Abstract
In this paper, one-dimensional rod-shaped nano zinc oxide (ZnO) and Ag-In co-doped ZnO complexes with different doping ratios were prepared by hydrothermal method with C6H12N4, Zn(NO3)2·6(H2O), AgNO3, and In(NO3)3 as raw materials. The samples were characterized by X-ray diffractometry (XRD), scanning electron microscopy (SEM), and ultraviolet protection factor (UPF). The results showed that the degradation rate of methylene blue (MB) in aqueous solution reached 90.17% after being dispersed with 10%Ag-4%In/ZnO and exposed to ultraviolet light for 120 min., furthermore, MB was almost completely degraded after 180 min. of UV irradiation, thus the ZnO sample was provided with splendid photocatalysis activity. After 10% Ag-4%In/ZnO processing on modified polyester fabric, UPF of the treated Dacron was 50+ in accordance with the standard GB/T18830-2009 and considered to possess excellent UV resistance.
Introduction
Zinc oxide (ZnO) is an important wide-gap semiconductor with a band gap of 3.37 eV and an exciton binding energy of 60 MeV at room temperature, which has potential value in the development of blue-green, ultraviolet, and other light-emitting devices. 1 In the process of improving the preparation of nano materials, researchers found that nano ZnO has characteristics such as photocatalysis activity, UV resistance, and antibacterial properties. 2 One-dimensional nano ZnO broadens the absorption band at different wavelengths, and displays exceptional shielding performance in the ultraviolet region; therefore, ZnO has been widely researched by scholars all over the world. Base materials such as Al2O3 and silicon wafer have been used as substrates for the growth of ZnO; however, this method is not applicable to fabrics because of uncontrollable ZnO growth orientation on fibers and the unsatisfactory bonding between ZnO and the textile. 3 In addition, one-dimensional nano ZnO has revealed other disadvantages, such as the excess of band gap width and the recombinational consumption of photogenerated charge carriers. 4
To solve the problems mentioned above, modified polyester fabrics with ZnO seeding layers were prepared by padding with KH560 as coupling agent and stoving. The growth of Ag-In/ZnO arrays was adjusted by dipping the treated fabric in a solution of Ag-In precursors with a morphology control agent (PEG-2000) and heating. 5 The epoxy groups in KH560 molecules can form covalent bonds with the hydroxyl groups of polyester molecules, and the alkoxy groups of KH560 molecules can form strong chemical bonds with ZnO molecules after hydrolysis. KH560 was selected to serve as the coupling agent to improve the binding fastness between ZnO and polyester fabric.
Experimental
Materials and Instruments
Polyester fabric (10 × 25 cm, Shanghai Songzhi Textile Fabric Co. Ltd.), chitosan (Shanghai Zhanyun Chemical Co. Ltd.), zinc acetate dihydrate (C4H6O4Zn·2H2O), zinc nitrate hexahydrate (Zn(NO3)2·6(H2O)), hexamethylenetet-ramine (C6H12N4), silver nitrate (AgNO3), indium nitrate (In(NO3)3·xH2O), magnesium chloride (MgCl2), hydrogen peroxide (H2O2), sodium hydroxide (NaOH), ethanol (C2H5OH), polyethylene glycol (PEG-2000), cetyltrimeth-ylammonium bromide (C19H42BrN), and γ-glycidyl ether oxypropyl trimethoxysilane (KH560) were used. All reagents were purchased from Sinopharm Chemical Reagent Co. Ltd.
An S-4800 scanning electron microscope (SEM, Hitachi Corporation of Japan), X'pert Powder X-ray diffractometer (XRD, Panaco Corp. of the Netherlands), UV-3600 ultraviolet visible spectrophotometer (UV-Vis, Shimadzu Corp. of Japan), BL-GHX-V photochemical reaction instrument (Shanghai Bilang Instrument Manufacturing Co. Ltd.), and UV-2000 ultraviolet transmittance analyzer (Labsphere Corp of USA) were used.
Preparation of ZnO Nanorods
Preparation began with 0.025 M Zn(NO3)2·6(H2O) liquor and 0.025 M C6H12N4 aqueous solution added to 150 mL deionized water. After continuous stirring for 15 min., the well-mixed dispersive system was transferred to a reactor and heated at 95 °C for 12 h. After the reaction, the product was centrifuged after cooling action and the supernatant was removed. Then, the precipitate was washed twice with deionized water and ethanol respectively, dried at 80 °C for 2 h, and fully ground to obtain ZnO powder.
Preparation of Ag-Doped ZnO
Aqueous solution of 0.025 M zinc nitrate hexahydrate solution and 0.025 M hexamethylene tetramine were added to 150 mL deionized water. Silver nitrate liquor was mixed into the dispersion according to certain molar ratios (x = nAg/ nZn = 2.5%, 5%, 7.5%, 10%). 6 The well-dispersed mixture was transferred to a reactor and the product was obtained through hydrothermal reaction at 95 °C for 12 h. After washing with water and ethanol, the precipitate was dried and ground, then the ZnO sample could be gathered.
Preparation of In-Doped ZnO
Using the method in the previous section, silver nitrate was replaced by indium nitrate solution with certain molar ratios (x = nIn/nZn = 1%, 2%, 3%, 4%) to obtain In/ZnO material.
Preparation of Modified Polyester Fabric
Glacial acetic acid and 30% H2O2 were gradually mixed into dispersion of chitosan and deionized water at 60 °C, continued heating for 2 h to prepare chitosan gel. Then pH of the gelatin was adjusted to 8–9 and low molecular weight chito-san powder was obtained by stoving after suction filtration.
Polyester fabric was pretreated by being immersed in an aqueous solution of NaOH and C19H42BrN, vibrated continuously at 90 °C for 1 h, then dried and treated by plasma for 7 min.
The pretreated polyester fabric was immersed in a solution of low molecular weight chitosan and 1% acetic acid liquor at 50 °C for 1 h, then stoved to obtain the modified polyester fabric after double-dipping-double-nipping.
Preparation of ZnO-Doped Fabric
For the nano ZnO soliquid, C4H6O4Zn·2H2O was mixed with anhydrous ethanol in a three-port flask and stirred at 60 °C. A mixture of NaOH and ethanol were dropped into the flask and stirred constantly for 2 h.
The modified polyester fabric was dipped in aqueous solution of KH560 and MgCl2 for 30 min., pre-dried at 80 °C and post-cured at 150 °C for both 3 min. After being immersed into the ZnO sol for 5 min., the fabric was dipped-nipped twice, stoved, and post-cured to attain polyester fabric with a ZnO seeding layer.
The modified polyester fabric with ZnO seeding layer was immersed in mixed solution of Zn(NO3)2·6(H2O), C6H12N4, PEG-2000, AgNO3, and In(NO3)3·xH2O and reacted for 2 h at 90 °C. At the end of the reaction, the textile was washed and dried to obtain modified polyester fabric doped with Ag-In/ZnO.
Characterization
The morphologies of ZnO were observed using an S-4800 SEM from Hitachi Corp., Japan.
The XRD analyzer was used to determine the crystal structures of ZnO with scanning range of 25° to 80° and scanning speed of 5°/min.
The UV-Vis diffuse reflectance of ZnO powder was analyzed by UV-Vis spectrophotometry.
In this paper, the photocatalysis abilities of ZnO photocata-lysts were evaluated by the degradation effects of 10 mg/L methylene blue liquor under irradiation of an ultraviolet lamp at 80 watts. 7
The UV transmittance and ultraviolet protection factor (UPF) of modified polyester fabrics was determined with a UV-2000 ultraviolet transmittance analyzer, and the anti-UV abilities were judged. 8 The relationship between UV protection grades and ultraviolet protection factors according to the GB/T18830-2009 standard, is displayed in Table I.
Relationship between UPF and UV Protection Grades
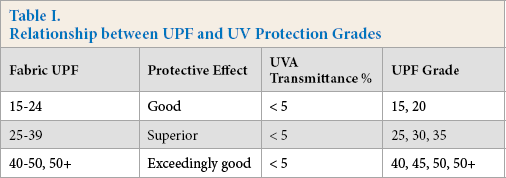
Results and Discussion
SEM Analysis of Ag-In/ZnO
The Ag-In/ZnO arrays on the surface of modified polyester fabric were tested by field-emission scanning electron microscopy, and the results are shown in Fig. 1. It can be seen in Fig. 1 that ZnO grown on the surface of polyester fabric in a low-temperature hydrothermal environment formed rod-shape structures with regular shape and uniform distribution. The mechanism of ZnO growth was that hexamethylene tetramine hydrolyzed to ammonia at the reaction temperature, thus increasing the concentration of OH– in the solution. Ten OH– and Zn2+ further formed a [Zn(OH)4]2– growth unit, which preferentially concentrated to the zinc-rich positive electrode. ZnO crystals grew along C-axis and formed rod-shaped structures.

SEM graphs of Ag-In/ZnO nanorods.
Crystal Structure Analyses of Ag/ZnO and In/ZnO
In Figs. 2 and 3, the diffraction peaks of ZnO at 31.76°, 34.51°, and 36.34° correspond to (100), (002) and (101) in the standard XRD spectra of ZnO with hexagonal wurtzite structure, respectively.9,10 Fig. 2 shows the XRD results of Ag/ZnO. When the doping ratio was 10%, the diffraction peaks at 31.76°, 34.51°, and 36.34° shifted significantly in the direction of high angles and the wave peaks grew noticeably. At doping ratios of 2.5%, 5%, and 7.5%, the intensities of diffraction peaks decreased and shifted to low angles. Accordingly, the optimal doping ratio of silver was determined to be 10%.
The XRD diagram of In/ZnO is displayed in Fig. 3. The half-height widths (FWHMs) of diffraction peaks in the spectral line were all minor, indicated decreasing grain sizes and increasing crystallinities. Among these samples, 4% In/ZnO not only showed a relatively small FWHM, but also represented a significant displacement to the direction of high angles. Thus, it could be speculated that indium had been doped into the lattices of ZnO, and 4% was the optimum doping ratio. 11
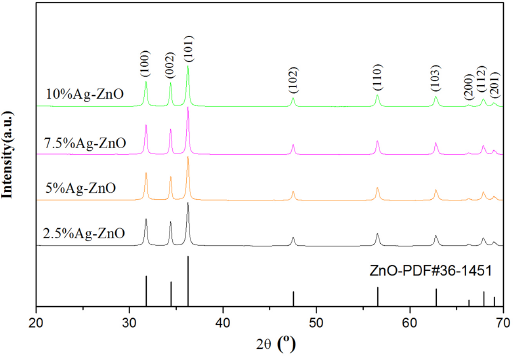
XRD pattern of Ag/ZnO nanorods.
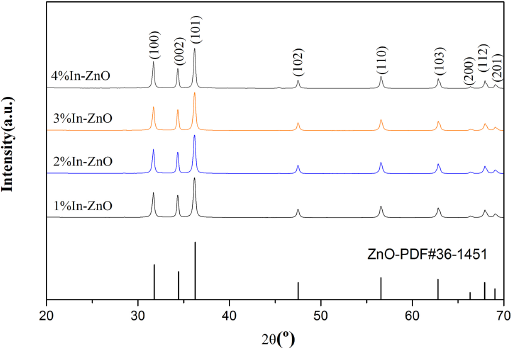
XRD pattern of In/ZnO nanorods.
Photocatalytic Activity of ZnO Nanorods
The degradation of methylene blue (MB) with Ag-doped ZnO under UV light is recorded in Fig. 4. When no pho-tocatalyst was added, the concentration of MB solution remained approximately constant with the extension of illumination time. As a result, self-decomposition of the indicator could be neglected.12–13
Fig. 4 demonstrates that the degradation rate of MB in the photoreaction significantly improved after adding ZnO, and the photocatalysis activity of Ag-doped ZnO was better than that of undoped ZnO. 14 The degradation rate increased with the increase of Ag doping from 2.5% to 10. When the UV irradiation was continued to 120 min., the degradation rates of MB with 2.5% Ag/ZnO, 5% Ag/ZnO, 7.5% Ag/ZnO, and 10% Ag/ZnO were 65.89%, 70.48%, 75.14%, and 88.33%, respectively. The degradation effect of 10% Ag/ZnO on the indicator was comparatively good, and reached 93.44% under UV irradiation for 180 min. Therefore, the optimal doping ratio of Ag was determined to be 10%.
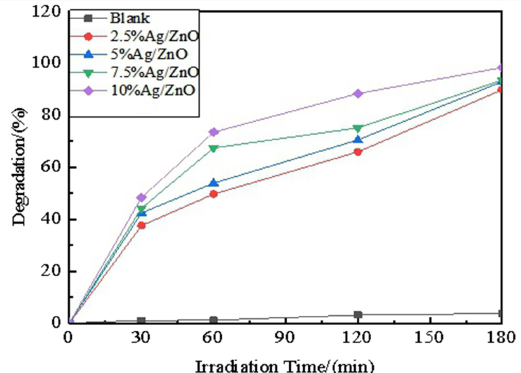
Photocatalytic activities of Ag/ZnO on MB.
The degradation of MB with In-doped ZnO irradiated by UV light is displayed in Fig. 5. Compared with undoped ZnO, the samples of In-doped ZnO worked more effectively in the test of photocatalysis. 15 The degradation rates increased with the increases of doping content in the range of 1%–4% within the same duration. Meanwhile, the degradation rates of MB with 1% In/ZnO, 2% In/ZnO, 3% In/ ZnO, and 4% In/ZnO reached 75.48%, 65.40%, 80.63%, and 86.85% respectively when the UV irradiation was prolonged to 120 min. Among the samples, the degradation rate of MB with 4% In/ZnO attained 96.38% under UV irradiation for 180 min. Thus the optimal doping ratio of In was determined to be 4%.
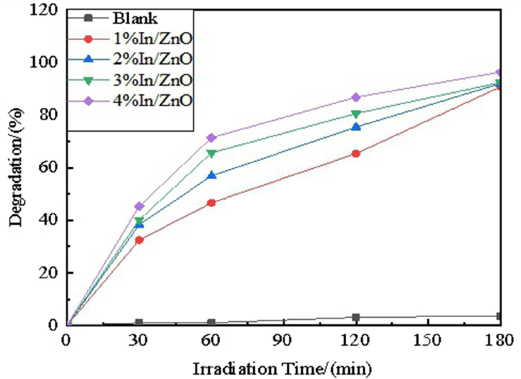
Photo catalytic activities of In/ZnO on MB.
The degradations of MB with ZnO, 4% In/ZnO, 10% Ag/ ZnO, and 10% Ag-4% In/ZnO under UV light are shown in Fig. 6 collectively. The photocatalysis effect of Ag-In co-doped ZnO significantly exceeded those of Ag/In single-doped ZnO. 16 The degradation rate of MB with 10% Ag-4% In/ZnO reached 90.17% after 120 min. of UV irradiation and 99.99% after 180 min.
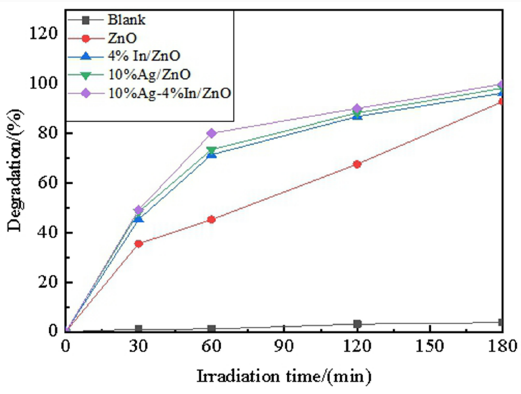
Photo catalytic activities of ZnO samples on MB.
The UV-Vis diffuse reflectance spectroscopy (DRS) analysis of ZnO arrays on the surface of modified polyester fabric is displayed in Fig. 7. Compared with undoped ZnO, Ag/In single and Ag-In co-doped ZnO represented more signifi-cant effects of absorption in the ultraviolet region.17–18 When the illumination on ZnO exceeded the energy of its band gap, photogenerated charge carriers were generated by electron transition, which could further form OH– radicals, and improve the photocatalytic activity. 19 In Fig. 5, 10% Ag-4% In/ZnO had the highest intensity of absorption in the ultraviolet region; therefore the sample possessed a satisfactory photocatalysis property. 20
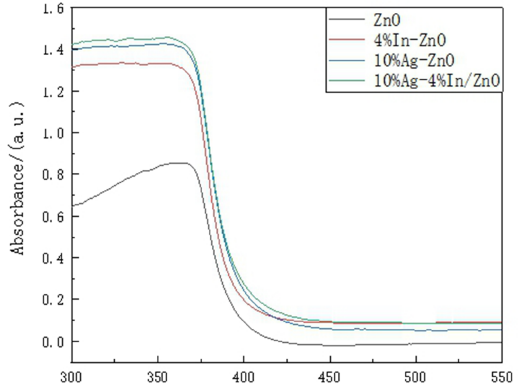
DRS results of ZnO samples.
UV-resistant properties of the modified polyester fabrics with no ZnO, 4% In/ZnO, 10% Ag/ZnO, and 10% Ag-4% In/ ZnO were tested and characterized by UPFs and transmittances of ultraviolet light. According to Table II, the average UPF of the undoped textile was 21.36, which indicated that polyester fabric possessed certain anti-UV ability, however, the effect was demonstrated to be unsatisfying. 21 The average UPF of 10% Ag/ZnO doped polyester fabric was 938.45, and that of 4% In/ZnO treated polyester fabric was 732.48. The UV-protection factors of modified polyester fabrics increased obviously after being doped with ZnO and the processed samples had excellent UV resistant properties. As for 10% Ag-4%In/ZnO doped modified polyester fabric, the UPF was 1452.64, indicating that the improvement of UV resistance of polyester fabric treated with Ag-In co-doped ZnO was significantly higher than those of Ag/In single-doped ZnO.
UV Protection Factors of Modified Polyester Fabrics
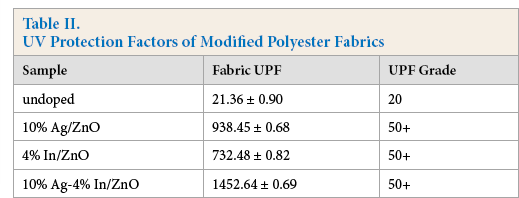
The ultraviolet transmittance curves of polyester fabric and its modified versions are displayed in Fig. 8. The modified polyester fabric treated with 10% Ag-4% In/ZnO represented relatively satisfactory UV resistance. For the doped polyester fabrics, when the wavelength was less than 380 nm, the transmittances of ultraviolet light were observed to be inferior. With the enhancement of wavelengths in the range of 380–400 nm, transmission effects of ultraviolet light on the modified polyester fabrics increased substantially. When the wavelength was about 400 nm, the increase in transmittances began to slow. Ultimately, the polyester fabric treated with 10% Ag-4% In/ZnO demonstrated its superior shielding effect in the whole ultraviolet region and UV protection factor of the fabric reached 50+ according to the standard GB/T18830-2009, consequently, the sample was considered to possess an exceptional anti-UV property.
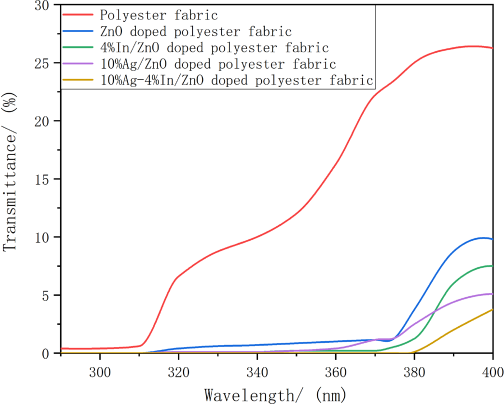
Polyester fabric transmittances of UV light.
Conclusions
Distinct rod-shaped nanostructures could be prepared by using a low temperature hydrothermal method and controlling the reaction conditions with Zn(NO3)2·6(H2O) and C6H12N4 as raw materials. The molecular architectures of ZnO nanorods were detected to be hexagonal wurtzite.
Composites were obtained by doping ZnO with certain molar ratios of Ag-In. According to the characterizations of XRD and XPS, no significant variation occurred in the crystal structures of the compounds. The photo catalysis activities of ZnO were obviously enhanced after doping and the optimal doping ratios of Ag/In were demonstrated to be 10%:4%, respectively. Compared with those of Ag-In single-doped ZnO, the photo catalytic activity of Ag-In co-doped ZnO represented a qualitative improvement that made the degradation rate of MB reach 99.99% under UV irradiation for 180 min., demonstrating the exceptional photocatalyst property of the sample.
The modified polyester fabric was treated with 10% Ag-4% In/ZnO and characterized by a UV transmittance analyzer. The test result indicated that the ultraviolet protection factor of the sample was 50+.
