliminated the impairment associated with the highest dose tested. Thus, these effects appear to have been localized in the area of the rostral intralaminar injection site. Comparable effects were observed for rats with and without prefrontal lesions.

Effects of drugs microinjected into the rostral intralaminar nuclei on delayed matching to position (DMTP) performance (data from Mair and Hembrook, 2008). Results are compared for rats with and without prefrontal lesions. Inhibiting neurons with the GABAA agonist muscimol produced dose dependent DMTP impairment. Injection of the 1.0 nmol dose at an anatomical control site (Anat Cnt) had no effect on performance. Exciting neurons with the negative allosteric GABAA modulator n-methyl-β-carboline-3-carboxamide (FG7142) produced an inverted-U dose response function. Exciting with orexin A enhanced DMTP at the longest delays tested for the highest dose tested (0.1 nmol).
To increase central thalamic activity n-methyl-β-carboline-3-carboxamide (FG-7142) was injected into the rostral intralaminar site. FG-7142 is an inverse benzodiazepine agonist that has an allosteric effect reducing the affinity of GABAA receptors for GABA and thus decreasing the level of inhibition. Previous studies have shown evidence of an inverted-U dose response curve when FG-7142 was administered systematically in rats performing the DMTP task (Cole and Hillman, 1991; Burk et al., 1999). Here similar results were observed for intrathalamic injections (Figure 3). DMTP performance improved up to a dose of 0.1 nmol and fell off when doses were increased to 1.0 and 10 nmol. Again comparable effects were observed for rats with and without medial prefrontal lesions. Taken together, the effects of muscimol and FG-7142 show a clear example of hormesis. Decreasing activity by stimulating GABAA receptors with muscimol produced dose dependent impairment, while increasing activity by reducing GABAergic tone with FG-7142 improved performance at low doses and impaired performance as doses increased beyond an optimal level.
Results for drug manipulations of orexinergic and cholinergic activity were more equivocal although again prefrontal lesions did not interact with any of the drug effects. Microinjection of orexin A was associated with a slight increase in performance at the highest dose tested (0.1 nmol). Here significant benefits were limited to the longest memory delay tested. By contrast, the cholinergic agonist carbachol produced signs of impairment at all doses tested. While these results may simply reflect opposite effects of Orexin A and carbachol on performance, the possibility remains that testing a wider range of doses (higher for Orexin A, lower for carbachol) may have provided evidence of an inverted-U relationship consistent with results for FG-7142.
CENTRAL THALAMIC REGULATION OF PREFRONTAL FUNCTION: DEEP BRAIN STIMULATION
Deep brain stimulation (DBS) is a treatment in which specific sites of the brain are stimulated with trains of electrical impulses through permanently implanted electrodes. DBS has been shown to have substantial therapeutic benefits for otherwise treatment-resistant cases of Parkinson's disease, essential tremor, and dystonia as well as chronic pain, affective disorders, and obsessive-compulsive disorder (Rabins et al., 2009). DBS in the pedunculopontine tegmental nucleus, an important source of cholinergic innervation of central thalamus, was recently reported to activate prefrontal cortex and enhance executive function in a small group of Parkinsonian patients (Alessandro et al., 2010). A notable clinical case report described behavioral recovery in a minimally conscious patient associated with DBS of the rostral intralaminar nuclei using a double blind alternating crossover design to confirm the relationship between stimulation and recovery (Schiff et al. 2007). DBS of the rostral intralaminar nuclei in the rat has been found to induce immediate-early gene expression in cerebral cortex and to enhance measures of object memory in spontaneous exploration (Shirvalkar et al., 2006).
DBS has obvious advantages for targeting functionally distinct clusters of central thalamic nuclei although questions remain about its utility for activating neurons in these areas. The precise mechanism of action of DBS is not well established. The premise for many current applications is that prolonged periods of DBS will temporarily disrupt activity in abnormally functioning areas of the brain and thus reduce unwanted symptoms. Recordings of neurons near the stimulating site or terminal fields innervated by these neurons have revealed excitation, inhibition, and more complex responses to DBS. High frequency stimulation used for DBS is commonly thought to activate axons leaving the area of stimulation while producing effects that mirror those of lesions damaging the tissue stimulated (Perlmutter and Mink, 2006; Kringelbach et al., 2007; Gradinaru et al., 2009). Recent results, however, suggest that the effects of DBS are more complex than this interpretation would suggest. Quinkert et al. (2010) report that DBS in hippocampus or central thalamus in mice can produce behavioral signs of arousal and changes in cortical EEG. Interestingly the behavioral effects of this stimulation varied with the temporal pattern of stimulus pulses used for DBS. Hamani et al. (2010) found that subcallosal cingulate DBS has antidepressant-like effects on forced swim test performance. Here the effects of DBS varied with stimulus amplitude following an inverted-U relationship: the strongest effect was observed for 200 μA with smaller effects at higher or lower levels. Taken together, these results suggest that the effects of DBS vary in a complex fashion with stimulation parameters in a manner inconsistent with simple monotonic disruption of neuronal function.
Mair and Hembrook (2008) carried out studies to address two hypotheses about the utility of central thalamic DBS. The first hypothesis concerns the level of current used to stimulate thalamus. The effects of FG-7142 in central thalamus provides evidence of an inverted-U relationship between the extent of neural activation and the benefits for working memory (see above). To test the hypothesis of an inverted-U relationship for DBS, bipolar electrodes were implanted to stimulate the rostral intralaminar nuclei in rats trained to perform DMTP. Performance was compared for sessions in which DBS stimulating current was varied between sessions. For each rat initial studies were conducted to determine a low level of current that appeared to enhance performance and a higher level of current that appeared to impair performance without affecting the fluency of movement. DBS in this study consisted of relatively brief (1 s) trains of high frequency current pulses (0.2 ms pulses at 120 Hz).
The second hypothesis concerns the temporal dynamics of stimulation: Does it matter when DBS is applied during a working memory task? The traditional view of central thalamus has focused on its role in regulating tonic behavioral state (Glenn and Steriade, 1982; Kinomura et al., 1996; Paus, 2000) and has led to the suggestion that prolonged periods of DBS might approximate normal levels of central thalamic activation during periods of enhanced vigilance and thus facilitate behavioral and cognitive function (Schiff, 2009). Functional imaging studies in human subjects (Manoach et al., 2003; Nagai et al., 2004) and electrophysiological recordings of neurons in monkeys (Matsumoto et al., 2001; Kunimatsu and Tanaka, 2010) indicate that central thalamic neurons also respond phasically on a much shorter timescale to brief environmental stimuli and response-related events. This suggests that beyond regulating cortical activity as a function of vigilance, central thalamic neurons may also trigger momentary activations of forebrain circuits. Analyses of event-related fMRI in human subjects (Manoach et al., 2003; Burianova and Grady, 2007) and recordings of neuronal activity in monkeys (Watanabe and Funahashi, 2004; Wyder et al., 2004; Tanaka, 2007) suggest that central thalamic activity corresponds to delay- and retrieval-related events in working memory tasks. It was thus hypothesized that the effects of central thalamic DBS would be most pronounced during these phases of working memory. To test this hypothesis, DBS sessions consisted of a random mix of five conditions including: i. stimulation when the back lever extended at the start of a trial (initiation), ii. stimulation when one of the two front levers was extended as the sample for the trial (sample), iii. stimulation when the back lever extended for the memory delay (delay), iv. stimulation after the delay ended and both front levers extended for the choice response (choice), or v. trials in which no stimulation was delivered. Results were compared for DMTP at three delays (1, 13, or 23 s) to vary the time lag between the choice responses, and when initiate, sample, or delay period DBS was delivered. Each delay was tested separately for a series of sessions in which current level (low current, high current, or no current) was varied randomly between sessions using a complete within subject counterbalanced design.
The results of the stimulation studies (Figure 4) provided support for both hypotheses. Stimulation delivered at the start of the delay and choice phases had significant effects on performance, while stimulation at the time of trial initiation or the sample phase did not. Increasing the length of the delay from 1 to 23 s did not eliminate the effects of stimulation at the start of the delay period. Thus the time lag between stimulation and the choice response does not provide a convincing account for why stimulation during the initiation and sample phases had no effect on performance. The lack of an effect during trials in which no stimulation was delivered, or when stimulation was delivered at the time of the initiate or sample phases, demonstrates that the effects of these brief periods of stimulation were short lived. Averaged across delays, low level stimulation produced a significant improvement, while high level stimulation produced significant impairment in DMTP performance. The benefits of low level stimulation were more pronounced at longer delays when performances tended to be worse (allowing more room for improvement). The deficits produced by high level stimulation were more pronounced at short delays when performances tended to be better (leaving more room for impairment).
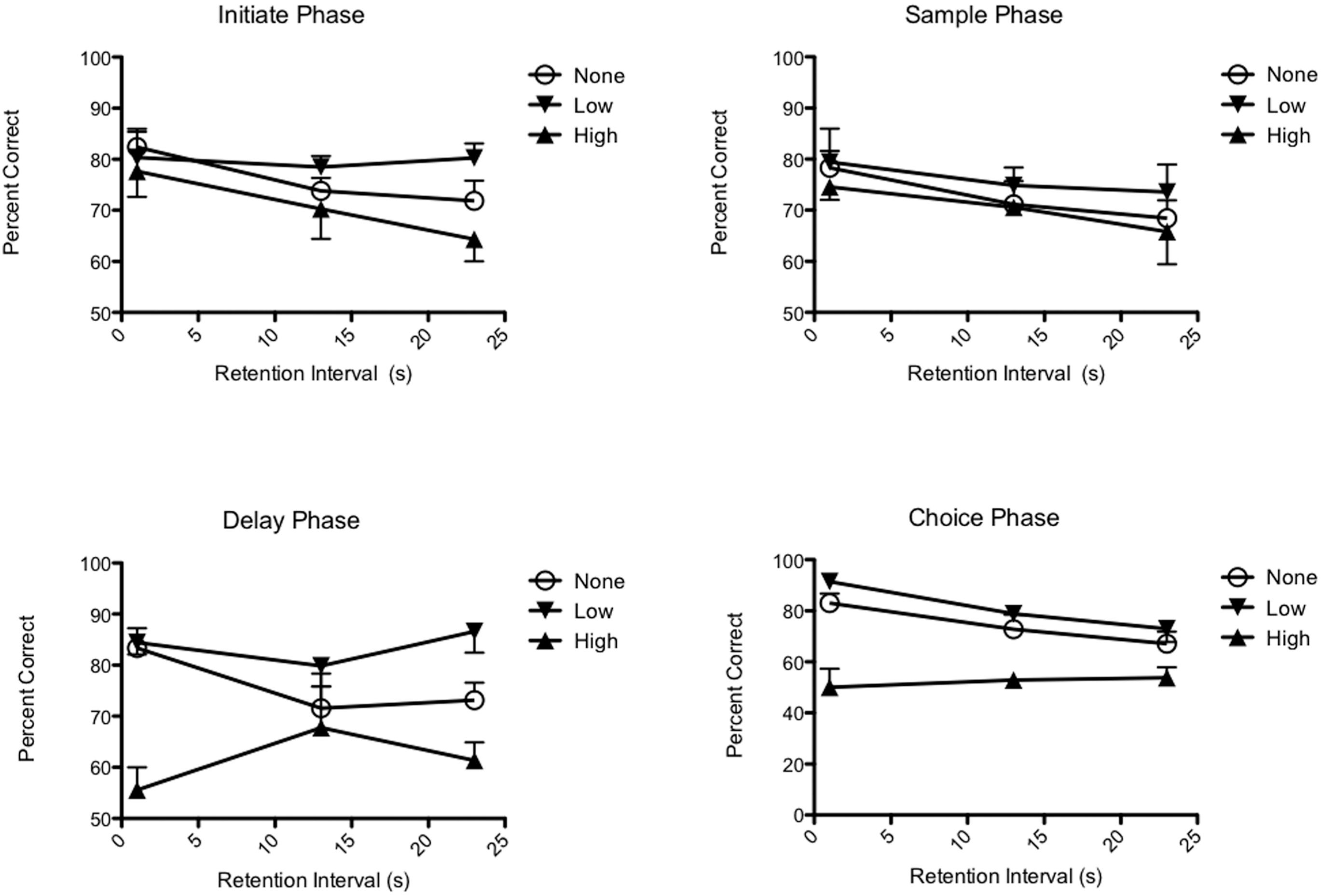
Effects of event-related deep brain stimulation (DBS) in the rostral intralaminar site on DMTP performance. Stimulation at the start of the delay and choice phases, but not the initiation and sample phases, significantly affected performance. The effects of delay period stimulation did not decrease when the delay was increased from 1 to 23 s. This provides evidence that temporal proximity to the choice response cannot account for the effectiveness of delay, compared to initiate or sample stimulation. Low levels of stimulation produced significant improvement (averaged across conditions) while high levels of stimulation impaired performance.
CENTRAL THALAMIC STIMULATION AND PREFRONTAL FUNCTION: HORMESIS OR ARTIFACT?
Hormesis has been defined as “a dose response relationship in which effects at low doses are opposite to those at high doses” (Hoffmann, 2009). The effects of stimulating the rostral intralaminar nuclei with either microinjected drugs or event-related DBS are consistent with hormesis. Low levels of FG-7142 were associated with improved DMTP performance while higher doses were associated with impairment (Figure 3). Similarly low levels of DBS current were associated with improvement and high levels with impairment (Figure 4). These effects are consistent with evidence that lesions damaging the rostral intralaminar site impair DMTP performance as do lesions in anatomically-related areas of prefrontal cortex, ventral striatum, and ventral pallidum. The temporal specificity of DBS is consistent with evidence that central thalamic activity correlates with the delay and retrieval phases of working memory tasks (Manoach et al., 2003; Watanabe and Funahashi, 2004; Wyder et al., 2004; Burianova and Grady, 2007; Tanaka, 2007). On a broader level, the possibility of hormesis in central thalamic stimulation is consistent with evidence that working memory is enhanced at optimal levels of PFC activation by catecholamine systems (Arnsten, 2009) and evidence going back to Yerkes and Dodson (1908) of an inverted-U relationship between arousal and decision making in many species.
Nevertheless, there are confounding factors that can provide alternative accounts for these results. First, impairments observed at high doses of FG-7142 or high levels of DBS current could result from desensitization of receptors or intracellular signaling unrelated to mechanisms producing low dose benefits (Kelly et al., 2008). Comparison of the effects of microinjected drugs and electrical stimulation provides evidence that neural activation by different means produces comparable low dose benefits and high dose deficits. Further, the short-lived effects of electrical stimulation seem difficult to reconcile with desensitization as a cause of high dose impairment. A second potential confound concerns the spread of drug or current following high dose treatments and the possibility that this could affect more distant nuclei or fiber tracts. By this argument, activation of local neurons might account for low dose benefits and activation of more distant neurons might account for high dose deficits. There are several counterarguments to this possibility. First, lesion studies have implicated the rostral intralaminar nuclei (Bailey and Mair, 2005) and areas innervated by these nuclei in medial prefrontal cortex (Mair et al., 1998) and ventral and dorsomedial striatum (Burk and Mair, 2001; Porter et al., 2001) as specifically affecting the DMTP task. Second, anatomical controls provided evidence that the effects of muscimol are localized in the area of the rostral intralaminar nuclei. Third, the finding of DMTP deficits with low and high doses of muscimol and high doses of FG7142, treatments having opposite effects on GABAergic tone, seem difficult to reconcile with an explanation based on spread of drug to adjacent areas. Fourth, the beneficial effects of orexin A are indicative of an effect localized to the rostral and intralaminar nuclei. The third potential confound is that high levels of drugs or electrical current could activate less sensitive receptors or neural processes that have higher thresholds for activation. While this is certainly true, it seems unlikely that high doses of FG7142 and high levels of DBS current would activate the same receptors or processes. While high doses of FG7142 would seem likely to affect GABAA receptors in a wider area of central thalamus, higher levels of DBS current should activate axons with higher thresholds of activation (Perlmutter and Mink, 2006; Kringelbach et al., 2007; Gradinaru et al., 2009).
