Abstract
The current system of radiation protection for humans is based on the linear-no-threshold (LNT) risk-assessment paradigm. Perceived harm to irradiated nuclear workers and the public is mainly reflected through calculated hypothetical increased cancers. The LNT-based system of protection employs easy-to-implement measures of radiation exposure. Such measures include the equivalent dose (a biological-damage-potential-weighted measure) and the effective dose (equivalent dose multiplied by a tissue-specific relative sensitivity factor for stochastic effects). These weighted doses have special units such as the sievert (Sv) and millisievert (mSv, one thousandth of a sievert). Radiation-induced harm is controlled via enforcing exposure limits expressed as effective dose. Expected cancer cases can be easily computed based on the summed effective dose (person-sievert) for an irradiated group or population. Yet the current system of radiation protection needs revision because radiation-induced natural protection (hormesis) has been neglected. A novel, nonlinear, hormetic relative risk model for radiation-induced cancers is discussed in the context of establishing new radiation exposure limits for nuclear workers and the public.
INTRODUCTION
The current system of limiting human exposure to ionizing radiation is based on the premise that the risk of deleterious stochastic effects such as cancer increases as a linear-no-threshold (LNT) function of the absorbed radiation dose (i.e., radiation energy deposited in tissue divided by the tissue mass). This is known as the LNT hypothesis and has no scientific basis. The linearly increasing risk function is also often called the LNT model. Such a linear relationship, if correct, means that doubling the radiation dose doubles the risk of harm. Conversely, reducing the dose one million-fold is supposed to reduce the risk by the same factor.
Some basic terminology is explained below to facilitate following the later sections of this paper.
High- and Low-LET Radiations
Two types of radiation (high and low linear energy transfer [LET]) are usually distinguished in characterizing radiation risks to humans. High-LET forms include alpha particles, neutrons, and heavy ions that produce intense ionization patterns when interacting with biological tissue. Considerable energy is deposited when traversing a narrow thickness of tissue. Low-LET forms include x and gamma rays and beta particles that deposit far less energy when traversing a narrow thickness of tissue.
Units for Expressing Radiation Doses
Radiation dose is expressed in different ways depending on the intended usage. A fundamental unit is the absorbed radiation dose, which is a measure of energy deposited in tissue (or other material) divided by the mass irradiated. Typical units of absorbed dose are the gray (Gy) which is equal to 1 joule/kg, and the milligray (mGy), which is one thousandth of a gray. These units can be applied when characterizing any type of radiobiological damage.
For regulating radiation exposure of humans (e.g., setting radiation exposure limits) and for low-dose risk assessment, special radiation dose units have been established that are based on the linear-no-threshold [LNT] hypothesis. These units are the result of applying statistical weights called
Radiation Dose Limits
Human radiation exposures are limited for nuclear workers, the public, and other groups based on limiting the effective dose. For example, the effective dose limit for nuclear workers is 50 mSv/y and for the public is 1 mSv/y based on U.S. Department of Energy and Nuclear Regulatory Commission regulatory policies (Metting 2005). The U.S. Environmental Protection Agency's regulatory policy limits on release of radioactivity to air is based on limiting the effective dose to humans to 0.1 mSv/y, and for public drinking water the corresponding limit is 0.04 mSv/y. For a point of reference, natural background radiation doses in the United States are associated with an effective dose of about 3 mSv/y (radon exposure included) (Metting 2005). For Ramsar, Iran, the corresponding dose associated with natural background radiation is about 200 mSv/y. Interestingly, such high background radiation doses appear to be associated with radiation hormesis-related protection against cancer (Frigèrio and Stowe 1976; Nambi and Soman 1987), i.e., a reduction in cancers.
Low Dose/Dose Rate Cancer Risk Assessment within the LNT Framework
Under the LNT risk assessment framework, effective doses for individuals can be added to obtained person-sievert (a collective dose) for population exposure, and the collective dose can be used to calculate the expected number of cancers among an irradiated population. Similarly, effective dose can be used to assign an individual specific cancer risk. However, low doses are often delivered at low rates and a correction is made for a reduction in harm after low-rate exposure as compared to high-rate exposure. For low doses and dose rates, a low-dose and dose-rate effectiveness factor (
Hormetic Dose-Response Curves
With hormesis, low doses of radiation protect against cancer, leading to a negative slope in the low-dose region for the dose-response curve. High doses, however, inhibit protection causing risk to then increase as dose increases. This yields what has often been called a U- or J-shaped dose-response curve (Calabrese and Baldwin 2001; Calabrese 2004, 2005; Calabrese et al. 2006).
DIFFERENT CLASSES OF RADIATION-ASSOCIATED HORMESIS
This paper distinguishes three classes of radiation hormesis based on the recent recommendations of Calabrese et al. (2007):
Sheldon Wolff's group (Olivieri et al. 1984; Wolff 1989, 1996) were the first to demonstrate and publish radiation conditioning hormesis data. When human lymphocytes were cultured with tritiated thymidine, which was a source of low-level chronic beta radiation, and then briefly exposed to 1500 mGy of x rays, the yield of chromatid aberrations from the x-ray exposure was suppressed. In a 1988 publication (Wolff et al. 1988) by his group, it was also demonstrated that human lymphocytes exposed to low doses of ionizing radiation (mild stress) became refractory to chemical mutagens that induced double-strand breaks in DNA. Howard Ducoff (1975) was the first to demonstrate radiation hormesis in insects. This author benefited greatly by participating in some of Dr. Ducoff's research as a graduate student at the University of Illinois. Members of this research group are now known as the
T.D. Luckey, in his 1991 book entitled
Ullrich et al. (1976) were the first to demonstrate a pronounced radiation hormesis effect (for lung cancer) in gamma-ray irradiated female RFM mice. The mice had a
Ullrich and Storer (1979) apparently attributed the radiation-hormesis-like observation for lung cancer in mice to systematic errors in lung cancer detection based on the methodology used. However, such a systematic error should operate at all dose levels, including those for the controls; and thus correcting such an error would not be expected to eradicate the hormetic dose-response curve shape. This can be demonstrated by assigning an arbitrary large systematic error (e.g., 50%) to each dose group including the controls and correcting the data. When evaluating relative risk, the correction is canceled so the hormetic curve shape remains. In addition, the study by Ullrich et al. (1976) not only demonstrated radiation hormesis for lung cancer, but it was also demonstrated for reticulum cell sarcoma for both gamma-ray and neutron exposures.
Edouard Azzam (Azzam et al. 1996) and colleagues were the first to demonstrate radiation hormesis
The indicated radiation-associated hormesis publications and others collectively demonstrate that low doses/dose rates of low-LET radiation:
Activate protective apoptosis signaling pathways and stimulate immunity.
Protect against spontaneous chromosomal damage, mutations, neoplastic transformation, and cancer.
Protect against high dose chemical- and radiation-induced cancer.
In spite of these now widely published hormetic effects, regulatory agencies still use the LNT-based system for regulating human exposure to ionizing radiation and for low-dose cancer risk estimation. Use of the LNT-based system is considered justifiable by many outside the hormesis community in light of publications such as the BEIR VII Report (NRC 2006), published by the U.S. National Research Council/National Academy of Science. The BEIR VII report concluded that the LNT approach to low-dose risk assessment was valid and essentially dismissed radiation-associated hormesis. A corresponding French Academies report did not come to the same conclusions (Tubiana 2005; Tubiana et al. 2005) when examining essentially the same data that were reviewed in the BEIR VII report. The French report found hormesis to be plausible and the LNT risk function to be invalid for low-LET radiation doses < 100 mGy and especially for doses < 10 mGy.
In the next section, three epidemiological tricks are discussed that when used helps to justify continued use of the LNT framework for low-dose-radiation risk assessment. An approach for accounting for radiation-associated hormetic effects in regulating radiation exposure is then discussed.
EPIDEMIOLOGICAL TRICKS THAT FAVOR A LNT DOSE-RESPONSE CURVE
Trick #1: Throwing Away Radiation Dose
With many previous epidemiological studies of radiation-induced cancer, the researchers somehow came to the conclusion that radiation dose was wasted. Thus, in order to correct for the so-called wasted dose, one has to lag (throw away) some of the dose. However, if the dose-response curve is indeed of the LNT type, then each fixed infinitesimally small increment,
When studying DNA double-strand break induction by radiation, one usually observes a LNT-type dose-response curve at low doses (NRC 2006). This seems to be the basis for the expectation by many experts that cancer risk is also a linear function of dose. Interestingly, no dose lagging is used when evaluating DNA double-strand break dose-response curves; possibly because the inappropriateness of doing so would be immediately realized by many if not most radiation researchers.
To illustrate how radiation hormesis can be hidden by this dose lagging trick, data are presented in Figure 1 for
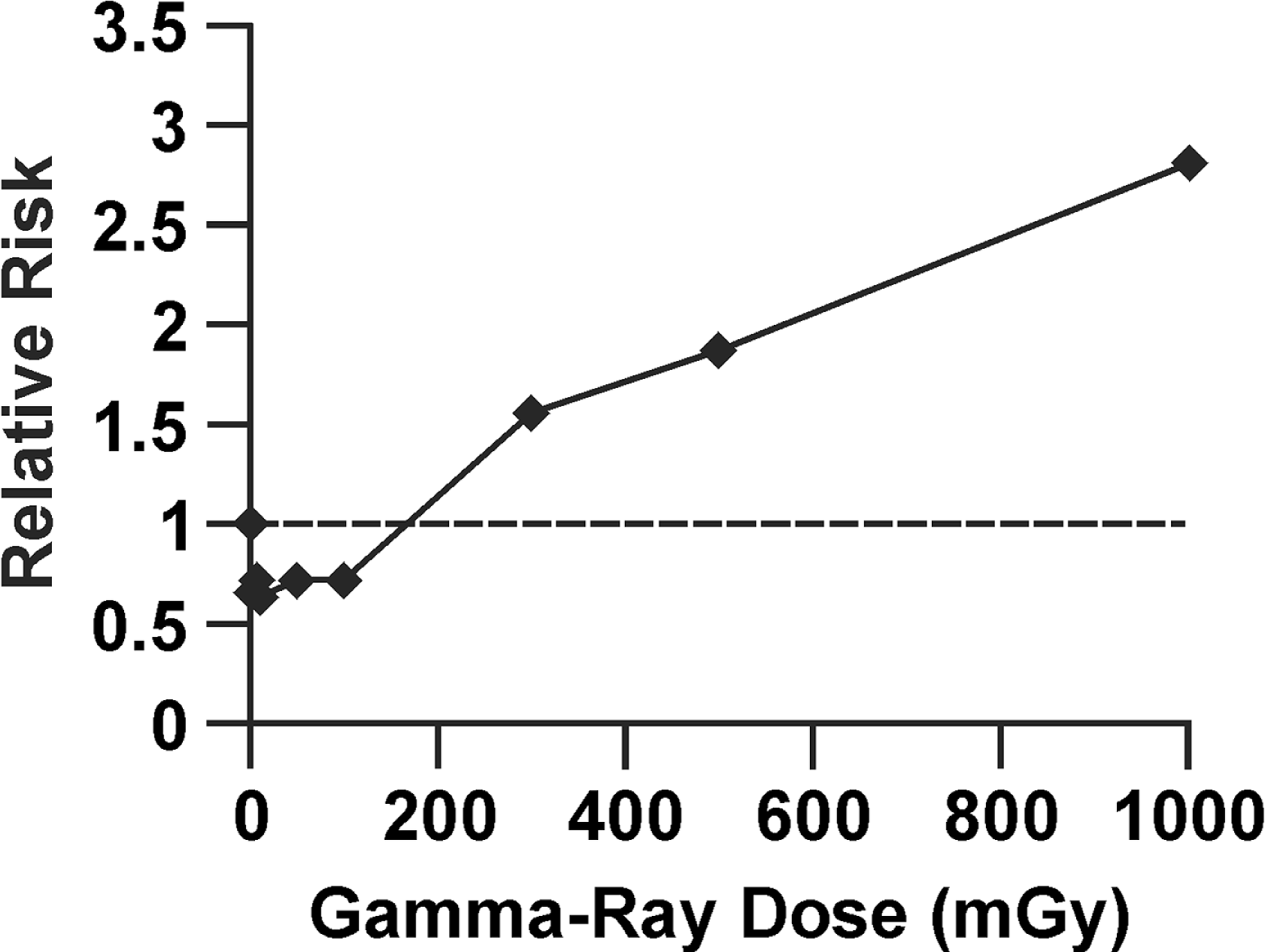
Relative risk dose-response relationship for gamma-ray induced neoplastic transformation of HeLa x skin fibroblast human hybrid cells by brief high-rate exposure, based on

Application of dose lagging (100 mGy) to the data in Figure 1. Analysis based on data from Redpath et al. (2001).
Trick #2: Eliminating the Hormetic Zone via Averaging over Dose Groups
The second trick relates to forming dose groups comprised of persons having received widely varying radiation doses (i.e., the minimum and maximum doses [often reconstructed] for each dose group differs greatly). Such dose groups are usually necessary in case-control studies and are also often used in cohort studies of irradiated populations. Here the focus is on case-control study design and the use of odds ratio (
The neoplastic transformation frequency data used for the
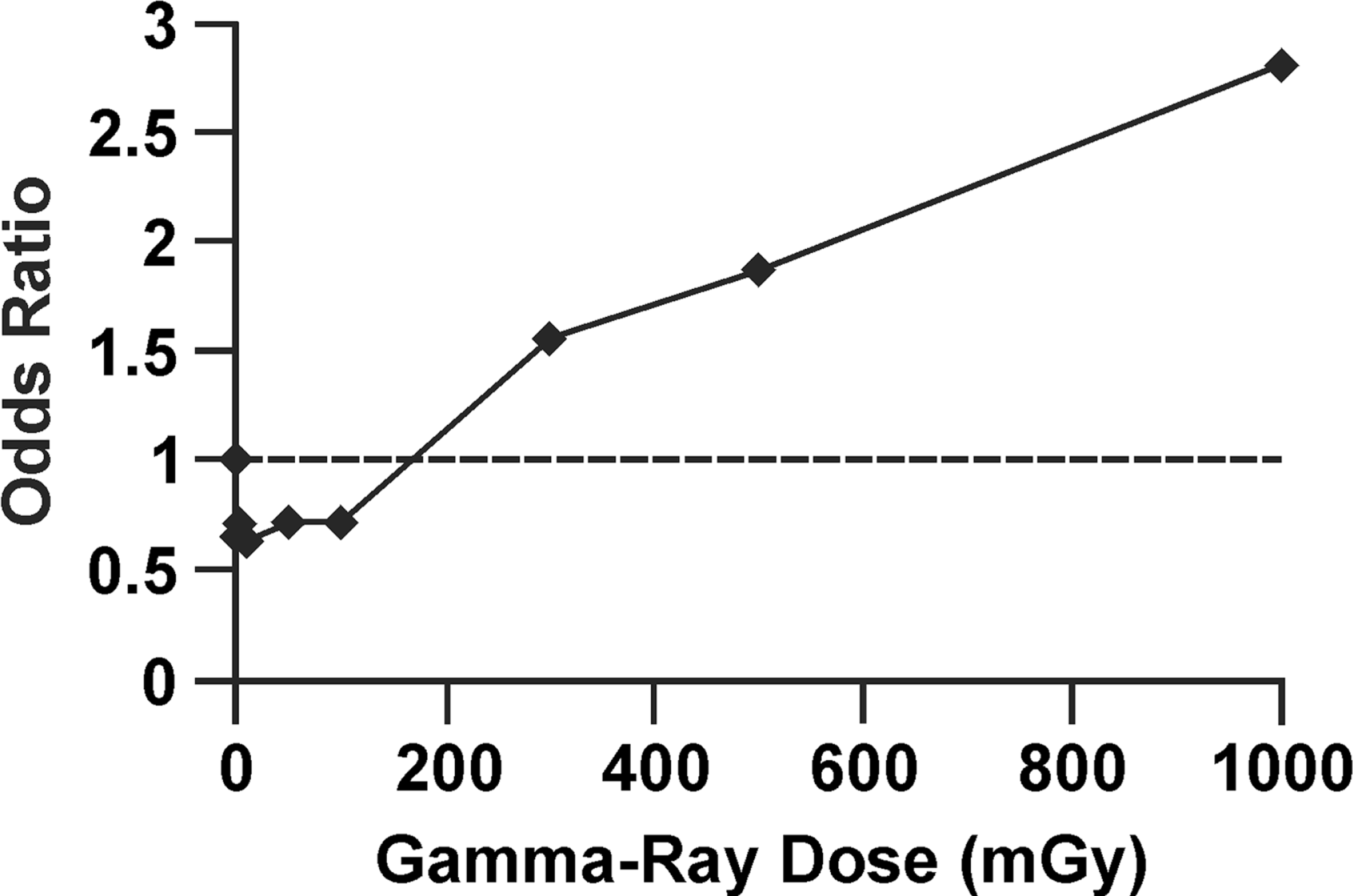
Odds ratio relative to controls for the neoplastic transformation data presented in Figure 1 for gamma-ray exposure of HeLa x skin fibroblast human hybrid cells.

Ratio of dose-interval-specific average odds for neoplastic transformation based on data in Figure 3. Ratio of average odds evaluated relative to the lowest dose group. The lowest dose group was plotted at dose = 0 mGy. Other data plotted at the midrange of the dose intervals are used.
Dose-grouping in cohort studies of radiation-induced cancer can also vanish the hormetic zone when persons who received low doses are included among the control group (representative of unexposed individuals). This is because the study design has reduced power for demonstrating suppressed risk at low doses when irradiated persons with radiation doses in the hormetic zone are included in the control group (used to represent unirradiated persons).
Trick #3: Constraining the Slope of the Cancer Risk Dose-response Curve to Always Be Positive
A trick often employed in cohort and case-control studies is to constrain the slope of the dose-response curve to be positive while including high-dose, high-risk data in the analysis of the dose-response curve fit. This is especially true when a LNT function has been presumed to apply at low doses by the researchers. Irrespective of the low-dose data, an increase in risk is predicted as dose increases for all such studies. The conclusion that any dose is harmful then follows. Low-dose hormetic (U- and J-shaped) data departing from the LNT characteristic is often simply ignored. It is wrong to portray such data as part of a LNT curve! Low-dose risk assessments should account for the hormetic shape to the dose-response curve.
HORMESIS IMPLICATIONS FOR REGULATORY POLICY
In Zbigniew Jaworowski's 1997 article,
Adaptive stimulating effects of ionizing radiation occur at near natural doses. This disagrees with linear, no-threshold hypothesis on the dose/effect relationship, which is a basis of the current radiation protection. Vast literature demonstrates that such effects, usually known as hormetic ones, occur at molecular, cellular and population levels, and often result in increased longevity and decreased cancer incidence…. After the Chernobyl accident, adverse health effects and vast material losses were induced in the former USSR by practical implementation of the ICRP radiation protection recommendations. A revision of the current approach to managing the risk of ionizing radiation is needed for the public interest.
Here, an approach to regulating radiation exposure is recommended that allows for the existence of a hormetic dose zone just above natural background radiation. The approach relates to the hormetic relative risk (HRR) model previously developed by this author (Scott 2007a,b,c), which is summarized below in a more general form.
Hormetic Relative Risk Model
With the HRR model for low-dose radiation-induced cancer, doses at or slightly above normal monthly natural background low-LET radiation levels are presumed to fall within the what is currently considered the hormetic zone. This hormetic zone starts at natural background radiation and spans a relative wide dose range, possibly exceeding 1000 mGy of low-LET radiation when radiation dose is delivered at a low rate. However, protective effects associated with hormesis may also occur at below current natural background levels for some individuals. For low-LET radiation doses in the hormetic zone, cancer
The protective signaling, presumed activated with low doses and dose rates of low-LET radiation, relates to removal of aberrant cells from the body via p53-dependent and independent apoptosis signaling pathways and stimulated immunity (Scott 2007a,b,c; Scott and Di Palma 2007; Scott et al. 2007). The protective signaling can also involve DNA repair pathways if a damage threshold is exceeded (Rothkamm and Löbrich 2003). Possible exceptions to full hormetic protection are the very young and children who may not have significant burdens of genomically unstable cells that participate in the signaling associated with protective p53-independent apoptosis (Scott and Di Palma 2007).
Stochastic thresholds (StoThresh) that vary between different individuals are required in the HRR model for activating the protective signaling. However, somewhat higher doses (also StoThresh) inhibit protection causing an increase in the

Schematic representation of the hormetic relative risk model. The model is presented as a function of the total absorbed radiation dose
The mathematical functions discussed in this paper relate to radiation doses equal to or greater than natural background radiation. For this dose range, the indicated nonlinear hormetic

Equation 1 is used to characterize the population average
For just above natural background radiation exposure
What has traditionally been considered the hormetic zone comprises the above natural background range of radiation doses for which
Transition Zone A in Figure 5 is where StoThresh for activating protective signaling are progressively exceeded as radiation dose increases. When protective signaling is activated in all members of the population, then the
Over Transition Zone A,
Regulatory Threshold with Respect to Cancer Induction
It is beneficial to define a
One can then use the normalized stochastic effect dose,

for all tissues
This example does not account for genetic effects. However, it is widely known that genetic effects are much less likely to be induced than cancer (NRC 2006). Thus, limiting testicular and ovarian cancer occurrence would be expected to also limit genetic effects. There is also some evidence for dose-response relationships for genetic effects in humans being of the hormetic type with respect to low-rate exposure to gamma rays (Chen et al. 2007). Limiting both cancer and genetic effect occurrences would be expected to also limit shortening of life due to deleterious genetic effects and cancer.
The REGRADT as defined would apply both to population and individual exposures. New, funded research is needed in order to properly assign appropriate values for
CONCLUSIONS
There is abundant evidence for radiation-associated hormesis. However, dismissal of radiation-associated hormesis is in many instances based on epidemiological tricks that include dose lagging, odds averaging over wide dose ranges when evaluating
Its time for new, low-dose radiation risk assessment and regulatory paradigms that allow for hormesis. Normalized stochastic effects dose, based on radiation-, radiation-energy-, and dose-rate-specific REGRADTs could be used to limit radiation exposure. For
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Office of Science (BER), U.S. Department of Energy (DOE) Grant DE-FG02-03ER63657. I am grateful to Ms. Vicki Fisher and Ms. Dinese Leonard for editorial assistance and to Ms. Wendy Piper for graphic support. I am also grateful to Dr. Leslie Redpath for his assistance in using published data from his research group and to the journal reviewers for their constructive comments. The views and conclusions contained herein are those of the author and should not be interpreted as necessarily representing the official policies or endorsement, either expressed or implied, of the DOE or of Lovelace Respiratory Research Institute.
