Abstract
Nanocellulose is a kind of renewable green material that comes mainly from plants. Nanocellulose aerogels have high porosity, excellent flexibility, and low thermal conductivity. They have broad application prospects in building insulation, sewage treatment, energy storage, aerospace, and other fields. The structure of nanocellulose aerogels has a significant influence on their properties, which further affects their application. In this paper, the present regulation methods of nanocellulose aerogel structure are reviewed, and the most widely used fields of nanocellulose aerogel are introduced in detail. In addition, research for improving the structure and applications of nanocellulose aerogels in the future is proposed.
Introduction
Nanocellulose (NC) is a kind of nanomaterial extracted from cellulose resources. It generally refers to cellulosic materials with at least one dimension in the nanometer range. NC is categorized into two groups: cellulose nanocrystal (CNC) and cellulose nanofibril (CNF). The dimension of CNC ranges 2-70 nm in diameter and 100-250 nm in length. CNF is characterized by diameters between 5 and 60 nm and lengths on the micrometer scale and is comprised of both amorphous and crystalline regions of cellulose. NC not only has the characteristics of the wide raw resource, renewabil-ity, biocompatibility, and biodegradability of the ordinary cellulose, but also has some other excellent advantages, such as high mechanical properties, high crystallinity, large specific surface area, and easy modification. Aerogel material is a lightweight material composed of organic or inorganic nanomaterials and filled with pores of gaseous disperse medium. As a new generation of aerogels, nanocellulose aerogels have the multi-porosity, light weight, and good thermal insulation performance characteristics of traditional aerogel. Meanwhile, nanocellulose aerogels also provide regeneration, good biocompatibility, and easy biodegrad-ability of cellulose. Moreover, nanocellulose aerogels can form many different pore structures by hydrogen bonding, which can be used as templates, and can be assembled into composite materials with different incompatible and non-interacting components for different applications. Many excellent properties can be obtained by adjusting the structure of nanocellulose aerogels, which greatly expands the application field of aerogel to include for example, solar cells, flexible electronics, building insulation, aerospace, sewage treatment, medicine, medical, and other fields.1-4
Although there is much literature on the preparation of nanocellulose aerogels, there is still little on the structure regulation of nanocellulose. In this paper, the structure control methods of nanocellulose aerogels are introduced in detail, and several main application fields are introduced.
Structure Control of Nanocellulose Aerogels
Structure Control from Material
The density, porosity, and specific surface area of aerogels made from various kinds of nanocellulose were obviously different, even with the same preparation process (see Table I).5-8 The structure of nanocellulose aerogels is significantly affected by the kinds of nanocellulose. This is because the self-assembly process of nanocellulose of various sizes differs in the drying process. As a result, aerogel structure can be controlled by controlling the kinds of nanocellulose.
Structure of Nanocellulose Aerogels with Various Kinds of Nanocellulose
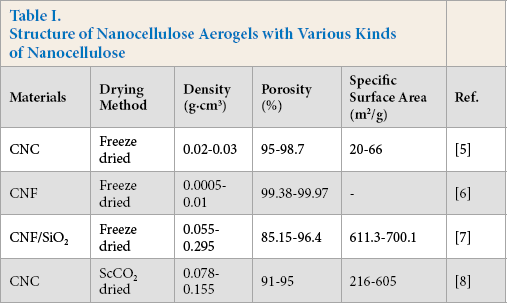
Meanwhile, the size of nanocellulose is closely related to the raw material and extraction process. The sizes and properties of nanocellulose prepared by different materials and different extraction methods are quite different. In consequence, the structure of nanocellulose aerogel can be adjusted by controlling the raw materials.
Structure Control from Preparation Method
There are many ways to control the structure of nanocel-lulose aerogels from the preparation method to improve its properties to meet the needs of different fields.
Different temperature gradients can be set to control the structure. During the drying process, different temperature gradients can affect the self-assembly process of nanocel-lulose to realize the structural control of aerogel. The process of freeze-drying is to sublimate the frozen solvent crystals in the sample through freezing, thus obtaining the unique porous structure of the sample. The porous structure of aerogels is almost a direct replica of the frozen solvent crystals. The porous structure of the aerogel is primarily defined by the morphology of the growing solvent crystals. Therefore, different temperature gradients lead to different morphology of solvent crystals during the freeze-drying process, resulting in different pore structures of aerogels. In this way, the structure of aerogel can be regulated by adjusting the temperature gradients. Jimenez-Saelices et al. used 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) oxidized nanocellulose as the raw material to prepare nanocellulose aerogel through a freeze-drying process. 9 Two different molds were used to produce ice templates to change the cooling rate and temperature gradient of the hydrogel during freeze drying. The influence of freeze-drying conditions on the structure of nanocellulose aerogels and the intrinsic relationship between microstructure and thermal insulation properties were discussed. The results showed that freeze-drying conditions had a significant effect on the microstructure of aerogels, which can be seen in Fig. 1. Zhang et al. set up several higher temperature gradients. 10 A pure physical method was adopted to heat the nanoscale hydrogel in distilled water at 60 °C, 70 °C, 80 °C, and 90 °C, to change the structure of aerogels. After freezing and drying, a nanocellulose aerogel with spherical structure was prepared. The results showed that the number of pores in the aerogel increased with the increase of heat treatment temperature. Especially at 90 °C heat treatment, the pore size of nanocellulose aerogel was concentrated at about 20 nm, which become more uniform, and its thermal insulation performance improved correspondingly. This was because after being treated at high temperatures, the hydroxyl groups on the surface of nanocellulose were gradually lost, and the surface groups polymerized again, resulting in a more uniform and compact structure, agglomeration of nanocellulose, and an increase in porosity.

SEM images of nanocellulose aerogels prepared at different temperature gradients. Reprinted with permission. 9
Inorganic salt inducement also affects the structure. Adding inorganic salts to the preparation process can accelerate the self-assembly rate of nanocellulose and change the aerogel structure of nanocellulose. Liu et al. titrated cellulose gel into a CaCl2 solution with a concentration of 0.5 mol/L by glass injector, after preparing nanoscale cellulose hydrogel with different CNF mass fraction. 11 As CaCl2 solution can accelerate the agglomeration of nanocellulose and improve the formation rate of gels, spherical CNF aerogels were prepared. This aerogel was a mesoporous material with a three-dimensional network structure. Its density was 0.0248 ∼ 0.0427 g/cm3, porosity was more than 97.33%, and pore size was less than 19.4 nm. As can be seen from Fig. 2, pore structures of the spherical CNF aerogels with differ-ent mass fractions are different. With the increase of mass fraction of CNF, the pore structure composed of filamentous nanofibers in CNF aerogel gradually changes into a lamellar honeycomb structure.

SEM micrographs of spherical CNF aerogels with various CNF mass fractions (× 20,000). Reprinted with permission. 11
The composite metal organic framework may be used to control the structure. Metal organic framework (MOF) is a new kind of porous material, which has abundant micro-pores and hybrid structures, and is connected by metal nodes and organic ligands through coordination bonds. Zhou et al. prepared CNF-MOF composite aerogels by coating and crosslinking cellulose nanofibers with continuous MOF nano layers. 12 The results showed that crosslinking made aerogels with higher mechanical strength and super elasticity. In addition, the light aerogel had a network structure and a stratified porosity, resulting in relatively low thermal conductivity (about 40 mW∣M−1K−1). Fig. 3 shows the SEM images of CNF-MOF aerogel at various magni-fications. The aerogel skeleton consisted of a network of interconnected cells with a diameter of about 10 μm. This typical cellular structure was developed during the phase separation of CNF-MOF nanofibers and water during the freeze drying process.

SEM images of CNF-MOF aerogel at various magnifications. The circled areas show the joints between the cross-linked nanofibers. MOF: metal-organic framework. Reprinted with permission. 12
Applications of Nanocellulose Aerogels
Thermal Insulation Field
Due to its three-dimensional porous network structure and gas filled pores, nanocellulose aerogels have low thermal conductivity and great potential in the field of thermal insulation. Lazzari et al. prepared a nanocellulose/biochar composite aerogel by adding a certain proportion of biochar to the nanocellulose suspension. 13 The structure and properties of the aerogel were fully studied. The results showed that the porosity of the composite aerogel was 93%∼97%, and the density was 0.01-0.027 g/cm3. The addition of 40 wt% of biochar can increase the compressive strength of aerogel by 60%. In addition, the thermal conductivity of aerogels was not affected by the addition of biochar, and the thermal conductivity of the composite aerogel is 0.021∼0.026 mW∣M−1K−1.
Han et al. used NaOH/urea aqueous solution as a template to prepare three-dimensional nanocellulose hydrogel by means of cell dissolution and coagulation, and combined Mg(OH)2 nanoparticles (MH-NP) with nanocellulose aero-gels. 14 The results showed that with the increase of MH-NP content, the density of aerogels increased, and the porosity decreased slightly. At the same time, the thermal conductivity of the composite aerogel increased from 0.056 to 0.081 mW∣M−1K−1, indicating excellent heat insulation performance of the nanocellulose aerogel.
Adsorption Field
Due to its porous structure, large specific surface area, and low density, nanocellulose aerogels have high adsorption properties for water, oil, and organic solvents. Secondly, nanocellulose aerogels are easy to modify due to the large number of hydroxyl groups on the surface. By combining with various materials, the surface roughness of cellulose aerogels can be raised, which can improve the hydrophobic-ity and oil affinity of nanocellulose aerogels. Therefore, the hydrophobicity and lipophilicity of nanocellulose aerogels can be significantly improved.
Jiao et al. successfully manufactured ultralight nanocellulose aerogels by means of chemical treatment, ultrasonic dispersion, and freeze-drying. 15 The research showed that the aerogel had ultra-low density and good hydrophobic effect. It had a good adsorption efficiency for various organic solvents and waste oils, and the oil adsorption rate can reach 53-93 g∣g−1. In addition, this kind of aerogel also exhibited good adsorption and recovery performance. The initial adsorption efficiency can maintain more than 80% after five cycles.
Fig. 4 shows the morphology of nanocellulose aerogels formed by the self-assembly of abundant cellulose nanofibrils (CNF). The cross-linked 3D network can be identified. Moreover, the inset of Fig. 4 shows that the nanocellulose aerogel with nanoscale pore structure is successfully prepared.

SEM images of CNF aerogels. Inset is the corresponding enlarged image, Reprinted with permission. 15
Energy Storage Field
Due to its high capacitance, nanocellulose aerogels can be used as supercapacitors, which have a good application prospect in the field of energy storage.
Lin et al. prepared porous nanocellulose composite aerogel electrode materials by combining nanocellulose with reduced graphene oxide (RGO) and polyaniline (PANI). 16 Nanocellulose composite aerogel supercapacitors were prepared by using polyvinyl alcohol/sulfuric acid (PVA/ H2SO4) as the electrolyte. Because of the porous structure of aerogels and the hydrophilicity of nanocellulose, the diffusion distance of the electrolyte can be shortened, and the area of the active layer of RGO infiltrated by electrolyte ions can be significantly increased. Therefore, a composite aerogel superconductor with stable structure and excellent electrochemical performance can be obtained.
The above three application fields are the most widely used fields for nanocellulose aerogels. To broaden the application of nanocellulose aerogels, it is necessary to continue to explore ways to optimize the structure of nanocellulose aerogels and prepare aerogels with better properties to meet the needs of other fields.
Conclusion and Prospects
Nanocellulose aerogels not only have the environmentally friendly, renewability, biocompatibility, and biodegradability of cellulose, but also have excellent properties such as low density, high porosity, and high specific surface area. Due to their excellent properties, nanocellulose aerogels have broad application prospects in many fields.
The structure of nanocellulose aerogels affects their properties and applications. This paper mainly introduces the specific methods of controlling nanocellulose aerogel from the two perspectives of raw material and preparation process. The applications of nanocellulose aerogels in thermal insulation, adsorption, and energy storage are introduced in detail.
There are still some problems in the preparation and modification of nanocellulose aerogels. First, the size of the nanocellulose cannot be precisely controlled. Its size is significantly affected by the raw materials and preparation methods. In addition, the nanofibers are easy to self-assemble in the drying process. At present, the size can only be roughly divided into cellulose nanocrystal (CNC) and cellulose nanofibril (CNF) according to the length. Secondly, some modification methods of nanocellulose aerogels (such as silane coupling agents) are more complex and expensive, thus cannot meet the requirements of industrial production. Thirdly, the strength and stability of nanocellulose aerogels, such as thermal stability and repeated adsorption, are still not available in many practical applications. Therefore, in future research, it is necessary to better understand the relationship between the size and properties of nanocellulose and cellulose materials and production technologies, as well as the influencing factors of nanocellulose aerogel structure. This is because, as with all natural products, difference among batches will always exist. Therefore, shortening the preparation cycles, reducing the preparation cost, simplifying the production process, improving the strength and structure of nanocellulose aerogels, and more tightly controlling the structure of nanocellulose aerogels to improve their properties will be the focus of future research.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Project No. 51903152), the Shanghai Science and Technology commission (Project No. 19YF1417900), and the Research Initiation Funds of Shanghai University of Engineering Science (Project No. E3-0507-19-05164).
