Abstract
Background
Transfusion of cold intravenous fluids (IVF) can exacerbate hypothermia. Civilian and military guidelines recommend heated IVF for hypothermic patients; however, there is currently no ideal IVF heating system for use in resource-limited settings.
Objective
Development of a system that uses flameless ration heaters (FRH) and an insulated sleeve for the consistent delivery of IVF at physiologically appropriate temperatures (40°–42°C) over the range of ambient conditions typical of the prehospital and wilderness environments.
Methods
The temperatures of 0.9% normal saline (NS) 1-L bags were measured under 3 ambient conditions: 3°C, 10°C, and 20°C. The IVF was placed in an insulated pouch along with a predetermined number of activated FRH (5 FRH for 3°C, 4 FRH for 10°C, and 3 FRH for 20°C) for 10 minutes before removing the FRHs. The insulated IVF bag was drained through 280 cm of intravenous tubing at a flow rate of 77 mL/min. Raw temperature data for internal and delivery temperatures were collected and analyzed.
Results
The temperature of the IVF throughout the delivery of 1 L of NS under the 3 ambient conditions was as follows (mean ± SD): at 3°C ambient, 47° ± 2.1°C internal and 42.6°C ± 1.4°C at delivery; at 10°C ambient, 52.3° ± 2.7°C and 45.2° ± 1.6°C; and at 20°C ambient, 45.5° ± 1°C and 39.7° ± 0.7°C.
Conclusions
The IVF heating system described here reliably delivered physiologically appropriate temperature intravenous fluids in 2 of the 3 ambient treatment conditions. With the appropriate number of FRH for the ambient conditions, this system enables the delivery of warmed IVF to provide active warming, which may be clinically beneficial in the prevention and treatment of hypothermia.
Introduction
Accidental hypothermia, an involuntary drop in core body temperature to less than 35°C (95°F), is a potentially lethal condition common in prehospital medical situations. Hypothermia adversely affects patient outcomes. There is a 7% mortality associated with core temperatures between 35°C and 32.2°C on emergency room admission and a 23% mortality associated with core temperatures less than 32.2°C. 1 Individuals with core temperatures below 35°C on admission have an 8 times greater risk of mortality compared with normothermic individuals. 2 Hypothermia in trauma victims is common, and severe hypothermia is associated with poorer clinical outcomes as well as higher mortality. 3 Delivery of room temperature or cold intravenous fluids (IVF) can reduce the core body temperature by 0.3°C or more, potentially exacerbating the hypothermic condition. 3
It is recommended that circulatory support in moderate and severe hypothermia be maintained with boluses of IVF warmed to 40°C to 42°C to prevent further core cooling. 4 Although delivery of warm IVF as an isolated clinical intervention cannot reverse severe hypothermia, in situations with limited resources the infusion of warmed IVF may be the only available means for internal rewarming of a hypothermic victim despite its limitations. 2 In prehospital settings, IVF are usually maintained at ambient temperature before use. 5 Current IVF heating systems using battery packs are available but their weight to energy ratio and limited battery life render them inefficient for most field applications. 2 Flameless ration heaters (FRHs) from meals ready-to-eat (MREs) have been studied as potentially useful heat sources for warming IVF in the field.5,6 However, current models lack standardization that can consistently deliver warmed IVF. The aim of this study was to establish a method for heating IVF to recommended temperatures over a range of ambient conditions using an insulated pouch and commercially available FRHs.
Methods
Device
The device consisted of a closed-cell foam insulating pouch to contain a 1-L IVF bag and up to 5 FRHs. The pouch was fabricated of 8-mm-thick cloth-finished neoprene. A 50 × 40 × 1.2 cm (approximately 19″ × 16″ × 0.5″ length × width × thickness) piece of neoprene was cut and folded into a 30 × 15 × 5 cm (approximately 12″ × 6″ × 2″) rectangle with overlapping sides (Figure 2). Pieces of hook and loop fabrics (Velcro) were attached to the sides of the perimeters of the neoprene to enable closing the structure (Figure 3). The flaps that formed the base and top of the enclosure were pierced to enable attachment of the intravenous (IV) tubing assemblage to the IVF (base). A hook to hang the assembly from a drip stand was attached (top). The neoprene material was laid flat for loading with a 1-L IVF bag and FRH, and once activated, the insulating pouch was closed using the hook and loop fabric.

Neoprene insulating pouch in open configuration containing 1 L of intravenous fluid and flameless ration heaters.

Neoprene insulating pouch in closed configuration containing 1 L of intravenous fluid and flameless ration heaters.
We measured IVF temperatures at 2 sites, inside the storage bag and at the point of delivery at the distal end of the IV tubing, using thermocouple probes (Mon-a-Therm 503-0028, Mallinckrodt Medical, Inc, St. Louis, MO) that had been stripped of their outermost coats of insulation. The tip of the thermocouple probes were threaded through 16G syringe needles (PrecisionGlide 16G1, Becton, Dickinson and Company, Franklin Lakes, NJ) so that the tips of the thermocouples reached but did not extend past the beveled end of the needle. The probes were then epoxied in place. The temperature of the IVF in the storage bag was measured by inserting a 16G needle encasing a thermocouple probe directly through the rubberized fill port of the IVF bag. The temperature of the IVF at the delivery site was measured by inserting a 16G needle encasing a thermocouple probe through the most remote access port in the IV tubing so that the tip of the thermocouple probe was positioned in the IVF stream. The thermocouple probes were connected to a laptop-based thermocouple transducer/data collection system (GEC Instruments, Gainesville, FL) that recorded temperature data at 1-second intervals. At the conclusion of each trial, the temperature data were downloaded to a central spreadsheet (Microsoft Office Excel for Windows 2003, Microsoft, Inc, Redmond, WA) for analysis.
General Methods
The experimental trials were conducted in a 2.4 × 3.3 × 2.4 m (width × length × height) temperature-controlled chamber designed to maintain ambient temperature within 1°C of the set-point temperature. The ambient conditions inside the chamber for these trials were temperatures of 3°C, 10°C, and 20°C, and a relative humidity of 20% to 35%. All equipment and supplies were stored in the chamber under the test conditions for a minimum of 24 hours before a test run. Additionally, the IVF storage bags used in the 3°C ambient temperature trials were placed in a 3°C temperature-controlled water bath for a minimum of 6 hours before a test run. The 1-L bags of 0.9% normal saline (model NDC 0409-7983-09, Hospira, Inc, Lake Forest, IL) were infused through a 280-cm-long standard IV tubing kit (Primary Symbiq Set, Hospira) with an 18G angiocatheter (Model 38170, Becton, Dickinson and Company) attached to the distal end. The insulated pouch containing the IVF bag was suspended 150 cm above the angiocatheter tip during the infusion. All IV fluids were run wide open. Flow rate was determined before the experimental trials as approximately 77 mL/min (approximately 1 L/13 min).
The number of FRHs used in the different ambient conditions had been determined in a previous set of exploratory trials. Five replicated tests were conducted in each of the 3 ambient conditions. In the 3°C ambient temperature condition, 5 FRHs were used; in the 10°C ambient condition, 4 FRHs were used; and in the 20°C ambient condition, 3 FRHs were used. The FRHs (Innotech, Cincinnati, OH) were activated with 30 mL of water immediately before placement in the pouch. The activated FRHs were housed in the pouch with the 1-L IVF bag for 10 minutes. After 10 minutes, the FRHs were removed (to prevent overheating of the IV fluid and eliminate potential spillage of the heat pack contents during fluid delivery). The IVF delivery was initiated 12 minutes after FRH activation. This 2-minute interval between removing the FRHs and starting the IVF infusion allowed time for setting up and priming the IV tubing.
Results
In the 3°C trial, the internal storage bag temperature at the end of the 10-minute heating period was 48.2° ± 1.9°C (mean ± SD). Once the IV tubing was primed appropriately (approximately 1 minute after initiation of the drip) the temperature of the IV fluid at the point of delivery remained relatively constant throughout the drainage at 42.6° ± 1.4°C (Figure 1, top). In the 10°C trial, 4 FRHs were used for heating. The internal storage bag temperature at the end of heating was 54.0° ± 3.3°C. The temperature of the IV fluids at the point of delivery for minutes 1–12 of the drip was 45.6° ± 1.4°C (Figure 1, middle). In the 20°C trial, 3 FRHs heated the IVF bag to 46.3° ± 1.7°C at the end of the 10-minute heating period. For minutes 1–12, the temperature of the IV fluids at the point of delivery was 39.7° ± 0.7°C (Figure 1, bottom). There was minimal loss of temperature for the IVF both inside the bag and at the point of delivery throughout the drainage of the bags of fluid (Figure 1).
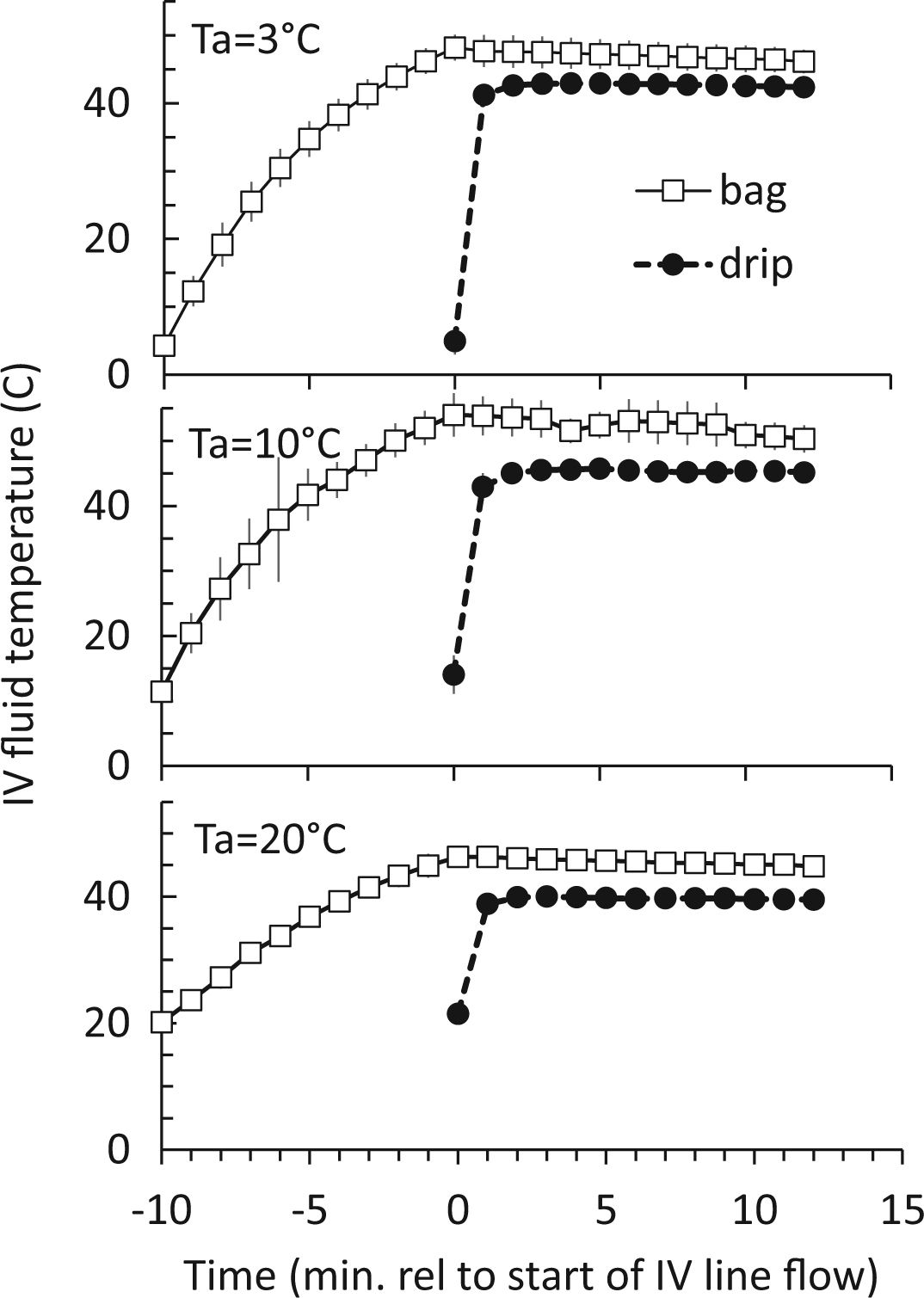
Intravenous (IV) fluid (mean ± SD) vs time at 3 ambient temperatures (Ta). Time is marked relative to the start of the draining of the IV fluid storage. Flameless ration heaters (FRHs) were placed in an insulated pouch along with the 1-L IV fluid bag 10 minutes before the start of the drip. The FRHs were removed at time 0. Five FRHs were used in the top panel, 4 FRHs in the middle panel, and 3 FRHs used in the bottom panel.
Discussion
This method provided a consistent delivery of heated IVF (range, 39.7°–45.6°C) under a variety of environmental temperatures. The system reached physiologically appropriate temperature IV fluids in 2 of the 3 ambient treatment conditions. By controlling the rate of energy delivery and exposure time for each FRH, we developed an algorithm correlating ambient temperatures with number of FRHs needed. These results confirm and expand on previous studies that FRHs can warm IVF.5,6 These prior findings have not been standardized to allow ease of reproducibility; one method used a fleece jacket for insulation of the infusion apparatus, 6 the other measured temperatures in the IVF bag rather than at delivery. 5 Furthermore, neither of these studies provided delivery temperatures considered to be warm enough to treat hypothermia with the use of FRHs. We demonstrated that a small, insulated pouch minimized environmental heat loss to cold environments and enabled thermal stability of IVF at the delivery point throughout the duration of a 1-L bolus of IVF.
The system described adds a modicum of weight (170–190 g not including water and IVF) to the typical supplies carried by medics. The insulating sleeve is made of 8-mm neoprene, weighing approximately 5 ounces (142 g). When not in use, the sleeve can be laid flat and used as an insulating pad for other applications (eg, a sitting pad or a preparation area). The surface area of the pad when laid flat is approximately 1850 cm2 (2 sq ft). During transport the pad can be rolled and attached to the outside of a pack or folded and placed inside a backpack. This system requires no external energy source and only small amounts of water (90–150 mL of water per use). This low-cost, lightweight, and simplistic FRH-based insulated system for heating IVF makes this a potentially valuable tool in a wilderness medicine resuscitation kit.
Using FRHs as heat sources is attractive because of their low weight (9.5 g per FRH), their effective exothermic reaction (approximately 50 kJ of heat energy at about 80 W), and their availability in wilderness settings. There has been concern that FRH-based heating of IVF is ineffective, inconvenient, or logistically impractical,5,6 and the reliability of using FRHs due to their uncontrolled release of energy has been questioned. 2 We minimized the potential variability in energy release rates by controlling the volume of water used to activate the FRH as well as standardizing the time of exposure of the IVF to the heating element.
Heat loss occurs across the walls of uninsulated IV bags and IV tubing, making insulation of these components integral for the delivery of a constant-temperature infusate. Intravenous tubing heat loss is predictable based on tubing length, fluid flow rate, and ambient temperature. 7 Heat loss will reflect the laws of physics and be greater with longer tubes and at lower ambient temperatures and slower flow rates. This environmental heat loss was factored into our experimental design when calculating the number of FRHs required for the different test temperatures. Insulating or shortening the line can reduce environmental heat loss from the IV tubing. We chose an alternative solution in which we heated the IVF bags to a higher temperature to accommodate the environmental heat loss under a given ambient condition.
Current recommendations are for IVF to be heated to 40°C to 42°C at the point of delivery to reduce the risk of venous endothelial damage and hemolysis that could occur with higher temperatures.4,8 The upper limit of temperature of IVF at delivery reached in this study overshot this recommendation by 3.6°C (range, 39.7°C–45.6°C). No reliable human studies exist looking at the delivery of IVF at temperatures higher than 42°C. However, dog studies have reported no adverse effect and negligible endothelial damage to dogs that received IVF up to 65°C. 8 Similarly, a small pilot study found that humans undergoing operative burn wound debridement and given 54°C IVF had no evidence of hemolysis, coagulopathy, or ill effect. 9 Based on these observations it may be reasonable to infer that the upper delivery temperatures observed in this current study are unlikely to cause local damage to the venous endothelium or intravascular hemolysis. Further human trials are needed to confirm this inference.
One concern of directly heating IVF storage bags is the possibility of thermally mediated chemicals leaching from the plastic bag liners into the fluids. Such leaching has never been documented at temperatures below which thermal damage to living tissues occurs. Commercially available IVF storage bags exposed to microwave radiation for up to 30 minutes did not leach chemicals into the contained fluids. 7 These results from the microwave heating reports cannot be directly translated to the thermal consequences using FRHs to warm IVF, but we speculate the effects of FRHs on chemical leaching may be the same. A simple chemical assay of the contents of the storage bags for polycarbonate materials after the heating protocol could be conducted for further risk assessment. Optimization of the IVF warming system could include the addition of a thermal sticker (colored thermometer sticker) to the surface of IVF storage bags to determine accurate temperatures of the contained fluids to direct the corresponding amount of FRHs.
The oxidation-reduction reaction of a magnesium salt with water produces heat (the primary function of an FRH) and hydrogen gas, which is released by the FRHs to the surrounding environment. The US Department of Transportation and the Federal Aviation Administration conducted studies showing that if enough hydrogen gas is released into a confined space (such as the cockpit of an airplane or storage container), it may becomes a source of ignition and potential fire hazard. 10 Based on these findings, we recommend that use of FRHs to heat IVF be performed in a well-ventilated space. We did not find that the levels of heat attained by FRHs in this study caused any melting or damage to the materials used (the neoprene pouch or IVF bags); however, cautious handling of FRHs during and after activation is recommended to prevent thermal burn injury to skin.
Limitations
The study was performed in controlled conditions; it is anticipated that the results will be more varied in real-world conditions. We used a cold room and cold fluids to simulate the cold environment. Multiple variables exist in the outdoors that are impossible to recreate in a laboratory setting. In particular, the method was not tested in subfreezing conditions. Although we attempted to consider the potential environmental variability by testing our model at a multitude of starting temperatures, wind chill factors and ranges of flow rates were not considered in this study.
We did not directly measure flow rates during the trials. However, we did measure flow rates through the system before conducting the trials. All flow rates were close to 77 mL/min as the 1-L IVF bags were consistently drained of fluids in 13 minutes. Slower infusion rates will alter the temperature profiles, lowering the temperatures of delivered fluids because of an increase in environmental heat loss at both the bag and IV tubing. The infusion rate in this study was set to deliver a large volume of IVF in a short period (1 L in 13 minutes) based on recommendations that out-of-hospital IVF be delivered as rapidly as possible to avoid cooling of fluid or freezing of transfusion lines. 4 Future studies that examine this method of heating IVF should take into consideration using smaller volumes of IVF (such as 250 mL and 500 mL) based on the most current trauma resuscitation guidelines.
We did not use insulated IV tubing in this study for the previously stated reasons. Many current field IV systems use insulated tubing to prevent heat loss to the environment at the level of the tubing. This proposed system would benefit from future investigations using insulated IV tubing as it would likely result in higher delivery temperatures and may alter the number of FRHs required to appropriately heat the IVF. Adjustments would have to be made in future systems based on the use of insulated IV tubing.
We chose to wait 10 minutes after activating the FRHs to begin delivery of IVF. A 10-minute delay in the initiation of rewarming of a patient is a concern; however, the alternative of delivering ambient temperature fluids is less attractive. Initiating the IVF heating immediately on making contact with the victim could mitigate this 10-minute delay. This would allow the medical provider a 10-minute window to assess the patient, obtain IV access, and address any other emergent medical or procedural interventions as the fluid undergoes warming. Furthermore, if it is anticipated that multiple bags of IVF were needed, additional IVF bags could be warmed simultaneously as the first bags are drained.
Conclusions
The IVF heating system described here reliably delivered physiologically appropriate temperature IVF in 2 of the 3 ambient treatment conditions. This system provides a cost-effective, reliable, lightweight, and easy-to-use method for heating IVF in field settings, and may be beneficial when used in combination with other recommended external rewarming procedures in the prevention and treatment of hypothermia in wilderness medicine scenarios.
