Abstract
A patient with severe hypothermia presented with an initial rectal temperature of 28.3°C coupled with a hemoglobin of 2.2 g dL−1 and acute pancreatitis. Although hypothermia decreases oxygen and substrate consumption by tissues and can be cerebro-protective, the ideal rewarming strategy is unclear when the oxygen-delivery system is profoundly deficient, as with severe anemia. In this patient, truncal active external rewarming with a forced-air system, heated inhalation, and slow warmed transfusion yielded a 1.5°C h−1 rate of rewarming and a good outcome. We discuss the numerous protective and detrimental factors affecting oxygenation and ventilation during hypothermia coupled with profound anemia and the possible etiologic explanations for coexistent hypothermia and pancreatitis.
Introduction
Drastic pathophysiologic changes are associated with hypothermia, defined as a core temperature below 35°C. 1 Primary-exposure hypothermia occurs when the environment overwhelms a previously healthy patient's ability to conserve heat via respiratory, conductive, convective, evaporative, or radiative mechanisms. 2 Herein we report a case of severe hypothermia (28.3°C) in a middle-age woman with an underlying profound anemia. Under “normal hypothermic conditions,” a patient at this temperature is poikilothermic and requires careful resuscitation to avoid precipitating a nonperfusing cardiac rhythm during aggressive rewarming. 3 Rapidly accelerating the rate of rewarming per se does not improve outcome,4,5 and the ideal rate of rewarming regarding concomitant severe anemia and impaired tissue oxygenation is conjectural.
Case report
A 47-year-old woman presented via emergency medical services after her social worker discovered her unresponsive in her apartment. The woman's medical history included both seizures and a schizoaffective disorder. Her medications included carbamazepine, phenytoin, and trihexyphenidyl. The indoor ambient temperature was not measured by emergency medical services but was described as chilly.
On arrival, the paramedics noted the patient to be unresponsive, cool, dry, and very pale. Cardiac monitoring and peripheral venous access were initiated, and normal saline was infused. Per standing protocol, passive external rewarming was initiated, coupled with slow, gentle “Code 1” (normal speed, no red lights or sirens) transport to our emergency department resuscitation area. Field core temperature measurements were not attempted.
The patient's vital signs on arrival were as follows: rectal core temperature 28.3°C, blood pressure 104/44 mm Hg, pulse 67 beats/min, and an initial spontaneous respiratory rate of 5 breaths/min. Respirations were assisted by bag-valve-mask ventilation. The emergency department staff recognized the patient and recalled that 2 weeks before this presentation she had left the department against medical advice with an unexplained hemoglobin of 6 g dL−1.
On physical examination, the patient was very pale and without evidence of external trauma. No shivering was observed. Her initial Glasgow Coma Scale score was 6. She withdrew from pain but lacked purposeful movements. Her pupils were 6 mm bilaterally and minimally reactive. Cardiopulmonary examination was unremarkable, and her abdomen was soft with hypoactive bowel sounds. There was no occult blood in the stool on rectal examination.
The patient lacked protective airway reflexes. Because neuromuscular blockade is largely ineffective at body temperatures below 30°C, and because the metabolism and clearance of paralytic agents is unpredictable in this setting, rapid-sequence intubation was not attempted.
6
Cold-induced trismus prevented opening the mouth sufficiently to accomplish orotracheal intubation. The patient was, therefore, fully preoxygenated and nasotracheally intubated. Mechanical ventilation was begun with synchronized intermittent mandatory ventilation at a base rate of 14 breaths/min, pressure support of 10 cm H20, tidal volume 650 mL, and an Fi
We chose a conservative approach to active core rewarming by including heated humidified oxygen at 40°C via the patient's endotracheal tube and slow infusion of warmed 35°C normal saline mixed with packed red blood cells. 7 –9 Modified truncal active external rewarming with a Bair Hugger forced-air warming system (Augustine Medical Inc, Eden Prairie, MN) was also initiated. 10 –12
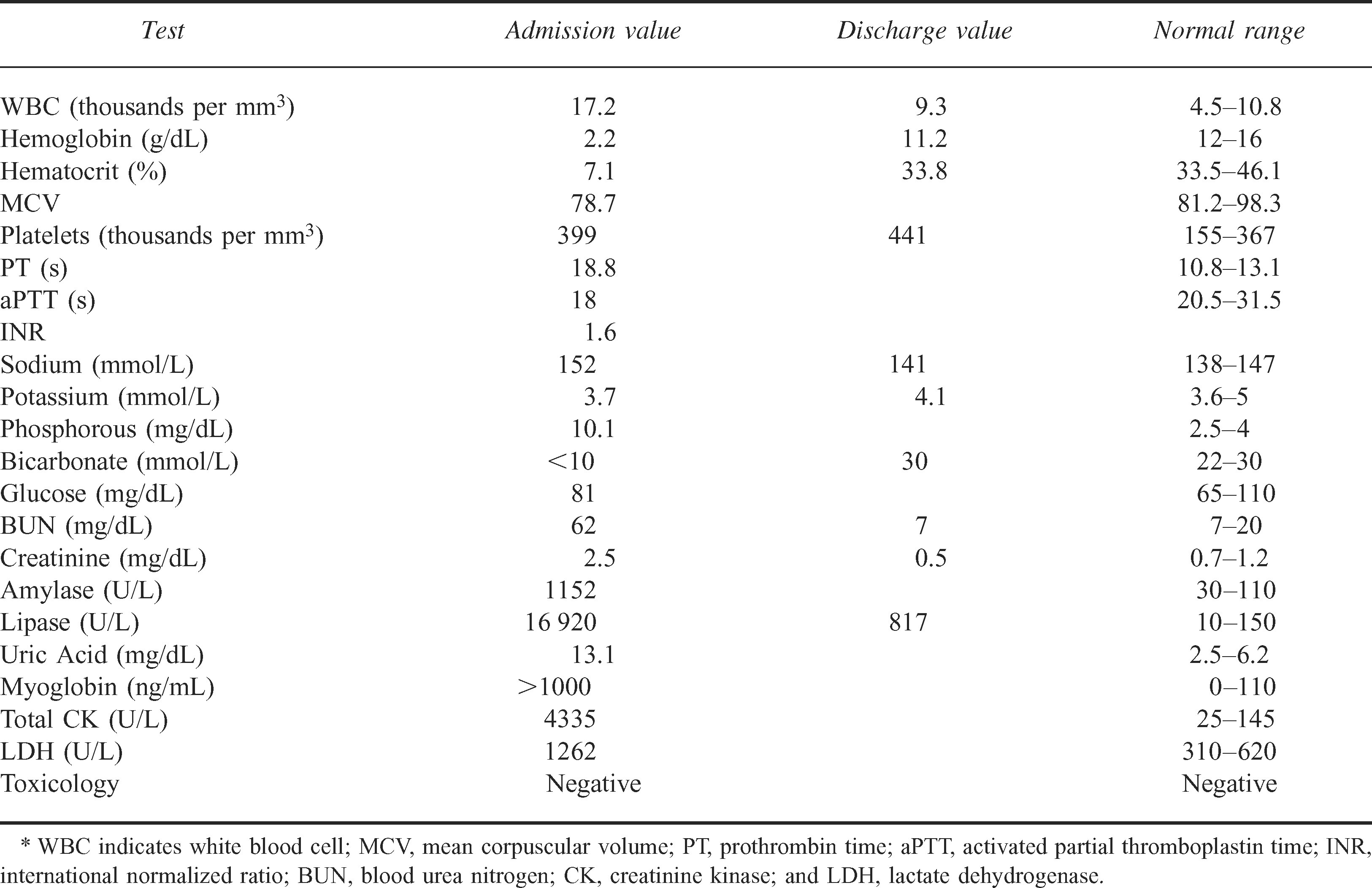
The patient's initial laboratory values are shown in Table 1. In addition to her profound anemia and multiple other laboratory abnormalities, it was noted that she had evidence of severe pancreatitis.
Initial admission and discharge laboratory values*
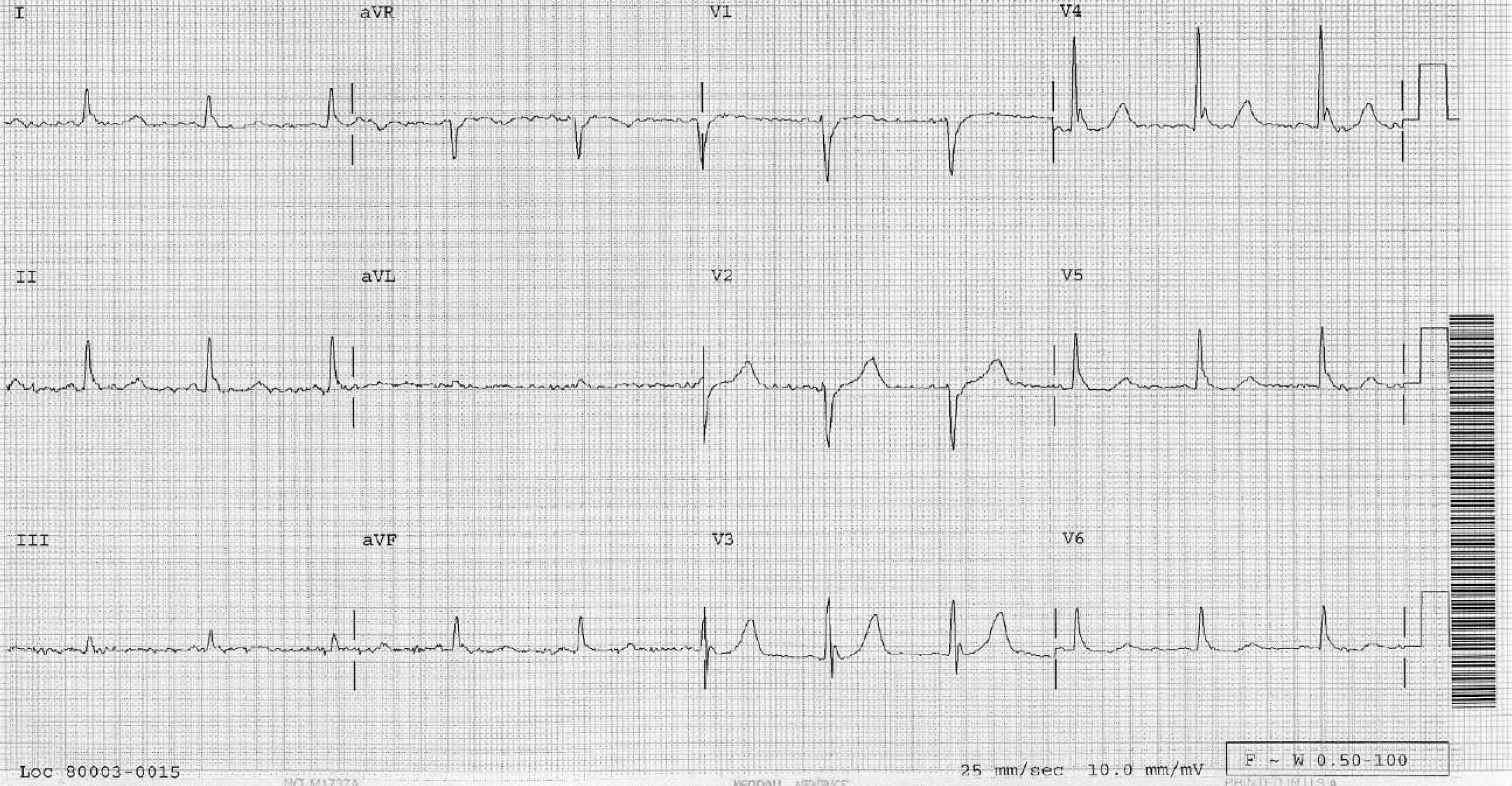
The patient's presenting electrocardiogram (Figure) displayed Osborn or J-waves, 14 and cranial tomography and chest radiography revealed no pathology.
Our choice of active rewarming measures achieved an average 1.5°C h−1 rise in body temperature in this patient. Continuous cardiac monitoring during rewarming and transfusion revealed no ventricular ectopy.
Work-up of the patient's severely elevated amylase and lipase included an abdominal computed tomography that excluded necrotizing pancreatitis. Once her body temperature reached 37°C, she underwent esophagogastroduodenoscopy, which revealed a gastric ulcer measuring 5 × 5 cm with 2 visible vessels at its base.
The patient gradually improved, and by hospital day 5 her lipase was down to 777 U/L, amylase was down to 120 U/L, and total creatine kinase was down to 338 U/L. She was extubated on hospital day 6 but subsequently developed an aspiration pneumonia and bacteremia. After an extended antibiotic course, she was discharged without apparent sequelae on hospital day 43. Ultimately, it was determined that she had no history of ethanol abuse, cholelithiasis, or pancreatitis.
Discussion
The patient appeared to have a combination of primary and secondary factors that led to loss of body heat. 15 –17 Her apartment was poorly heated, and at the time of her presentation the ambient outdoor temperature was 2.8°C. Her secondary risk factors for hypothermia included the hemorrhagic gastric ulcer with profound anemia and her psychiatric medications, which are known to jeopardize thermostability.
Multiple processes can contribute to decreased tissue oxygenation in patients with hypothermia (Table 2). 19 Early on, hypothermia increases myocardial oxygen demand because of cold-induced tachycardia and an increase in systemic vascular resistance caused by peripheral vasoconstriction. The tachycardic effects progressively wane as the core temperature approaches 28°C. During this transition there is a decrease in spontaneous pacemaker depolarization, resulting in bradycardia that is often profound. 20
Tissue oxygenation considerations during hypothermia*
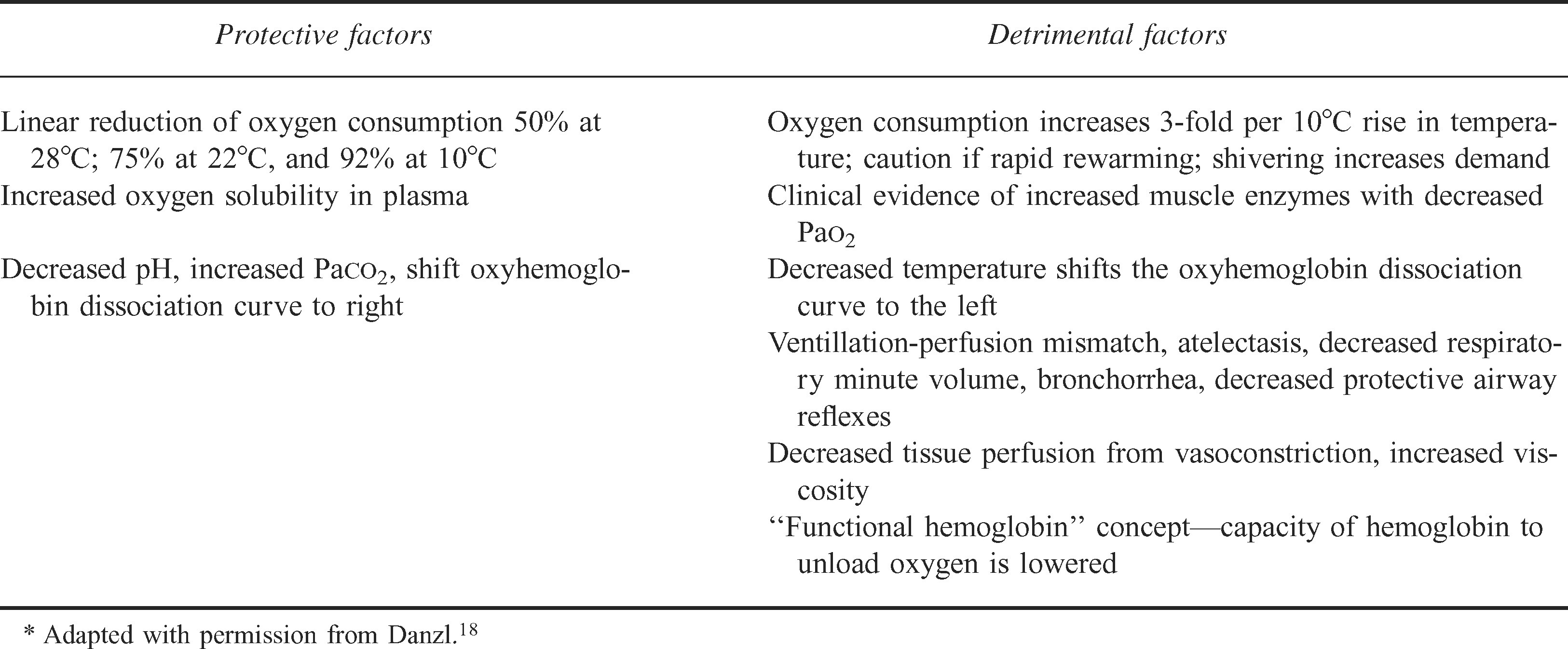
There are many significant hematologic effects from hypothermia. Red blood cells stiffen and are capable of less cellular deformity to facilitate perfusion. As a result of the decreased plasma volume, the hematocrit appears to increase 2% per 1°C fall in temperature. 21
Because this patient was hemodynamically stable, we elected to actively rewarm her by using noninvasive measures, including the use of a Bair Hugger forced-air rewarmer; administration of warm, humidified oxygen; and warmed blood transfusion to augment tissue oxygenation. Previous studies confirm the feasibility and safety of conservative noninvasive methods of rewarming in patients with hypothermia who have a perfusing rhythm. 5
Cardiac effects of hypothermia include decreased conduction velocity and increased action potential duration of myocytes. Unlike normothermic bradycardia, systole is prolonged more than diastole in hypothermia. 22 To complicate the clinical scenario, pre-existent ventricular ectopy can be suppressed during hypothermia and reappear during rewarming. 20 Common putative explanations for the development of a nonperfusing rhythm during resuscitation include tissue hypoxia, therapeutic manipulations, acid-base fluxes, autonomic dysfunction, mechanical jostling of the patient, sudden vertical positioning, core temperature afterdrop, coronary vasoconstriction with increased blood viscosity, and surges in metabolic demands.2,3,6
Vital signs can be deceptive in hypothermia. Normally there is a linear decrease in heart rate as body temperature declines. In this case report, the patient's relative tachycardia of 67 beats/min at 28.3°C suggests severe tissue oxygen deprivation. A tachycardia disproportionate to the degree of hypothermia warrants the consideration of hypovolemia, anemia, hypoglycemia, or a toxic ingestion. 20
Tissue oxygenation is hindered in hypothermia. Below core temperatures around 30°C, the respiratory minute volume is severely depressed, which inhibits gaseous exchange. Although carbon dioxide production de Note the prominent J-waves. They are currently not computer interpretable, as the deflections resulting from hypothermia are often misattributed to injury current. Thrombolytic therapy during the coagulopathic state of hypothermia is ill advised (from Danzl and O’Brien
13
).
Several other factors also impede oxygenation. The thorax is less compliant in patients with severe hypothermia, and this inhibits thoracic excursion. Noncardiogenic pulmonary edema may also develop below 30°C. 1 Profuse bronchorrhea can often result from cold stress, and, compounded by decreased ciliary motility, pulmonary clearance is impaired.
Offsetting decreased delivery of oxygen to the tissues, as body temperatures drop to 35°C there is an incremental decrease in tissue oxygen demand. This ranges from a 25% decrease at 32°C to a 92% decrease at 10°C.20,23 Carbon dioxide retention decreases tissue pH, shifting the oxygen dissociation curve to the right. This facilitates oxyhemoglobin dissociation. In addition, at lower core temperatures the solubility of oxygen in plasma increases (Table 2). 23
According to her medical history, the patient in this case report had developed her profound anemia gradually. This would initially stimulate cardiac output and regional blood flow alterations in an effort to compensate for a global loss in oxygen-carrying capacity. Intracellular levels of 2,3-diphosphoglycerate would rise, favorably shifting the dissociation curve to the right. This, however, can compensate for only a maximum 3 g dL−1 deficit in hemoglobin concentration. 3 Further compensation would involve shunting and protection of oxygen delivery to vital organs.
There are 3 possible reasons for the coexistence of hypothermia and pancreatitis: 1) pancreatitis might occur secondary to ischemia caused by microcirculatory shock during hypothermia, 2) severe primary pancreatitis develops and hypothermia occurs as a sequela, and 3) pancreatitis and hypothermia occur independently (eg, both as consequences of ethanol abuse). 24 Whether hypothermia-induced pancreatitis is a true entity is debated. 25 Some authors point out that in patients undergoing pancreatic transplant the donor pancreas is often intentionally cooled on ice for up to 24 hours before implantation, and ultimately pancreatic function returns to normal. Obviously there are inherent differences between intentional cooling of the pancreas under controlled in vitro conditions and in vivo accidental hypothermia. Donated organs are frequently flushed and washed in special preservative solutions, which does not adequately model in vivo conditions. 26
Conclusion
There is no optimal rewarming strategy for perfusing patients with accidental hypothermia below 30°C. Some authors prefer invasive techniques, including extracorporeal rewarming, whereas others prefer a less invasive approach.5,6,8 The significant comorbidities in this patient with severe anemia and pancreatitis prompted us to adopt a conservative, noninvasive strategy combining active core and external rewarming, for cardiopulmonary bypass facilities were immediately available should they have been needed.
