Abstract
New research data for low-dose, low- linear energy transfer (LET) radiation-induced, stochastic effects (mutations and neoplastic transformations) are modeled using the recently published NEOTRANS3 model. The model incorporates a protective, stochastic threshold (StoThresh) at low doses for activating cooperative protective processes considered to include presumptive p53-dependent, high-fidelity repair of nuclear DNA damage in competition with presumptive p53-dependent apoptosis and a novel presumptive p53-independent protective apoptosis mediated (PAM) process which selectively removes genomically compromised cells (mutants, neoplastic transformants, micronucleated cells, etc.). The protective StoThresh are considered to fall in a relatively narrow low-dose zone (Transition Zone A). Below Transition Zone A is the ultra-low-dose region where it is assumed that only low-fidelity DNA repair is activated along with presumably apoptosis. For this zone there is evidence for an increase in mutations with increases in dose. Just above Transition Zone A, a Zone of Maximal Protection (suppression of stochastic effects) arises and is attributed to maximal cooperation of high-fidelity, DNA repair/apoptosis and the PAM process. The width of the Zone of Maximal Protection depends on low-LET radiation dose rate and appears to depend on photon radiation energy. Just above the Zone of Maximal Protection is Transition Zone B, where deleterious StoThresh for preventing the PAM process fall. Just above Transition Zone B is a zone of moderate doses where complete inhibition of the PAM process appears to occur. However, for both Transition Zone B and the zone of complete inhibition of the PAM process, high-fidelity DNA repair/apoptosis are presumed to still operate. The indicated protective and deleterious StoThresh lead to nonlinear, hormetic-type dose-response relationships for low-LET radiation-induced mutations, neoplastic transformation and, presumably, also for cancer.
INTRODUCTION
The risk of induced stochastic biological effects in humans (e.g., mutations, neoplastic transformations, and cancers) after low doses of ionizing radiation of any type is generally assessed based on the linear-no-threshold (LNT) model whereby risk of harm increases linearly without a threshold (NRC 2005). The LNT model is used in establishing radiation protection guidelines for nuclear workers and the general public, and is often applied in epidemiological studies of radiation-induced cancer. In 2001, the National Council on Radiation Protection and Measurements published Report 136 entitled “Evaluation of the Linear-Nonthreshold Dose-Response Model for Ionizing Radiation” (NCRP 2001). A major but controversial conclusion expressed in the report was as follows:
In keeping with previous reviews by the NCRP …, the Council concludes that there is no conclusive evidence on which to reject the assumption of a linear-nonthreshold dose-response relationship for many of the risks attributable to low-level ionizing radiation although additional data are needed.… However, while many, but not all, scientific data support this assumption …, the probability of effects at very low doses such as are received from natural background … is so small that it may never be possible to prove or disprove the validity of the linear-nonthreshold assumption.
I have omitted reference citations from the above quotation where ellipses appear. The above quote is a reflection of what I have called the low-dose extrapolation fallacy associated with the LNT model (Scott 2005a). Some radiation experts were quite surprised that such a conclusion could be made in light of the growing body of evidence against the general validity of the LNT model (Higson 2004; Tubiana et al. 2005). Evidence against the general validity of the LNT model has also been discussed in many publications (Ootsuymaya and Tanooka 1993; Howe 1995; Azzam et al. 1996, 2004; Rossi and Zaider 1997; Yamamato et al. 1998; Kondo 1999; Tanooka 2000; Yamamato and Seyama 2000; Redpath et al. 2001, 2003; Calabrese and Baldwin 2003a, 2003b; Mitchel et al. 2003; Scott et al. 2003, 2004; Chen et al. 2004; Feinendegen et al. 2004; Higson 2004; Ko et al. 2004; Scott 2004, 2005a, 2005b; Aurengo et al. 2005; Feinendegen 2005).
Don Higson in his recent article, “The Bell Tolls for LNT,” wrote the following (Higson 2004):
The linear no-threshold (LNT) model has been a convenient tool in the practice of radiation protection, but it is not supported by scientific data at doses less than about 100 millisievert or at chronic dose rates up to at least 200 millisievert per year. Radiation protection practices based on the LNT model yield no demonstrable benefits to health when applied at lower annual doses. The assumption that such exposures are harmful may not even be conservative and has helped to foster an unwarranted fear of low-level radiation.
The recent report by the Academy of Sciences, National Academy of Medicine (French) (Tubiana et al. 2005), written after an extensive review of available literature on radiation-induced stochastic biological effects, stated the following:
In conclusion, this report raises doubts on the validity of using LNT for evaluating the carcinogenic risk of low doses (<100 mSv) and even more for very low doses (<10 mSv). The LNT concept can be a useful pragmatic tool for assessing rules in radioprotection for doses above 10 mSv; however since it is not based on biological concepts of our current knowledge, it should not be used without precaution for assessing by extrapolation the risks associated with low and even more so, with very low doses (<10 mSv) …
The French report also states the following:
Decision makers confronted with problems of radioactive waste or risk of contamination, should re-examine the methodology used for evaluation of risks associated with very low doses and with doses delivered at a very low dose rate. The report confirms the inappropriateness of the collective dose concept to evaluate population irradiation risks.
In the present article I present support for each of the following assertions:
Stochastic thresholds (StoThresh (Scott et al. 2004)) can cause hormetic-type dose-response relationships for radiation-induced mutations, neoplastic transformation, and, likely, also cancer.
At doses on the order of 1 mSv of low-LET radiation, neoplastic transformation and cancer risks are orders of magnitude more likely to decrease than to increase by an amount as would be expected based on the LNT model.
Low doses of low-LET radiation may prevent cancer occurrence and extend life expectancy for adults that already have precancerous cells (e.g., heavy, long-time smokers).
Low doses of low-LET radiation, when used in conjunction with apoptosis-sensitizing agents, could possibly cure cancer.
Background low-LET radiation may be suppressing cancer occurrence and other mutations-related diseases among the human population.
RADIATION-RESEARCH RELATED CONCEPTS
Radiation Absorption in Tissue or Cell Cultures
X-rays, gamma rays, and beta radiation are examples of low-LET radiation. Neutrons, alpha particles, and heavy ions (which are encountered in space travel) are examples of high-LET radiation. High-LET radiation, such as alpha particles, does not penetrate very far in matter. For example, alpha particles can be stopped by a piece of paper. However, low-LET gamma rays can penetrate the entire body of a human. Low-LET beta particles have an intermediate penetration between that for alpha particles and gamma rays. Low-LET X-rays and gamma rays have similar characteristics. Low-LET radiation is therefore more penetrating than high-LET radiation.
The absorption of ionizing radiation in biological tissue or cell cultures involves stochastic interactions with constituent atoms and molecules, and generates energy deposition (track) events accompanied by bursts of reactive oxygen species (ROS) (Feinendegen 2005). Induced radiogenic DNA damage increases as radiation dose increases. The ROS are similar to those that arise constantly by normal oxidative metabolism. Endogenous ROS alone induce about a million DNA oxyadducts per cell per day compared to 5 × 10−3 total DNA-damaging events per average cell per day from background radiation exposure at 1 mGy over a year (Feinendegen 2005).
In the case of penetrating low-LET radiation, particle tracks arise stochastically throughout the exposed tissue with relatively low density at low doses. For low doses of high-LET radiation, the distribution of ionized molecules and of ROS bursts is more heterogeneous (Feinendegen 2005). Intercellular communication after radiation damage probably depends on the spatial distribution of the hit cells as well as the cellular environment. Biological response to irradiation of humans is known to depend on the spatial distribution of the radiation hits in the body, the total radiation dose, and how rapidly the dose is delivered (i.e., dose-rate history).
Stochastic Radiobiological Effects
For ionizing radiation doses in the range 0 – 100 mGy, biological effects of interest include induced genomic instability, mutations, neoplastic transformation, and cancer (Scott 2004). These effects, along with genetic effects, are called stochastic, since their occurrence is governed by probabilistic considerations.
Three forms of genomic instability have been distinguished in the context of modeling low-dose radiation-induced stochastic effects (Scott 2004; Scott et al. 2004): normal minor instability (NMI), transient problematic instability (TPI), and persistent problematic instability (PPI). The transient instability can be eliminated via cooperative repair processes (e.g. high-fidelity, p53-dependent DNA repair/apoptosis). However, misrepair of damage can lead to PPI (mutant cells) which can be passed to cell progeny increasing their chances for undergoing spontaneous neoplastic transformation. These forms of instability are features of the NEOTRANS3 model discussed later.
Protective and Deleterious Bystander Effects
Radiation hits in cells may cause non-hit neighboring cells to become affected by signaling substrates from hit cells (bystander effect) (Azzam et al. 2004). The bystander effect can either be deleterious (enhancing the net biological damage) or protective (suppressing the net biological damage). Protective effects appear to predominate over the dose range of 1 – 100 mGy after brief exposure at a high rate to low-LET photon radiation. Three cooperative processes are now considered to contribute to protection in the indicated dose zone: activated presumptive p53-dependent high-fidelity DNA repair in competition with presumptive p53-dependent apoptosis, and the previously introduced novel presumptive p53-independent protective apoptosis mediated (PAM) process. The PAM process (a bystander effect) is thought to be mediated via ROS and cytokines such as transforming growth factor β, and selectively removes genomically compromised cells including mutants and neoplastically transformed cells (Bauer 1995, 1996; Scott 2004, 2005a, 2005b). The PAM process was first demonstrated experimentally by German researchers (Jürgensmeier et al. 1994; Bauer 1995, 1996, 2000; Langer et al. 1996; Hipp and Bauer 1997).
Deleterious bystander effects may predominate over the ultra-low-dose range of 0 – 0.01 mGy so far as inducing inversion mutation in mice (Hooker et al. 2004). This implicates an ultra-low-dose zone of activated low-fidelity DNA repair. The indicated inversion mutations results implicate a threshold on the order of 0.01 mGy for activating the cooperative protective processes. At doses from 250 – 1000 mGy and higher delivered at a high rate, there is no evidence for the PAM process (Scott 2004). It is either inhibited or is just not activated in the indicated dose range. Here the phrase “inhibition of the PAM process” is used in a very general way to simply imply that it does not occur since it is not clear whether a true inhibition is invoked.
When the dose rate is low and exposure time is spread over a large time interval, cooperative protective processes may occur over a much wider dose range, being continually activated (or reactivated) during the protracted exposure (Scott 2004, 2005b). In such cases the zone of suppression of stochastic effects may range from very low doses up to more than 400 mGy (Scott 2004, 2005b). Further, the magnitude of suppression may significantly increase after such protracted exposures (Scott 2004).
Adaptive Response
The classical two-dose, adaptive-response study involves exposing cell cultures or animals to a very small low-LET radiation dose and after a few hours or longer irradiating them with a much larger dose that is expected to produce easily measurable enhanced biological effects (Wolff 1998). For the comparison group, only the higher dose is given. The hypothesis is that the adapting low dose will protect the cells from damage from the subsequent high dose (e.g., by inducing enhanced repair capacity). Thus, if the frequency of biological effect (e.g., mutations, neoplastic transformations) is lower in the group that received both the adapting and larger test dose than for the test dose alone, then one concludes that the small dose caused the cells to adapt so that they were less affected by the subsequent high dose.
Azzam et al. (1996) and Redpath et al. (2001) introduced a novel experimental single-dose protocol whereby only the small adapting dose was administered. The yield of biological effect (neoplastic transformation
I have previously applied the NEOTRANS3 model (Scott 2004) to neoplastic transformation data of Redpath et al. (2001). Here the model is applied to mutation data of Hooker et al. (2004) for inversion mutation in spleen of pKZ1 mice exposed to 250 kVp X-rays and also to data for 28 kVp X-ray- (Ko et al. 2004) and 60 kVp X-ray- (Redpath et al. 2003) induced neoplastic transformation
The NEOTRANS3 model and how it can be used to clarify issues related to the shape of the dose-response curves for specific stochastic effects at very low doses is discussed in the section that follows.
NONLINEAR DOSE-RESPONSE RELATIONSHIPS
Deterministic Threshold and Nonlinear Dose-Response Curves
The risk for specific radiation-induced deterministic effects (e.g., death from injury to the hematopoietic system) is known to be a nonlinear function of dose with an associated threshold (i.e., minimal tolerance dose for a population). Dose-response curves generally have a sigmoidal shape.
Stochastic Thresholds and Nonlinear Dose-Response Curves
Many stressor-induced biological processes likely have dose thresholds. It is a common view that DNA repair activation requires a damage threshold (Tubiana et al. 2005) which implicates a StoThresh. Because different individuals respond differently to a given amount of radiation, the indicated StoThresh likely varies for different individuals having a characteristic distribution that may depend on other covariates (e.g., age, gender, genetic background). Examples of molecular processes suspected of having StoThresh are oncogene activation, suppressor gene inactivation, stress protein induction, and induced activated p53-dependent high-fidelity DNA repair/apoptosis.
For replicate
The recently published NEOTRANS3 model incorporates StoThresh for radiation-induced stochastic effects such as mutations and neoplastic transformation (an early step in cancer induction) (Scott 2004). The model has been used to characterize nonlinear (or linear) dose-response relationships for mutation or neoplastic transformation induction by low doses of ionizing radiation (Scott 2004). With the model, nonlinearity arises from protective and deleterious StoThresh. StoThresh distributions associated with specific radiation-induced stochastic effects (mutation and neoplastic transformation induction) are estimated in this paper.
NEOTRANS3 Model and Stochastic Thresholds
The NEOTRANS3 model is presented in Figure 1 and applies to low doses and to a small number of hypersensitive cells among a much larger number of resistant cells. The model features radiation-induced transitions between instability states NMI, TPI, and PPI. Doses above a threshold activate presumed p53-dependent, high-fidelity DNA repair/apoptosis and the presumed p53-independent PAM process. Features in Figure 1 relate to dose ranges over which the indicated protective processes are activated. When activated the PAM process selectively removes genomically compromised cells (e.g. mutants and neoplastically transformed cells). In the present applications of the NEOTRANS3 model, after ultra-low doses of low-LET radiation, high-fidelity repair/apoptosis is presumed not to be activated. Rather low-fidelity DNA repair/apoptosis is presumed activated. This facilitates characterizing the dose-response curve presented later for X-ray induced recombination mutations in mice.

Modified NEOTRANS3 model for low-dose-induced stochastic effects. See main text for more detail.
The parameter α
Typical units for α
With the NEOTRANS3 model (as with its predecessor NEOTRANS2, model) the p53-independent PAM process (Jürgensmeier et al. 1994; Hipp and Bauer 1997) and p53-dependent high-fidelity DNA repair/apoptosis, when jointly activated, work together in guarding against propagation of genomic instability and subsequent stochastic effects. The p53-dependent apoptosis removes damaged cells before mutations and neoplastic transformations arise and competes with error-free repair and misrepair pathways. When error-free repair occurs it eliminates DNA damage but not damaged cells. The PAM process (considered p53-independent) removes mutants and neoplastic transformants, as well as other aberrant cells that are present. Thus, the indicated team of biological protectors when jointly activated provides powerful protection against adverse stochastic effects of exposure to genotoxic agents.
After moderate doses, protection is also presumed to be provided by high-fidelity p53-dependent DNA repair/apoptosis. Such protection could be revealed via inhibition of these protective processes, in which case the frequency of stochastic effects such as mutations and neoplastic transformation is expected to significantly increase.
Yield of Biological Effects after Low-Dose Irradiation
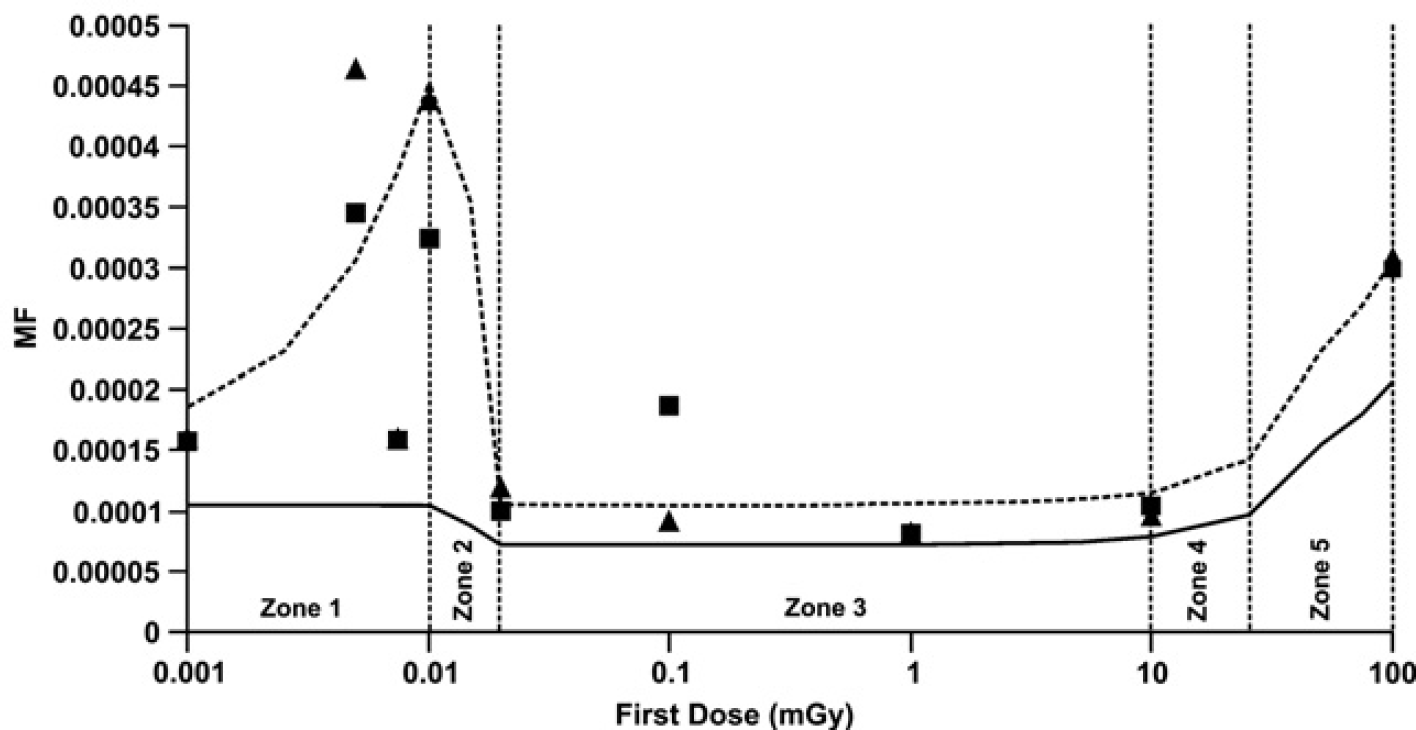
For brief exposure to small or moderate doses,
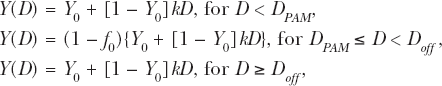
where the slope parameter

Note that for
The parameter
The parameters
The parameter ∼1 accounts for both unrepaired and misrepaired damage. Here, the fundamental model parameters discussed are allowed to change values over different dose zones. This is achieved via allowing the model parameter
For mutations induction, Ω represents the probability for the occurrence of mutation of interest among those induced by irradiation. For neoplastic transformation, Ω represents the probability that a cell with radiation-induced PPI (mutant cells) will produce transformed progeny during a specified follow-up time.
With the current NEOTRANS3 model, induced enhanced p53-dependent high-fidelity DNA repair/apoptosis can reduce the slope parameter
Removal of precancerous cells via the PAM process would be expected to have the same impact on the cancer induction dose-response curve. Thus, for cancer induction, risk could decrease after low doses of low-LET radiation. A similar reduction may also occur after exposure to low doses of other toxic agents that activate the PAM process. Immune system stimulation may also contribute to the reduction.
Therefore, for protracted exposure of humans to low-LET radiation at very low rates over a prolonged period (e.g., years), the cancer incidence may be greatly reduced as has been reported (Chen et al. 2004). The title of the cited paper by Chen
Specifications of Dose Zones
To facilitate modeling of data for low-LET radiation-induced stochastic effects (mutations, neoplastic transformation), five dose zones are considered, as indicated below, with respect to brief high-rate or protracted low-rate exposure to low-LET radiation (e.g., X-rays, gamma rays).
Zone 1 (Ultra Low-Doses Zone)
After ultra low doses (close to daily natural background radiation), it is assumed that high-fidelity DNA repair is not activated. Rather low-fidelity, error-prone repair is presumed to occur in cooperation with apoptosis. This is based on inversion mutation data (Hooker et al. 2004) for brief exposure at a high rate to 250 kVp X-rays from doses near daily background up to 0.01 Gy. These data are presented later.
Zone 2 (Transition Zone A)
This is a relatively narrow low-dose zone just above Zone 1 in which StoThresh for activating the cooperative protective processes (p53-dependent high-fidelity DNA repair/apoptosis and the PAM process) are considered to occur. The indicated protective StoThresh is represented by the notation
Zone 3 (Zone of Maximal Protection)
This dose zone is just above Zone 2 and is the zone where maximal protection against mutations and neoplastic transformation is afforded. For irradiation of animals or humans, each irradiated member has the cooperative protective processes activated. For
Zone 4 (Transition Zone B)
This dose zone is just above Zone 3 and is the zone where the StoThresh for inhibiting the PAM process occur. The indicated deleterious StoThresh is represented by the notation
Zone 5 (LNT Zone)
This dose zone is just above Zone 4 and is the zone where the PAM process is maximally inhibited. For irradiation of animals or humans, each individual does not have the PAM process activated. However, high-fidelity DNA repair/apoptosis is presumed to be activated in this zone as well as in Zone 4. For doses in this zone, the LNT model is expected to adequately represent the frequency of stochastic effects.
Definitions of higher dose zones may also prove beneficial when studying high-dose effects. However, the focus here is on low to moderate doses. For the NEOTRANS3 model applications to mutation induction and neoplastic transformation that follow, the slope parameter
The mutation data presented later span all five dose zones. The neoplastic transformation data presented later span Zones 3 – 5 but provide no information on the shape of the dose-response curve over Zones 1 and 2.
Impact of StoThresh on the Shape of the Dose-Response Curve for Mutation Induction
Figure 2 shows results of fitting the NEOTRANS3 model to data of Hooker et al. (2004) for 250 kVp X-ray-induced inversion mutations in spleen of irradiated pKZ1 mice. The model has been applied using previously described Bayesian methods implemented via Markov Chain Monte Carlo to facilitate working with StoThresh (Scott 2004; Scott et al. 2004). The steep initial rise in the fitted curve in Figure 2 is due to the slope parameter

Application of the NEOTRANS3 model to data of Hooker et al. (2004) for 250 kVp X-ray-induced inversion mutations in the spleens of irradiated mice. Dose zones are explained in the text.
The protective StoThresh,
For doses in the range 50 – 100 mGy (portion of LNT zone; no PAM process), the LNT model applies and has a slope given by
Impact of StoThresh on the Shape of the Dose-Response Curve for Neoplastic Transformation
While the inversion mutation frequency data in Figure 2 span dose Zones 1 – 5, there are no data for neoplastic transformations with doses below 0.1 mGy except for control animals. Thus, it is not known if a similar curve shape below 0.1 mGy, as shown in Figure 2, also applies to low-LET radiation-induced neoplastic transformation. Previously the NEOTRANS3 model was applied to data of Redpath et al. (2001) for gamma-ray induced neoplastic transformation of HeLa × skin fibroblast human hybrid cells exposed
Figure 3 shows results of applying the NEOTRANS3 model to data from Redpath et al. (2003) for 60 kVp X-ray induced neoplastic transformation of HeLa × skin fibroblast human hybrid cells. Bayesian methods as previously described (Scott 2004) were used to fit the data. Data only span Dose Zones 3 – 5. The shape of the dose-response curve over these zones is essentially the same as for the inversion mutations in Figure 2.
Application of the NEOTRANS3 model to data of Redpath et al. (2003) for 60 kVp X-ray induced neoplastic transformation in HeLa × skin fibroblast human hybrid cells.
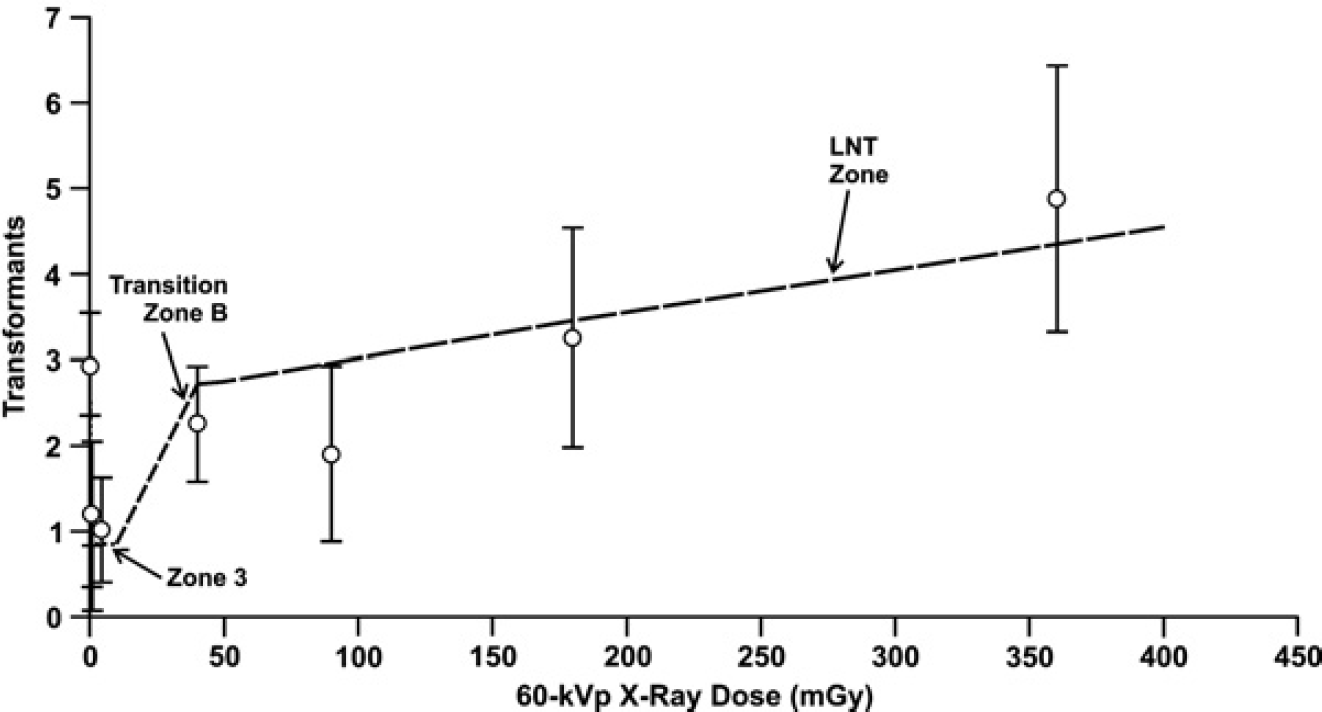
Figure 4 shows similar results for 28 kVp X-ray induced neoplastic transformation of HeLa × skin fibroblast human hybrid cells based on data from Ko et al. (2004). Posterior means and associated standard deviations for NEOTRANS3 model parameters
Bayesian posterior means and standard deviations for NEOTRANS3 model parameters and Transition Zone B boundary estimates

Application of the NEOTRANS3 model to data of Ko et al. (2004) for 28 kVp X-ray-induced neoplastic transformation of HeLa × skin fibroblast human hybrid cell.
Table 2 provides relative efficiencies of these radiation sources for inducing genomic instability (based on
Relative efficiency for different low-LET radiation sources for inducing stochastic effects in HeLa × skin fibroblast hybrid cells

Similarity in Relative Risks for Neoplastic Transformation in Vitro and Cancer Induction in Vivo
Redpath and colleagues (2001) have shown that dose-response relationships for the relative risk (
For gamma-ray induced cancer in humans, and for
Predictions for Protracted Exposure to Low-LET Radiation
During low-dose-rate protracted exposure to low-LET irradiation the PAM process can be repeatedly activated, thereby intensifying and prolonging its protective effect. For low-dose-rate exposure of humans over years, it is expected that the cancer incidence could be greatly suppressed below the level for unirradiated persons. The ratio
Regarding protracted exposure to gamma rays, Chen and colleagues' (2004) recently published a paper “Is Chronic Radiation an Effective Prophylaxis Against Cancer?”, in which they described their study of cancer occurrence among approximately 10,000 residents of 180 apartment buildings in Taiwan that were built with cobalt-60 contaminated steel. Cobalt-60 is a gamma-ray source. The building inhabitants resided there from approximately 9 – 20 years, during which they unknowingly received radiation doses that averaged about 400 mGy (same as 400 mSv for gamma rays) to the total body. The cancer incidence among this population was reported by Chen
The experience of these 10,000 persons suggest that long-term exposure to radiation, at a dose rate of the order of 50 mSv (5 rem) per year, greatly reduces cancer mortality, which is a major cause of death in North America.
Background Radiation Considerations
We are all exposed to background radiation. The average dose from background radiation is about 3 mSv annually. The dose unit mSv is used for mixed high- and low-LET radiations. For low-LET X-rays, gamma rays, and beta particles, 1 mSv = 1 mGy. However, for alpha particles and neutrons, this is not the case. For a 1 mGy alpha or neutron dose, the corresponding dose in mSv is significantly larger than 1 mSv. Here, our focus is on low-LET radiations such as gamma rays and beta radiation. Background doses of low-radiation > 0.01 mGy could activate the PAM process. If so, mutant and neoplastically transformed cells already present in the body could be triggered to undergo apoptosis via the PAM process.
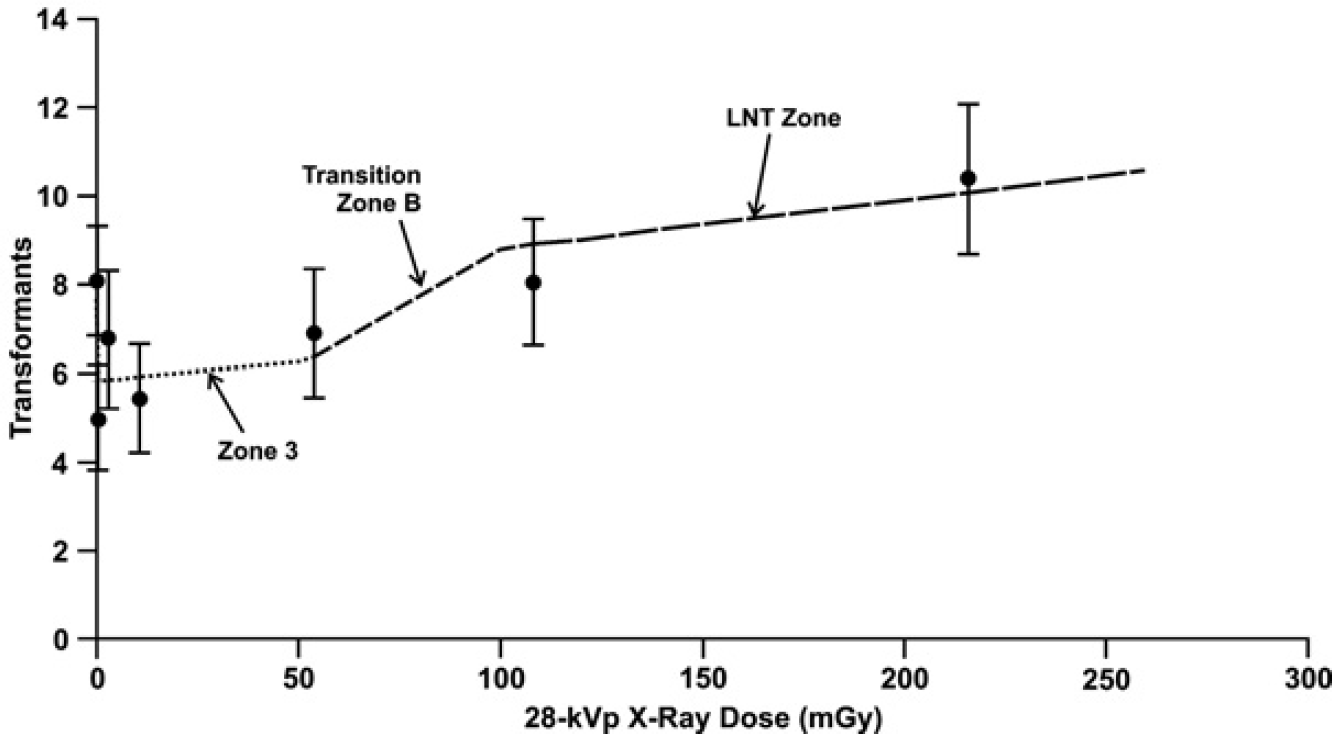
This can be seen from results of applying the NEOTRANS3 model to a two-dose exposure scenario involving 250 kVp X-ray irradiation of pKZ1 mice. Here, the 250 kVp X-rays are considered as representative of a low-LET environmental radiation source (e.g., beta or gamma radiation). For this evaluation, the first dose is any of the doses along the dose axis in Figure 2. The second dose is fixed at 1 mGy and is given 4 hours after the first dose. The second dose (representative of background radiation after having had previous exposure to another radiation source) is expected to activate the PAM process irrespective of whether it was previously activated or inhibited by the first dose. Thus, the frequency for inversion mutations in the indicated two-dose group would be expected to be less than for a group receiving only the first dose. The predicted reduction is indicated in Figure 5, where a second dose-response curve (two-dose study) has been added to the results presented in Figure 2. Thus, subsequent exposure to elevated background low-LET radiation after first exposure to a mutation-inducing dose would be expected to suppress (via activation of the PAM process) the initial rise in the dose-response curve presented in Figure 2. This has been reported for the pKZ1 mouse inversion mutation assay (P. Sykes
A comparison of dose-response curves for a two-dose and single-dose induction of inversion mutations in 250 kVp X-ray exposed pKZ1 mice. The prediction for the two-dose study (lower curve) is based on the NEOTRANS3 model. The single-dose results (upper curve) are the same as for the fitted curve in Figure 2. Doses are in mGy. Dose zones are explained in the text.
The PAM process, if activated by background radiation, should also protect from low-dose high-LET radiation and chemically induced stochastic effects (Scott 2005b). These results have interesting implications for low-dose cancer risk, pointing to a possible major role of background radiation in suppressing cancer occurrence. Background radiation may also suppress other mutation-related diseases.
Cancer Prevention Possibilities
Consider the hypothetical case of a 25-year smoker who based on a computed tomography (CT) scan has precancerous (neoplastically transformed) cells in their lung due to their smoking. Could a little low-LET X- or gamma rays to the lung protect from cancer via activating the PAM process? Possibly, if not likely! Repeated exposures to low doses of low-LET radiation in Dose Zone 3 could possibly amplify the level of protection. Induced immune system stimulation would be expected to add additional protection against cancer. Note that the indicated form of cancer prevention if successful would eliminate the need for surgery (which is currently controversial for small precancerous lesions). Agents other than ionizing radiation may also activate the PAM process and stimulate the immune response (e.g., sunlight, environmental chemicals, workplace chemicals, cosmic rays [low-LET component] received by airline flight attendants, etc).
Low Dose Cancer Therapy Implications
The PAM process also could be exploited related to developing a novel, low-dose therapy for cancer (Scott 2005b). Cancer cells are considered to be resistant to undergoing the PAM process. However, new research may lead to the discovery of apoptosis sensitizing agents (e.g., arsenite in small amounts has this characteristic). If so, then use of such sensitizing agents in combination with low-dose (or low-dose-rate) low-LET radiation could lead to eliminating some cancers.
Cautionary Note
Not everyone may benefit from the PAM process. Persons not bearing significant numbers of genomically compromised cells (e.g., mutation-bearing cells, neoplastically transformed cells, micronucleated cells, etc.) would not be expected to benefit from the PAM process in normal circumstances. Whether the PAM process would remove cells with significant mitochondrial DNA damage is unclear. This is an area where new research may prove fruitful.
CONCLUSIONS
Stochastic thresholds
At doses on the order of 1 mSv of low-LET radiation, neoplastic transformation and cancer risks are orders of magnitude more likely to decrease than to increase by an amount as would be expected based on the LNT model.
Low doses of low-LET radiation may prevent cancer occurrence and extend life expectancy for adults that already have precancerous cells (e.g., heavy, long-time smokers).
Low doses of low-LET radiation, when used in conjunction with apoptosis sensitizing agents, could possibly cure cancer.
Background low-LET radiation may be suppressing cancer occurrence and other mutations-related diseases among the human population.
Footnotes
ACKNOWLEDGEMENTS
This research was supported by the Office of Science (BER), U.S. Department of Energy (DOE) Grants DE-FG02–03ER63671 and DE-FG03ER63657. I am grateful to Ms. Vicki Fisher and Ms. Jennifer Di Palma for editorial assistance and to Ms. Wendy Piper for graphic support. I am also grateful to Dr. Leslie Redpath for his assistance in using published data from his research group and to Dr. Pamela Sykes and Ms. Tanya Day for useful discussions related to their inversion mutation data. The views and conclusions contained herein are those of the author and should not be interpreted as necessarily representing the official policies or endorsement, either expressed or implied, of the DOE or of Lovelace Respiratory Research Institute.
