Abstract
Objective
Numerous studies support the use of warmed intravenous fluids in hypothermic patients. The most effective method to accomplish this goal in a cold prehospital, wilderness, or combat setting is unknown. We evaluated various methods of warming intravenous fluids for a bolus infusion in a cold remote environment.
Methods
One liter and 500 mL bags of intravenous fluid at 5°C were heated using various methods in a 5°C cold room. Methods included attachment of 3 types of chemical heat packs and heating the fluid in a pot on a camping stove. For all methods, fluids were run at a wide-open rate through an intravenous line with an 18-gauge catheter attached to the end to simulate a bolus infusion. The temperature of the fluid at the end of the intravenous line was measured. Each method was tested twice. Equipment weight and setup times are reported. Mean infusion temperatures for the various methods are compared.
Results
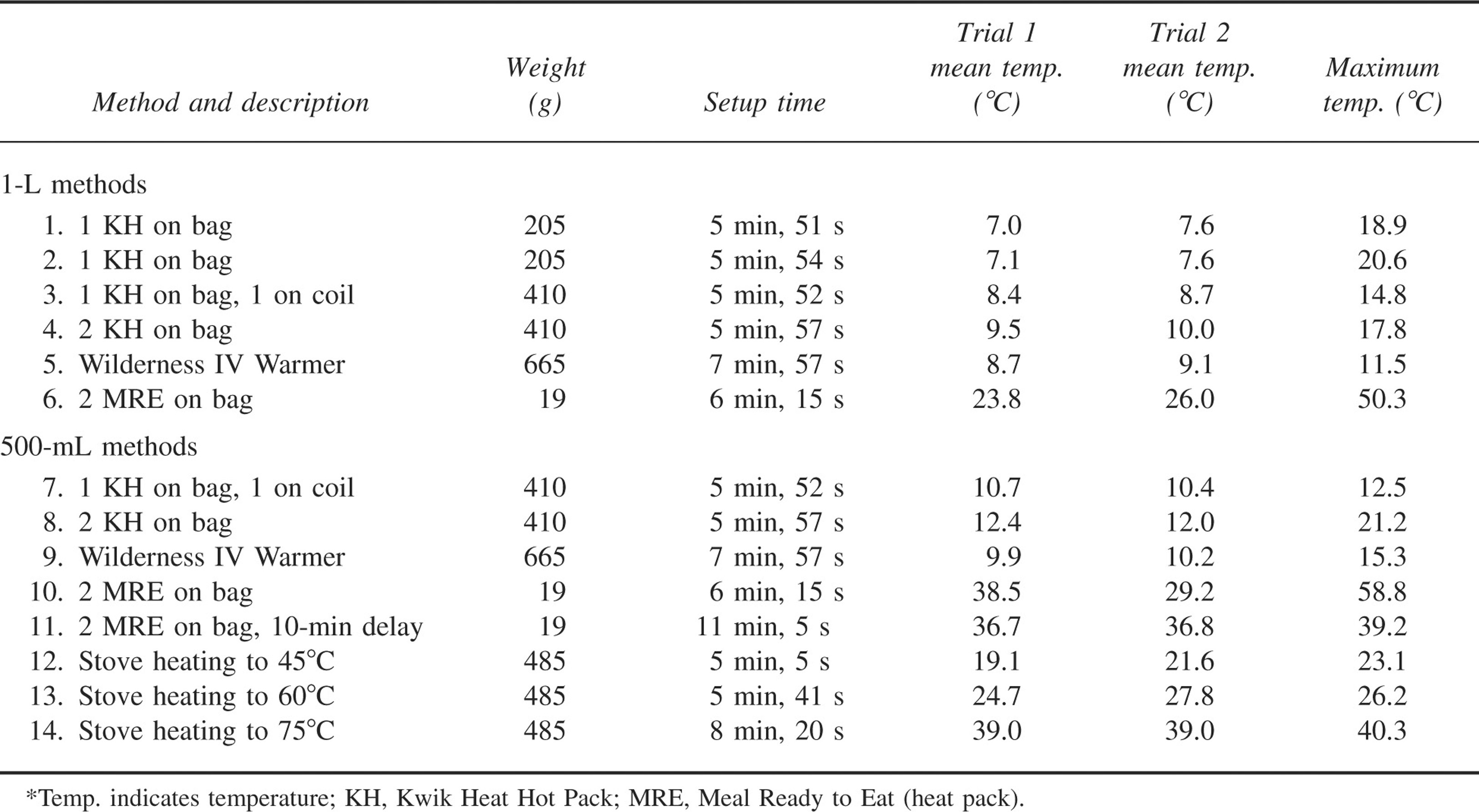
Equipment weights ranged from 19 to 665 gm. Setup times ranged from 5 to 11 minutes. The 2 methods which achieved the desired mean infusion temperature of 35 to 42°C without excessive maximum temperatures were 1) 2 Meal Ready to Eat hot packs attached to a 500 mL bag of fluid for 10 minutes prior to infusion, and 2) a camping stove heating the surface of a 500 mL bag of fluid to 75°C prior to infusion. Other methods, including the use of commonly available heat packs and a commercially available IV fluid warmer were ineffective, with mean infusion temperatures ranging from 7 to 12°C.
Conclusions
Heating of cold intravenous fluids in a cold environment is possible using either Meal Ready to Eat heat packs or a camping stove. Further study is needed to evaluate the ability of either method to consistently produce an appropriate fluid temperature given various ambient and initial fluid temperatures.
Introduction
Primary hypothermia accounts for an estimated 700 deaths per year in the United States. 1 The incidence of secondary hypothermia, which typically occurs in the setting of illness, trauma, or intoxication, is unknown and probably exceeds that of primary hypothermia. Cases of hypothermia occurring in remote environments constitute a small but important subset of hypothermia cases. The American Alpine Club reported 138 cases of hypothermia occurring in the United States between 1984 and 2004. 2 This number is limited to cases of hypothermia occurring in association with mountaineering.
Hypothermia can be lethal, but early appropriate therapy may be lifesaving. A 1987 survey of 428 cases of hypothermia found a mortality rate of 23% for patients with an initial temperature ≤32.2°C and a mortality rate of only 7% for patients with an initial temperature between 32.2°C and 35°C. 3 For trauma patients with initial temperatures ≤32°C, the mortality rate approaches 100%. 4 Two prospective studies have found that aggressive treatment of hypothermia improves outcomes.5,6 Transfusing a liter of fluid at room temperature (21°C) into an average-sized adult who cannot increase heat production to compensate causes a decrease in body temperature of 0.3°C. 7 The cooling effect of ambient-temperature intravenous fluids has also been demonstrated in a swine model of hemorrhagic shock. Swine treated with 80 mL/kg of crystalloid had a core temperature, following the infusion, that was 2.8°C lower than that of untreated swine. 8 For these reasons, the American College of Surgeons recommends the use of warmed intravenous fluids in the treatment of trauma patients. 9
In the Emergency Department, optimal treatment of hypothermia depends on the temperature of the patient. For patients with a temperature above 32°C, current recommendations include active external warming, typically with forced heated air circulating through a blanket. 10 For patients with a temperature less than 32°C, active core rewarming techniques are recommended; these techniques include peritoneal and bladder lavage and extracorporeal blood rewarming. 10 Intravenous fluids given to hypothermic patients are typically warmed to between 40°C and 42°C. 10 Administering 65°C intravenous fluid via a multiport balloon-tipped central venous catheter has been demonstrated to successfully accelerate the warming of hypothermic dogs without adverse effect.11,12 However, we were unable to find human studies or case reports describing the administration of intravenous fluids heated above 42°C for the treatment or prevention of hypothermia.
For patients in remote environments and combat settings, there is no standard method of warming fluids prior to administration. Heating fluids on a stove or against a care provider's body are practiced methods, but we found no published studies evaluating these methods. Two flameless heat packs from Meal Ready to Eat (MRE) packages have been shown to increase the temperature of a 1-L bag of fluid from 10°C to 38°C in 8 minutes, 13 but other types of heat packs and the effect of MRE heat packs on fluid temperature during infusion have not been studied.
Not all ill or injured patients in remote environments require intravenous fluids. However, the administration of intravenous fluids is a central component in the treatment of hypotension, shock, or dehydration, and these conditions do occur in remote settings. Adult patients requiring fluids for the treatment of either trauma or medical illness without clinical evidence of pulmonary edema would typically be treated with a 1-L bolus of isotonic fluid. Following the administration of this fluid, reassessment of the patient's clinical status and vital signs would guide further fluid administration.
We conducted an experimental study to evaluate various methods of warming intravenous fluid for a bolus infusion in a cold environment with lightweight equipment that could easily be available to a wilderness rescuer. Specifically, we wanted to identify methods of heating 5°C intravenous fluid to an infusion temperature of between 35°C and 42°C.
Materials and methods
All tests were performed in a 5°C cold room, and all equipment was stored in the cold room for at least 24 hours prior to testing to ensure equilibration with the ambient temperature. One-liter and 500-mL bags of 0.9% sodium chloride intravenous fluid (Hospira Inc, Lake Forest, IL) were infused through a 254-cm–long, 3.05-mm–internal diameter intravenous line (Kawasumi Laboratories Inc, Tampa, FL) and were heated using various methods. Heat packs studied included 1) Kwik-Heat Instant Hot Packs (Cardinal Health Inc, Dublin, OH) containing anhydrous sodium thiosulfate, 2) Wilderness IV Warmer heat packs (Ultimate Hot Pack Inc, Lander, WY) containing calcium chloride, and 3) MRE heat packs (TrueTech Inc, Riverhead, NY) containing a magnesium-iron alloy.
Heat packs were activated either by squeezing the heat pack to combine the active substance with water contained in a separate pouch within the heat pack (Kwik-Heat Instant Hot Packs) or by adding 5°C water, per manufacturer instructions (Wilderness IV Warmer and MRE heat packs). For studies using the Wilderness IV Warmer, 16 ounces (473 mL) of water were added to the bladder containing 1 chemical heat pack. For the MRE heat packs, water was added to the outer bag to a level between 2 lines imprinted on the bag by the manufacturer. This quantity corresponded to approximately 100 mL of water. The inner heat pack was left in the bottom of the bag and the bag of intravenous fluid was also fitted inside this bag.
Heat packs were attached to fluid bags using 1-inch– wide plastic Transpore tape (3M, St Paul, MN) (Figure 1a). To decrease heat loss from the intravenous line, 150 cm of excess intravenous tubing was coiled and attached to the side of the fluid bag or to a separate hot pack (methods 3 and 7; see Table). The coil was concentric, flat, and formed of approximately 7 loops, with the radius of the coil increasing from 2 cm to 5 cm. The decision to coil a portion of the intravenous line and to attach it to the heat source was an attempt to reduce heat loss, which has been demonstrated during flow of fluid through uninsulated intravenous tubing. 14 –16 The remaining 100 cm of tubing was left uncoiled. The only method for which the intravenous tubing was not coiled was for the Wilderness IV Warmer, which comes with an intravenous tubing insulator that covers the entire intravenous line. Additional insulation for all methods, other than those using the Wilderness IV Warmer, was created by wrapping an adult, large, synthetic fleece jacket around the fluid bag, heat packs, and intravenous tubing coil (Figure 1b). The height of the bag of fluid above the end of the intravenous tubing was 50 cm for all trials. Fluids were infused through an 18-guage catheter in the wide-open setting to simulate a bolus infusion. We did not shake the bags of fluid prior to or during infusions for any of the methods.
Pictures showing
Equipment weight, mean setup time, and mean and maximum temperatures at site of infusion for the 14 methods*
Stove methods employed a Pocket Rocket stove and an IsoPro (isobutane) fuel canister (Mountain Safety Research Inc, Seattle, WA) to heat a 500-mL bag of fluid inside a metal pot with a diameter of 15 cm, a height of 8 cm, and a volume of 1400 mL. One-liter bags of fluid were not studied using the stove method because they are too large to fit in commonly used backpacking pots. After placing the 500-mL bag of fluid inside the pot, we added 800 mL of 5°C water, almost filling the pot. No lid was used. The stove was run at the maximum level. A 15-cm–long mercury thermometer with the bulb held against the intravenous fluid bag was used to measure the temperature of the fluid in the bag during the stove trials (Figure 1c). Although all the equipment for heating the fluid on the stove was stored at 5°C, because of safety concerns, the heating of the fluid on the stove occurred under a ventilated hood just outside the cold room. After warming the bag of fluid on the stove, the bag was removed from the pot, brought back into the cold room, attached to the intravenous infusion setup (including the coiled section of intravenous line), wrapped in the fleece jacket for insulation, and infused.
Temperature was measured using Omega Scientific Type-T Thermocouples (Omega Scientific Inc, Tarzana, CA). For all trials, the temperature of the infused fluid was measured with a thermocouple inserted through a side port and positioned inside the intravenous tubing 3 cm from the end of the tubing. The temperature probe was placed at the end of the intravenous tubing so that measurements would correspond to the temperature of fluids that would be received by a patient. During each trial, concurrent ambient temperature measurements were obtained from a thermocouple placed inside the cold room. Temperatures were converted from analog to digital using an InstruNet model 100B converter (GW Instruments Inc, Somerville, MA) and were recorded each second on a Microsoft Excel file on an Apple PowerBook computer.
Warming fluids using heat packs takes time. Preliminary trials using 2 Kwik-Heat Instant Hot Packs against a 1-L bag of fluid (method 4) demonstrated minimal increases in the infused fluid temperature, up to a maximum of 12.5°C over the course of a 30-minute infusion. The maximum infusion temperature of 17.8°C listed in the Table for method 4 was recorded at the beginning of the infusion, when fluid in the coil had been sitting next to the heat pack; this temperature was not reached during the rest of the infusion. Based on these results and similar results obtained during a preliminary 30-minute infusion using the Wilderness IV Warmer, it was felt that delays of greater than 5 minutes before beginning the infusion would not significantly increase the mean infusion temperature. Therefore, we chose to wait 5 minutes after activating and attaching heat packs to the fluid before beginning the infusion. An exception was made for the second set of trials using the MRE heat packs, for which preliminary trials demonstrated a substantial increase in fluid temperatures during the course of an infusion. The stove heating was not stopped at a pre-set time, but rather the heating ended when the surface temperature of the 500-mL bag of fluid reached the specified temperatures of either 45°C, 60°C, or 75°C. These temperatures were chosen based on preliminary trials in which we attempted to reach our goal infusion temperature of 35°C to 42°C.
1-L METHODS 1 Kwik-Heat Instant Hot Pack attached to 1 L of fluid 1 Kwik-Heat Instant Hot Pack attached to 1 L of fluid plus insulation 1 Kwik-Heat Instant Hot Pack attached to 1 L of fluid and 1 Kwik-Heat Instant Hot Pack attached to a coil of intravenous tubing plus insulation 2 Kwik-Heat Instant Hot Packs attached to 1 L of fluid plus insulation Wilderness IV Warmer attached to 1 L of fluid 2 MRE hot packs attached to 1 L of fluid plus insulation 500-mL METHODS 1 Kwik-Heat Instant Hot Pack attached to 500 mL of fluid and 1 Kwik-Heat Instant Hot Pack attached to a coil of intravenous tubing plus insulation 2 Kwik-Heat Instant Hot Packs attached to 500 mL of fluid plus insulation Wilderness IV Warmer attached to 500 mL of fluid 2 MRE hot packs attached to 500 mL of fluid plus insulation 2 MRE hot packs attached to 500 mL of fluid plus insulation with a 10-minute delay and removal of fluid from hot packs prior to infusion Stove and metal pot filled with water to heat 500 mL of fluid to 45°C Stove and metal pot filled with water to heat 500 mL of fluid to 60°C Stove and metal pot filled with water to heat 500 mL of fluid to 75°C
Equipment weight and mean setup time for each method are reported. Setup time is defined as the time beginning with unassembled cold equipment until the beginning of fluid infusion. For methods using heat packs, setup time includes the 5-minute or 10-minute (method 11) delay to allow heat packs to warm the fluid. Similarly, for stove methods, the setup time includes the time required to heat the fluid. The mean infusion temperature for each trial was calculated by summing the temperature measured each second during the course of the infusion and dividing by the total number of seconds. Mean and maximum infusion temperatures are reported for each trial. Mean infusion temperatures for the various methods are compared using an analysis of variance (ANOVA) test.
Prior to data collection, a sample size calculation was performed. We identified a 5°C difference in the mean temperature of the intravenous fluid as the lower estimate of a significant difference in infusion temperature. This value was chosen based on our impression that a difference of 5°C in an infused fluid could result in a clinically relevant change in patient temperature. Assuming 14 treatment groups and a standard deviation of 1°C, 2 trials of each method were needed to give the study a power of 80% to detect a 5°C difference (α = .05; 2-tailed analysis). Primer of Biostatistics: The Program (McGraw-Hill Inc, Columbus, Ohio) was utilized for the power calculation. Six trials of method 1 were performed in order to allow an accurate calculation of the standard deviation of the mean infusion temperature for this method.
Results
Equipment weight and mean setup time for each method are reported in the TableTable. The weight of the fleece insulating jacket (340 g) is not included in equipment weights. The MRE heat pack (9.5 g) is much lighter than the Kwik-Heat Instant Hot Pack (205 g) or the heat pack for the Wilderness IV Warmer (336 g). Setup times ranged from 5 to 11 minutes and were longest for the 2 methods with the mean temperatures that achieved the most desirable temperature profiles (methods 11 and 14). The mean ambient temperature in the cold room for all trials was 4.9°C (SD 0.3; range 4.4°C to 5.4°C). The mean infusion time for methods with 1-L boluses was 29 minutes (range 28–31 minutes), and the mean infusion time for methods with 500-mL boluses was 14 minutes (range 12–18 minutes).
The standard deviation of the mean infusion temperature for the 6 trials of method 1 was 0.35°C. The largest difference in mean infusion temperature for 2 trials of the same method was 9.3°C for method 10. With the exception of method 10, the largest difference in temperature for 2 trials of the same method was 2.9°C.
The 1-way ANOVA found a significant difference between the mean temperatures of the various methods (P < .001). A Bonferroni multiple-comparisons test revealed that the mean infusion temperatures from trials of methods 10, 11, and 14 were significantly higher than temperatures obtained using all other methods (P < .05), except for method 13 (P = .28, .62, and .02 for methods 10, 11, and 14, respectively). Mean infusion temperatures for the trials of methods 10, 11, and 14 were not statistically different from one another.
Only methods 10, 11, and 14 produced mean infusion temperatures within the desired range of 35°C to 40°C. These methods used MRE heat packs (methods 10 and 11) or the stove, and all 3 methods used 500-mL bags rather than 1-L bags of fluid. The maximum temperatures of methods 6 and 10 were 50.3°C and 58.8°C, respectively. A graph of infusion temperature vs time for the first trial of methods 10, 11, and 14 illustrates the temperature profile over time for these methods (Figure 2). The peak temperatures in methods 6 and 10 occur at the beginning of the infusion, when the heat packs against the coiled intravenous tubing cause high temperatures in the coil prior to infusion, and at the end of the infusion, when the volume of fluid remaining in the bag is small. These high fluid temperatures do not occur with methods 11 and 14 because for these methods the fluid bag is removed from the heat source prior to infusion.
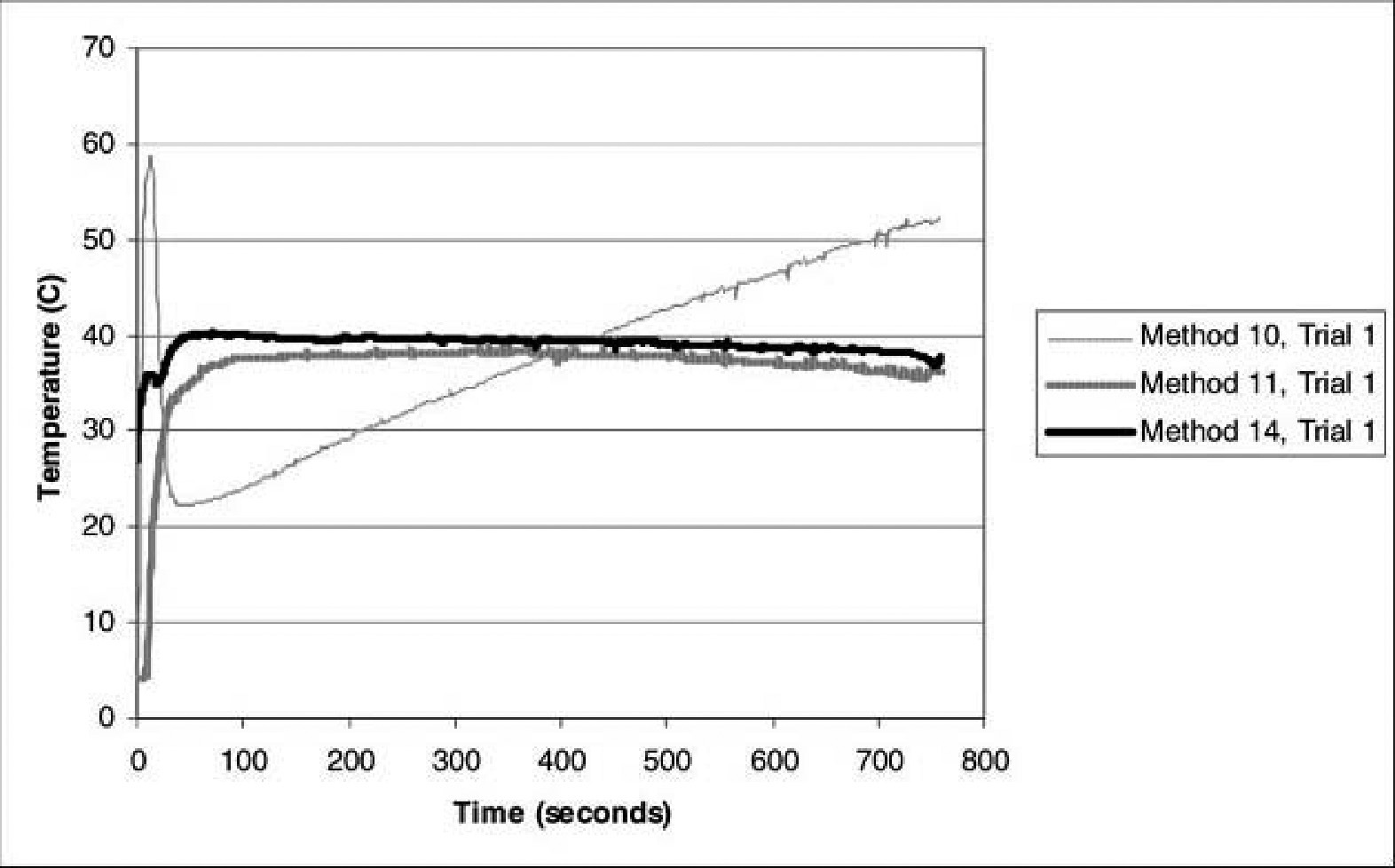
Temperature vs time for trial 1 of methods 10 (2 Meal Ready to Eat [MRE] packages on 500 mL), 11 (2 MRE on 500 mL for 10 minutes, then fluid removed from heat source), and 14 (stove heating 500 mL to 75°C, then fluid removed from heat source). Time zero represents the beginning of the infusion. Delays of 1) 6 minutes, 15 seconds; 2) 11 minutes, 5 seconds; and 3) 8 minutes, 20 seconds occurred prior to infusion for methods 10, 11, and 14, respectively. Note the spikes in the temperature associated with method 10 at the beginning (58°C) and the end (52°C) of the infusion.
Discussion
For both the 500-mL and 1-L fluid volumes, the Kwik-Heat Instant Hot Packs and Wilderness IV Warmer achieved only small increases in fluid temperature. The approximately 5°C increase in fluid temperature that occurred when these methods were applied to 500-mL bags of fluid may have some clinical value but falls far short of our goal of warming the fluid to between 35°C and 42°C. The fluid flow rates during our study were in excess of those recommended by the manufacturer of the Wilderness IV Warmer, and our results are not intended to discredit this product but only to see if it could perform this particular task of rapidly warming fluids for a bolus infusion. Only 2 of the methods employing the attachment of 2 MRE heat packs (methods 10 and 11) and 1 of the methods using an isobutane stove (method 14) were able to increase the fluid temperature to the desired level. Our findings are consistent with those of Garcia et al, who show that MRE heat packs can effectively heat a bag of intravenous fluid. 13 Their finding that 2 MRE heat packs can raise the temperature of 1 L of fluid to 38°C differs somewhat from our finding that the mean temperature of infusion when attaching 2 MRE heat packs to a 1-L bag of fluid is approximately 25°C. This difference is probably due to the longer time period over which they monitored fluid temperature (8 to 20 minutes) and their placement of the temperature probe inside the fluid bag rather than at the end of an intravenous line. Also, Garcia et al report adding 150 mL of water at an unspecified temperature to activate the heat pack. We added 5°C water, per manufacturer instructions, which corresponded to about 100 mL of fluid. These differences should not obscure the primary finding by Garcia et al, also supported here, that MRE heat packs can be used to warm fluids to a temperature appropriate for infusion. Our findings indicate that 2 MRE heat packs attached to a 500-mL bag of fluid for 10 minutes and then removed prior to infusion comprises a method that is likely to yield an infusion temperature in the desired range.
The high initial and final temperatures noted during method 10 (Figure 2) exceed the generally accepted maximum temperature for infused fluids of 42°C. 10 –12 Studies of the treatment of hypothermic dogs have demonstrated rapid and safe rewarming using 65°C intravenous fluids,11,12 but we were unable to find human studies or case reports of the treatment of hypothermia using fluid infusions at 65°C. At present the optimal safe temperature for fluid infusions in the treatment of hypothermia is unknown, but it likely exceeds 42°C. In the absence of human studies on this subject, we identify methods 11 and 14 as providing a desirable mean infusion temperature without excessive maximum temperatures.
Limitations
We used a 5°C cold room and fluids in order to simulate a cold environment. Much variation exists in the actual conditions encountered in prehospital settings, which may limit the field application of these results. Variations in wind, humidity, altitude, and sun exposure may affect the performance of the methods we describe. The ideal method of warming intravenous fluids would consistently and quickly warm fluids to an infusion temperature of at least 38°C regardless of the starting temperature of the fluids or the ambient temperature. Further testing of selected methods is needed to determine if this is possible.
We chose to study bolus infusions because this is the most important type of fluid administration in a remote prehospital setting. Bolus infusions allow care providers to effect a rapid intervention and to assess response to treatment. Nonbolus infusions have different thermal profiles than bolus infusions, and our results are not directly applicable to situations requiring maintenance fluids.
We used average temperature for each second of the infusion as a measure of the average infusion temperature. It would be more accurate to simultaneously measure fluid flow rates and calculate the average temperature per unit of fluid volume infused. In the presence of large variations in flow rates and infusion temperatures, the mean temperature over time during the course of the bolus would not accurately represent the mean temperature of the fluid received by a patient. We did not measure fluid flow rates. Based on observation and theory, we think that fluid flow rates were close to constant during the course of each bolus and that adjusting for changes in fluid flow rates during the infusion would not have significantly changed our results. Although most prehospital infusion setups do not allow for either precise measurement or control of infusion rate, we would expect infusion rates in most prehospital settings to be roughly constant during the course of a given bolus infusion and similar to the rates in our study.
We do not report a minimum infusion temperature because most infusions began with the infusion of a small amount of very cold fluid that was present in the intravenous line. As a result, the minimum temperature would be close to 5°C for all methods and would not provide important additional information about the methods. These initial cold values were included in the calculation of the mean infusion temperature but had a small effect on mean temperatures because of the brief period during which they occurred.
We chose to wait 5 minutes after activating the heat packs and before administering the infusions. This decision was based on preliminary trials that showed minimal changes in infusion temperatures with longer delays. The exception was method 11, for which we waited 10 minutes. This exception was made because the MRE heat packs were able to generate enough heat to significantly increase fluid temperatures, given their longer heating period. For the Kwik-Heat Instant Hot Packs and the Wilderness IV Warmer, we do not think that changes in wait times to optimize infusion temperature would have significantly changed the results. Adjustments in the wait time for the methods using the MRE heat packs probably are important in determining the temperature of the infused fluid and may prove to be a way to adjust the warming process for various ambient and initial fluid temperatures.
All trials used the same type of intravenous tubing, 18-gauge catheter, and height from the IV bag to the tip of the catheter. Variations in infusion times were thought to be due to variations in the resistance of the intravenous tubing resulting from the compression created by the attachment of the heat packs and fleece jacket. We used 254-cm–length tubing because that is the length currently used by parkmedics in Sequoia and Kings Canyon National Parks where the authors (GS and MS) serve as medical advisors. Handrigan et al 15 demonstrated a loss of 10°C in fluid temperature along the course of 280-cm–length tubing. They also demonstrated that higher flow rates result in less heat loss from the intravenous tubing; these findings are supported by several other studies. 14 –16 Extrapolating from the result of Handrigan et al, we estimate that because of higher flow rates in our study and the use of a fleece jacket to cover a 150-cm section of the tubing, our tubing resulted in a drop in fluid temperature of at most 5°C. We agree with the conclusion of Handrigan et al that shorter tubing length is preferable to minimize heat loss from the tubing. If we had used shorter tubing in this study, mean temperatures may have increased by a small amount, but we do not think that our conclusions about the various methods would have changed.
We only conducted 2 measurements of each method, which allowed us to efficiently evaluate a large number of different methods. The small differences in mean temperatures between the 2 trials of each method indicate that the mean temperatures we obtained are representative. The large difference (38.5°C vs 29.2°C) in mean temperature that occurred between the 2 trials of method 10 probably resulted from the fact that we added an inadequate amount of water to the MRE heat pack in the second trial. According to manufacturer instructions on the heat pack, water is added to the outer package of the MRE heat packs until it is between the lines on the heat pack. Perhaps because of how the bag was positioned, the fluid we added during the second trial may have been inadequate. After recognizing the large difference in temperature between the 2 trials of method 10, we repeated the method a third time and obtained a mean infusion temperature of 38.1°C, similar to the results of trial 1. For future studies, adding a specified quantity of fluid, as was done by Garcia et al, 13 may allow for more consistent heat pack activation than does adding fluid up to the level marked on the outer package, as per manufacturers instructions.
Because of safety regulations, we could not use the stove inside the cold room. Instead, we heated the fluid using the stove method under a hood vent at room temperature. It is our impression that the rate of heating of the fluid on the stove that we observed at room temperature would not have changed significantly if the heating had been done in the cold room.
For all stove methods, we measured the temperature of the bag of fluid by placing the bulb of a mercury thermometer against the surface of the bag inside the water. This method of measuring temperature is imperfect, because the measurement probably varies depending on the positioning of the bag and the thermometer within the pot. A more exact method would involve measuring the temperature of fluid inside the bag. However, since this method would violate the sterility of the bag and require additional temperature-measuring instruments, we feel that the use of a thermometer placed on the surface of the bag of fluid is a practical and adequate method of determining when to stop heating the fluid.
Method 14 used a stove to heat a 500-mL bag of fluid to 75°C. This method achieved a mean infusion temperature of 39°C and a peak temperature of 40.3°C. During the heating of the fluid, the temperature of the plastic was measured at 75°C and may have been hotter on the surface touching the bottom of the pot. Although intravenous fluids are commonly heated to 40°C without any known danger to the patient, higher temperatures may result in changes in the plastic that may release plastic compounds into the intravenous fluid. This concern also applies to methods employing the MRE heat packs, which, according to the manufacturer, can reach temperatures up to 125°C. After each trial, the plastic bag containing the intravenous fluid was examined by visual inspection. We were unable to identify damage to the plastic from any of the methods.
Conclusion
Our results demonstrate that it is easier to warm 500-mL bags of fluid than it is to warm 1-L bags of fluid. Since 500-mL bags of fluid are also as easy or easier to pack and carry, compared with 1-L bags, our findings indicate that 500-mL bags of fluid may be more suitable for use in remote settings in which warming intravenous fluid is likely to be necessary.
Of the methods we studied, only the MRE heat packs and the stove were able to heat the IV fluid to a temperature desirable for infusion. In comparing the 2 methods with the most desirable temperature profiles, method 11 (2 MRE heat packs on 500 mL of fluid) is lighter and less expensive but has a slightly longer setup time than method 14 (stove heating 500 mL of fluid to 75°C). Of these 2 methods, we regard the use of the stove as the preferable method, because we believe that the stove will allow for more control in the heating of fluids, given a variety of ambient and initial fluid temperatures. Further study of these 2 methods is needed to determine whether either approach can provide a consistent infusion temperature regardless of ambient and initial fluid temperatures.
Footnotes
Acknowledgments
We thank Dr David Grubbs at California State University, Fresno, for providing equipment and training for temperature measurement; Jan Wilson at University Medical Center for providing cold room access; Ronna Mallios for statistical analysis; and Leonard Stensaas for providing us with the Wilderness IV Warmer with heat packs.
Funding
Support for this project was provided by a grant from the Department of Emergency Medicine, University of California, San Francisco, Fresno.
*
Presented at the Central California Research Symposium, California State University, Fresno, Fresno, CA, April 21, 2006.
