Abstract
Especially for the potential use as sensors, but also in all other applications in which an interaction with the environment occurs, nanofibrous materials are advantageous due to their large specific surface area. An interesting material for electrospinning is the semiconductor zinc oxide (ZnO) which is often used in photoelectric or sensory applications. Nanofibers containing ZnO can be produced, for example, by electrospinning polyvinylpyrrolidone/zinc nitrate from a dimethylformamide/ethanol solution, followed by calcination to remove the organic phase. Alternatively, the polymer/semiconductor blended nanofibers can be used which are often less brittle, but on the other hand offer less contact between ZnO and the environment. Finally, decorating a nanofiber mat with ZnO offers another possibility to prepare nanofibers with ZnO surface. Possible applications of electrospun ZnO nanofibers or nanofiber mats include gas sensing, microwave absorption, photocatalytic degradation or enhancement of supercapacitor electrodes. This short review gives an overview of the most recent electrospinning and after-treatment techniques to create pure and blended ZnO nanofibers and presents the broad variety of possible applications of this well-known semiconductor with some still surprising properties.
Introduction
Electrospinning offers the possibility to create long nanofibers with diameters ranging from some 10 to some 100 nm from diverse materials. 1 The large surface-to-volume ratio of such nanofiber mats suggests using them in diverse areas where a large contact area with the environment is necessary, 2 such as filter materials,3,4 wound dressings,5,6 in tissue engineering or for other cell growth applications.7,8
While electrospinning necessitates a polymer solution or melt and thus typically creates polymeric fibers,9,10 it can also be used to prepare blends of polymers and other materials, for example, by embedding metallic 11 or semiconducting nanoparticles, 12 or to produce fibers from a chemical solution which allows for complete mixing of polymeric and other components and thus for calcinating the polymeric part after electrospinning, in this way retaining pure ceramic or other non-polymeric nanofibers. 13 By such techniques, the possible applications can widely be extended, as compared to pure polymer nanofiber mats.
Especially zinc oxide (ZnO) is of high interest as a gas sensor material, 14 for microwave absorption, 15 as supercapacitor electrode, 16 for photocatalytic degradation 17 or as an electron injection support material in light-emitting electrochemical cells. 18 Here, we give an overview of the most recent developments in electrospinning pure and blended ZnO nanofibers and their possible applications.
Blended ZnO nanofibers
To prepare blended ZnO/polymer nanofibers, ZnO nanoparticles or similar small shapes of ZnO are often inserted into the polymeric spinning solution which is used as a spinning agent and afterwards as a matrix, stabilizing the semiconductor particles. As an example, ZnO nanorods were prepared by a sol-gel process from sodium dodecyl sulfate and sodium hydroxide (NaOH) and electrospun with a polyacrylonitrile (PAN) solution using needle-electrospinning. The resulting hybrid nanofibers had a significantly increased diameter with parallel oriented ZnO nanorods, as supported by transmission electron microscopy (TEM). These fibers revealed high photocatalytic performance in visible light. 19
To obtain nanofiber mats with self-cleaning properties, the photocatalytic activity of ZnO was used by embedding ZnO nanoparticles at different concentrations into the co-polyester poly(1,4-cyclohexanedimethylene isosorbide terephthalate). Using TEM images, the ZnO nanoparticles were found to be uniformly dispersed in the nanofibers, resulting in a high self-cleaning efficiency with 9% ZnO nanoparticles. 20
Chen et al. 21 prepared PAN-ZnO/Ag composite fibers with diverse ZnO morphologies by single-capillary electrospinning, followed by hydrothermal ZnO synthesis and Ag reduction, and found different photocatalytic activity and ultraviolet (UV)-shielding efficiency, depending on the varying morphologies.
With electrospun polyaniline/polyethylene oxide (PAni/PEO) and PAni/ZnO/PEO nanofibers, Patil et al. 22 investigated the sensing properties for liquefied petroleum gas (LPG) and found advantages for the latter in comparison to pure ZnO or polyaniline.
Trabelsi et al. 23 showed that PAN/ZnO nanofibers mats, electrospun with a needle-based system, stayed much more elastic after carbonization than pure PAN nanofiber mats which may be useful in future medical or biotechnological applications.
Calcinated ZnO nanofibers
Preparation of pure ZnO nanofibers by calcination of the electrospun nanofiber mat and in this way evaporating the polymer component is usually performed by electrospinning a Zn precursor in combination with a polymer solution.
To create pure ZnO nanofibers, Shingange et al. suggested electrospinning a solution of zinc nitrate hexahydrate (Zn(NO3)2·6H2O) with polyvinylpyrrolidone (PVP) as the spinning agent. After electrospinning, the fibers were taken off the substrate and annealed at temperatures between 500°C and 900°C in air to gain pure ZnO nanofibers. 24
Matysiak and Tanski used PVP/zinc nitrate in dimethylformamide and ethanol instead to prepare nanofibers which were calcinated afterwards at different temperatures to remove the polymer. It was shown that not only the morphology, but also the other physical properties, such as the complex refractive index and the complex dielectric permittivity, could be tailored, making this material useful for opto-electronic or photovoltaic applications. 13
By electrospinning PEO/Zn(CH3COO)2·2H2O, followed by a first annealing step at varying temperatures, then hydrothermally growing ZnO micro- and nano-arrays in a solution containing Zn(NO3)2·6H2O, and finally a post-annealing-process, Xia et al. 25 optimized the point defects in the ZnO micro/nano array. In this way, they achieved increased response times and recovery times of ZnO photodetectors.
Huang and Or produced Co-doped ZnO nanofibers by electrospinning zinc acetate dehydrate and cobalt acetate anhydrous with PVP from dimethylformamide and ethanol as the solvent. After calcination of the blended fibers, Co-doped ZnO nanofibers were gained which showed ferromagnetic properties and high dielectric and magnetic loss, combined with average microwave absorption. 26
ZnO on nanofiber substrates
Combining the electrospinning technology with the material ZnO can not only be performed in the above described ways. Samuel et al. prepared freestanding supercapacitor electrodes from PAN nanofiber mats which were decorated by ZnO doped with manganese, followed by carbonization. In this way, a carbon nanofiber mat creating a fast electron-transport path could be combined with the active manganese-doped ZnO sites, resulting in a specific capacitance of ~ 500 F/g which stayed nearly fully stable for 10,000 cycles. 27
Piezoelectric electrospun poly(3-hydroxybutyrate) (PHB) nanofiber mats were used as substrates for hydrothermal deposition of ZnO. In this way, a mixture of nanoparticles and nanorods were formed, while ZnO grew preferentially in the hexagonal wurtzite structure. The ZnO coating increased the piezoelectric charge coefficient by more than a factor of 4, as compared to pristine PHB nanofiber mats, and also the surface wettability. 28
Electrospun PAN nanofiber mats were decorated with ZnO/Ag heterostructure nanoparticles using reflux, blending and hydrothermal methods, in all cases uniformly dispersing the nanoparticles on the nanofiber surfaces. All three decoration methods resulted in good antibacterial properties against Escherichia coli (gram-negative) and Micrococcus luteus (gram-positive), suggesting this fast and inexpensive path to developing antibacterial nanofiber membranes for protective textiles and filtration. 29
Zhu et al. 30 used a hydrothermal process to coat electrospun and calcinated SnO2 nanofibers with ZnO nanoparticles which were transferred into ZnO nanorods or ZnO nanosheets, respectively, using a combination of thermal and chemical treatment which defined the ZnO shape (Figure 1).
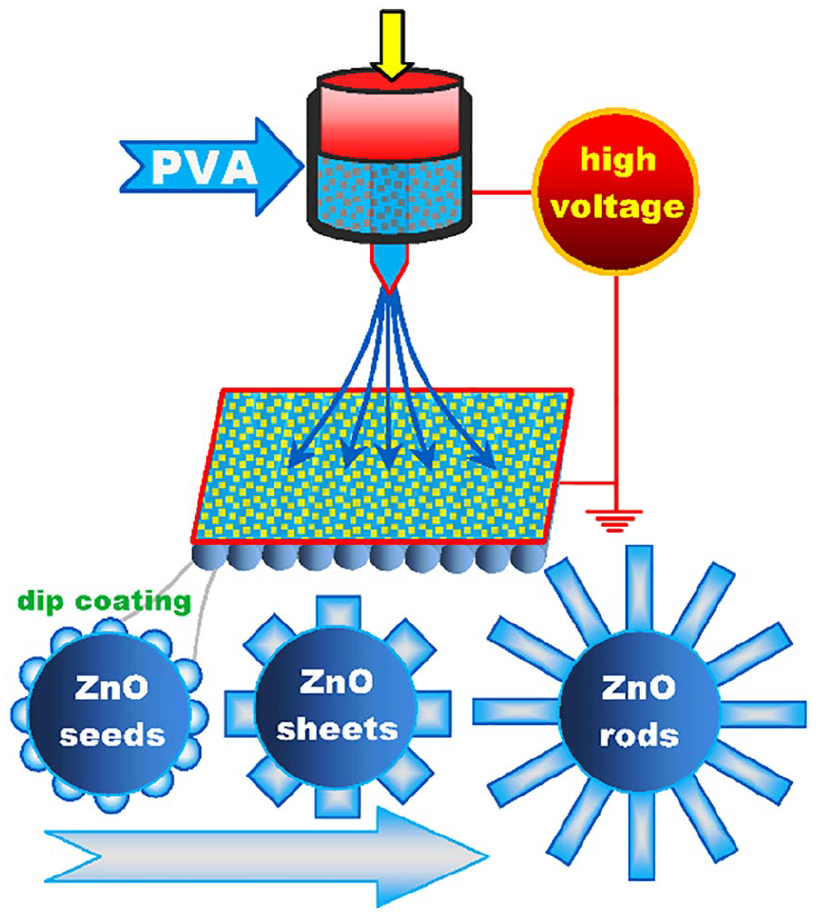
Production of ZnO nanorods and nanosheets on nanofibers according to Zhu et al. 30
Applications of ZnO nanofibers
After this short overview on recent possibilities to prepare ZnO nanofibers or coatings on nanofiber mats, here we give an insight into some of the numerous applications of ZnO nanofibers.
For the use as a highly efficient photo-detector, ZnO nanowires on highly aligned and on randomly oriented poly(vinylidene fluoride-co-trifluoroethylene) nanofiber mats were used. Kang et al. 2 found that photo-induced electrons moved approx. 4 times more effectively in the aligned electrospun mats, as compared to the randomly distributed nanofibers, and could thus increase the photo-sensitivity of the ZnO nanowire array by controlling fiber orientation and duration of the electrospinning process.
ZnO is also able to detect several gasses. Similar to other metal-oxide semiconductors, the sensing principle can be explained by trapping electrons at adsorbed molecules, followed by band bending in the semiconductor due to these charged molecules which results in a modification of the conductivity. 31 One problem of these sensors thus is the sensitivity to a specific molecule which is to be detected.
One of these molecules under investigation is hydrogen sulfide (H2S), a hazardous substance even at low concentrations. Especially when doped with La, electrospun ZnO nanofibers were found to detect H2S highly selectively and show low response times and recovery times. This effect could further be increased by tailoring the annealing temperature which defined whether the La was incorporated into the ZnO lattice or co-existed with the latter, and by optimizing the operating temperature. 24
Hydrogen peroxide (H2O2), on the other hand, could be detected by electrospun ZnO/CuO nanofiber mats on a carbon paste electrode. These electrodes were highly sensitive and selective toward H2O2 and could thus be used as H2O2 biosensors. 32
To detect H2, Kim et al. irradiated electrospun ZnO nanofibers with a high-energy electron beam of different doses. They showed that the highest dose significantly increased the selectivity toward H2 as compared to other gases, such as carbon monoxide (CO), C2H5OH, C6H6, and C7H8. In this way, the sensing performance of the ZnO nanofibers could be improved. 14
Doping electrospun ZnO nanofibers with Pd, followed by electron irradiation, also increased the response to hydrogen, which was attributed to metallization of ZnO, structural defects, the catalytic activity of Pd and the formation of ZnO-Pd heterojunctions. 33
Modification of electrospun ZnO nanofibers with praseodymium (Pr) resulted in a porous morphology and reduced ZnO crystallite sizes. This led to a doubled photoluminescence-based O2 sensing response at room temperature, making this material promising for utilization in optical gas sensing. 34
Besides sensing, ZnO can also be used for photocatalytic degradation. La-doped ZnO was grown on nanofiber mats and used for photocatalytic degradation of organophosphorus pesticides which are often used in agriculture to prevent crop from insects and rodents, but of course also harms animals and humans who are exposed to this toxin. Degradation of methyl parathion, one of these organophosphorus pesticides, by the optimized La-doped ZnO on nanofiber mats was completed after approx. 150 min, showing 100% efficiency. 35
Electrospun La-doped ZnO nanofibers were also successfully used for degradation of Congo-Red dye as a model molecule. In this study, Pascariu et al. 36 could show that recovering the catalyst, followed by thermal activation, made it applicable for dye photo-degradation again.
Rhodamine B was photodegraded from an aqueous solution by ZnO nanofibers, produced by electrospinning and subsequent calcination to remove the organic parts. 37 Adding fly ash from thermal power plants to ZnO during electrospinning resulted in increased adsorption and photocatalytic removal of methylene blue from water. 38
Similarly, PAN nanofiber mats doped with ZnO and partly additional TiO2 were used for adsorption of Cr(VI) ions from water. While doping with ZnO increased the equilibrium adsorption capacity of the PAN nanofiber mat, additional TiO2 further improved this value. Correspondingly, PAN/ZnO/TiO2 nanofiber mats could most rapidly adsorb the Cr(VI) ions from water. 39
ZnO was also suggested as part of a mixed matrix membrane, consisting from a PVA nanofiber mat on a functionalized cellulose-acetate substrate. The nanofiber mat was modified by embedding ZnO nanoparticles and sodium alginate. This composite could be used for reverse osmosis to reject 97% of the salt in sea water and was thus promising for seawater desalination. 40
Air filtration of ultrafine particles (300 nm dimension) was nearly 100% for nanofiber mats from PVA and konjac glucomannan, a polysaccharide extracted from Amorphophallus konjac with high water binding and gelation properties, 41 which were loaded with ZnO nanoparticles and thermally cross-linked. In addition to the good filtration efficiency—higher than that of commercial High Efficiency Particulate Air (HEPA) filters—the high photocatalytic activity allowed for nearly full decolorizing of methyl orange during 2 h of solar irradiation. This material was thus suggested for multifunctional usage. 42
In medical applications, electrospun gelatin/ZnO nanofibers were prepared for wound dressing. The ZnO particles, uniformly dispersed on the surface of the gelatin fibers, were not cytotoxic, but the whole nanofiber mat showed high antibacterial activity against Staphylococcus aureus and Escherichia coli. 43
Another topic in which ZnO is of interest is microwave absorption. Zhen et al. prepared porous carbon nanofibers by electrospinning PAN with ZnCl2, followed by annealing. During temperature treatment, ZnCl2 was gasified, leading to pores and formation of ZnO and Zn complexes. The high microwave absorption properties were attributed to the porous structures and the ZnO or Zn complexes increasing the dielectric loss. 15
Doping ZnO nanofibers with Co results in a bead-like structure with ferromagnetic properties and a high electromagnetic loss performance due to interfacial polarization and dipole polarization, while the magnetic loss can be attributed to magnetic hysteresis loss and residual loss. 44
In supercapacitors, carbon nanofiber mats decorated with ZnO can be used as electrodes. 27 On the other hand, electrospinning carbon precursors such as PAN, pitch and lignin with zinc acetate leads to a one-step preparation of nanofiber mats. These can afterwards be temperature-treated to create carbon nanofibers with a large specific capacitance and high energy density as well as good cycling performance. 17
Piezoelectric nano-generators were prepared from poly(vinylidene fluoride) (PVDF) with ZnO nanoparticles and nanorods as fillers. The piezoelectric properties of ZnO improved the piezoelectric nano-generators, as compared to pure PVDF, and offer a possible way to flexible self-powered electronic devices. 45 It could be shown that the solvents—here dimethylformamide and tetrahydrofuran—and their ratio influenced the output currents and voltages as well as the polymer and ZnO concentrations, the electrospinning intervals and the injection rates. 46
Similarly, piezoelectric acoustoelectric nano-generators were produced by PVDF/ZnO composite fibers which were found to produce higher voltage under low frequency sound and high sound pressure level and can thus be used for noise energy harvesting. 47
PEO can be applied as the base for a solid-state electrolyte in lithium-ion batteries. By adding TiO2 or ZnO as fillers in the electrospinning process, the ion conductivity could be increased as compared to the original unfilled electrolyte and also in comparison with film electrolytes. 48 Co-doped ZnO nanofibers were also used as photo-anodes for dye-sensitized solar cells, showing a high photo-conversion efficiency, photocurrent density and power conversion efficiency. 49
It should be mentioned that polymer/ZnO nanofiber mats can also be used as a coating for increased mechanical and chemical resistance. Polystyrene/ZnO nanofiber mats, for example, were electrospun on an aluminum alloy substrate and reduced the corrosion current of the aluminum substrate by two orders of magnitude, at the same time producing a superhydrophobic surface. 50 Similarly, PVC/ZnO nanofibers as a coating on an aluminum alloy were found to improve the corrosion resistance of the aluminum alloy. 51
Conclusion
This short review of the most recent developments and research results dealing with ZnO nanofibers or nanofiber coatings clearly shows the broad area of application for such nanostructured materials. While different methods can be applied to create nanofibers from pure ZnO, ZnO blended with a polymer or a ZnO coating around electrospun polymer nanofibers, the possible fields of application range from gas sensors to photocatalytic degradation and filter materials, from electrical applications such as solid state electrolytes or battery electrodes to anti-corrosive coatings. We hope that this brief overview will stimulate the readers to use ZnO in their own fields of research and develop further the here described methods of producing ZnO nanofibers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
