Abstract
The objective of this study was to determine if exposure to divalent cations, Cd2+, Ni2+, and Co2+ would lead to malformations in Xenopus laevis embryos, and whether addition of Mg2+ and Zn2+; separately and in combination, would reduce their toxicity and teratogenicity on the embryos of Xenopus laevis as assessed by 96-h FETAX tests. Results indicate that exposure to Cd2+, Ni2+ or Co2+ lead to an increase in toxicity and teratogenicity in embryos, whereas Mg2+, Zn2+, or a combination of them reduced the toxic and teratogenic effects of these divalent cations. Modulation of Cd2+, Ni2+ or Co2+ toxicity and teratogenicity by Mg2+ and Zn2+, varied with the metal. Zn2+ was observed to be a better suppressor of Co2+ toxicity and teratogenicity than Mg2+. In contrast, Ni2+, and Cd2+ teratogenicity was reduced more prominently by Mg2+. On the other hand, combination of Mg2+ and Zn2+ showed potentialization effect on all divalent cation toxicity and teratogenicity. We concluded that Mg2+ and Zn2+ reduced the toxicity and teratogenicity of Cd2+, Ni2+, Co2+.
Keywords
INTRODUCTION
Hill and Matrone (1970) first suggested that the biologically essential ‘bioelements’ interact with toxic metals due to their similar physical and chemical properties (Brzoska, and Jakonuik 2001a). In recent years, such interactions have been determined to exist among bioelements like Zn2+, Cu2+, Fe2+, Se2+, and Ca2+, and the toxic metals such as Cd2+, Ni2+, Co2+ (Brzoska and Jakonuik, 2001b). The antagonistic and synergistic interactions were evaluated with regard to their competition for metal-specific target molecules or the metalloproteins, metalloenzymes, and the binding sites on membranes (Güven, 1999).
Ni2+, Co2+ and Cd2+ have been demonstrated to be potentially teratogenic as shown on Xenopus laevis by the FETAX test (Plowman et al., 1991, TOX Probe, 1993, Plowman et al., 1994, Costa et al., 2001, Kasprzak, 2002). It has been found that addition of Mg2+ at various concentrations to Ni2+, Co2+, Cd2+ and Zn2+ decreased the embryotoxic and teratogenic effects of these cations in proportion with the Mg2+ concentration on Xenopus laevis (Luo et al.,1993). The same investigators speculated that this effect was the result of the competition between Mg2+ and these divalent cations for physiological process such as binding and intake into cells.
The interaction of Zn2+ with Cd2+ and Ni2+ have also been investigated on Bufo arenarum embryos (Herkovits et al., 1998, Herkovits et al., 2000, Waalkes, 2000, Fort et al., 2001). In these studies, Zn2+ was found to have a protective agent against retarded growth and lethal effects of these heavy metals on the embryos. However, in biological systems, the interactions between the heavy metals depend on the amount of material present, the tested species, concentration and length of exposure. In this manner, different results have been obtained from the exposure of fish and plants to different heavy metals (Herkovits et al., 1998).
In biological systems, Cd2+ and Zn2+ macromolecules bind to sulphur (S), oxygen (O) and nitrogen (N) and reacts avidly with the S−, O−, and N− donors. Cadmium and Zn2+ are bound preferentially to the same proteins in plasma, and proteins and metallothionein (MT) in tissues. Various studies have shown Cd2+ and Zn2+ to have high affinities for metallothionein and that their interactions lead to the induction of the synthesis of MT. MT has been shown to be synthesized in various tissues especially from the intestines, liver and kidneys (Pasky et al.,1996, Brzoska and Jakonuik, 1998). On the other hand, the binding of heavy metals by homeostatically-protective sulfur, oxygen, or nitrogen-based ligands were highly variable (Herkovits et al., 2000). In each case the intake of the metals into cells and their roles in other processes depend on their concentrations (Rainbow, 2002, Brzoska and Jakoniuk, 2001b, Falchuk, 1998a). The result of this is the tendency for Cd2+ to competitively bind intercellular sites resulting in the exclusion of Zn2+ from critical processes (Rink and Gabriel, 2001, Sanstead, 2000, Falchuk, 1998b, Pasky et al., 1996).
The influence of Zn2+ on to Ni2+ toxicity was suggested to be due to various factors such as metal intake, which leads to a fall in tissue concentration. This finding indicated the requirement of one metal for the intake of the other. Zinc applied at concentrations of 0.5 mg /l led to little or no effect which can be attributed to the presence of Ni2+. At higher concentrations of Zn2+ (2–30 mg/1) however, the intake of Ni2+ increased as a result of synergistic toxic effect. At much higher concentrations 60–100 mg/l Zn2+, by competing with Ni2+ for uptake into cells, may increase its intake. This result resembles a U-shaped dose—response curve. A typical U-shaped dose-response curve is a relation in which concentrations below a certain level for a particular chemical shows benefits and an opposite effect at higher doses (Arndt-Schulz law (Herkovits et al., 2000).
The beneficial effect of Zn2+ on the toxicity of Ni2+ is most likely due to its induction of the metabolic enzymes, GSH (reduced GSH) and GST (Glutathion—S-Transferase), and is likely to be similarly affected by Cd2+, Ag2+, Hg2+, Pb2+, and Se2+ which all are considered to be xenobiotics (foreign to the organism) (Herkovits et al.,1998). Glutathion conjugation has an important toxicological function especially in the excretion of reactive intermediate products (OH−, H2O2). Glutathion, is a tripeptide (glutamic acid, glycine, and cysteine) compound found in several mammalian tissues (particularly in the liver). Normally, the sulphydryl groups attack exposed reactive or electrophilic metabolites. These reactions are catalyzed by the glutathion transferases. The importance of this class of enzymes lies in their role in the metabolism of electrophilic groups like alkyl, aldehydes and ketones (Herkovits et al., 1998).
The frog, particularly the South African Claved frog Xenopus laevis, has been used as a model of embryonic development for more than 100 years. Alternative bioassys such as Frog Embryo Teratogenesis Assay: Xenopus (FETAX) provide a rapid, simple, and cost effective method for evaluating mechanisms of developmental toxicity on a preliminary basis. FETAX is a 96-h whole embryo, static renewal assay using Xenopus laevis embryos (Fort et al., 2001). It is essentially an organogenesis test, and organogenesis is highly conserved across amphibians and mammals. The first 96 hours of embryonic development in Xenopus parellels many major processes of human organogenesis. Thus, FETAX should be useful in predicting potential human developmental toxicants and teratogens (American Society for Testing and Materials (ASTM), 1998). The National Toxicology Program Interagency Center for the Evaluation of Alternative Toxicological Methods (NICEATM) assessed the validation status of FETAX as a screening assay for detecting potential human teratogens, and for its use in the ecotoxicological assessment of water /soil/sediment/samples (NICEATM, 2004).
The aim of this study was to evaluate embryotoxic and teratogenic effects of the xenobiotic divalent metals Cd2+, Ni2+ and Co2+, and the interactions of the presence of Mg2+, Zn2+ and their combination using the FETAX test.
MATERIALS AND METHODS
Xenopus laevis embryos (N: 13, n: 6660) were used in this study and were obtained by in vitro fertilization. According to ASTM FETAX Guideline (1998), in practice, 95% normal, live embryos should be obtained routinely. In the present study, at every phase approximately 700 embryos were squeezed, and the ratio of normal embryos were appropriate to ASTM guidelines.
CI− salts of Cd2+, Ni2+, Co2+, Mg2+, Zn2+ divalent cations obtained from Sigma chemical Co. (St.Louis, Missouri, USA) were used to make stock solutions. Stock solutions were prepared and the concentrations were confirmed using a Parkin-Elmer 3100 model Atomic Absorbtion spec-trophotometer (AAS). To prevent the precipitation of metallic hydroxides, the solutions were adjusted to a pH of 6.8. Stock solutions were stored at +4°C.
The tested concentrations of Cd2+, Ni2+, Co2+, Mg2+, Zn2 were prepared in FETAX solution and pipetted into a petri dish. After a period of five hours, embryos reached phases 7–8 of development; were placed in the test solutions, and were incubated at 23± 1°C. Solutions were changed every 24 hours. The number of normal, abnormal and dead embryos were evaluated at the 96th hour under a stereo microscope (Kyowa). Evaluations were conducted according to the Nieuwkoop's normal table atlas (Nieuwkoop, and Faber, 1994). Live, but not considered fit normal embryos were accepted as abnormal. Mortality at 96th (stage 45) hour was confirmed by an arrested heart in the transparent embryo. Dead malformed embryos were not included in the malformed count (ASTM, 1998, Boga et al., 2000).
The experiment was conducted in four stages. In the first stage, the xenobiotic cation solutions including Cd2+ (5, 7.2, 8.5,10 μmol/1), Ni2+ (10, 80, 100, 120 μmol/1), Co2+ (20, 56, 112, 120 μmol/1) were evaluated alone. A total of 1800 Xenopus embryos (from 5 females) were used in 5 replicated experiments. Five petri dishes each containing 20 embryos were used for each replicate.
In the second stage, a single concentration of magnesium (40 mmol/l) was added to solutions of different concentrations of the xenobiotic divalent cations including, Cd2+ (5, 7.2, 8.5, 10 μmol/l); Ni2+ (10, 80,100,120 μmol/l); Co2+ (20, 56, 112, 120 μmol/l). A total of 1800 Xenopus embryos (from 5 females) were used in 5 replicated experiments. Five petri dishes each containing 20 embryos were used for each replicate.
In the third stage, a single concentration of zinc (40 μg/l) was added to the solutions of different concentrations of the xenobiotic divalent cations including Cd2+ (5, 7.2, 8.5, 10 μmol/l), Ni2+ (10, 80, 100, 120 μmol/l), Co2+ (20, 56, 112, 120 μmol/l). A total of 1260 Xenopus embryos (from 3 females) were used in 3 replicated experiments. Three petri dishes each containing 20 embryos were used for each replicate.
In the fourth stage, a combination of magnesium (40 mmol/l) + zinc (40 μg/l) was added to solutions of xenobiotic cations including Cd2+ (5, 7.2, 8.5,10 μmol/l), Ni2+ (10, 80, 100, 120 μmol/l), Co2+ (20, 56, 112, 120 μmol/l). A total of 1800 Xenopus embryos (from 5 females) were used in 5 replicated experiments. Five petri dishes each containing 20 embryos were used for each replicate.
DATA ANALYSIS
The percentages of normal, malformed, and dead embryos were analyzed using the multivariated Logistic regression. SPSS 14.0 was used for data analysis. A multivariate logistic regression analysis was applied to assess the Odds Ratio (OR) for anomaly, death and anomaly or death. The OR's given for group and dose were adjusted for the effect of each other. Significance level was accepted as p≤0.05.
RESULTS
The level of risk for each teratogen dose and combination group was evaluated statistically. The risk of anomaly, death, anomaly/death of Cd2+ treatment are presented in Table 1. As can be seen from Table 1 anomaly, death, anomaly or death risk rose parallel to the increase in doses.
Odds Ratio (OR) (95%C.I) of Cd, Cd + Mg; Cd + Zn and Mg + Zn + Cd on Xenopus embryos, by using FETAX.

According to ref1 (Cd2+–5μmol/l), the risk of anomaly of the treatments of 7.2, 8.5, 10 μmol/l Cd2+ was 1.47 (1.04–2.07) p=0.029, 1.80 (1.26–2.55) p=0.001, 2.47 (1.68–3.63) p<0.001 respectively; the death risk was 2.42 (1.58–3.71) p<0.001, 1.87 (1.21–2.9) p=0.004, 3.77 (2.44–5.70) p<0.001 respectively, and the anomaly/death risk was 1.63 (1.20–2.21) p=0.001, 1.62 (1.19–2.220) p=0.002, 2.48 (1.81–3.4) p<0.001, respectively. Figure 1 shows the risk of anomaly, death, and anomaly or death together due to the Cd2+ doses on Xenopus laevis embryos. As seen in this figure; the highest death risk was with 10 μmol/l Cd2+.

Odds Ratio (OR) (95%C.I) of Cd2+ doses adjusted for group.
The risk of anomaly for groups T (Teratogen) + Mg2+, T + Zn2+, T + Mg2+ + Zn2+ compared to ref1 (Cd2+) was 0.067 (0.045–.099) p<0.001, 0.163 (.116–.229) p<0.001, 0.302 (.203–.443) p<0.001, the risk of death was 0.337 (.226–.501) p<0.001, 0.021 (.009–.049) p<0.001, 1.320 (.883–1.97) p=0.175, and the risk of anomaly/death was 0.143 (.103–.199) p<0.001, 0.128 (.092–.178) p<0.001, .556 (.396–.781) p=0.001, respectively (Table 1, Figure 2, 3, 4).
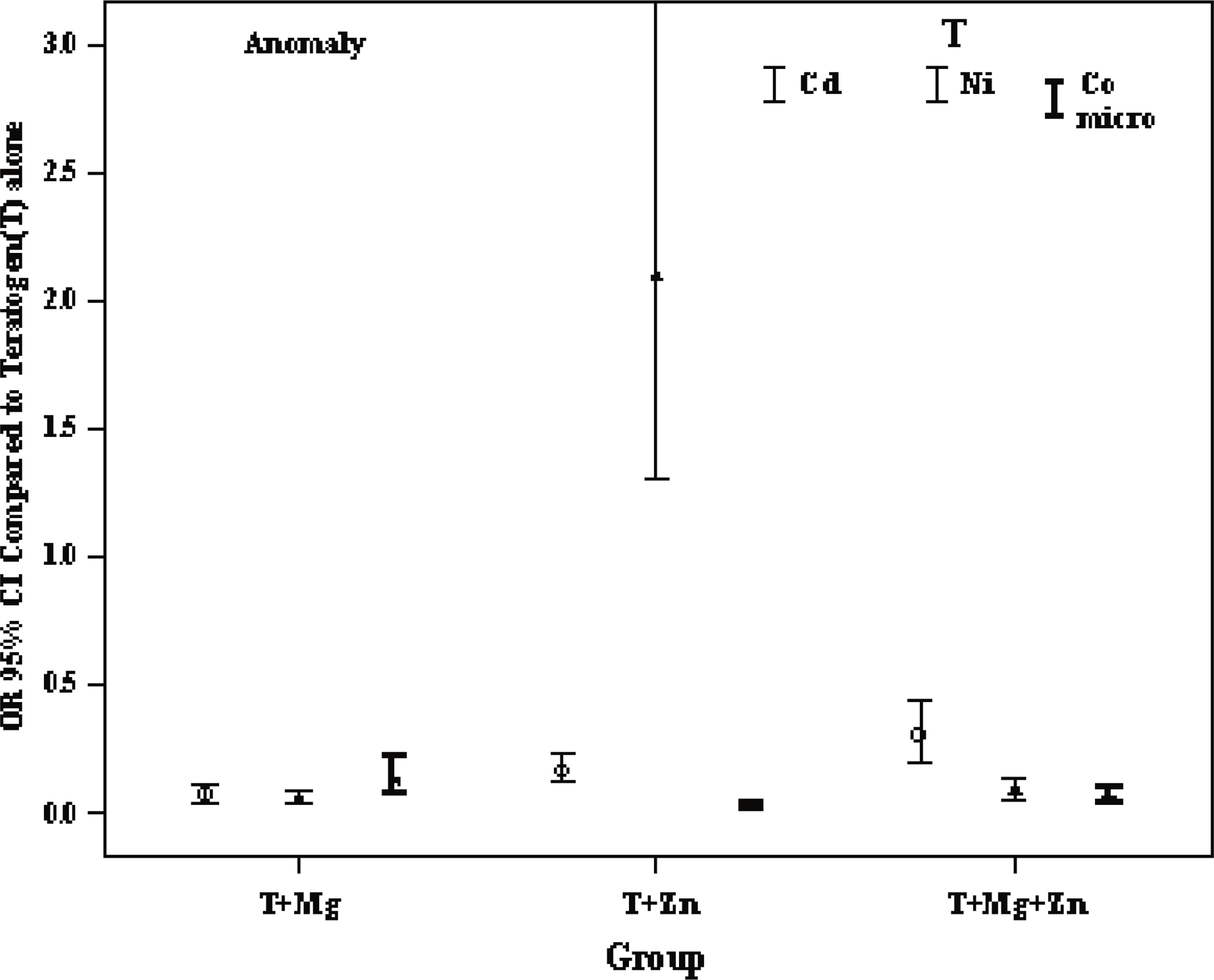
Odds Ratio (OR) (95%C.I) of Cd2+, Ni2+, Co2+ anomaly group adjusted for doses.

Odds Ratio (OR) (95%C.I) of Cd2+, Ni2+, Co2+ death group adjusted for doses.
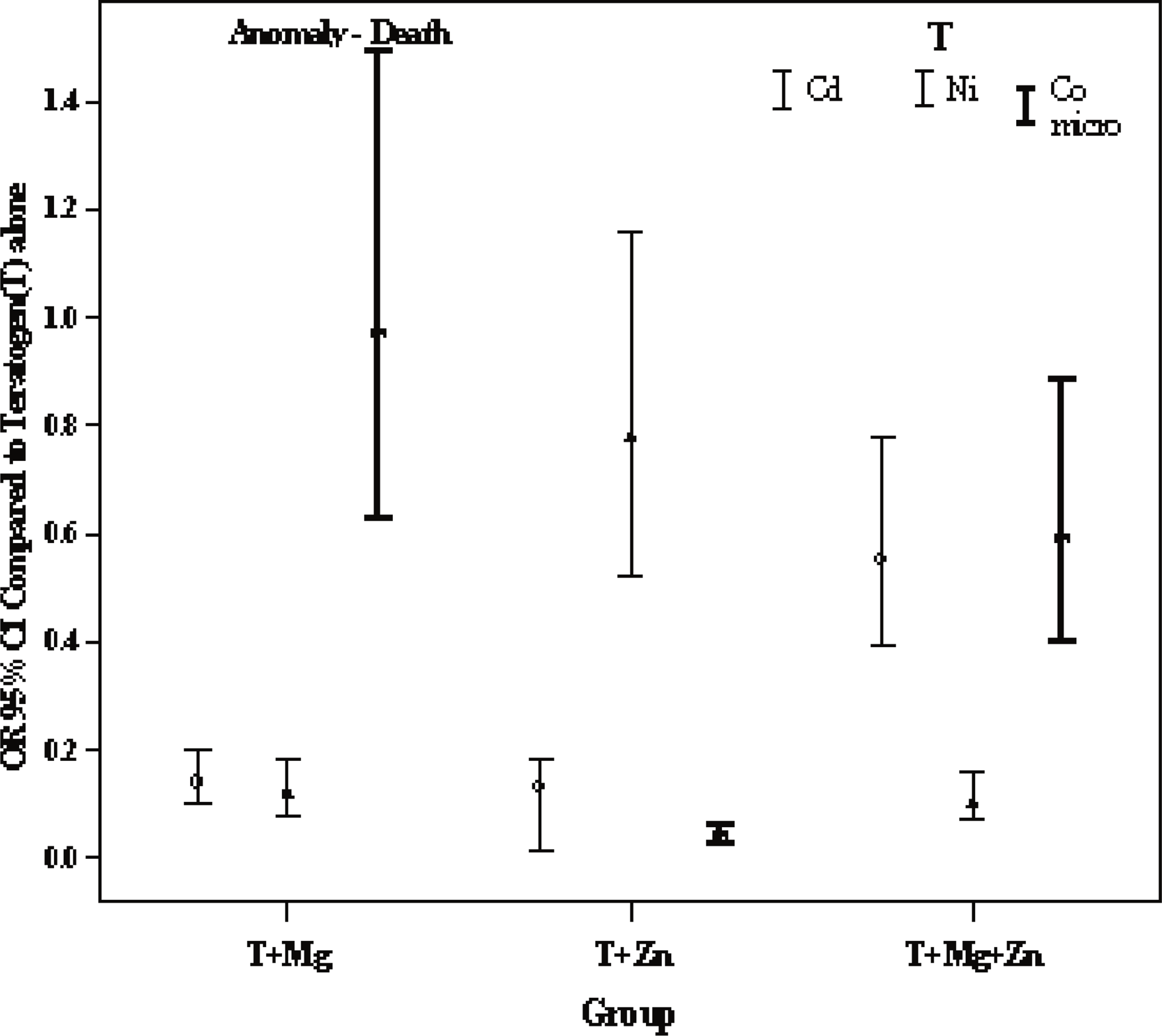
Odds Ratio (OR) (95%C.I) of Cd2+, Ni2+, Co2+ anomaly/death group adjusted for doses.
It appears that among all combinations, Cd2+ + Mg2+ 0.0067 (0.045–.099) p<0.001) induced the lowest risk of anomaly, and also appeared to decrease the risk of death 0.337 (.226–.501) p<0.001. The Cd2+ + Mg2+ + Zn2+ combinations induced significantly lower risk of anomaly 0.302 (0.203–0.443) p<0.001 but induced a higher risk of death 1.320 (.883–1.97) p=0.175 whereas this increase was not signifcantly higher than for Cd2+ alone. Zinc appeared to induce the lowest mortality 0,021 (.009–.049) p<0,001, and also decrease the risk of anomaly 0.163 (.116–.229) p<0.001 which may indicate that Zn2+ was found to be the most protective 0.128 (.092–.178) p<0.001, (Table 1).
The anomaly, death, anomaly/death risk of the Ni2+ treatment are presented in Table 2. Similar to the results illustrated in Table 1, we can see that the increase/decrease in anomally, death, anomaly/death risk also dependant on the doses. According to ref1 (Ni2+–10μmol/l), the risk of anomaly of the treatments of 80, 100, 120 μmol/l Ni2+, was 10.6 (6.61–17.0) p<0.001, 14.5 (8.95–23.6) p<0.001, 12.9 (7.64–21.9) p<0.001 respectively; the risk of death was 3.31 (2.21–4.96) p<0.001, 3.74 (2.48–5.64) p<0.001, 6.92 (4.58–10.45) p<0.001 respectively and the risk of, anomaly/death was 6.45 (4.57–9.12) p<0.001, 7.83 (5.50–11.1) p<0.001, 11.55 (7.97–16.7) p<0.001, respectively. Figure 5 shows the risk of anomaly, death, and anomaly or death together due to the Ni2+ treatments on Xenopus laevis embryos. As seen in Figure 5, higher anomaly risk was found in all doses when compared to ref1 (Ni2+).
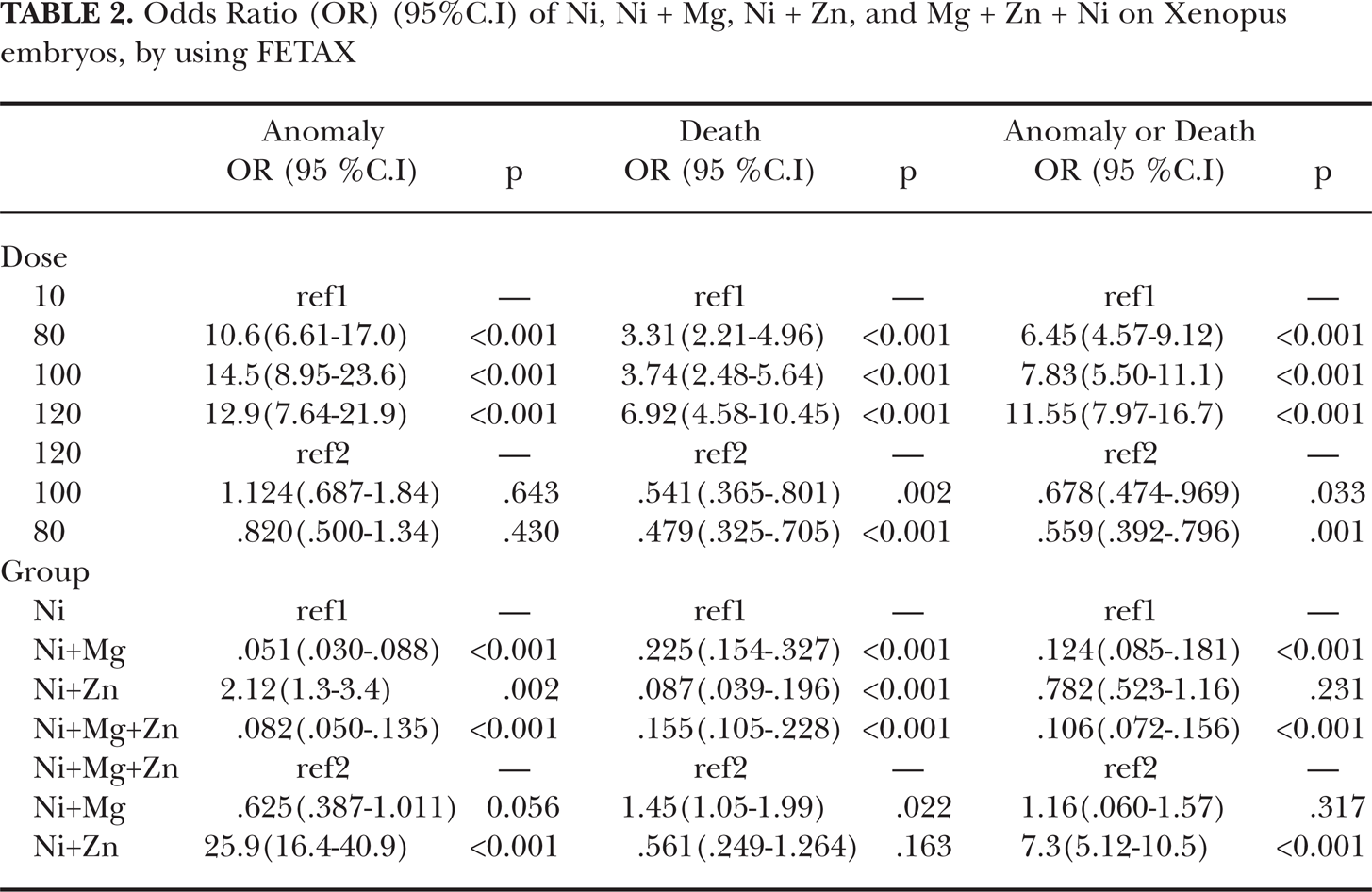
Odds Ratio (OR) (95%C.I) of Ni, Ni + Mg, Ni + Zn, and Mg + Zn + Ni on Xenopus embryos, by using FETAX
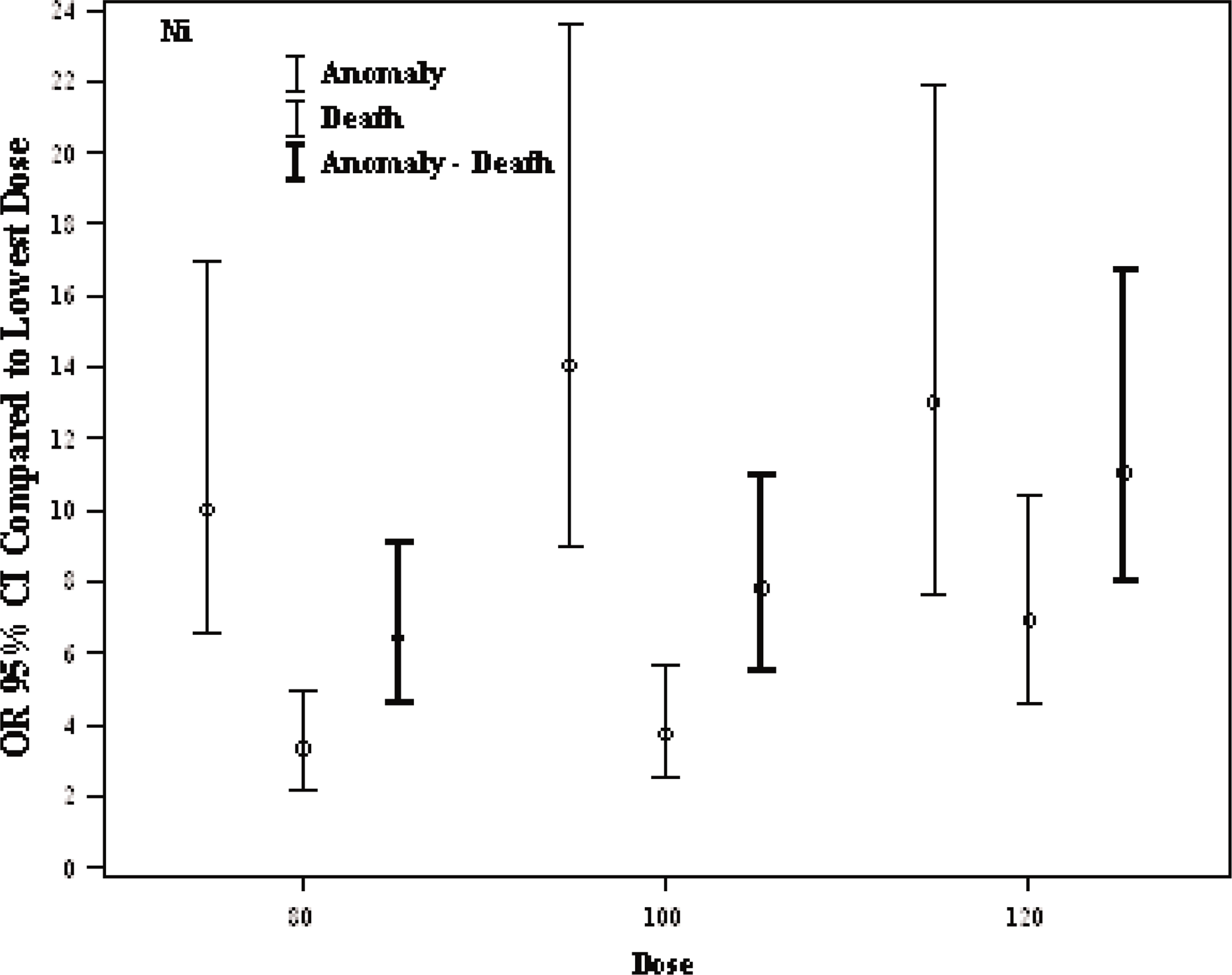
Odds Ratio (OR) (95%C.I) of Ni2+ doses adjusted for group.
The risk of anomaly for groups T (teratogen) + Mg2+, T + Zn2+, T + Mg2+ + Zn2+ compared to ref1 (Ni2+) was 0.051 (.030–.088) p<0.001, 2.12 (1.3–3.4) p:0.002, 0,082 (.050–.135) p<0.001, respectively, the risk of death was 0.225 (.154–.327) p<0.001, 0.087 (.039–.196) p<0.001, 0.155 (.105–.228) p<0.001 respectively, and the risk of anomaly/death was 0.124 (.085–.181) p<0.001, 0.782 (.523–1.16) p:0.231) and 0.106 (.072–.156) p<0.001, respectively (Table 2, Figures 2, 3, 4).
It appears that among all combinations, Ni2+ + Mg2+ 0.051 (.030–.088) p<0.001 induced the lowest risk of anomaly and the Ni2+ + Mg2+ + Zn2+ combination 0.106 (.072–.156) p<0.001 induced the lowest risk of anomaly/death. The difference in this of risk of anomaly/death was not statistically significant from that of the Ni2+ + Mg2+ treatment. Zinc appeared to induce the lowest mortality, due to the Ni2+ treatments 0.087 (.039–.196) p<0.001, but increased the risk of anomaly of the Ni2+2.12 (1.3–3.4), p= 0.002 (Table 2).
The anomaly, death, anomaly/death risk of Co2+ treatment are presented in Table 3. Here, again as in Table 1 and 2, increase in anomally, death, anomaly/death risk was also found to be closely related to the rise in doses. According to ref1 (Co2+-20μmol/l), the risk of anomaly of the treatment of 56, 112, 120 μmol/l Co2+ was 3.14 (1.88–5.2) p<0.001, 8.9 (5.1–15.5) p<0.001, 23.9 (13.5–39.8) p<0.001 respectively; the risk of death was 1.56 (1.04–2.33) p=0.029, 4.5 (2.8–7.2) p<0.001, 6.4 (3.5–11.6) p<0.001 respectively, and the risk of anomaly/death was 2.0 (1.4–2.8) p< 0.001, 4.7 (3.2–7.0) p<0.001, 7.3 (4.8–11.1) p<0.001, respectively. Figure 6 shows the risk of anomaly, death, and anomaly/death of the Co2+ doses. The OR for anomaly was highest at 120μmol/l.
Odds Ratio (OR) (95%C.I) of Co, Co + Mg, Co + Zn, and Mg + Zn + Cd on Xenopus embryos, by using FETAX.


Odds Ratio (OR) (95%C.I) of Co2+ doses adjusted for group.
As risk of anomaly compared to ref1 (Co2+) T (teratogen) + Mg2+, T + Zn2+, T+ Mg2+ + Zn2+ was 0.126 (.072–.221) p<0.001, 0.022 (.014–.036) p<0.001, 0.052 (.029–.095) p<0.001 respectively, the risk of death was 5.3 (3.21–8.9) p<0.001, 0.047 (.025–.088) p<0.001, 3.45 (2.1–5.5) p<0.001 respectively, and the risk of anomaly/death was 0.977 (.63–1.49) p=0,914, 0.039 (.026–.059) p<0.001, 0.594 (.399–.883) p=0.010), respectively (Table 3, Figure 2, 3, 4).
It appeared that among all exposure combinations, the combination of Co2+ and Zn2+ induced the lowest risk of mortality 0.047 (.025–.088) p<0.001, the lowest risk of anomaly 0.022 (.014–.036) p<0.001, and the lowest risk of anomaly/death 0.039 (.026–.059) p<0.001. The Mg2+ treatment appeared to increase the toxicity of Co2+ exposure. The OR value for Co2+ + Mg2+ + Zn2+ combination was lower than that for the Co2+ + Mg2+ combination, but higher than that for the Co2+ + Zn2+ combination, and it was significantly lower compared to the ref1 (Co2+).
It also appeared that there may have been some competetive interaction between Mg2+ and Zn2+ since the protective effects of Zn2+ were not clearly evident when Mg2+ was included.
DISCUSSION
Since the 1990s, various investigators have studied the effect of metallic ions on teratogenicity indices and mechanisms. Interactions that exist between the bioelements and the toxic metals have been discovered (Brzoska, and Jakonuik, 2001b, Kasprzak, 2002, Plowman et al., 1994, Costa et al., 2001, TOX Probe, 1993, Plowman et al., 1991, Miller and Landesman, 1978).
The protective effect of Mg2+ on all the divalent cations (Luo et al., 1993) and that of Zn2+ on Ni2+ (Herkovits et al., 2000) and Cd2+ (Waalkes, 2000, Brzoska and Jakoniuk, 2001b) have been investigated. In addition, Kasprzak (2002) and Costa et al. (2001) demonstrated the protective effect of Mg2+ against the the toxic and teratogenic effects of Ni2+ compounds. We evaluated not only the effect of Mg2+ against toxic effect of Cd2+ and Ni2+ but also the effect of Zn2+ alone and in combination with Mg2+ on the toxic effect of Cd2+, Ni2+ and Co2+. We found that these combinations reduced toxicity and teratogenicity of these xenobiotic metals.
According to Luo et al. (1993), a possible mechanism for the effect of magnesium on the other divalent cations is the competition between these ions for transport mechanisms; including absorption, intake into cells, or binding to critical molecules (e.g. DNA polimerase) (Luo et al., 1993). This finding may explain our results of Zn2+ supression of the teratogenic effect of Co2+, and Mg2+ supression of the teratogenicity of Cd2+ and Ni2.
The FETAX test seems to be, useful for rapid screening of samples as the method provide adequate information on the teratogenicity of various agents using a standardized procedure (Dawson and Bantle, 1987). Although, the FETAX test meets most of the foreseen criteria for testing in vitro teratogenesis, combination with Mixed Function Oxidase System (MFO) proves to be a more effective method in transforming proteratogenic compounds into teratogenic metabolites. By making use of the data from Kitchin and Woods (1979) who utilized whole rat embryo cultures, Dawson and Bantle (1987), employed recently induced rat liver micro-somes as the exogenous metabolic activator system (MAS) for the FETAX test. Potentially teratogenic substances that tested negative in these tests may become transformed into teratogenic forms causing congenital defects.
Zinc is a component of the metalloenzymes which plays a role in the metabolism of animals at all stages of their development. At all cell development stages, it plays regulatory and protective part in the process of protein synthesis. It also stabilizes biological membranes by competing with the redox active divalent metals such as copper, mercury and cadmium for binding onto cell membrane sites (sulfhydryl groups). It synthesizes metallothionein which also binds to the heavy metals to eradicate OH− radicals, which can be formed by cobalt as well as Ni2+, Cu2+, Cr2+, Fe2+ (Plowman et al., 1994, Tox Probe, 1993, Plowman et al., 1991). Zinc was also found to decrease the teratogenic effect of Co2+ thus, in turn, leading to the decrease of level of OH− radicals.
Deficiency of zinc has a negative impact on development and reproduction, causing development of abnormal eggs, changes in meiotic and ovulatory processes, abnormal spermatozoa and a high incidence of congenital anomalies. On the other hand, high zinc concentrations affect reproduction and viability. Zinc enables absortion of food, provides well-being, reproduction and longevity in invertebrates. No adverse effect was encountered in Bufo arenarum embryos exposed to a concentration of up to 130 mg/l Zn2+. In studies conducted, beneficial effects of Zn2+ on amphibian embryos were found to be a reduction in the rate of spontaneous anomalies and malformations, and protection against the retarded growth and development and death due to metals like Cd2+, Pb2+, Al2+, Hg2+, and Cu2+ (Rink and Gabriel, 2001, Sandstead, 2000, Falchuk, 1998a, Pasky et al., 1996).
The effect of cadmium is neutralized by the addition of zinc, which decreases absorption of Cd2+. Zinc deficiency leads to accumulation and increases toxicity of Cd2+. In the biological system, Cd2+ and Zn2+ bind specifically to macromolecules, specifically through sulphur (S), oxygen (O) and Nitrogen (N), and interact with the S−, O− and N donors. The ions of Cd2+ and Zn2+ compete for entry into various cells and in many biological processes Zn2+ may take the place of Cd2+. Such interactions can be either competitive or non competitive and largely occur in the intestines where MT and Cd2+ absorption occurs (Pasky et al., 1996, Brzoska and Jakonuik, 2001a). Zinc was also found to be more effective than magnesium reducing the toxic effect of Cd2+ in our study.
An earlier study, demonstrated that 0.5 mg/l of Zn2+ did not affect the toxicity of cadmium in zebra fish (Brachydanio rerio) (Küçükoglu, 1996). Contrary to this finding, we observed that zinc, even at μg/l concentrations, was enough to decrease the teratogenicity on Xenopus laevis embryos.
The toxicity of Ni2+ on embryos of Bufo arenarum was found to be lower compared to other metals (Cu2+> Cd2+> Hg2+> Al2+> Pb2+> Ni2+> Zn2+) whereas Zn2+ sulphate concentrations under 130 mg/l were found to have no lethal effect on the embryo. Embryos of amphibians, compared to other species such as daphnia, algae, and fish, are more tolerant to Zn2+ at least in their last developmental stage (Herkovits et al., 2000).
In the same study, the Ni2+–Zn2+ interaction forms a U shaped dose-response curve. While Zn2+ at 0.5 mg/l failed to suppress the toxicity of Ni2+, concentrations of this metal at 2–30 mg/l increased the death rate but desired beneficial effects were observed at higher concentrations of 60–100 mg/l. A synergistic effect of combined Zn2+ and Ni2+ was observed at concentrations of 2–30mg/l.
The mechanism of Ni2+ toxicity, similar to other metals (Cd2+, Co2+), increases lipid peroxidation, cellular injury and death. Inhibition of cellular defence against peroxidative injury is through the inhibition of protective enzymes against free radicals and/or through free oxygen radical production. On the other hand, Ni2+ under in vitro conditions binds to proteins and DNA while, under in vivo conditions, it binds to chromatins. By binding to macromolecules, Ni2+ compounds interfere with DNA synthesis, and hence lead to increased chromosomal abnormalities (Herkovits et al., 2000). In our study, Mg2+ at mmol/l concentrations significantly decreased the teratogenic effect of Ni2+. Thus, we suggest Mg2+ may have competed with Ni2+ for Ni2+'s target.
In conclusion, bioelements such as Zn2+, and Mg2+ can suppress teratogenic and toxic effects of xenobiotic cations such as Cd2+, Ni2+, Co2+. These interactions can be evaluated by FETAX, and therefore Xenopus laevis is an effective model for this evaluation. In the light of such studies, metal interaction research provides a scientific basis for establishment of water quality criteria for protection of humans from wildlife.
