Abstract
Routine diagnostic X-rays (e.g., chest X-rays, mammograms, computed tomography scans) and routine diagnostic nuclear medicine procedures using sparsely ionizing radiation forms (e.g., beta and gamma radiations) stimulate the removal of precancerous neoplastically transformed and other genomically unstable cells from the body (medical radiation hormesis). The indicated radiation hormesis arises because radiation doses above an individual-specific stochastic threshold activate a system of cooperative protective processes that include high-fidelity DNA repair/apoptosis (presumed p53 related), an auxiliary apoptosis process (PAM process) that is presumed p53-independent, and stimulated immunity. These forms of induced protection are called adapted protection because they are associated with the radiation adaptive response. Diagnostic X-ray sources, other sources of sparsely ionizing radiation used in nuclear medicine diagnostic procedures, as well as radioisotope-labeled immunoglobulins could be used in conjunction with apoptosis-sensitizing agents (e.g., the natural phenolic compound resveratrol) in curing existing cancer via low-dose fractionated or low-dose, low-dose-rate therapy (therapeutic radiation hormesis). Evidence is provided to support the existence of both therapeutic (curing existing cancer) and medical (cancer prevention) radiation hormesis. Evidence is also provided demonstrating that exposure to environmental sparsely ionizing radiations, such as gamma rays, protect from cancer occurrence and the occurrence of other diseases via inducing adapted protection (environmental radiation hormesis).
INTRODUCTION
Ionizing radiation spans the universe in which we reside (Bonner 2003). There are two basic forms of ionizing radiation: electromagnetic and particulate. Electromagnetic radiation is comprised of uncharged photons (entities without mass) that interact with electrons in matter causing ionizations if photon energy is high enough. Examples of ionizing electromagnetic radiations are X-rays and gamma rays. Examples of particulate ionizing radiation are alpha and beta particles emitted by radioisotopes and protons ejected from the sun. Neutrons do not directly cause ionizations but cause them indirectly through secondary charged particles such as protons (e.g., from water in biological tissue) that are dislodged by neutrons.
Natural background ionizing radiation on earth comes from the following three sources: the sun (solar radiation), outer space (cosmic rays), and terrestrial sources (e.g., radionuclides in our bodies and environment, and radon in the home) (NCRP 1997). While most solar radiation is electromagnetic, the sun also produces particulate radiation (solar cosmic rays), including protons, which vary with the solar cycle.
All organisms on earth are constantly bombarded by cosmic radiation from outside our solar system. This radiation is comprised of charged particles ranging in atomic mass from protons to iron nuclei. These particles interact in the atmosphere creating secondary radiation that rains down and includes X-rays, electrons, protons, alpha particles, neutrons, pions, and muons. Our exposure to cosmic rays increases each time we take an airline flight. Persons living at high elevations such as in Denver, Colorado, and Salt Lake City, Utah, receive higher exposures to cosmic rays than do persons residing in Miami, Florida, or New Orleans, Louisiana.
Natural radioactivity (the capacity to emit particulate or electromagnetic ionizing radiation forms) is everywhere on earth. All organisms on earth are continuously exposed to varying amounts of natural radiation. We humans are irradiated from: radioactivity in our bodies (e.g., associated with potassium-40), natural radioactivity in ingested foods (e.g., associated with carbon-14), exposure to radiation emanating from soils and rocks (e.g., from uranium and thorium isotopes), and exposure in our homes and businesses to radon and its radioactive daughter radionuclides. Thus, we humans are continuously exposed to radiation arising from naturally occurring terrestrial radioactivity and from the cosmos. This is true prior to birth and through one's entire life.
Diagnostic X-rays (chest X-rays and computed tomography scans) and other sources of radiation used in nuclear medicine diagnostic procedures are also sources for our exposure to low-level ionizing radiation. Both exposure to natural background sparsely ionizing radiation and exposure to diagnostic sparsely ionizing radiation sources are likely playing a beneficial role in the maintenance and preservation of life on earth through suppressing genomic-instability-associated diseases such as cancer. This topic is partly the focus of this paper. An additional focus is on the use of low doses and dose rates of sparsely ionizing radiation in curing existing cancer.
The potential for severe radiation damage is generally evaluated based on what is called linear energy transfer (LET), which is just the average energy loss when penetrating a small thickness of material (e.g., tissue). Low-LET radiations include X-rays, gamma rays, and beta particles. These radiation forms deposit relatively small amounts of energy when penetrating a small thickness of tissue. High-LET radiation (e.g., alpha particles and neutrons) deposit more energy in the indicated small thickness of tissue. High-LET radiations usually cause more biological damage locally in tissue than low-LET radiation.
It is important to be aware that small amounts of radiation kill only a few cells and those cells are generally replaced without harm in humans and other mammals. Radiation also produces sublethal damage to our cells, and most such damage is repaired without any significant error (e.g., error free). However, some cells commit repair errors (i.e., misrepair leading to mutations). Mutations represent a form of genomic instability. A certain amount of instability is tolerated by cells and the instability can propagate over subsequent cellular generations. Cells with threatening instability may commit suicide (apoptosis) or may be eliminated via the immune system. Uncontrolled instability in the genome can result in cancer and other diseases.
The oxygen we breathe is by far the greatest natural cause of cellular damage–many orders of magnitude greater than other natural causes (Pollycove and Feinendegen 2003). All living mammals have a system of protective processes that prevents, repairs, and removes cell damage. Radiation primarily affects the components of this protective system. Low doses activate protection resulting in fewer mutations, neoplastic transformation, and cancers, while high doses suppress some of the protection resulting in more of the indicated stochastic effects (Feinendegen et al. 2004; Scott 2005a,b, 2006a,b).
The recently released BEIR VII Report (Phase 2) has implicated diagnostic X-rays (e.g., chest X-rays, mammograms, CT scans) and nuclear medicine diagnostic procedures as causing harm through inducing excess cancers (NRC 2006). This view is based on the linear-no-threshold (LNT) hypothesis of radiation carcinogenesis, which states that cancer risk increases as a LNT function of radiation dose, no matter how small. Relative risk (
Cancer risk estimates based on the LNT hypothesis (Figure 1) are mainly based on extrapolating high-dose cancer mortality data acquired following the nuclear blasts that took place in Hiroshima and Nagasaki, Japan, to low doses (NRC 2006). A LNT cancer risk curve is fitted to the high-dose cancer frequency data, as was done in the BEIR VII Report (NRC 2006). For evaluating cancer risk after low doses and dose rates, a low-dose and dose-rate effectiveness factor (

LNT risk function which is usually based on data derived from high doses delivered at high rates from the atomic bombings in Hiroshima and Nagasaki, Japan. The high-dose LNT curve is reduced by a DDREF when evaluating the risks at low doses and dose rates. Even so, the slope of the dose-response curve can never be negative (i.e., a hormetic response curve). The notation
In contrast to the BEIR VII Report use of the LNT hypothesis when assessing low-LET radiation-associated cancer risk at low doses and dose rates, the recent French Academies report related to LNT dismissed the LNT hypothesis for low-LET radiation doses less than 100 mGy and found radiation hormesis to be plausible (Tubiana 2005; Tubiana et al. 2005).
This paper presents evidence that we are unlikely to be harmed by infrequent applications of diagnostic X-rays (from a chest X-ray machine, mammogram, or CT scan), by most routine nuclear medicine procedures or by elevated natural background radiation (including radon in our homes). More importantly, this paper provides evidence that low levels of low-LET radiation (e.g., X-rays or gamma rays) received from natural and medical sources protect us from cancer and other diseases via stimulating a system of known protective processes. Similar protection also appears to be associated with combined exposure to low doses and dose rates of alpha plus gamma radiation (as occurs for radon in the home).
LOW-DOSE/DOSE-RATE LOW-LET RADIATION-INDUCED SYSTEM OF PROTECTION
As previously indicated, low doses and dose rates of low-LET radiation activate a system of cooperative protective processes in the body. The protective processes include (1) defenses such as scavenging of reactive oxygen species and other toxins, (2) presumably p53 related activated high-fidelity DNA repair/apoptosis, (3) a novel auxiliary protective apoptosis mediated (PAM) process that selectively eliminates aberrant cells, and (4) induced immunity (Liu et al. 1994; Liu, 2004). The PAM process has been demonstrated to involve reactive oxygen and nitrogen chemical species, specific cytokines (e.g., transforming growth factor beta in the case of fibroblast cells), and can occur independently of the
In this article the idea is put forth that low doses and dose rates of diagnostic X-rays, gamma rays, and beta radiation can prevent cancer occurrence via stimulating selective removal of precancerous neoplastically transformed cells that could otherwise lead to cancer. In the next section, we briefly discuss publications which indicate that low doses of low-LET radiation are protecting us from mutations, neoplastic transformation, and cancer (including cancer metastasis) and other diseases.
EVIDENCE THAT LOW-DOSE RADIATION PROTECTS US
Low doses of low-LET radiation (gamma or X-rays) have been demonstrated to
Induce defense such as detoxification of reactive oxygen species (for review, see Feinendegen et al. 2004).
Induce high-fidelity repair of DNA damage (Joiner et al. 1999; Rothkamm and Löbrich 2003).
Protect from chromosomal damage from a subsequent high radiation dose (Wolff et al. 1988).
Protect from spontaneous mutations occurrence
Protect from spontaneous neoplastic transformation occurrence
Protect from spontaneous cancers in animals (Sakai 2003).
Extend tumor latency in cancer-prone mice (Mitchel et al. 2003; Mitchel 2004, 2005)
Activate the immune response (Liu et al. 1987; Makinodan and James 1990; Sakamoto et al. 1997; Liu 2003, 2004) and suppress lung and lymph node metastasis
Suppress spontaneous cancers in humans (Howe, 1995; Rossi and Zaider 1997; Scott 2005a, 2006a).
Protect from some diseases other than cancer (Luckey 1991; Wang et al. 2005).
The low-LET radiation doses that protect us fall into a presently not-well-defined dose zone which is dose-rate and exposure-duration dependent (Scott 2004, 2005a; Scott et al. 2006). For brief exposure at a high rate to X-rays (28-kVp, 60-kVp, or 250-kVp) and for neoplastic transformation the protective zone includes doses in the 0.5 mGy to 10 mGy range (Scott 2004, 2005a). The 28-kVp X-rays are representative of mammographic-energy X-rays (Ko et al. 2004). For high-energy, gamma-ray photons, the protective zone includes doses in the range 1 mGy to 100 mGy. For protracted exposure of humans, the zone is increased to include total doses over several hundred miligray as discussed later, related to multiple applications of fluoroscopy and mammography.
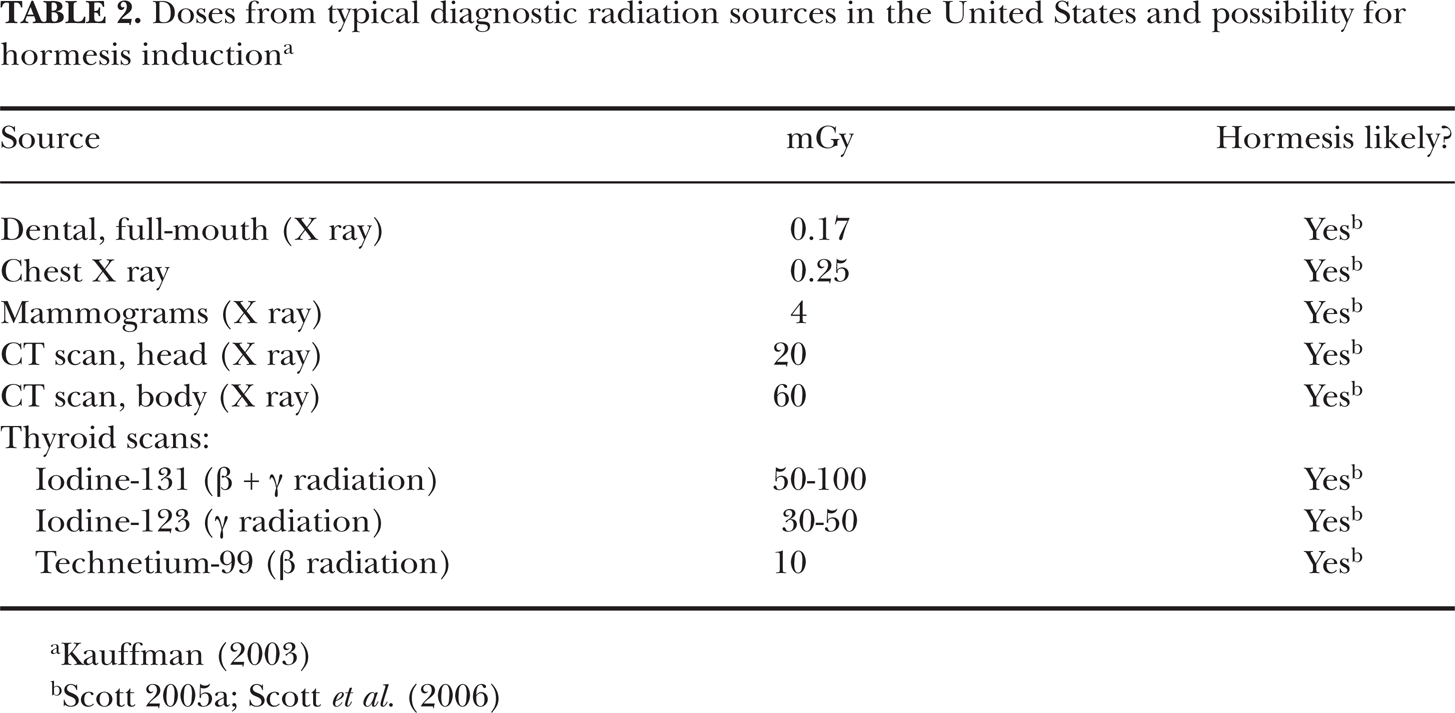
Doses currently associated with applications of X-rays and other routine diagnostic radiations fall in the protective zone (Tables 1 and 2) and therefore are likely protecting us from cancer and some other diseases. Unfortunately, because of the BEIR VII Report (NRC 2006) claim that any amount of radiation is harmful, many citizens are now terrified of having to undergo diagnostic chest X-rays, mammograms, CT scans, or nuclear medicine diagnostics. Using a LNT risk function, the BEIR VII Report (NRC 2006) concluded that such diagnostic treatments harm us through inducing cancers. To the contrary, research results presented in this paper and elsewhere (Scott 2005a, 2006a) suggest that some precancerous neoplastically transformed cells in the body disappear (medical radiation hormesis) as a result of the low-level, low-LET radiation exposure associated with diagnostic X-rays. Multiple X-rays (e.g., from CT scans, mammograms, chest X-rays) at appropriate intervals (not yet determined) would be expected to increase the efficiency of removal of the neoplastically transformed cells as well as other genomically unstable cells. Repeated low doses of X-rays likely over and over stimulate the transient PAM process and immunity. The indicated low-dose-radiation-induced system of protection is illustrated in Figure 2. The protection factor (
Doses from routine diagnostic X rays and possibility of hormesis induction

Doses from typical diagnostic radiation sources in the United States and possibility for hormesis induction a

Low-dose, low-LET-radiation-induced system of protection against spontaneous cancers. The indicated protective components are features of the HRR model. Increasing DNA fidelity influences the slope parameter
HORMESIS-BASED VS. LNT-BASED RELATIVE RISK
As already indicated, cancer
Figure 3 shows the expected

Basic features of the HRR model. Doses
The dose zone [
For Transition Zone A, changes in
The related cancer

for background radiation exposure (

for doses > background.
Here
The
Regarding Equation 1, for low doses and dose rates (near natural background levels), the term (

For exposure only to low-LET X-rays or gamma rays,
Moderate and high doses can inactivate the PAM process and suppress (rather than stimulate) the immune system (Scott 2006a,b; Hashimoto et al., 1999) leading to increased radiation-associated cancers. Thus, the increased incidence of cancer at moderate and high doses relates to the loss of protection against stochastic effects. The
Both
For exposures at below natural background radiation levels, the
The HRR model is pragmatically applicable to all types of exposures, be they acute or protracted or fractionated, and is based on high- and low-LET absorbed radiation doses, rather than on a weighted combination of these doses.
IMPLICATIONS OF THE DOSE-INDEPENDENT ZONE
With the HRR model, there is a relatively large dose region (Figure 3) over which
With the HRR model and for low doses delivered at low rates over prolonged periods, the dose-independent region of suppressed
DATA SUPPORTING THE HRR MODEL
A similar curve shape as in Figure 3 (HRR model) for doses >

Lung cancer
The dose-response curve shape in Figure 4 has also been demonstrated for lung cancer induction in humans exposed to fractionated low-LET radiation for absorbed doses up to about 1,000 mGy of diagnostic X-rays as shown in Figure 5 (Canadian Fluoroscopy Cohort Study, Howe 1995). Data points are presented separately for males and females. Published dose bins (Howe 1995) were used with the data points plotted at the midrange of the bins. The low-LET data used were based on multiple applications of diagnostic X-rays given to TB patients. The data in Figure 5 are consistent with the notion that fractionated exposures to diagnostic X-rays over and over stimulate the removal of precancerous neoplastically transformed cells from the lung and thereby reduce the risk of lung cancer (medical radiation hormesis). There is no evidence that fractionated diagnostic X-ray doses ≤ 1,000 mGy are causing excess lung cancers as would be predicted based on the LNT hypothesis! Instead, there is strong evidence for medical radiation hormesis.

Applications of the HRR model to lung cancer data for humans (TB patients) exposed to fractionated X-ray (diagnostic) doses, based on data reported by Howe (1995). Data for males and females were jointly analyzed. The two dashed curves indicate the 95% confidence region
A similar curve shape has also been used (Scott 2004) to characterize lung cancer risk in Mayak plutonium facility workers chronically exposed at low rates over years to gamma radiations based on data reported by Khokhryakov et al. (1996). The results presented here were corrected for exposure to alpha radiation (Scott 2006a) using the HRR model. The dose-response curve and 95% confidence region is presented in Figure 6 and shows the high degree of protection that appears to be associated with exposure over years at low rates to gamma rays. The average value for

Applications of the HRR model to lung cancer mortality data for Mayak workers chronically exposedover years to gamma and alpha radiation at low rates. Results presented were adjusted for the influence of alpha irradiation (Scott 2006a). Only the gamma-ray dose (for an arbitrary dose range) is therefore indicated. The notation
Small X-ray doses have been demonstrated to suppress lung metastasis of squamous carcinoma cells transplanted into mice (Sakamoto 2004). The dose-response curve shapes for suppressing lung metastasis
ADDITIONAL EVIDENCE THAT RADIATION EXPOSURE OF HUMANS IS PREVENTING CANCER
Values of
Central estimates of the radiation-hormesis-related protection factor (
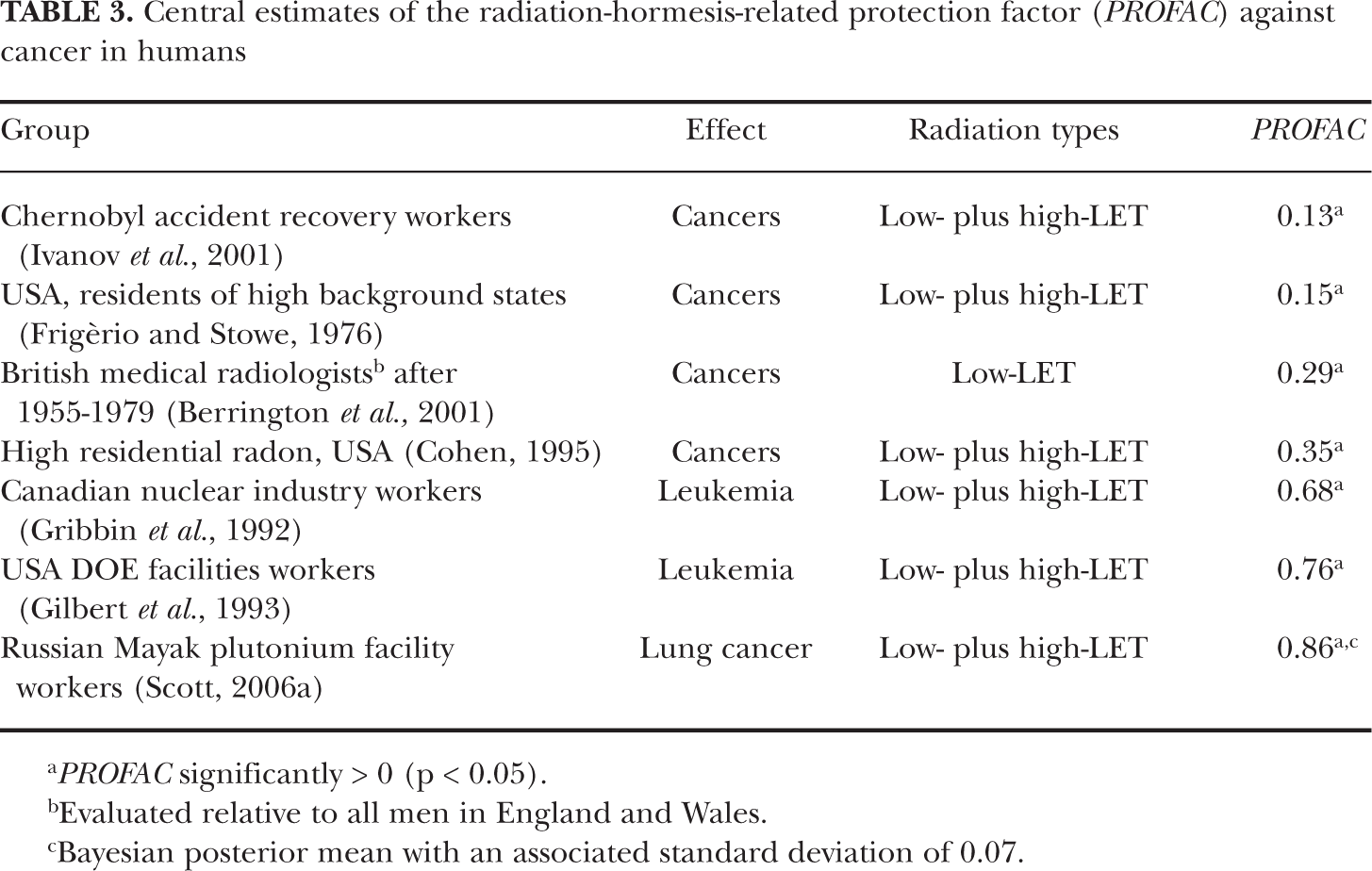
Evaluated relative to all men in England and Wales.
Bayesian posterior mean with an associated standard deviation of 0.07.
As previously indicated, the product 100 ×
EXPECTED IMPACT OF AGE AT EXPOSURE ON THE PROFAC
DNA repair fidelity is known to be reduced with increasing age (Szczesny et al. 2003). Thus, genomic instability is expected to increase as we age because of reduced DNA fidelity. However, increasing genomic instability would be expected to be associated with an increased role of the PAM process and immune system stimulation in protecting against genomic-instability-associated diseases such as cancer. The PAM process involves signaling between normal and aberrant cells. The higher the concentration of genomically unstable cells the stronger the signaling associated with the PAM process is expected to be, once signaling is initiated (Scott 2004). Thus, one would expect the
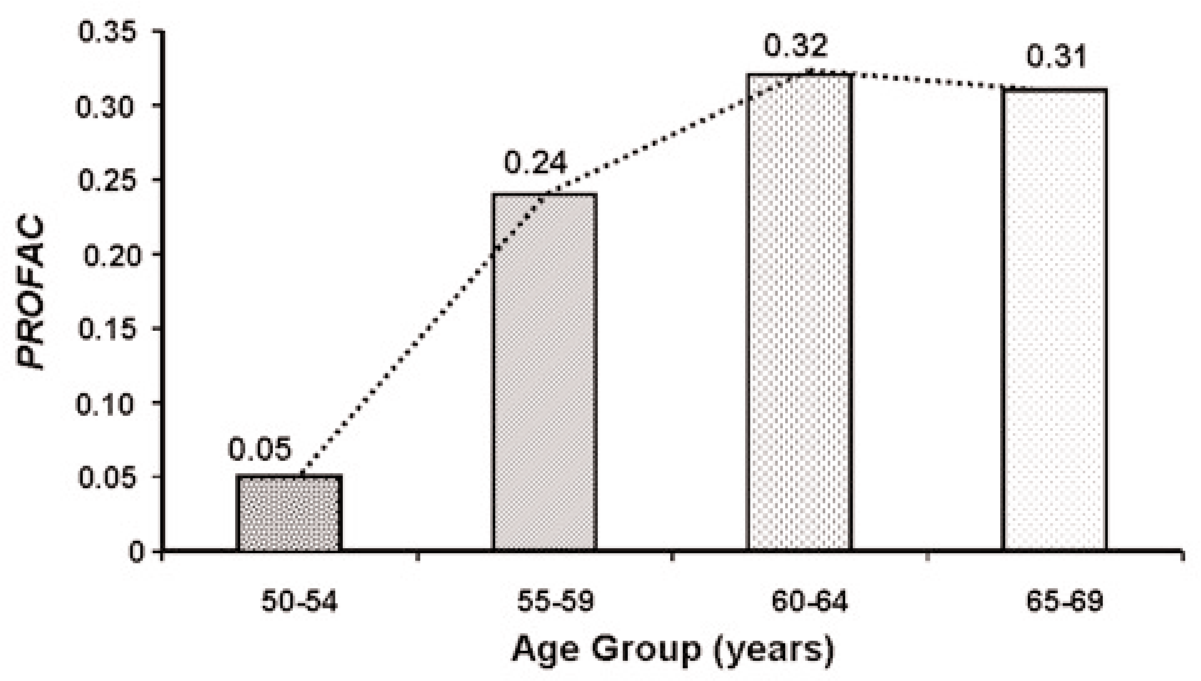
Proportion of breast cancer prevented (
THERAPEUTIC RADIATION HORMESIS IMPLICATIONS
The relatively large
Resveratrol has been shown to potentiate the apoptotic effects of gamma radiation, cytokines (e.g., TRAIL), and chemotherapeutic agents (Aggarwal et al. 2004). In addition to beneficial cardiovascular effects, resveratrol exhibits anticancer properties, as suggested by its ability to suppress proliferation of a wide variety of tumor cells, including myeloid and lymphoid cancers; multiple myeloma; cancers of the stomach, prostate, breast, colon, thyroid and pancreas; melanoma; squamous cell cancinoma in the head and neck; ovarian carcinoma; and cervical carcinoma (Aggarwal et al. 2004).
The cancer suppressive effects of resveratrol involve signaling through multiple pathways (e.g., to apoptosis) and are mediated via the following (Aggarwal et al. 2004): (1) cell cycle arrest; (2) upregulation of
The ability of resveratrol to trigger apoptosis (likely the PAM process) has been established in different human tumor cell lines (Hsieh and Wu 1999; Clement et al. 1998; Surh et al. 1999; Ahmad et al. 2001; Dorrie et al. 2001; Tinhofer et al. 2001). Joint applications of fractionated or protracted low-dose irradiation (low-LET) in combination with applications of resveratrol may lead to enhanced selective killing of cancer cells (genomically unstable cells selectively removed via the PAM process). Such low-dose combined therapy would likely be preferred by cancer patients over current high-dose radiation and chemotherapy which are associated with severe side effects. However, new research is needed to develop optimal dosing schemes. Common low-LET radiation sources used in medical diagnostics could be used in this form of combined therapy, including those used in nuclear medicine. Further, low-dose/dose-rate radioimmunotherapy could be employed in combination with applications of apoptosis-sensitizing agents (for selectively sensitizing cancer cells), such as resveratrol, in curing cancer while minimizing side effects. Other plant polyphenols that also sensitize cancer cells to undergoing apoptosis are genistein, curcumin, emodin, and flavopiridol (Garg et al. 2005). Multiple pathways to apoptosis may be associated with the indicated sensitizers since multiple pathways are known to be associated with resveratrol-induced apoptosis (Aggarwal et al. 2004). These pathways include the Fas pathway, mitochondrial pathway, Rb-E2F/DP pathway, p53-activation pathway, the ceramide-activation pathway, the tubulin-polymerization pathway, and the Adenyl-cyclase pathway.
Low-dose radiation therapy (i.e., therapeutic radiation hormesis) has already been reported to be successful for some types of cancer (Chaffey et al. 1976; Choi et al. 1979; Sakamota et al. 1997; Richaud et al. 1998; Cuttler et al. 2000; Cuttler and Pollycove 2003; Sakamota 2004; Kaminski et al. 2005). Fractionated, low-dose, total-body, and half-body external beam therapy has been used successfully by several medical groups in treating non-Hodgkin's lymphoma (Chaffey et al. 1976; Choi et al. 1979; Richaud et al. 1998; Cuttler et al. 2000, Cuttler and Pollycove 2003; Sakamota 2004). Small individual doses (called fractions) are administered after designated time intervals over a given time period. Dose fraction sizes used in treating non-Hodgkin's lymphoma have been relatively large, e.g., 100 to 150 mGy (Cuttler et al. 2000). Our research results indicate that much smaller fraction sizes may be equally effective and, if so, would allow for considerable extension of the total period over which dose fractions were given.
Therapeutic radiation hormesis has also been successfully employed to treat ovarian, colon, and hematologic cancer, with no symptomatic side effects (Cuttler and Pollycove 2003; Sakamoto 2004). Low-dose, low-dose-rate radioimmunotherapy (a form of radiation hormesis involving beta radiation) has also been used successfully in treating follicular lymphoma (Kaminski et al. 2005).
The PAM process is expected to be more efficiently activated by low-dose-rate and fractionated exposures than by high-dose-rate and single exposures. The time interval between the dose fractions could be quite critical. For new research, biweekly or once monthly fractions could be initially investigated. The number of fractions could be large without serious side effects, so long as small fraction sizes (e.g., 0.5 to 1 mGy) were used.
ENVIRONMENTAL RADIATION HORMESIS
Numerous studies have demonstrated that environmental exposures to ionizing radiation can suppress cancer and other diseases (environmental radiation hormesis). Indeed, immune responses have been found to be upregulated among inhabitants of high natural background radiation areas (Luckey 1991; Safwat 2000; Kojima et al. 2002). Table 4 shows
Central estimates of high-level, radon-associated

Presumed to be associated with environmental radiation hormesis.
With our HRR model, one can calculate the expected impact of reducing natural background ionizing radiation to zero. For such calculations, it is convenient to use normalized dose

where

Expected effects of reducing natural background radiation on cancer mortality, based on solid cancer mortality data for Yangjiang, China, reported by Wei and Sugahara (2002). Central (from constrained linear regression), and subjective upper and lower bound curves are presented. Normalized doses
Similar results are shown in Figure 9 for cancer among inhabitants of various cites and states in India, based on data from an ecological study conducted by Nambi and Soman (1987). Normalized dose was evaluated relative to an annual gamma-ray dose of 850 μGy (a relatively large dose) from natural background radiation. Thus, only gamma-ray doses were used. Cancer

Expected effects of reducing natural background radiation on cancer relative risk based on data for various cities and states of India reported by Nambi and Soman (1987). Central (from constrained linear regression), and subjective upper and lower bound curves are presented. Normalized doses
For calculations associated with Figures 8 and 9, it was assumed that high-fidelity DNA repair is not lost at near absolute zero background radiation, which may not be the case.
CONCLUSIONS
Environmental radiation hormesis associated with radon in our homes and with elevated background radiation (low- or low- plus high-LET) appears to be preventing many cancer deaths.
Medical radiation hormesis associated with routine applications of diagnostic chest X-rays, mammograms, and CT scans may be preventing cancer occurrence through stimulating the removal of precancerous neoplastically transformed cells. Medical and environmental radiation hormesis may also be preventing metastasis of existing cancer.
Low-dose therapeutic radiation hormesis associated with fractionated exposure to small X-ray doses has been used to successfully treat non-Hodgkin's lymphoma and ovarian, colon, and hematologic cancer.
Low-dose, low-dose-rate therapeutic radiation hormesis associated with application of radiolabeled antibodies (beta radiation source) has been used successfully to treat follicular lymphoma.
Low-dose therapeutic radiation hormesis in combination with apoptosis-sensitizing agents such as resveratrol could be used to successfully cure cancer.
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Office of Science (BER), U.S. Department of Energy (DOE) Grants DE-FG02-03ER63671 and DE-FG02-03ER63657. We are grateful to Vicki Fisher and Cynthia Herrera for editorial assistance. The views and conclusions contained herein are those of the authors and should not be interpreted as necessarily representing the official policies or endorsement, either express or implied, of the DOE or of Lovelace Respiratory Research Institute.
