Abstract
The percentage of cells with chromosome aberrations or micronuclei induced by low doses of acute (dose rate of 47 cGy/min) or chronic (dose rate of 0.01 cGy/min) gamma-irradiation was studied
INTRODUCTION
The linear no-threshold (LNT) hypothesis is the basis for radiation protection standards at low doses. However, questions about the validity of the LNT model remain. Validation of the LNT model is the most important issue related to radiation risk assessment. Studies of cytogenetic effects can provide key information related to the validity of the LNT model. Cytogenetic effects of ionizing radiation have been studied
The aim of the present study was to investigate the shape of the dose-response relationship for specific cytogenetic effects after low doses of low-linear-energy-transfer (LET) ionizing radiation using mammalian and plant cells.
MATERIALS AND METHODS
In our previous studies, we initiated investigations of biological responses to ionizing radiation exposure using the occurrence of chromosomal rearrangements as the endpoint scored (Zaichkina and Ganassi, 1984; Zaichkina et al., 1992, 1995). Based on results of those studies, we have subsequently used different radiation dose ranges for the different bioassays associated with our studies of radiation-induced stochastic effects.
In our present study, Chinese hamster fibroblasts (clone 431) were cultivated at 37°C, in a humidified atmosphere under 5% CO2. Cell culture was grown in Dulbecco's Modified Eagle Medium containing 10% calf serum and antibiotics. The cells were irradiated with acute doses of 0.05–2 Gy of 60Co gamma-rays at a dose rate of 28.2 Gy/h. The chronic gamma-irradiation was carried out for 1, 2, 3, 4, 5, and 6 days at room temperature at a dose rate of 0.0061 Gy/h, with cumulative doses of 0.14, 0.28, 0.42, 0.56, and 0.84 Gy, respectively. The technique for preparation of samples for the micronucleus test has been previously described (Zaichkina and Ganassi, 1984).
When the cells were fixed immediately after chronic irradiation, no damage was observed. To detect cytogenetic damage, the irradiated and control cells kept at room temperature were stored for 24 h at 37°C before fixation. Several experiments were performed for each radiation dose. About 10,000 cells were analyzed for each radiation dose point.
Whole human blood from ten essentially healthy males was irradiated with acute doses of 0.1–0.5 Gy at a dose rate of 28.2 Gy/h. Other blood samples were also chronically irradiated at a dose rate of 0.0061 Gy/h at a 37°C temperature during 0.5, 1, and 2 days with cumulative doses of 0.07, 0.13, and 0.27 Gy, respectively. Whole blood (0.2 ml) was cultivated by the micromethod (Zaichkina et al., 1997). Cytogenetic analysis was carried out based on 150–200 metaphase cells scored from each blood sample. All structural chromosome-type and chromatid-type changes were recorded. Preliminary experiments showed that the conditions of blood-sample irradiation affected neither the background level of chromosomal damage nor radiosensitivity (Zaichkina et al., 1997).
Air-dried seeds of
Air-dried seeds of
All cytological slides were coded blind and analyzed by the investigators independently. Experiments were repeated at least three times. In the figures, results are presented as means +/− standard error.
RESULTS AND DISCUSSION
The dose dependence of the yield of cytogenetic damage in Chinese hamster cells after acute and chronic gamma-irradiation is presented in Figure 1. The dose-response curve used is discontinuous. At very low doses, the dependence of cytogenetic damage on dose was fitted by a linear regression with a zero intercept. In the dose range of about 0.5–1 Gy, there is a plateau, and above this range the curve again appears linear but with a different slope. There was no statistically significant difference between the results for the low doses of chronic and acute irradiation for the investigated dose range.

Dose-response curve for micronuclei induction in Chinese hamster fibroblasts by acute (28.2 Gy/h) and chronic (0.0061 Gy/h) gamma-irradiation. Results (means ± standard errors) of six experiments with acute and four experiments with chronic exposure are shown.
Similar results were obtained for human lymphocyte culture. The dose dependence of the yield of chromosome aberrations in human lymphocytes induced by acute and chronic gamma-irradiation is presented in Figure 2. It can be seen that the obtained high- and low dose rate data for chromosome aberrations were adequately described by single, discontinuous dose-response relationships with an initial linear increase followed by a plateau. Thus, as was the case with Chinese hamster fibroblasts, for the low-dose region, no influence of dose rate was found.

Dose-response curve for chromosomal aberration induction in human lymphocytes. Whole blood received either acute (28.2 Gy/h) or chronic (0.0061 Gy/h) gamma-irradiation. Each point represents the mean ± standard error obtained from blood lymphocytes from 10 healthy donors. The experiment was repeated at least three times.
The dose-response curve for micronuclei induction in root cells of
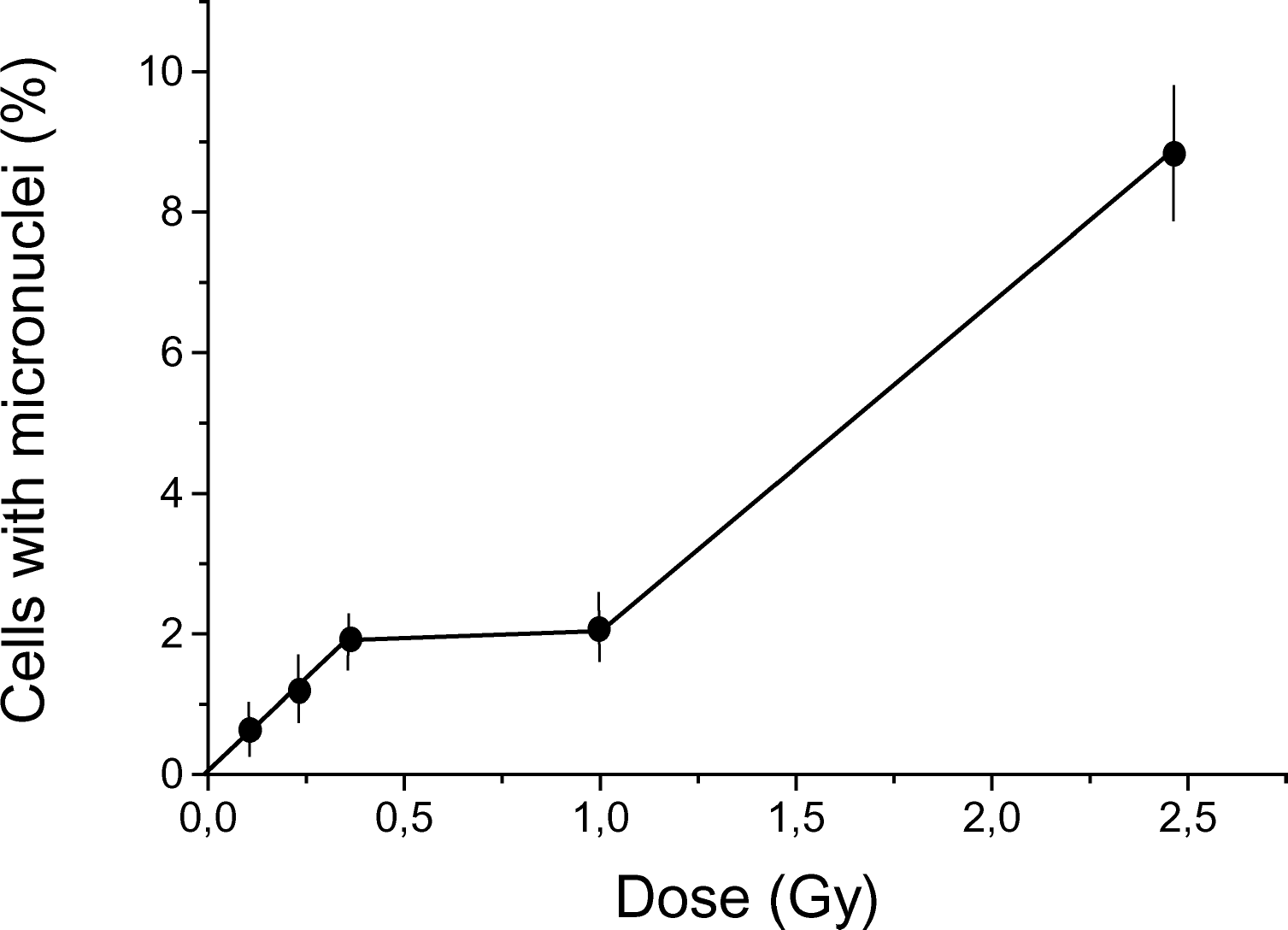
Dose-response curve for micronuclei induction in root cells of
Our data demonstrate that the dose-response curves for micronuclei induction in Chinese hamster fibroblasts, human lymphocytes, and root cells and dry seeds of
We hypothesized that the indicated complex shape of the dose-response curves relates to the status of DNA repair processes. Previously we have shown that caffeine, an inhibitor of DNA repair, increases the number of cells with micronuclei in linear segment 2 (Zaichkina et al., 1992, 1995). To verify the assumption that low doses of gamma-radiation do not induce repair processes, we studied the influence of the DNA repair inhibitor caffeine on the dose dependence of the induction of chromosome aberrations in Chinese hamster fibroblast cultures. Figure 4 shows the dose-response curves for micronuclei induction in fibroblasts irradiated with low doses of gamma-radiation in the presence of caffeine and without caffeine. Caffeine was added to the culture medium (final concentration 0.08 M, pH 6.5–6.8) prior to irradiation. The cells were kept in the medium with caffeine after irradiation up to fixation. There was no modifying effect of caffeine for linear segment 1; however, for the plateau region and for linear segment 2, caffeine caused significant changes in the dose-response curve. More specifically, caffeine treatment led to the disappearance of the plateau (effectively extending linear segment 1).

Dose-response curve for micronuclei induction in Chinese hamster fibroblasts by acute (28.2 Gy/h) gamma-irradiation with and without caffeine treatment. Caffeine was added to culture medium prior to irradiation at a final concentration of 0.08 M, pH 6.5–6.8. Results (means ± standard errors) of six experiments without caffeine and three experiments with caffeine treatment are shown.
Another approach for validating our hypothesis was to investigate the action of low doses of secondary radiation from 70 GeV protons and neutrons (90%) and of chronic monoenergetic neutrons generated by the Serpukhov charged particle accelerator on cytogenetic damage in mammalian cells. Using inhibitors of DNA repair we have shown that high doses of gamma-radiation induced repair, in contrast to neutrons which inhibit repair of cytogenetic damage (Zaichkina et al., 1995). It was found that, in contrast to the gamma-irradiation, the dose-response curves after exposure to high-LET neutrons for all the cytogenetic endpoints were linear; that is, the plateau was absent (Rozanova et al., 2000). These results suggest that the induction by gamma-rays of DNA repair processes occurs in the dose range corresponding to the plateau. Thus, it can be assumed that the induction of repair by low-LET radiation occurs only after a threshold level of cytogenetic damage and that the increased yield of cytogenetic damage per unit dose at low radiation doses is attributable to an insignificant contribution or the absence of DNA repair. Recently, Rothkamm and Lobrich (2003) have shown a lack of DNA double-strand-break repair in primary human fibroblasts exposed to very low doses (approximately 1 mGy) of ionizing radiation.
Figure 5 shows the dose-response curves for micronuclei induction in the culture of Chinese hamster fibroblasts irradiated in the presence and without b-mercaptoethylenamine (MEA), a well-known radioprotector. MEA was added (final concentration 0.1 mM) to the culture medium 15 min before irradiation and the cells were kept in the medium with MEA for 2 h after irradiation. As with caffeine, no modifying effect of MEA in linear segment 1 was observed but the plateau region was extended from just above 1 Gy to about 2 Gy. This is direct evidence that a plateau exists and that our interpretation of the experimental results are plausible.
Recently in our study involving irradiated mice, we showed that the
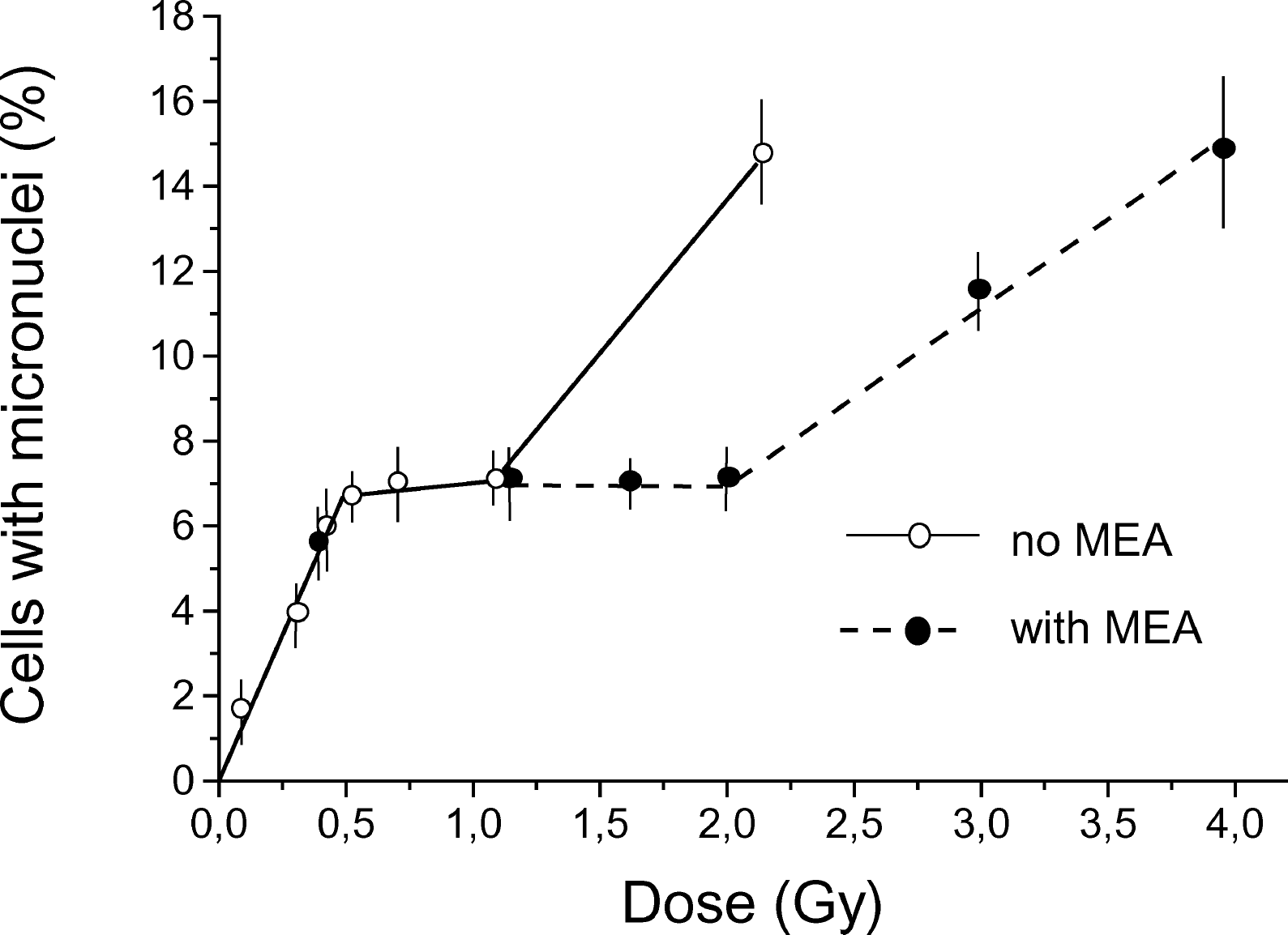
Dose-response curve for micronuclei induction in Chinese hamster fibroblasts by acute (28.2 Gy/h) gamma-irradiation with and without MEA treatment. MEA was added at a final concentration of 0.1 mM to the culture medium 15 min before irradiation and the cells were kept in the medium containing MEA for 2 h after irradiation. Results (means ± standard errors) of six experiments without MEA and three experiments with MEA treatment are shown.
The results we have presented indicate that dose-response curves both in mammalian and plant cells are nonlinear and reflect hypersensitivity of cells to low radiation doses that is not predicted by back-extrapolation of cytogenetic response from higher doses. Although further studies are required to reveal the molecular mechanisms associated with the observed dose-response relationships, our results suggest that the hypersensitivity may be attributed to a lack of induced DNA repair at very low radiation doses. Altogether, our results provide additional evidence that low doses of low-LET radiation could have more significant genetic consequences than would be expected based on extrapolating from high to low doses based on the LNT model.
