Abstract
Objective
During August of each year, thousands of Nepalese religious pilgrims ascend from 2050 m to 4500 m in 1 to days. Our objectives were to evaluate the incidence of acute mountain sickness (AMS) among this large group of native people, to explore changes in serum electrolytes as subjects ascend to high altitude, and to attempt to determine whether decreased effective circulating volume is associated with the development of AMS.
Methods
This was a prospective study with 2 parts. In the first part, demographic, physiologic, and laboratory data were collected from a cohort of 34 pilgrims at both moderate (2050 m) and high altitude (4500 m). Changes that occurred with ascent were compared in subjects who did and did not develop AMS. The second part was a cross sectional study of a different group of 57 pilgrims at the high-altitude site to further determine variables associated with AMS.
Results
In the cohort of 34 subjects, Lake Louise score, heart rate, respiratory rate, blood urea nitrogen (BUN), BUN:creatinine ratio, and pH increased at high altitude, whereas oxygen saturation, bicarbonate, creatinine, and P
Conclusions
Fifty-two percent of the subjects developed AMS. With ascent to altitude, subjects showed some evidence of decreased effective circulating volume, though there were no clinically significant changes. The data did not show whether decreased circulating volume is a significant risk factor in the development of AMS at high altitude.
Introduction
Altitude-related illnesses afflict a large number of people every year. Of these illnesses, acute mountain sickness (AMS) is the most common. 1 Despite the significant incidence of AMS, our understanding of its pathophysiology is incomplete. By filling in gaps in our knowledge, we can expect to develop more effective ways of preventing and treating AMS.
Previous studies on AMS have focused primarily on subjects’ self-reported symptoms, vital sign and physical exam findings, and demographic and acclimatization data. These studies have led to many hypotheses regarding risk factors for the development of AMS. 2 Studies evaluating either the exposure of general populations to high altitude or the blood gas and electrolyte changes that occur with ascent to altitude and the subsequent development of AMS are lacking.
Despite frequent recommendations that travelers to high altitude stay well hydrated, the effect of dehydration or hypovolemia on the risk of AMS is not known. 3 Both dehydration and hypovolemia may impair acclimatization by interfering with the bicarbonate diuresis that occurs during ascent.4,5 Alternatively, both dehydration and hypovolemia may be protective by reducing the cerebral and pulmonary edema that are the hallmarks of high-altitude cerebral edema (HACE) and high-altitude pulmonary edema (HAPE) and that also may characterize the development of AMS. 6 The associated mechanism for the protective effect may be within the hypotheses that AMS and HACE are on the same continuum, with cerebral edema as a common late pathway. Only the extent of the cerebral edema, and the resultant ataxia and mental status changes, separate the 2 illnesses. Similarly, HAPE occurs secondarily to capillary leak as well. 1 Dehydration is associated with decreases in free water within the circulation and thus potentially a relative hypernatremia, which would balance oncotic pressure differences more in favor of the intravascular spaces. Hypovolemia may lessen the hydrostatic forces that contribute to capillary leak and edema, both in the lung and in the brain, thus reducing the development of AMS, HACE, and HAPE.1,2,7
Dehydration and hypovolemia are associated with various physiologic and laboratory abnormalities. Increased heart rate (especially with standing), decreased serum bicarbonate concentration, decreased venous pH, and increased anion gap are associated with hypovolemia. The latter 2 are thought to be related to lactic acidosis from decreased tissue perfusion. Increased hematocrit, increased blood urea nitrogen (BUN), increased BUN:creatinine ratio, and hypernatremia have been associated with dehydration. 8 –13 Some of these associations (heart rate changes in particular) have been questioned. 14
In this study, we had 3 objectives. First, we evaluated the incidence of AMS among a large group of native people. Second, we collected laboratory data by using a portable device at both moderate and high altitudes to define changes in serum electrolytes as subjects ascend. Finally, we evaluated the association between surrogate markers of dehydration and decreased effective circulating volume with the development of AMS.
Methods
Each year thousands of Nepalese people make a pilgrimage to Lake Gosinkunda during a Hindu festival at the time of the first full moon in August. The lake is located north of Kathmandu in the Langtang region of the Nepal Himalaya at an elevation of 4500 m. Most of these pilgrims ascend from approximately 2000 m in anywhere from 1 to 3 days.
We conducted a prospective study of this population. Permission to conduct this study was obtained from institutional review boards at the University of New Mexico and Sinai Hospital of Baltimore, as well as from the Nepal Medical Review Board. Informed written consent was obtained from all subjects in Nepali or English. The Himalayan Rescue Association provided logistical and personnel support. A team of physicians delivered medical care during all phases of the study as was clinically indicated. Evacuation and follow-up medical care were made available.
Data collection was accomplished in the town of Dunche (elevation 2050 m) and at Lake Gosinkunda (elevation 4500 m). Many of the pilgrims and all the cohort subjects made their way to the lake via Dunche, many arriving there via bus from Kathmandu (elevation 1400 m). All subjects resided at an elevation less than or equal to that of Dunche. The research team also arrived at Dunche from Kathmandu ahead of most of the pilgrims and then separated into 2 groups. One group ascended to the lake over the course of 3 days. The second group stayed in Dunche for 3 days to enlist volunteers and collect data on the cohort group. Nepali speakers randomly approached people heading toward the lake via the main street in Dunche. Informed consent was obtained and a questionnaire was completed that included age; gender; medical history; smoking history; medications; home village or town; and focused questions to allow Lake Louise scoring for AMS, HAPE, and HACE. Each subject was assigned a number to facilitate comparisons at higher elevation. Vital signs that were taken included oral temperature, heart rate, blood pressure, and respiratory rate. Heart rate was taken both while supine after at least 2 minutes and after standing for at least 2 minutes to assess orthostatic changes. Pulse oximetry (NONIN Medical, Inc, Plymouth, MN) was performed on each subject twice, and results were averaged. A physician in the team performed a focused physical exam. Willing subjects were phlebotomized from a peripheral vein after site preparation with an alcohol swab. Blood was analyzed with an I-stat device (I-stat Corporation, East Windsor, NJ, distributed by Abbott Laboratories) with multiple cartridge types. These handheld devices are able to analyze blood for various chemical markers according to the cartridge type that is used and then display a value. Finally, each subject was given a card with a unique number on it and was asked to find the medical group upon arriving at the lake, where he or she would then undergo the same procedure and receive a free T-shirt.
When the first group of researchers reached the lake, they occupied a centrally located permanent structure and hung a sign to facilitate subjects originally tested at the lowland site as well as new subjects in locating them. The researchers followed the same procedure described above both on subjects presenting to the hut with cards given to them by researchers in Dunche as well as on noncohort subjects who presented to the hut for either a free T-shirt or medical care or who were randomly approached at the lake by our Nepalese researchers and were asked to participate. Demographic, vital sign, and laboratory data were collected as described above. Additional information collected at the lake included symptoms of illness experienced on the hike, duration of ascent (measured in number of nights spent at intermediate locations between Dunche and the lake), total liters of water consumed since leaving Dunche, amount of time since last urination, time of arrival at the lake, and expected time of departure. For cohort subjects, number cards were collected and each subject received a free T-shirt.
A Magellan XL2000 Global Positioning System (Alexandria, Virginia, USA) with map correlation was used to ascertain altitude. The I-stat was calibrated daily and was operated at manufacturer-recommended temperatures. The I-stat device is certified to give accurate readings above a barometric pressure of 300 mm Hg. 15 The lowest barometric pressure we recorded was 340 mm Hg.
Acute mountain sickness was defined according to the Lake Louise criteria as the presence of headache with at least 1 other symptom of either nausea, dizziness, insomnia, or fatigue. 16 A Lake Louise score (LLS) was calculated for each subject for symptom severity comparison. For each symptom, a score of 0 = no symptoms, 1 = moderate symptoms, and 2 = severe symptoms. These scores were summed to arrive at the LLS.
Statistical Methods
Two subject groups were studied. The cohort group (n = 63) was enrolled at Dunche (2050 m), with 38 (60%) proceeding to have a completed assessment at the lake (4500 m). According to Lake Louise criteria, these 38 subjects were categorized into those with and without AMS. Of these subjects, 4 could not be evaluated for the development of AMS and were omitted from data analysis: 1 scored as having AMS at 2050 m (due to presence of headache and 1 other symptom, though unlikely truly AMS given the elevation), and the remaining 3 did not have LLSs completed at the 4500 m site.
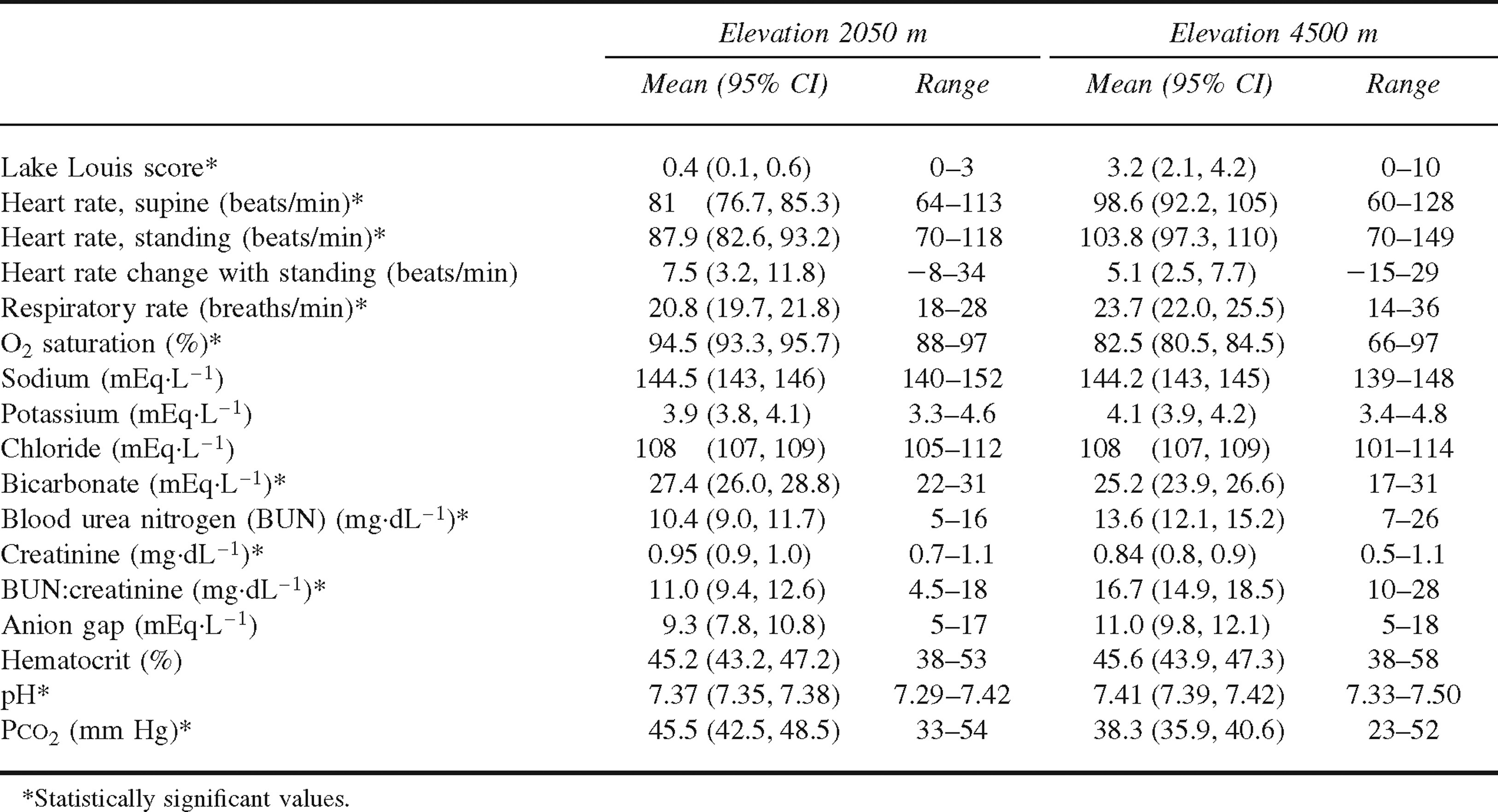
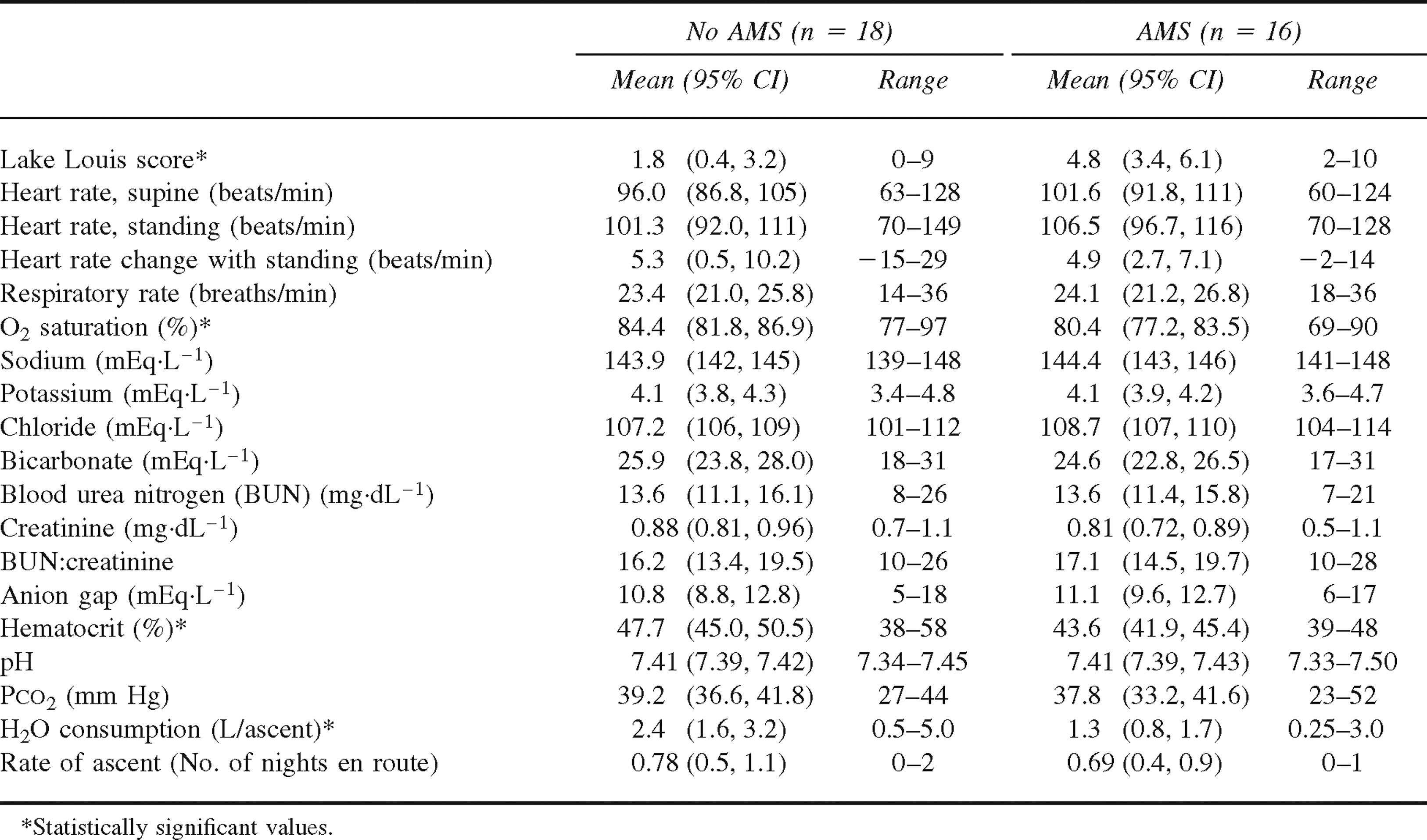
The data from the remaining cohort group (n = 34) were analyzed by a paired analysis. This allowed us to compare changes from moderate to high altitude within the same subject population. To gain a better understanding of the laboratory and vital sign data, we calculated mean values at moderate and high altitudes. For statistical inference, we used a paired t test (Table 1). We compared summary measures of the continuous variables at both moderate and high altitudes (Table 2) by using an unpaired 2-sample t test, comparing results from subjects who developed AMS with results from those who did not develop AMS.
Comparison of mean physiologic and laboratory variables at moderate and high altitudes in cohort of 34 subjects
Comparison of mean physiologic and laboratory variables at high altitude in cohort of 34 subjects with and without acute mountain sickness (AMS)
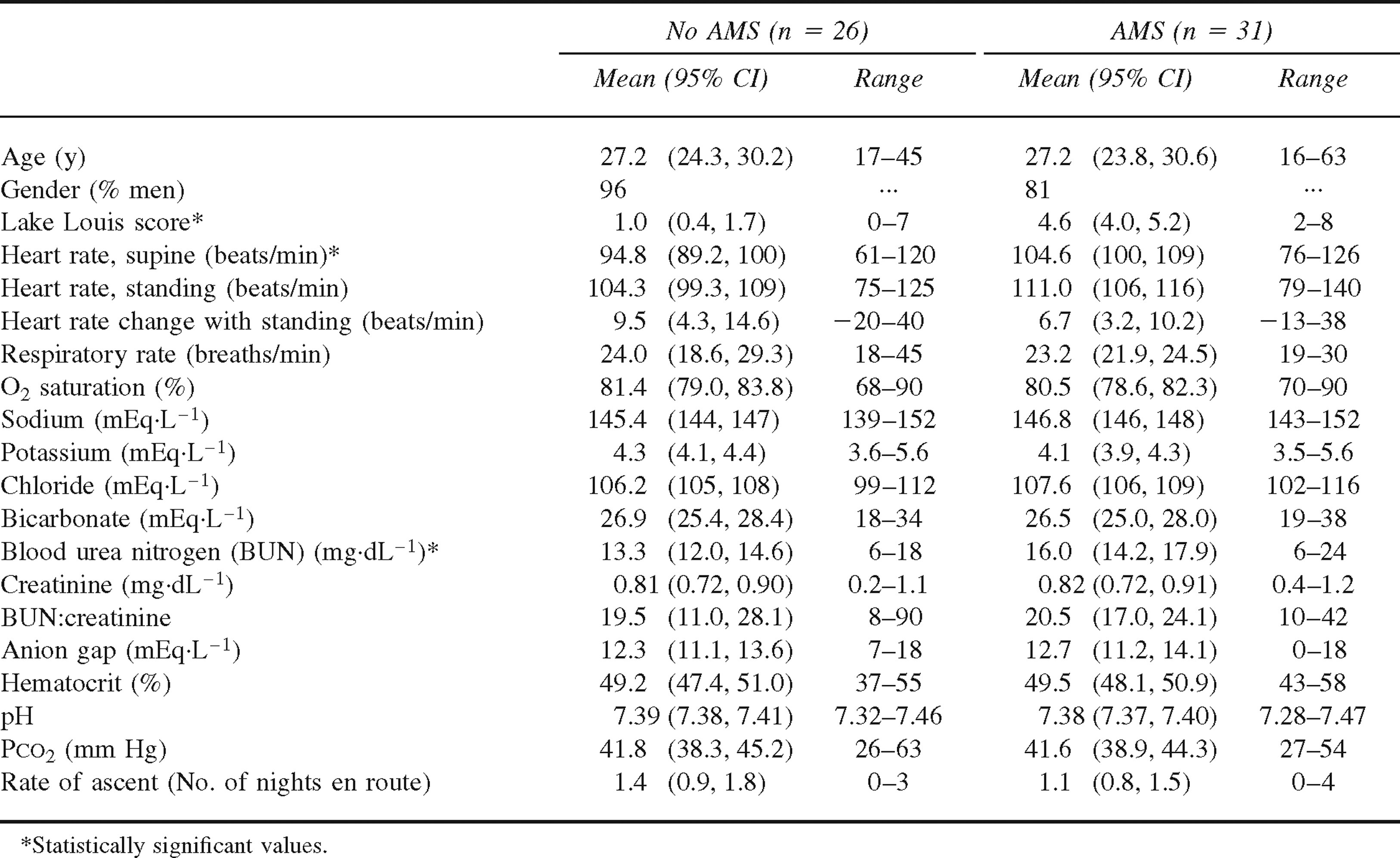
We enrolled subjects seen only at the lake in the cross sectional group (n = 57). We analyzed these cross sectional data only from subjects at high altitude who were not part of the cohort group. For most analyses, we again used an unpaired 2-sample t test (Table 3).
Comparison of mean variables for 57 subjects at high altitude with and without acute mountain sickness (AMS)
Data were entered in a Microsoft Access database and then analyzed by using a commercially available statistical package (SAS version 9.0, SAS Institute, Cary, NC). For all analyses, we used a 2-tailed critical P value of .05 to determine statistical significance. Means and 95% CIs are reported in Tables 1 through 3.
This study was conducted simultaneously with another, previously published study. 7 Some subjects participated in both studies.
Results
All enrolled subjects were self-reported to be healthy at baseline, and all denied use of medications. The 34 subjects in the cohort group had a mean age of 39.7 years (range 19–65 years, 95% CI 34.8–44.6). Twenty-six (76%) of the 34 subjects were men. The mean rate of ascent, or number of nights spent en route, was 0.74 nights (range 0–2 nights, 95% CI 0.6–0.9), and the median rate of ascent was 1 night, indicating that most of the pilgrims made the ascent during a 2-day period with 1 night spent en route. The mean water consumption was 1.9 L per ascent (range 0.25–5 L, 95% CI 1.4–2.4). With ascent to altitude, LLS (P < .001), heart rate (P < .001), respiratory rate (P = .004), BUN (P = .002), BUN: creatinine ratio (P < .001), and venous pH (P < .001) increased. Oxygen saturation (P < .001), serum bicarbonate concentration (P = .023), creatinine (P = .006), and venous P
Sixteen of 34 subjects (42%) in the cohort group were diagnosed with AMS. At moderate altitude, there was no difference in demographic, vital sign, or electrolyte data between the groups who did and did not develop AMS later on at high altitude. At high altitude, those with AMS (mean LLS 4.8) had lower oxygen saturation (P = .042), lower hematocrit (P = .011), and lower self-reported water consumption (1.3 L vs 2.4 L, P = .025) than those without AMS (mean LLS 1.8). All other variables, including demographic data and rate of ascent, were not significantly different (Table 2). The median rate of ascent for both groups was 1 night spent en route.
Of the 57 subjects enrolled at 4500 m in the cross sectional group, 31 (54%) were diagnosed with AMS. In the cross sectional group, 6 of the 7 women (12.3%) had AMS, whereas 25 of the 50 men had AMS. The subjects with AMS (mean LLS 4.6) had higher heart rates when supine (P = .0059) and higher BUNs (P = .022) than did their non-AMS counterparts (mean LLS 1.0). All other variables, including demographic data and rate of ascent, were not significantly different (Table 3). Again the median rate of ascent for both groups was 1 night spent en route. Water consumption data were not collected.
Discussion
This study found a prevalence of AMS similar to observations in previous studies conducted within the same population in previous years.5,17 We found that some of the laboratory values changed significantly with ascent from moderate to high altitude. Both BUN and BUN: creatinine ratio increased. Venous bicarbonate and creatinine decreased. Particularly, the increased BUN and BUN:creatinine ratio are associated with dehydration8,9,11 and are not clearly explained by other mechanisms. Although the degree of the changes in BUN and BUN:creatinine ratio are not great enough to indicate that subjects were clinically dehydrated, the changes suggest a decrease in circulating volume with ascent to altitude. The decrease in venous bicarbonate, though associated with hypovolemia in previous studies on gastroenteritis in children,
8
–11 may also be explained by the bicarbonate diuresis that occurs secondary to exposure to hypoxia.
18
The creatinine decrease was minimal and of no clinical significance. The observed increase in heart rate with ascent may reflect a decrease in effective circulating volume or could be secondary to increased physical and physiologic stress. The increased venous pH and decreased P
If we examine all the data from both the cohort and cross sectional groups, we find that subjects with AMS had lower self-reported water consumption, lower oxygen saturation, higher heart rate, less hemoconcentration, and higher BUN, though these changes were not always consistent within both groups. Furthermore, all the other variables, including most of the physiologic and laboratory markers of dehydration and hypovolemia, did not differ significantly between those with and without AMS in either the cohort or cross sectional group. The lower hematocrit in cohort subjects with AMS is unlikely related to a decreased ability to increase red blood cell count. Although this is an important adaptation to altitude, the effect is not seen until after at least a few days to weeks at altitude. 19 The subjects in our study were at altitude for only 1 to 2 days. Another possibility is that the greater increase in hematocrit seen in the cohort subjects without AMS was secondary to a more effective altitude-related diuresis, leading to a relative decrease in effective circulating volume. Diuresis is seen in those who acclimatize well and in those who take acetazolamide, and is associated with higher oxygen saturation.1,2,7 This diuresis may aid acclimatization by the excretion of excess base, thus allowing for increased ventilatory response, as well as by both dehydration increasing intravascular oncotic pressures and hypovolemia decreasing hydrostatic forces. It is possible that this dehydration and hypovolemia may be protective against the development of AMS. 6
In this study, however, other markers of dehydration and decreased circulating volume were not associated with protection from AMS. Although a significant difference in self-reported hydration patterns between healthy and affected subjects was noted, it is unclear what significance the lower self-reported water intake by those in the cohort group with AMS had. Even if self-reported intake were reliable, whether it contributes to AMS or is simply secondary to the associated nausea is unclear. Westerterp et al 20 showed a decrease in water intake in individuals with AMS. Those in the cross sectional group with AMS showed a higher BUN. Their BUN:creatinine ratios, however, were not different. The increased heart rate in those in the cross sectional group with AMS points to a relative hypovolemia. It is possible, though, that this increase may instead be related to the relative distress that those with AMS commonly experience, secondary to increased catecholamines and other neurohormonal mediators. This is supported by the data in that those in the cross sectional group with AMS had higher resting heart rates while supine, but they did not show any greater orthostatic changes than those without AMS.
This study was limited by the difficulties inherent in conducting field research, language and cultural barriers, an imperfect randomization process, the large number of pilgrims in the cohort group who were lost to follow-up (40%), a relatively small sample size, the subjective nature of the symptom questions, and the suspected insensitivity of dehydration and hypovolemia markers.11,14 Also, though some of the vital sign and laboratory variables were found to be significantly different either between moderate and high altitude or between subjects with and without AMS, the differences are not necessarily clinically significant (eg, minor differences in creatinine). It is not known what happened to subjects who were lost to follow-up or whether they were less ill or more so than the subjects enrolled. Subjects who were lost to follow-up likely arrived at the lake but did not present themselves to us perhaps because of lack of time or fear of blood draw, or they may have never arrived at the lake because of fatigue or illness. Once at the lake, we may have seen a larger proportion of subjects with AMS because of the use of subjects who self-presented or whom we approached outside our clinic.
In this study, we describe laboratory values for a group exposed to high altitude. We believe that it is the first time a portable laboratory device, like the I-stat, has been used extensively in altitude research. Through this work, we were able to address our 3 objectives. First, we found a high prevalence of AMS among this group of native people when they ascended to high altitude. Second, some of the changes in serum electrolytes, though not clinically significant, support previous hypotheses that ascent to altitude is associated with both dehydration and decreased effective circulating volume, which is likely secondary to diuresis. Finally, our study does not find a clear association between the common markers of dehydration, hypovolemia, and the development of AMS. More research with larger sample sizes and more comprehensive follow-up are needed to delineate these physiologic processes. Also, future studies may provide clarification by measuring volume intake and excretion as subjects ascend to altitude.
Footnotes
Funding
Financial and intellectual support was obtained from the Department of Emergency Medicine at the University of New Mexico through the help of Dr David Sklar, Department Chair, and special thanks go to Drs Judith Brillman and Darryl Macias.
Acknowledgments
We thank the volunteers and staff of the Himalayan Rescue Association for help in research, translation, and patient care activities. Mr Robert Casias of Abbott Diagnostics was instrumental in securing an I-stat device for our use on this project. We would finally like to thank all the Nepalese pilgrims we met for their willingness to participate in this work.
*
Presented in part at the Wilderness Medical Society Annual Winter Conference, Jackson Hole, WY, February 13–18, 2003.
1
Offprints will not be available from the corresponding author.
