Abstract
Objective
So far there are still no effective immediate-early markers for assessing the efficacy of Stereotactic Body Radiation Therapy (SBRT). To find effective biomarkers for accurate assessment of the efficacy of SBRT in patients with primary liver cancer, we conducted this study including retrospective part and prospective part.
Material and Methods
589 patients with primary liver cancer were included at Ruikang Hospital affiliated to Guangxi Medical University from January 2012 to December 2018. Follow-up was conducted, clinical information and a total of 17 patients with 51 blood samples (before SBRT, before discharge and 2 months after SBRT) were collected. mRNAs profiles on 2 patients with 6 blood samples were detected by high-throughput sequencing, followed by qPCR verification on 15 patients with 45 blood samples.
Results
The commonly used serum biomarkers such as AFP, CEA, and CA125 shown low prognostic value in distinguishing survival group and death group, indicated by low AUC (less than .7) and Youden indexes (less than .5). Based on high-throughput sequencing of test group and qPCR detection of another verification group, we found 16 up-regulated and 12 downregulated genes after SBRT. Among them, ADIPOR1 and EPB42 showed significantly different between effective and ineffective group after SBRT, ROC suggested that based on the optimal threshold of .5838, ADIPOR1 shown a sensitivity of 100% and a specificity of 83.33% to distinguish effective from ineffective group. And EPB42 had a sensitivity of 75% and a specificity of 100% at the optimal threshold of 1.3817. In addition, GSEA showed that high expression of ADIPOR1 was mainly related to Mismatch repair, Circadian rhythm, Protein processing in endoplasmic reticulum, DNA replication, and Fanconi anemia pathways.
Conclusion
ADIPOR1 in whole blood is a promising candidate to act as prognostic biomarker for predication of SBRT outcomes in primary liver cancer patients.
Introduction
Liver cancer is expected to be the seventh most frequently diagnosed cancer and the second leading cause of cancer death around the world in 2020, with 905 677 new cases and 830 180 deaths per year. 1 It is also one of the most common malignant tumors in China, showing No. 5 of morbidity and No. 2 of mortality among malignant tumors in Chinese. 1 Hepatocellular carcinoma (HCC) is the leading pathologic subtype contributing 85–90% of primary liver cancer. 2 The main risk factors for liver cancer are chronic hepatitis B virus HBV or hepatitis C virus HCV, heavy alcohol intake, aflatoxin-contaminated foods, obesity, smoking, etc. Among all cancers in China, liver cancer has the poorest survival except for pancreatic cancer and the age-standardized 5-year relative survival is only 14.1%. 3 In 2022, it is estimated that there will be 431,383 new liver cancer cases and 412,216 new liver cancer deaths in China. 4 Invasion, metastasis and recurrence are the primary factors that affect clinical treatment and prognosis. 5 Besides, the insidious onset of liver cancer is another reason, for the majority of liver cancer patients are diagnosed at a late stage when it is too far advanced to be cured. 6 Being highly malignant with rapid progression, the treatment for advanced liver cancer is difficult.
At present, there are various treatment methods for liver cancer, including surgical treatment, such as radical surgical resection and liver transplantation; non-surgical treatment, such as local ablation therapy, arterial chemoembolization, gene molecular targeted therapy, systemic chemotherapy, and radiotherapy. 7 Surgical resection has proven to be the optimal treatment for long-term survival of liver cancer patients. 8 However, more than 70% of patients with liver cancer are unable to undergo liver resection due to the location, size, number of liver tumors, and impaired liver function. Therefore, the status of non-surgical therapy in the treatment of liver cancer is self-evident. For patients with liver cancer who are unable to undergo surgery, the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN guidelines, Version 1.2022) recommend radiation therapy as one of the treatments. 9
Improvements in diagnostic imaging, treatment planning, and treatment delivery have made radiation therapy more accurate and precise. 10 Beginning with the three-dimensional conformal radiation therapy, radiation therapy is increasingly being used for the treatment of liver cancer. At present, radiotherapy for liver cancer includes a series of advanced technologies, such as intensity-modulated radiation therapy, body stereotactic radiotherapy, and particle therapy. Current precision external exposure techniques ensure that the tumor is locally administered with high doses while protecting the remaining normal liver tissue from exposure to low doses, thereby limiting the risk of radiation-induced liver damage. In addition, external beam radiation therapy (EBRT) is applied to tumors in almost all locations of the liver. SBRT is an advanced technique of EBRT that delivers large ablative doses of radiation. Increasing evidence supported the usefulness of SBRT for patients with unresectable, locally advanced, or recurrent liver cancer.11-13 Additionally, NCCN also recommends that SBRT can also be used as an alternative to ablation/Transarterial Chemoembolization (TACE) treatments, options for treatment after ablation/TACE failure, or treatment options for liver cancer patients with ablation/TACE contraindications.
However, to date, the evaluation of radiotherapy efficacy relies mainly on imaging data and the calculated local control based on complete response (CR), partial response (PR), stable disease (SD), progressive disease (PD), such long-term index as overall survival (OS), disease-free survival (DFS), progression-free survival (PFS), etc. are also used.14,15 In addition, some serum molecules have been used for biomarkers, for example, AFP has long been used for estimation of the liver cancer progression, but even in the advanced stage 15–30% of patients with a normal AFP levels. 16 So far there are still no effective immediate-early markers for assessing the efficacy of radiotherapy. Therefore, finding biomarkers with prognostic value for radiotherapy is still a focus that researchers are paying attention to. Genetics affects the occurrence and development of tumors, whether the change of mRNA profiles in venous blood after radiotherapy could be used as prognostic biomarkers, is our concern. In the current study, we mainly focused on evaluating the prognostic value of serum biomarkers, both the commonly used (AFP, CEA, CA125, CA153, CA199, and CA724) and novel (screened from high-throughput sequencing data) biomarkers, for HCC patients treated by SBRT.
Adiponectin is the most abundant fat specific hormone, which functions by binding to its specific receptors adiponectin receptor protein 1 (ADIPOR1) and adiponectin receptor protein 2 (ADIPOR2). 17 ADIPOR1 is widely expressed in skeletal muscle and liver. The activation of ADIPOR1 plays an important role in regulating glucose metabolism and fatty acid catabolism. 18 High-fat alcoholic diet downregulated the protein levels of hepatic ADIPOR1. 19 In recent years, it has been found that ADIPOR1 also plays an important role in tumorigenesis, invasion, and metastasis.20-23 As for ADIPOR1 and liver cancer, it reported that overexpressed miR-221 promoted epithelial-mesenchymal transition (EMT) in hepatocellular carcinoma by targeting ADIPOR1, 24 and low expression of ADIPOR1 was associated with increased risk of recurrence and death in patients with liver cancer. 25 In this study, we identified ADIPOR1 as a prognostic marker for SBRT in patients with liver cancer through clinical information, high-throughput sequencing and qPCR analysis.
In short, the main purpose of this study has two points: 1. To evaluate the prognostic value of commonly used tumor markers in patients with primary liver cancer treated with SBRT; 2. Screen for new and effective biomarkers for patients with primary liver cancer treated with SBRT based on sequencing data. This is the first report for screening prognostic biomarkers for liver patients treated with SBRT.
Material and Methods
Patient Selection and Medical Record Collection
The primary liver cancer patients involved in this study were divided into two parts. In the retrospective study, the clinical records of all primary liver cancer patients who underwent SBRT at Ruikang Hospital affiliated to Guangxi Medical University from January 2012 to December 2018 were retrospectively reviewed and follow-up was conducted in the following years, and the last follow-up date was April 2019. Patient information collected included gender, age, SBRT treatment time, discharge time, reexamination time, tumor marker examination results, and 1–5-year follow-up results. The inclusion criteria for the retrospective study were as follows: (1) First SBRT treatment without previous treatment history; (2) No other combined treatment such as surgery, intervention, chemotherapy, etc. The exclusion criteria were: (1) Patients with previous treatment history; (2) Patients with other malignant tumors. In the prospective study, we collected blood samples from HCC patients who received SBRT from December 2017 to December 2018. Three blood samples for each patient were collected, that is,, before SBRT, before discharge and 2 months after SBRT. In addition, the inclusion criteria for the prospective study were as follows: (1) Size less than 10 cm, with Child-Pugh score A or B liver function; (2) First SBRT treatment without previous treatment history, 80 Gy < BED <100 Gy (BED: Biologically Effective Dose); (3) No other comprehensive treatment such as chemotherapy that seriously affects the blood index. The exclusion criteria were: (1) Lost to follow-up and failed to complete three blood collections as scheduled; (2) Received other treatments within two months of SBRT. Patients were all informed of the study and signed a written informed consent form. All patients provided informed written consent and all research and related activities involving human subjects were approved by the Ethics Committee of the first and second hospital affiliated to Jilin University and performed in accordance with guidelines and regulations and the Declaration of Helsinki. Trial registration: ResMan, number: ChiCTR1800015499. Registered 20 April 2018, www.medresman.org.
Specimen Collection and High-Throughput Sequencing
A total of six blood samples from two HCC patients were collected for RNA-sequencing. Blood samples were collected before SBRT (first sample), before discharge (second sample) and 2 months after SBRT (third sample). In this study, PAXgene Blood RNA Tubes (PreAnalytiX, Qiagen BD, Valencia, CA) (abbreviated as BRT) were used to collect blood samples for quick protection of the RNA from degradation. PAXgene Blood RNA Kit (PreAnalytiX, Qiagen BD, Valencia, CA) was used for subsequent experimental studies. The whole blood samples (5 mL) were transported to Novogene (Novogene, Beijing) for RNA isolation, quality control, library preparation, and sequencing.
Quantification of mRNAs by RT-qPCR Analysis
A total of 45 blood samples from 15 HCC patients were collected for RT-qPCR. Blood sample collection and RNA extraction were all carried as previously described. For the reverse transcription reaction, the RT reaction solution was prepared on ice according to the following components: 2 μL of 5×PrimeScript RT Master Mix (Perfect Real Time), 500 ng of total RNA, followed by RNase Free dH2O up to 10 μL. The reaction mixtures were incubated at 37°C for 15 min, followed by 85°C for 5 sec and saved at 4°C. qRT-PCR was performed using the ABI StepOnePlusTM Real-Time PCR System (Applied Biosystems, Foster City, CA).
All primer set were designed and synthesized by Wcgene Biotechnology Corporation (Shanghai, China). The sense and antisense primers of ADIPOR1 are: TCCTGCCAGTAACAGGGAAG and GGTTGGCGATTACCCGTTTG; the sense and antisense primers of EPB42 are: ACTTGTTGAA CCAGAATGGT CTC and TCCACTTCTC TACCTGCTTG TC; and so on. GAPDH (forward primers: CAATGACCCC TTCATTGACC and reverse primers: GACAAGCTTC CCGTTCTCAG) was used as the reference control. We used the TB GreenTM Premix Ex TaqTM II (Tli RNaseH Plus) (Takara, Code No. RR820 A) as the QPCR test kit following the manufacturer’s recommendations. The 2−ΔΔCt method was performed to calculate the relative levels of mRNAs.
Statistical Analysis
Mann–Whitney U test and Student’s
Results
Patient Specimens
In the retrospective study, we collected a total of 589 medical records of primary liver cancer patients admitted to hospital for SBRT between January 2012 and December 2018. After excluding 55 patients who were lost for follow-up, a total of 534 patients were involved in the following research. In the prospective study, 51 blood samples from 17 patients were collected, the outcome after SBRT were followed up and recorded, the relationship between markers and prognosis, the ROC and assessment of prognostic value were analyzed.
Prognostic Analysis of Commonly Used Serum Tumor Biomarkers
Analysis of the difference in the expression of tumor markers in patients before treatment, 3 months after treatment and 6 months after treatment.
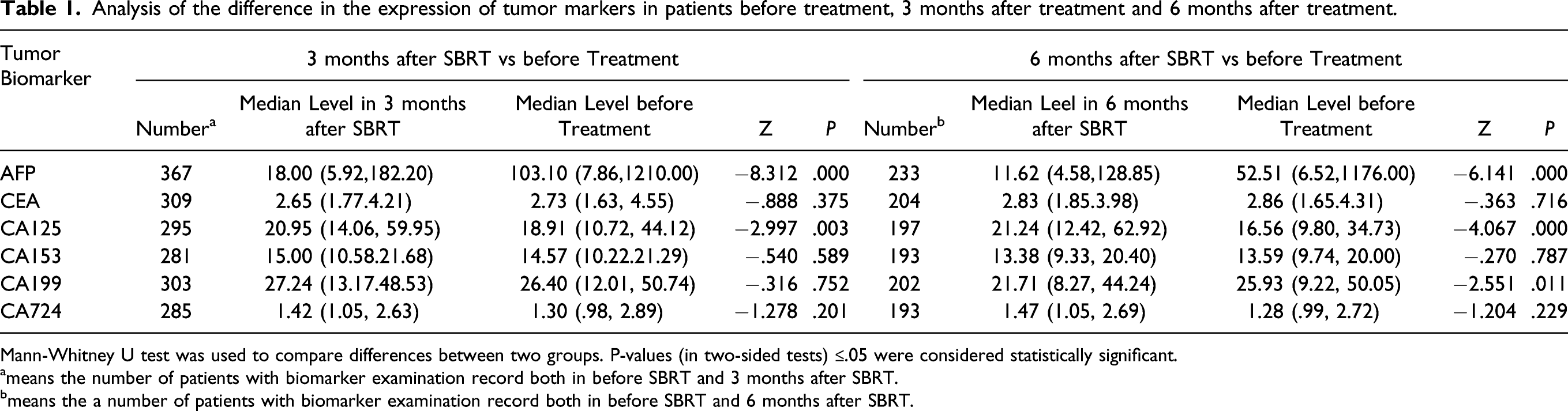
Mann-Whitney U test was used to compare differences between two groups. P-values (in two-sided tests) ≤.05 were considered statistically significant.
ameans the number of patients with biomarker examination record both in before SBRT and 3 months after SBRT.
bmeans the a number of patients with biomarker examination record both in before SBRT and 6 months after SBRT.
Analysis of the difference in the proportion of tumor markers in the survival group and the death group.

Evaluation of predictive efficacy in distinguishing survival group and death group of the tumor markers.

AUC: Area under receiver-operating characteristic (ROC) curve. Medcalc Software, Version 11.5.0.0 (MedCalc Software, Mariakerke, Belgium) Was Used to Calculate AUC and Cut-Off.
High-Throughput Sequencing for Gene Profiles
By the high-throughput sequencing, we obtained gene expression profiles before and after SBRT, the visualization of the heatmap of the HCC samples are shown in Supplementary Figure 1. Then, we compared the changes in gene expression before discharge (group 2) with pre-treatment (group 1), 2 months after SBRT treatment (group 3) with pre-treatment (group 1), and also group 3 vs group 2. Genes with
Evaluation of the Differential Genes for SBRT Efficacy in HCC
Gene expression levels before and after SBRT treatment.

Mann-Whitney U test was used to compare differences between 2 groups.
Change ratio of differentially expressed genes in the effective and ineffective groups of liver cancer at 3 months after SBRT.

Mann-Whitney U test was used to compare differences between 2 groups.
The evaluation results of the prognostic value of the change ratio of ADIPOR1 and EPB42 (2vs1) for liver cancer patients in 3 months after SBRT.

*2vs1, mean a pre-discharge vs before SBRT
Function Analysis Based on TCGA
In order to further explore the biological functions of the potential prognostic marker ADIPOR1, we used the TCGA database to perform further biometric analysis. By using 371 tumor tissues and 50 paracarinoma tissues in liver hepatocellular carcinoma (LIHC), we found that ADIPOR1 was significant higher expressed in tumor tissues (shown in Supplementary Figure 5(A)). Further, by using Gene Set Enrichment Analysis (GSEA), we found that high expression of ADIPOR1 is mainly related to the following pathways: mismatch repair, Circadian rhythm, protein processing in endoplasmic reticulum, DNA replication, and Fanconi anemia pathway (top 5), the GSEA plot were shown in Supplementary Figure 5(B).
Discussion
Tumor biomarkers, such as AFP, CEA, CA199, CA153, and CA125, are commonly used for estimation of the disease progression. By detecting the levels of these biomarkers, clinician can make a preliminary assessment of the occurrence and progress of tumors. Jung J et al 26 reported that liver cancer patients had a better prognosis when AFP levels returned to normal levels 3 months after SBRT, Uemotok et al 27 found that the risk of recurrence was associated with the elevated AFP level. Whether those biomarkers are with prognostic value is our concern. Previous study reported that high AFP were associated with worse survival for liver patients who receiving Cyberknife treatment, 28 however, all patients they included are in advanced or terminal stage of HCC. In our study, we evaluated the prognostic values of those existing tumor biomarkers, and found that those widely used tumor biomarkers such as AFP, CEA, and CA199. could not precisely predict the outcome of liver cancer patients treated by SBRT due to insufficient sensitivity or specificity, for the Yoden index was less than .5, indicating that these commonly used biomarkers do not provide a good assessment of the therapeutic efficacy of SBRT. Actually, although AFP is a well-known biomarker, the use of AFP as a screening indicator for HCC has been canceled by the 2010 American Association for the Study of Liver Diseases guidelines due to its low sensitivity. 29 Therefore, it is necessary to find more effective biomarkers for accurate assessment of the efficacy of SBRT in patients with liver cancer. To this end, we used blood samples from liver cancer patients before and after radiotherapy to discover new and effective biomarkers.
In our research, we explored potential biomarkers in mRNA level for the immediate-early assessment of prognosis after SBRT in patients with primary liver cancer through high-throughput sequencing and PCR. By using strict screening criteria, we finally got 28 differentially expressed mRNAs that were commonly increased or decreased after SBRT. Of all the 28 mRNAs, we found that the change ratio of ADIPOR1 (
Previous studies revealed that the expression of ADIPOR1 was significantly lower in liver cancer than non-neoplastic hepatic tissues, 30 suggested an inverse relation of ADIPOR1 to malignancy. In our study, ADIPOR1 is elevated after SBRT and had a good sensitivity and specificity in assessing the efficacy of SBRT after 3 months. Similarly, EPB42 was also found to be a good prognostic biomarker for liver cancer patients treated by SBRT. Furthermore, when the change of ADIPOR1 was lower than .5838 in pre-discharge compared with that before SBRT, patients had a better prognosis. And the sensitivity is 100%. That is to say, by detecting the changes of ADIPOR1 before discharge compared with those before SBRT, the efficacy of the patients three months after SBRT could be effectively predicted. In addition, routine blood collection before treatment and before discharge, did not increase burden for patients, it is convenient to monitor the changes of ADIPOR1 and help to estimate whether the patient has a good prognosis, whether the patient needs further radiotherapy, and consequently providing a reliable reference for the determination of the overall clinical treatment plan.
In addition, there are only two studies on ionizing radiation (IR) and ADIPOR1 response, one is on IR and mouse intestine, suggesting that ADIPOR1 in mouse colon tissue has a significant decrease 12 months after 2 Gy irradiation 31 ; one is a recent report by our research group, we found that ADIPOR1 decreased significantly in hepatoma cells after 15 Gy irradiation, and knocking down ADIPOR1 could enhance radiation sensitivity. 32 These results suggested that ADIPOR1 was related to the progression of liver cancer, and IR could cause significant changes in ADIPOR1. Targeting ADIPOR1, combined with IR, may improve the efficacy of radiotherapy in patients with liver cancer.
There are still some limitations in this study. First, although ADIPOR1 shown promising prognostic value for SBRT, it still needs more samples and further validation; Secondly, how does ionizing radiation cause changes in ADIPOR1 and then affects the prognosis of patients with liver cancer, and what’s the underlying mechanism, remain to be studied. In addition, the GSEA showed that high expression of ADIPOR1 was mainly related to Mismatch repair, Circadian rhythm, Protein processing in endoplasmic reticulum, DNA replication and Fanconi anemia pathways, perhaps those results may provide some theoretical basis for future mechanism research.
Footnotes
Author Contributions
Zhenzhen Liang: Data analysis, original draft.
Chang Xue and Guanghui Li: Data analysis.
Qing Chen, Mengke Li: Data cleaning.
Hao Feng and Yi Liu: Data collection.
Xiaodong Liu and Shumei Ma: Conceptualization, review & editing. Information on the authors responsible for statistical analysis
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81972969, 81872558 and 81773363).
Ethical Approval and Consent to Participate
The study design was approved by the Ethics Committee of the certain hospitals.
Consent for Publication
The authors agree to publication.
Availability of Data and Material
The data used and analyzed in this study are available from the corresponding author upon reasonable request.
