Abstract
Flame resistant cotton fabric is usually dyed first, and is then treated with a flame retardant by the pad-dry-cure technique. In this research, cotton fabric was treated with 2-(2-aminoethyl hydrogen phosphite)-4,6-dichloro-1,3,5-triazine (APDCT). APDCT contains s-triazine groups, which are the same used by reactive dyes. This process allows cotton fabric dyeing and flame retardant treatment to occur simultaneously, while decreasing treatment temperature, improving efficiency, and saving energy. Optimal treatment was determined by the percent dye uptake, fixation, and fabric flame resistance. The color fastness to rubbing of the treated cotton fabric was almost unchanged. The tensile strength of the treated cotton fabric was slightly reduced.
Introduction
One of the most widely used fibers, cotton, is flammable. Thus, the flame resistance of cotton fabric is especially important. Greater attention has been paid to the use of halogen-free flame retardants on textiles in the last two decades.1–5 The durability, toxicity, and formaldehyde release of flame retardants should be considered. Research indicates that multi-element flame retardants can play a synergistic role in fabric flame resistance.6–10
Most of the flame retardants used to treat cotton fabric are applied by the traditional pad-dry-cure technique, leading to fabric strength decrease and yellowing.11–19 The temperature of this process is usually higher than 140°C, dehydrating glucose residues in the cellulose, decreasing the degree of polymerization, and increasing the number of carbonyl and carboxyl groups. It also causes other chemical changes. 20 In addition, use of crosslinking agents leads to cotton fabric yellowing after high temperature treatment. To solve this problem, researchers have designed flame retardants that can react with cotton fabric at lower temperatures. 21
Flame resistant fabrics usually need be treated with a flame retardant after dyeing. The two-step method consumes water resources and is costly. With the emphasis on energy saving, emission reduction, and environmental protection, shortening the finishing process has become an important research topic for researchers. Cao et al. treated cotton fabric with reactive dyes and reactive ultraviolet absorbents in one bath by making use of the same reaction mechanism. 22 This method improved the production efficiency and simplified the dyeing and finishing process. However, little research has been done on the process of combining dyeing with flame resistance for cotton fabric. Therefore, it is of great significance to propose a facile and low-cost process to produce flame resistant fabric.
Reactive dyes have become widely used in cellulosic fabric dyeing, as it is a low cost, simple dyeing method, leading to good wet fastness. 23 They can react directly with the hydroxyl group on the cellulose fiber, thereby fixing the dye on the fabric. Triazine groups have excellent dyeing properties, but they are seldom used in the synthesis of flame retardants at present.
We synthesized the flame retardant 2-(2-aminoethyl hydrogen phosphite)-4,6-dichloro-1,3,5-triazine (APDCT). It contains s-triazine groups, which are the same as the active groups of reactive dyes. 24 Due to synergistic effects, cotton fabrics treated with APDCT have excellent flame resistance. APDCT can react with cotton fabrics, and thereby improve the durability of the attached flame retardant. The strength and whiteness of the finished fabric changed very little. In addition, this type of flame retardant does not have the problem of formaldehyde release.
In this study, cotton fabrics were treated with APDCT during the dyeing process. This method can significantly shorten the production process and reduce production costs. In addition, this method can effectively reduce the production of wastewater in the dyeing and finishing process. 25 Optimal process conditions were determined by the study, including the amount of APDCT, the amount of C.I. Reactive Red 2, the dosage of sodium chloride, and the dosage of sodium carbonate.
Experimental
Material
APDCT was synthesized in our lab, which has been reported previously (Fig. 1). 24 Sourced and bleached 100% plain-woven cotton fabric was supplied by Weifang Qirong Textiles Co. Ltd. Sodium carbonate was purchased from Tianjin Ruijinte Chemical Reagent Co. Ltd. Sodium chloride was supplied from Fine Chemical Factory of the Laiyang Economic and Technological Development Zone. C.I. Reactive Red 2 was supplied by Shanghai Pudong Dyeing Co. Ltd.
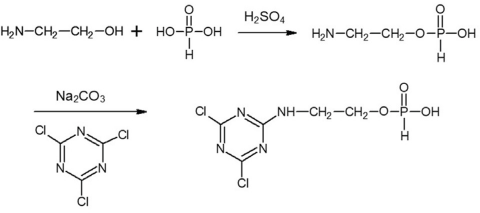
Synthesis of APDCT.
Dyeing Process
APDCT and Reactive Red 2 were added to the finishing bath at room temperature (RT). Sodium chloride was added to the finishing bath 15 min later. And then sodium carbonate was added into it. The dyeing process is shown in Fig. 2. The treated cotton fabric was then rinsed and soaped. Finally, it was rinsed again and dried. The reaction equation is shown in Fig. 3.
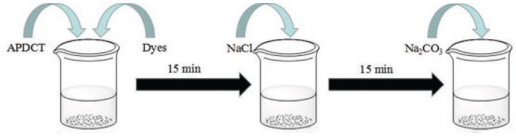
The process of treating cotton fabric with APDCT and Reactive Red 2.
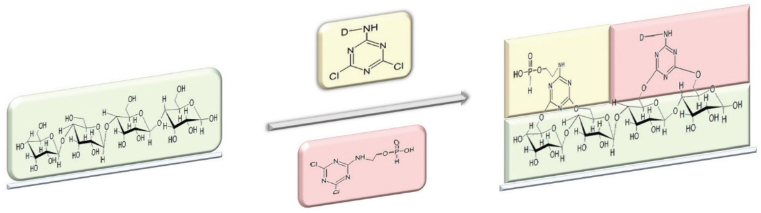
The reaction of treating cotton fabric with APDCT and Reactive Red 2.
In the presence of alkali, cotton fibers form cellulose fiber hydroxyl anion (Cell-O−) which can displace the chlorine atoms in triazine by nucleophilic addition, forming a covalent bond. Chlorine atoms of triazines are eliminated under alkaline conditions and at higher temperatures. Thus, Reactive Red 2 covalently reacts with cotton fiber, and APDCT, having the same reactive triazine group, does the same. APDCT competes with reactive dyes for bonding to cotton fibers during the finishing process.
Analysis
Fourier Transform Infrared
Fourier transform infrared (FTIR) analysis was conducted by using a Nicolet 5700 Fourier transform infrared spectrometer (Termo Nicolet Corporation, USA) using the KBr pressed-disk technique.
Limiting Oxygen Index
The limiting oxygen index (LOI) was used as per GB/T 5454-1997. The sample clip with cotton fabric was made according to the required size and was placed in the combustion cylinder. The proportion of oxygen and nitrogen was adjusted, and then the specific igniter ignited the cotton fabric. When the fabric extinguished just after 2 min or the damage length of the cotton fabric was 4 cm, the oxygen volume fraction was the limit oxygen value of the sample. The greater the LOI value, the better the flame resistance of the cotton fabric.
Tensile Strength
The tensile strength test was determined as per GB/T 3923.1-2013, using an LFY-201D multifunction fabric power machine.
Color Fastness
Color fastness was determined as per GB/T 3920-2008, using an LFY-304 textiles rubbing fastness tester.
Percent Dye Uptake
The maximum absorption wavelength of Reactive Red 2 was determined using a 721 spectrophotometer. The absorbance of the finishing bath before and after finishing was tested at the maximum absorption wavelength. The formula for calculating the dye uptake percentage
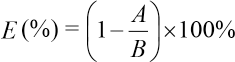
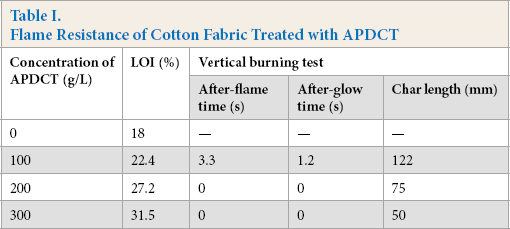
Flame Resistance of Cotton Fabric Treated with APDCT
Fixation
The treated cotton fabric was put into a solution containing 2 g/L of soap chips and 2 g/L of Na2CO3 at 95°C with a bath ratio of 1:30. The same solution was prepared as the standard soap solution. After soaping, the absorbance of the mixture of soaping residue and washing liquid and standard soap liquid were measured. The formula for calculating the fixation
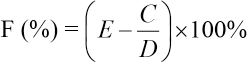
Results and Discussion
The synthesis of APDCT was reported previously (Fig. 1). 24 The flame resistance of treated cotton fabric was investigated by LOI and vertical burning tests, as shown in Table I. 24 The cotton fabric treated with APDCT has good flame resistance.
FTIR Analysis
The structure of APDCT was investigated by FTIR analysis (Fig. 4). The absorption peak at 3440 cm–1 was due to the N-H stretching vibration. The absorption peak at 3370 cm–1 was due to the -OH stretching vibration. The absorption peak at 2460 cm–1 was assigned to the P-H stretching vibration. The peaks at 1720, 1610, 1400, 825, and 770 cm–1 were attributed to triazine. The absorption peak at 1150 cm–1 was assigned to the P=O stretching vibration. The absorption peak at 1067 cm–1 was assigned to the P-O-C stretching vibration. These results confirmed that APDCT was synthesized successfully.
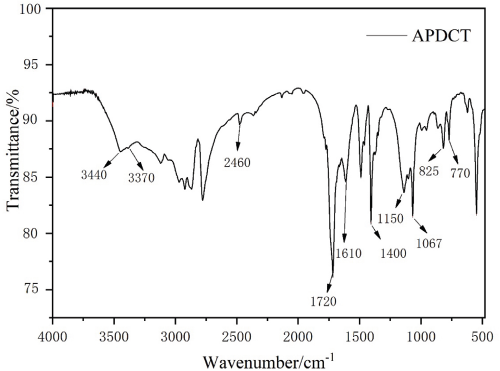
FTIR spectra of APDCT.
Effect of Reactive Red 2 and APDCT Concentrations on Dyeing and Flame Resistance of Cotton Fabric
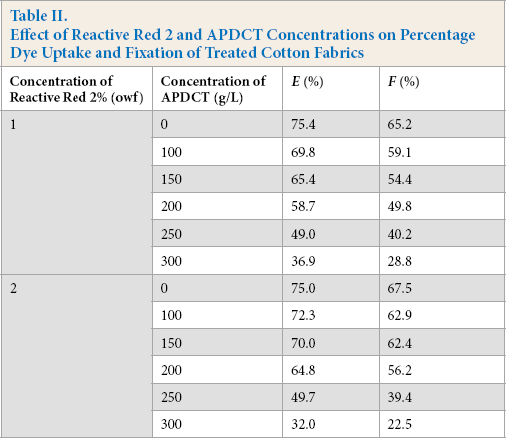
The effect of various concentrations of Reactive Red 2 and APDCT on the dye uptake percentage and fixation (Table II), and flame resistance of cotton fabrics, were studied. With increased APDCT concentration, the dye uptake percentage and the fixation of Reactive Red 2 decreased. When the concentration of APDCT was low, the dye uptake percentage and fixation rate for 2% Reactive Red 2 were slower than for 1% Reactive Red 2. The dye uptake percentage and fixation of 2% Reactive Red 2 decreased with increased APDCT concentration. Since the reactive group of Reactive Red 2 and APDCT are the same, there is competition between the two in the finishing bath, leading dye uptake percentage and fixation decreases. As the APDCT concentration increased, relative increases in the LOI value became smaller (Fig. 5). Overall, the optimal concentrations of Reactive Red 2 and APDCT were 2% and 200 g/L, respectively.
Effect of Reactive Red 2 and APDCT Concentrations on Percentage Dye Uptake and Fixation of Treated Cotton Fabrics
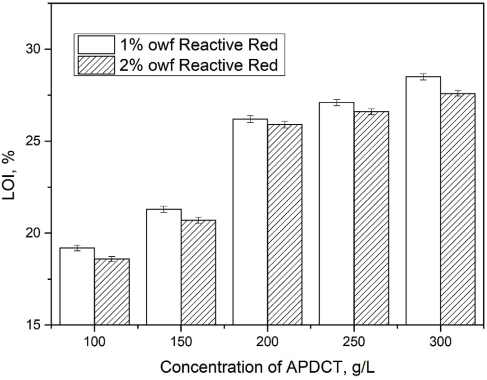
Effect of Reactive Red 2 and APDCT concentrations on flame resistance of treated cotton fabric.
Effect of Sodium Chloride Concentration on Dyeing and Flame Resistance of Cotton Fabric
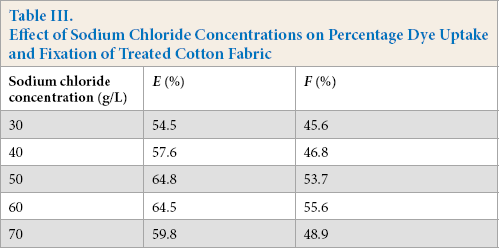
With increased sodium chloride dosage, the percentage dye uptake and fixation of Reactive Red 2 first increased, and then decreased (Table III, Figs. 6 and 7). When the concentration of sodium chloride was 50 g/L, the percentage dye uptake was 64.8%. When the concentration of sodium chloride was 30–50 g/L, the increase in flame resistance was more prominent. Due to the addition of sodium chloride, the electrolyte concentration in the finishing solution increased, improving the ionic strength of the finishing system, and accelerating the reaction, thus promoting dyeing. The optimal concentration of sodium chloride was 50 g/L.
Effect of Sodium Chloride Concentrations on Percentage Dye Uptake and Fixation of Treated Cotton Fabric

Effect of sodium chloride concentrations on dyeing of treated cotton fabric. (a) 30 g/L, (b) 40 g/L, (c) 50 g/L, (d) 60 g/L, and (e) 70 g/L.
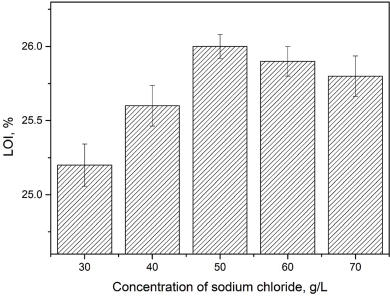
Effect of sodium chloride concentrations on flame resistance of treated cotton fabric.
Effect of Sodium Carbonate Concentration on Dyeing and Flame Resistance of Cotton Fabric
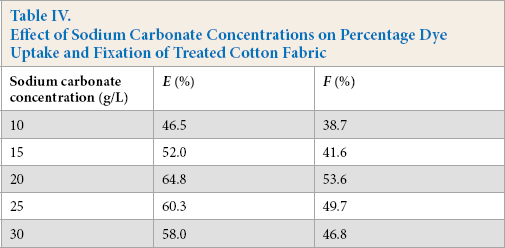
Sodium carbonate had a distinct fixation effect on the dyeing process of Reactive Red 2 (Table IV, Figs. 8 and 9). When the concentration of sodium carbonate was increased from 10 to 20 g/L, the percentage dye uptake and fixation were greatly improved. Cellulose, containing hydroxyl groups, in cotton is transformed into alkali cellulose under alkaline conditions, facilitating the reaction with triazine. However, greater amounts of sodium carbonate also accelerates the hydrolysis of triazine chlorine groups that otherwise would react with the alkali cellulose. This hydrolysis has a negative effect on the dyeing and flame resistance of cotton fabric in the same bath. The optimal concentration of sodium carbonate was determined to be 20 g/L.
Effect of Sodium Carbonate Concentrations on Percentage Dye Uptake and Fixation of Treated Cotton Fabric

Effect of sodium carbonate concentrations on dyeing of treated cotton fabric. (a)10 g/L, (b) 15 g/L, (c) 20 g/L, (d) 25 g/L, and (e) 30 g/L.
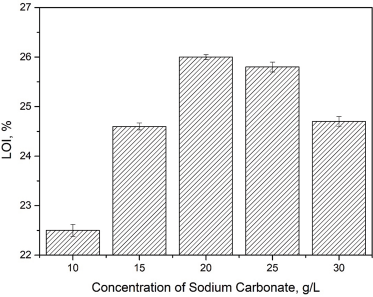
Effect of sodium carbonate concentrations on flame resistance of treated cotton fabric.
Color Fastness and Tensile Strength of Treated Cotton Fabric
The color fastness of the cotton fabric changed little with finishing (Table V). Finishing has little effect on the color fastness to friction of cotton fabric. The warp tensile strength of the cotton fabric with flame-retardant finishing decreased from 655.0 N to 623.4 N, and the weft tensile strength decreased from 355.5 N to 335.8 N. Covalent bonds can be introduced between cotton fiber macromolecules, because the triazine active group in Reactive Red 2 and APDCT can react with cotton. Compared with untreated cotton fabric, the mobility of cotton fiber after the same bath finishing is reduced. There is inhomogeneous force of cotton fiber in the presence of external force. The tensile strength of treated cotton fabric therefore is reduced.
Color Fastness to Rubbing and Tensile Strength of Treated and Untreated Cotton Fabric

Conclusions
Cotton fabric was treated with APDCT in a Reactive Red 2 dyeing process. This one-step process can significantly shorten the production process and reduce production costs. However, there is competition between Reactive Red 2 and APDCT in the finishing bath. According to test methods for determining the percentage dye uptake, fixation, and LOI, the optimal concentration of Reactive Red 2, APDCT, sodium chloride, and sodium carbonate are 2%, 200 g/L, 50 g/L, and 20 g/L, respectively. The dye uptake percentage and fixation of treated cotton fabric under these conditions were 64.8% and 56.2%, respectively. The LOI value of the treated cotton fabric increased to 31.0%. The process had little effect on the color fastness to friction of the treated cotton fabric. The tensile strength of treated cotton fabric was slightly reduced.
Footnotes
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFB0309001). It also got the financial support of the Natural Science Foundation of Shandong Province (ZR2018MEM026).
