Abstract
Poly[tetra(tetramethylcyclosiloxyl-piperazin)-phosphinic acid methyl ether) (PNCTSi) was successfully synthesized, and its chemical structure was investigated by Fourier transform infrared spectra (FTIR) and nuclear magnetic resonance (1H-NMR and 13C-NMR) spectroscopy. The flammability and thermal decomposition behaviors of cotton fabrics treated with PNCTSi were systematically evaluated by limiting oxygen index (LOI), vertical burning test (UL-94), thermogravimetric analysis (TGA), and cone calorimetry. As a result, the LOI value reached 30.1% and the char length decreased to 10.3 cm when cotton fabrics were treated in 300 g/L of PNCTSi solution for 30 min. According to TGA results, the residual rate of treated cotton fabrics at 800 °C increased to 16.4%. The peak heat release rate (PHRR) and CO2/CO ratio significantly decreased.
Introduction
Cotton fabrics are widely used in many fields, including apparel and industrial textiles, due to its superior performance such as comfort, softness, and durability.1-3 However, flammability is a main drawback of cotton fabric, which limits further applications in some special fields. Therefore, flame retardant treatment of cotton fabric is desirable for some uses.
Halogen-containing flame retardants have superior flame retardant efficiency, but poisonous gases, such as hydrogen halide and dioxins, can be released in the process of thermal degradation, so it is gradually being replaced by some natural biomolecules, including polysaccharides, lignin, deoxyribose nucleic acid (DNA), and proteins.4-6
Phosphorus-containing flame retardants can generate phosphoric acid or polyphosphoric acid during thermal degradation. These can subsequently undergo esterification reaction with hydroxyl groups on the surface during combustion, which leads to cotton fiber dehydration, formation of a carbonized layer on the surface of the cotton fibers, and isolation of oxygen and heat transfer.
As nitrogen and phosphorus have synergistic inhibitory effects, nitrogen-containing flame retardants can accelerate the oxidation of phosphorus during thermal decomposition, and generate non-combustible gases, such as ammonia and nitrogen oxides, which are able to dilute the oxygen concentration.7-10 For example, Alongi et al. reduced the flammability of cotton fabrics by using sol-gel treatment and the phosphorus-nitrogen synergy.11,12
Polysiloxane, an organic compound with an -Si-O- bond in the main chain and a large number of side groups, has flame retardancy. Similar to the flame retardant mechanism of phosphorus, it can produce a silicon char layer during thermal decomposition, providing anti-melting drops and little smoke.13-15 The softness of cotton fabrics does not change after treatment. In addition, because of the side groups, it is easy for cotton fabrics to modify.
Recently, some compounds containing Si/P/N have received much attention due to their excellent char-forming ability. For example, Chen et al. synthesized a linear piperazine/phosphorus/polysiloxane copolymer (a, ω-di[(4-butoxypiperazin-1-yl)-phosphinic acid methyl ether]-terminated linear polysiloxane) and applied it to cotton fabrics. 16 As a result, the LOI value and char residue of treated cotton fabrics were increased to 30% and 22%, respectively. Dong et al. synthesized a linear guanidyl-containing and phosphorus-containing polysiloxane flame retardant, which can reduce the release of volatiles and accelerate the formation of char layers.17,18 However, studies on flame retardants based on cyclic polysiloxanes have rarely been reported.19,20 Recently, Liu et al. synthesized a novel polysiloxane quaternary ammonium salt (NCTSi) and used it as an antibacterial and hydrophobic agent for cotton fabrics, 19 inspiring the use of cyclic polysiloxanes as flame retardants. Cyclic polysiloxanes have four active sites,20,21 which can be connected to more phosphorus- or nitrogen-containing groups, increasing the content of phosphorus and nitrogen in the flame retardant, so that the condensed- and gas-phase flame retardancy can be better imparted during combustion. At the same time, the cyclic polysiloxane skeleton (as a flame retardant matrix) can form a “cake” molecular structure, which can be crosslinked with each other during burning, which is more conducive to forming a dense SiO2 film to prevent transfer of heat and oxygen.
In this study, cotton fabrics were treated with poly[tetra(tetramethylcyclosiloxyl-piperazin)-phosphinic acid methyl ether] (PNCTSi). The resulting flame resistance and thermal stability of treated cotton fabric were evaluated by limiting oxygen index (LOI), vertical burning test, thermogravimetric analysis (TGA), and cone calorimetry (CONE). The surface morphology of the fabric was evaluated by scanning electron microscopy (SEM). The elemental composition was determined by energy dispersive spectrometry (EDS). It was found that the treated cotton fabrics displayed both gas phase and condensed phase flame resistant mechanisms.
Experimental
Materials
Scoured and bleached 100% plain woven cotton fabrics (14.75 × 14.75 tex, 122 g/m2) were supplied by Weifang Qirong Textiles Co. Ltd, Weifang, China. Phosphorus oxychloride (POCl3), triethylamine (Et3N), 1,2,3,4-tetracarboxylic acid (BTCA), and piperazine were obtained from Sinopharm Chemical Reagent Co. Ltd. n-Hexane, tetrahydrofuran (THF), and methanol were obtained from Tianjin Fu Yu Fine Chemical Co. Ltd. Methyltriethoxysilane was supplied by Shanghai Macklin Biochemical Co. Ltd. Chloromethyl dimethylchlorosilane was purchased from Hunan Huateng Pharmaceutical Co. Ltd. THF was distilled from Na/benzophenone. Other reagents were used without further purification.
Measurements
LOI tests of untreated and treated cotton fabrics were measured according to GB/T 5454-2009, 22 with a LYF-606B digital display oxygen index apparatus. The vertical flammability of cotton fabrics were tested based on the GB/T 5455-2014 standard 23 using an LYF-606B vertical burning tester.
TGA was investigated using a TGA851 thermal analyzer. Samples were tested under a heating rate of 10 °C/min from 25 °C to 800 °C. The combustion character of the samples was tested on a cone calorimeter (Fire Testing Technology Ltd.) under a heating flux of 35 kW/m2 according to the ISO 5660-1 standard. 24 Two measurements were made to get the average. Whenever the first two deviated from each other by more than 20% in any characteristic parameter, a third sample was measured.
SEM was performed using a JSM-6010LA SEM apparatus (JEOL) to study the surface morphologies of treated cotton fabrics and the residue of treated cotton fabrics after combustion. A JSM-6700F (JEOL) elemental analyzer was used to confirm the elemental composition of the cotton fabrics.
Synthesis of PNCTSi
Poly[tetra(tetramethylcyclosiloxyl-piperazin)-phosphinic acid methyl ether] (PNCTSi) was prepared using chloromethyl dimethylchlorosilane, methyltriethoxysilane, phosphorus oxychloride, and piperazine. 25 The structure of PNCTSi is shown in Scheme 1 and was verified by 1H-NMR, 13C-NMR, and FT-IR spectroscopy. 1H-NMR: δ 0.07 (inner cyclic siloxane groups of SiCH3), 0.08 (outer cyclic siloxane groups of SiCH3), 1.39 (CH2), 3.077 (CH2 on piperazine), 3.6 (P-O-CH3). 13C-NMR: δ -3.11 (inner cyclic siloxane groups of SiCH3), -1.84 (outer cyclic siloxane groups of SiCH3), 0.76 (Si-CH2-N), 8.37 (carbon on piperazine), 45.60 (P-O-C). FT-IR (cm−1): 1592 (C-N), 1447 (CH2), 1261 (P=O), 755 (P-N), 1017, 839 (Si-O-Si).
Cotton Fabric Treatment
PNCTSi was dissolved in acetone/water (1:1, v/v) to prepare a 300 g/L PNCTSi solution. Urea and BTCA were then added to it as a solubilizer and crosslinking agent, respectively (the percentage of urea was 5% (w/w), and the percentage of BTCA was 10% (w/w), cotton:bath = 1:10). Then, cotton fabrics were soaked in the bath for 10, 20, and 30 min, respectively. Next, two dips and two nips were used to create a wet pickup of ∼80%, the cotton fabrics were dried in an oven at 90 °C for 20 min, and cured at 110 °C for 5 min.
Results and Discussion
Flammability
The value of the LOI and vertical burning test results of the treated cotton fabric with PNCTSi are illustrated in Table I and Fig. 1. The LOI value of the treated cotton fabrics was greatly improved compared with the untreated cotton fabrics, showing excellent flame resistance. Among them, the LOI value of the cotton fabrics treated with PNCTSi for 30 min reached 30.1% and the char length decreased to 10.3 cm without after-flame time and after-glow time. While the LOI value of the untreated cotton fabrics was 18%, the vertical burning test could not be completed. These results were mainly due to the synergistic flame retardant effect of Si/P/N, indicating that PNCTSi imparted excellent flame resistant properties to the treated cotton fabrics.
Flame Resistance Test Data for Treated Cotton Fabric
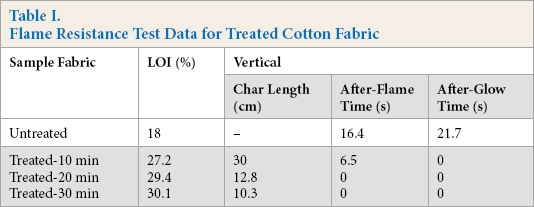

Photographs of cotton fabrics treated at various treatment times after vertical burning tests.
Combustion Behavior of Cotton Fabrics
The TGA and DTG curves of untreated and PNCTSi-treated cotton fabrics under air and nitrogen atmospheres are shown in Fig. 2, and some detailed data are listed in Table II. Under air atmosphere (Figs. 2a and c), the onset degradation temperature (T5%) of untreated cotton fabrics was ∼297 °C. Its main mass loss stage was between 300 °C to 370 °C, resulting from the depolymerization of cellulose through transglycosylation reactions. 26 The maximum-rate degradation temperature appeared at 344 °C. With increased temperature, the residue of the untreated cotton fabrics continued to decrease and almost no residue remained at 800 °C. However, there were two stages in the thermal decomposition process of the treated cotton fabrics, and the initial decomposition temperature of each stage was less compared to that of the untreated cotton fabrics. The decomposition temperature of the first stage was from 205 °C to 246 °C, which may be due to the dehydration of cotton fabrics and breakage of the chemical bond in PNCTSi. The second stage of weight loss was from 263 °C to 315 °C, and the rate of thermal weight loss of the treated cotton fabrics was significantly less than that of the untreated cotton fabrics, because phosphorus can promote the dehydration of cotton fabrics into char during thermal decomposition. Cyclic polysiloxane can produce a layer of SiO2 char on the surface of cotton fabrics, which has the function of insulating air. In addition, nitrogen generates non-combustible nitrogen oxides, which acts to dilute the combustible gas and air. The untreated cotton fabrics were completely burned at 494 °C, while the residue ratio of the treated cotton fabric at 800 °C was 16.4%. The results were more pronounced in a nitrogen atmosphere (Figs. 2b and d).
TGA Data of Untreated and 30 min PNCTSi-Treated Cotton Fabrics
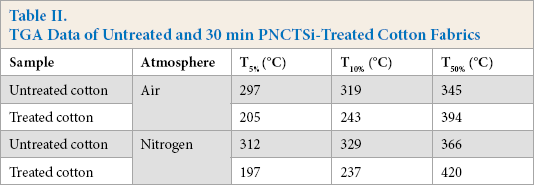
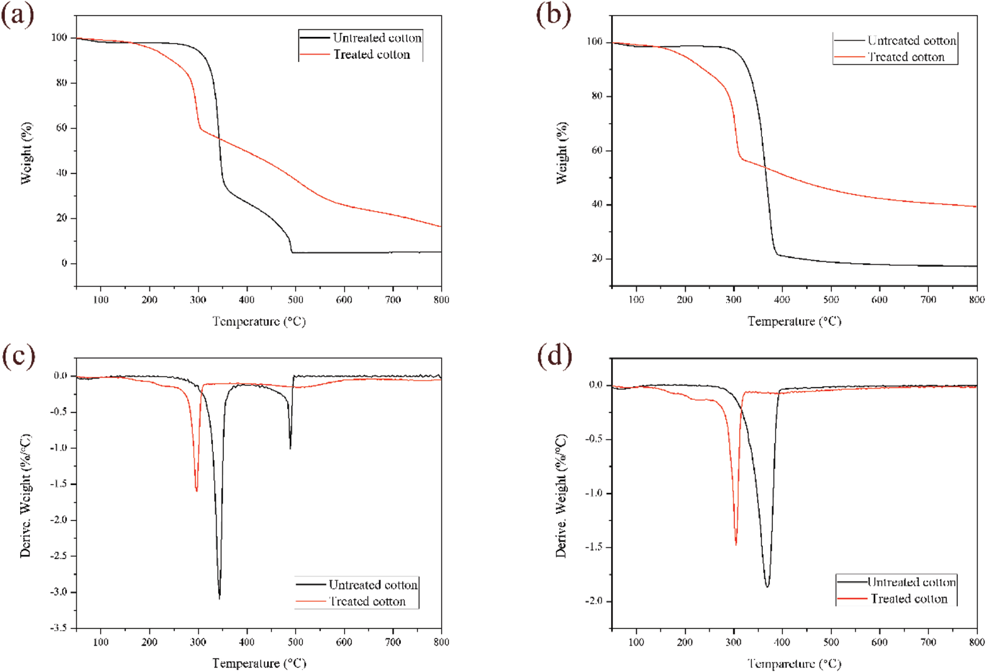
TGA and DTG curves of untreated and treated (30 min) cotton fabrics in air (a and c) and nitrogen (b and d) atmospheres.
Although the initial decomposition temperature of the two stages was almost the same as in the air atmosphere, the char residue of the treated cotton fabric reached 39.1% at 800 °C, which was greater than that in air. The above results indicate that PNCTSi improved the stability of the cotton fabrics.
In this work, cone calorimetry (CONE) tests are used to assess the combustion behavior of treated fabric. Various parameters obtained from the cone calorimetry tests are summarized in Table III, and the curves of heat release rate (HRR), total heat release (THR), effective heat of combustion (EHC), and mass loss are illustrated in Fig. 3. As shown in Fig. 3, the test time of the cotton fabrics treated with PNCTSi was slightly longer than that of the untreated cotton fabrics. With the introduction of PNCTSi, the HRR and the THR were less than those of the untreated cotton fabrics at the same stage, and the peak of heat release rate reduced from 142.1 to 65.8 kW/ m2. The formation of the char layer is believed to be primarily responsible for reducing the HRR during the CONE test. 27 The char layer can isolate heat and prevent oxygen exchange, so the more char residue formed during combustion, the better the barrier protection effect. It is reasonable to regard the flame resistant mechanism of treated cotton fabrics as a condensed phase mechanism in terms of the HRR and THR results. This conclusion is consistent with the TGA result.
CONE Data for 30 min Treated and Untreated Cotton Fabric a
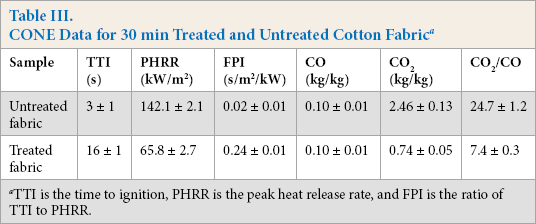
TTI is the time to ignition, PHRR is the peak heat release rate, and FPI is the ratio of TTI to PHRR.
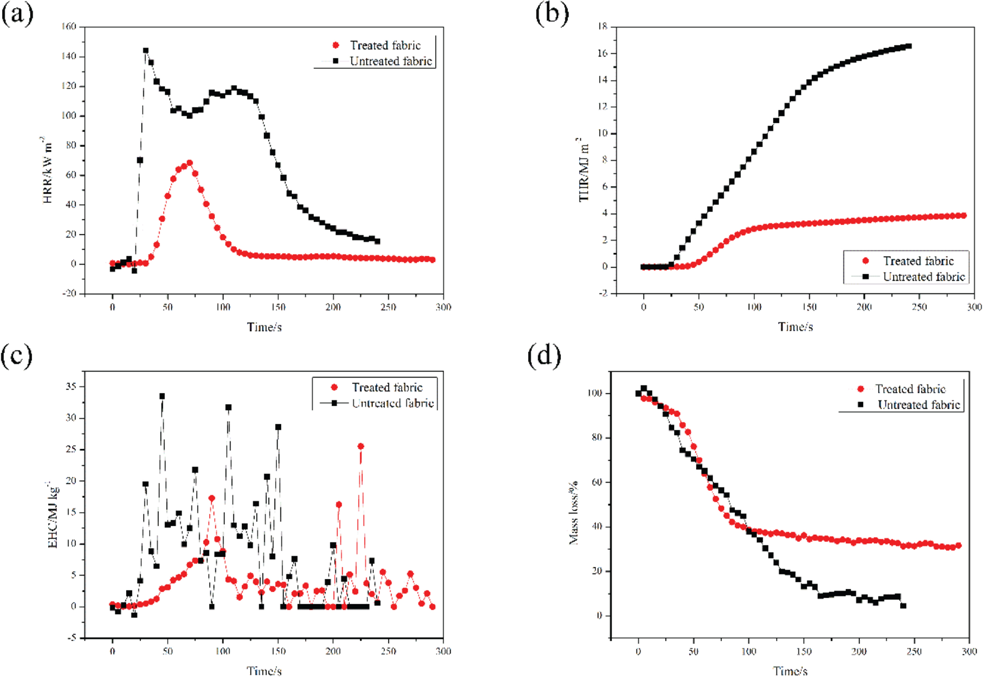
Conical calorimetry test curves of cotton fabrics. (a) HRR curve of cotton fabrics, (b) THR curve of cotton fabrics, (c) EHC curve of cotton fabrics, and (d) mass curve of cotton fabrics.
EHC is defined as the ratio of HRR to mass loss rate, which is an important parameter to evaluate the burning degree of the volatile species generated from the materials during combustion. As is well-known, the smaller amount of combustion heat generated during combustion is ascribed to the incomplete combustion of volatile species produced by materials, implying a gas-phase fire-inhibition activity. The EHC values of treated cotton fabric decreased to some extent as compared to that of untreated cotton fabric. The mass loss curve shows that the residues of the treated cotton fabric was significantly greater than that of untreated cotton fabric. FPI, the ratio of the time of ignition (TTI) to the peak heat release rate (PHRR), is one of the important parameters to evaluate flame resistance. The higher FPI (0.24) indicated a better flame resistance. At the same time, the CO release of untreated cotton fabric and treated cotton fabric were basically the same, but the difference in CO2 emissions was huge. The ratio of CO2/ CO decreased from 24.7 to 7.4 compared with the untreated cotton fabric. This meant that PNCTSi prevented most of the reaction between CO and O2, thereby reducing the CO2 production. 28 The above results indicated that PNCTSi can impart superior flame resistance to cotton fabrics.
SEM and EDS Analysis
SEM analysis was used to learn more about the surface morphology of the treated cotton fabric. It is observed from Figs. 4a and e that the surface of the untreated cotton fabric was relatively smooth, while a film-like structure was adsorbed uniformly on the surface of the treated cotton fabric, which was due to the adhesion of PNCTSi to the surface of the cotton fabric (Figs. 4b and f). Figs. 4c and g presented the SEM morphology of the char residue of the untreated fiber. The fiber bundle was completely burned, while the original integrated shape of the cotton fiber remained after combustion for the treated cotton fabric (Fig. 4d). At high magnification (Fig. 4h), a change from an expanded bubble structure to the floc on surface of char was observed. A possible reason for this phenomenon is that the nitrogen oxide gas generated by the thermal decomposition of PNCTSi caused bubbling of the phosphorus-silicon char layer formed on the surface of the cotton fiber, and a part of the bubbles was broken to form flocs.16,29,30 The above results indicate that PNCTSi had a synergistic flame resistance effect.

SEM images of untreated cotton fabric (a and e), treated cotton fabric (b and f), residues of untreated cotton fabric (e and g), and treated cotton fabric (d and h).
The elemental composition of the cotton fabric was confirmed by EDS. It can be seen from Figs. 5 and 6 that the elements contained in PNCTSi were evenly distributed on the surface of treated cotton fabric, while only C and O exist on the surface of the untreated cotton fabric (other scanning signals are impurities), as shown in Fig. 7. A change in the composition of the elements can be found by comparing the EDS images of cotton fabrics before and after treatment. Fig. 7a shows the presence of only C and O in cotton fabric, while the presence of Si, P, and N were observed in the treated cotton fabric. The proportion of each element was consistent with the element ratio of PNCTSi, as shown in Fig. 7b, which demonstrates that PNCTSi was successfully applied to the cotton fabrics.

EDS mapping of treated cotton fabric with PNCTSi.
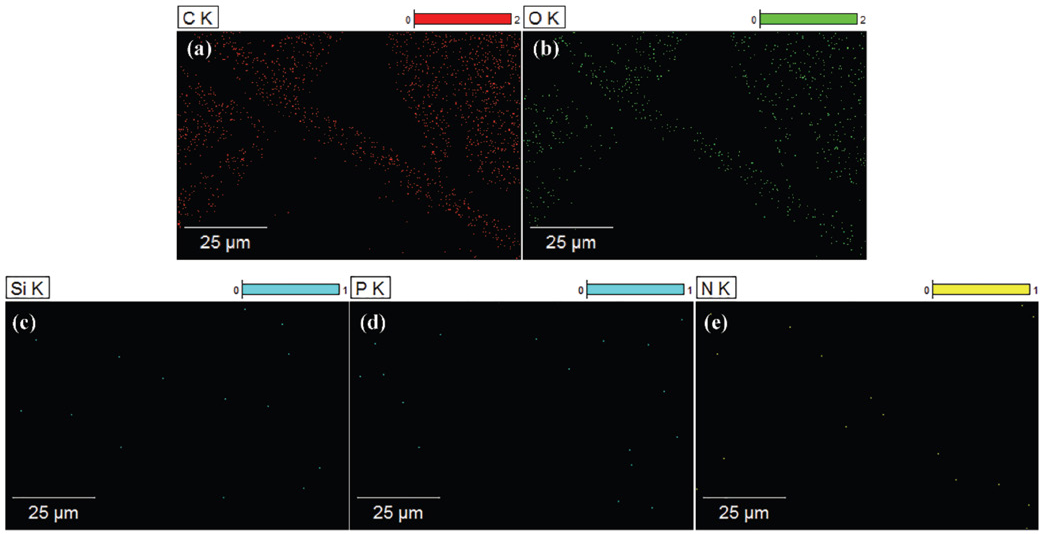
EDS mapping of untreated cotton fabric.
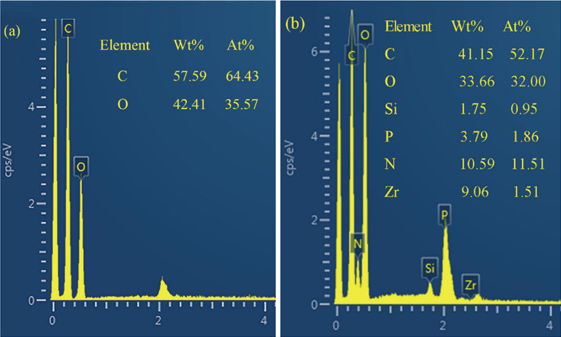
EDS images of untreated (a) and treated (b) cotton fabric with PNCTSi.
Washing Durability
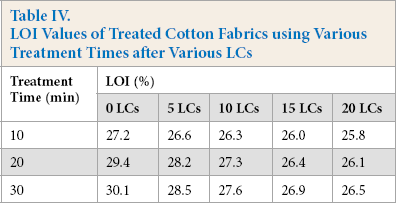
The flame resistance of treated cotton fabric after various laundering cycles (LCs) is shown in Table IV. Although the LOI decreased with increasing washing cycles, treated cotton fabric still achieved excellent washing durability. After washing 10 times, the LOI value of cotton fabric treated with 300 g/L PNCTSi for 30 min was 27.6%, and the LOI value still remained above 26% after 20 washing cycles. The above data demonstrated that PNCTSi had excellent flame resistant durability.
LOI Values of Treated Cotton Fabrics using Various Treatment Times after Various LCs
Conclusion
A novel flame retardant (PNCTSi) was successfully synthesized using triethoxymethylsilane, anhydrous piperazine, and phosphorus oxychloride. With the incorporation of PNCTSi, the flame resistance of cotton fabrics was greatly improved. When cotton fabrics were treated in 300 g/L of PNCTSi solution for 30 min, the LOI showed a favorable increase to 30.1%. Meanwhile, the results of TGA and CONE indicated that the flame resistance of cotton fabrics was improved significantly and the burning risk of cotton fabrics was effectively reduced because of the addition of PNCTSi. Its fame-resistance mechanism was well proved through SEM, showing that PNCTSi exerted predominant fire inhibition activity via generation of phosphoric acid, polyphosphoric acid, and nitrogen-containing volatiles. In summary, the PNCTSi is a flame retardant agent for treatment of cotton fabric that had excellent performance.
