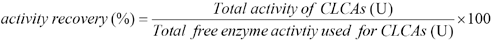Abstract
Cellulase was immobilized by cross-linked enzyme aggregation to improve the stability of cellulase. The prepared cross-linked cellulase aggregates (CLCAs) and ice silicone oil were used for the cool finishing of cotton fabric. The results showed that the CLCAs extended the cellulase stability compared to free cellulase. The surface softness, smoothness, moisture permeability, and air permeability of the cotton fabric increased after CLCAs and ice silicone oil treatment. Shearing rigidity of the treated sample was 0.44 cN/(cm·deg), bending rigidity was 0.0069 cN cm, and the drape coefficient was 29.3%. Coefficient of kinetic friction of the treated sample was 0.186. The capillary effect of the treated fabric was 12.1 cm/(30 min). Air permeability was 354.3 L/(m2·s). Moisture penetrability was 3.912 g/(m2·d). The thermal and water-vapor resistance were 0.0194 m2·°C/W and 4.691 Pa·m2/ W, respectively.
Introduction
In recent decades, enzymatic technology has been widely used in textile processing.1,2 The enzymatic process has the merits of substrate specificity, low energy consumption, eco-friendliness, and mild operating conditions.3,4 Notwithstanding all these advantages, enzymes often lack operational and storage stability that have limited their widespread application. 5
Cellulase is the most investigated enzyme in the textile field. Cellulase can catalyze the degradation of cellulose and remove the hairiness on the surface of cotton fabrics. 6 After cellulase treatment, the surface of cotton fabric becomes smooth and clear. At the same time, the pilling resistance, softness, and drapability of the fabric are improved.
To improve the stability and economics of cellulase, immobilization technology has been studied extensively.7,8 At present, there are two types of methodology for immobilization: carrier-bound and carrier-free immobilizations. Carrier-bound immobilization can be performed by entrapping the enzyme into polymeric matrices or binding the enzyme onto carrier materials. 9 A distinct disadvantage of carrier-bound enzymes in general is the dilution of catalytic activity owing to the large amounts of non-catalytic ballast. Therefore, there is increasing interest in carrier-free immobilized enzymes.
Recently, cross-linked enzyme aggregates (CLEAs) have attracted increased attention. 10 CLEAs are formed by precipitation and cross-linking of enzyme molecules. CLEAs generally exhibit high activity retention, enhanced thermal stability, and better tolerance to organic solvents.11,12 These properties are largely ascribed to the rigidification of the tertiary structure of the enzyme, prevention of multimeric enzyme dissociation, and prevention of enzyme denaturation by the multipoint attachment of the enzyme molecules. However, CLEAs also have some drawbacks, such as poor mechanical strength and ease of breakage during repeated use, resulting in enzyme leakage and difficulties in recovery. 13
To overcome these drawbacks, polyethyleneimine (PEI) was introduced into the preparation of cross-linked cellulase aggregates (CLCAs) in the present work. PEI acts as a protective agent to prevent over-crossing of enzyme activity sites. At the same time, the silica coating was formed on the outside surface of CLCAs via the induction of PEI. The stability and reusability of the CLCAs were then measured.
Ice silicone oil is a block copolymerized organosiloxane that imparts smooth, silky, and icy hand, as well as improved drapability to the treated fabrics. In this study, the CLCAs and ice silicone oil were used for the preparation of cool feeling cotton fabric. The properties of the prepared fabrics were investigated by Fourier transform infrared spectrometer (FTIR), thermogravimetric analysis (TGA), a Kawabata evaluation system (KES) fabric style instrument (KATO Tech Co. Ltd.), air permeability, moisture penetrability, thermal resistance, and water-vapor resistance testing.
Experimental
Materials
Cotton plain stitch knit fabric (18.3 tex, 125 g/m2) was kindly provided by Shaoxing Bangcai Textile Technology Co. Ltd. (Zhejiang, China). Cellulase was purchased from Yanjin Biotechnology Co. Ltd. (Shanghai, China). Ice silicone oil was provided by Guangdong Dry Chemical Co. Ltd. (Guangdong, China). Carboxymethyl cellulose (CMC), glutaraldehyde (GA), polyethyleneglycol (PEG), polyethyleneimine (PEI), and tetramethoxysilane (TMOS) were purchased from Luyue Chemical Reagent Co. Ltd. (Shandong, China) and the chemicals purchased were of analytical grade.
Preparation of CLCAs
Various amounts of PEI were thoroughly mixed with free cellulase (10 mg) and TMOS (0.05 M) in a disodium hydrogen phosphate-sodium dihydrogen phosphate buffer solution (pH = 6.0). Afterwards, different concentrations of GA and PEG were added to the mixture and stirred at 40 °C. Subsequently, the reaction mixture was centrifugated at 5000 rpm. The supernatant was removed and the mixture was washed with distilled water successively for removing the loosely adsorbed enzyme. Finally, the CLCAs were dried and stored at 4 °C.
Preparation of Cool Cotton Fabric
Cotton fabrics (20 g) were first treated by impregnation with 7% owf CLCAs at 50 °C for 30 min. Then the sample was soaked in 70 g/L ice silicone oil solution (1 L) until thoroughly wet, padded with a wet pickup of 80%, dried at 90 °C for 5 min, and cured at 150 °C for 2 min. The treated fabrics were then washed with distilled water and dried at ambient temperature.
Cellulase Activity
The activity of cellulase was determined according to the standard QB 2583-2002 Cellulases: Appendix A. 14 Filter paper was used as the substrate. The enzyme substrate mixture was incubated at 50 °C for 10 min and the reaction was stopped by boiling for 5 min. The amount of glucose was measured by a polarimeter (SWG-1 type, Shanghai Yinfu Industrial Co. Ltd., China). One unit of cellulase activity is defined as “the amount of enzyme that produces 1 μmole of glucose in one minute.” The activity recovery in CLCAs was calculated as given in Eq. 1.
The relative activity was calculated by the ratio of the residual activity to the initial activity.
Stability of Free-Cellulase and CLCAs
Thermal stabilities of free-cellulase and CLCAs were measured by incubating enzyme samples in aqueous buffer (pH 6.0) at various temperatures (30-70 °C) for 1 h. The activity of free-cellulase and CLCAs was determined by the same procedure as described above. The effect of pH on free-cellulase and CLCAs was studied by measuring activities over a range of pH values (from 4.0 to 8.0) at 50 °C for 1 h.
Reusability of CLCAs
The reusability of CLCAs was measured by adding CLCAs into the substrate solution and shaking for 10 min at 50 °C. The CLCAs were recycled by filtration ten times. After each run, the CLCAs were separated by filtration and used for successive cycles. The relative activity was calculated by the ratio with the initial activity.
Surface Morphology of CLCAs
The surface morphology of CLCAs was observed by a JSM-5600LV scanning electron microscopy (JEOL, Japan) and recorded as photographs. The magnification of the image was set in the range of 18 to 300000×.
Particle Size Measurement
The particle sizes of CLCAs were measured using a Nano ZS90 laser particle size analyzer (Malvern Panalytical Ltd., UK).
Measurements of Fabric Properties
Softness was evaluated comprehensively by determining the shearing, bending and draping properties of the fabrics. The shearing rigidity of the fabrics was tested by using a KES-FB1 tensile shear analyzer (CATO Technology Co. Ltd., Japan). The bending rigidity of the fabrics was tested using a KES-FB2 flexural performance tester (CATO Technology Co. Ltd., Japan). The drape coefficient of the fabrics was tested using a YG (B) 811E drape tester (Wenzhou Darong Textile Instrument Co. Ltd., China) according to GB/T 23329-2009. 15 The dynamic friction coefficient, average deviation of friction coefficient, and surface roughness of fabrics were measured using a Kawabata KES-FB4 fabric surface friction analyzer (CATO Technology Co. Ltd., Japan).
Thermogravimetric analysis (TGA) was conducted using a TG/DTA6300 analyzer (NSK, Japan) with a temperature range of 30 °C to 800 °C in a static air atmosphere at a heating rate of 10 °C/min. Sample weight was in the range of 3 to 5 mg.
The formation of new bonds of the samples was monitored by an IRprestige-21 FTIR spectrometer (Shimadzu, Japan).
The heat and humidity transitivity of the fabrics were tested using iSGHP-10.5 thermal and water-vapor resistance apparatus (MTNW, USA) according to GB/T 11048-2008. 16
The moisture permeability of the fabrics was measured with a YG501D moisture permeable tester (Wuxi Sanhuan Instrument Co. Ltd., China) according to GB/T 12704.1-2009. 17
The air permeability of the fabrics was determined by a Y561 fabric air permeable instrument (Ningbo Textile Instrument Factory, China) according to GB/T 5453-1997. 18
Results and Discussion
Preparation of CLCAs
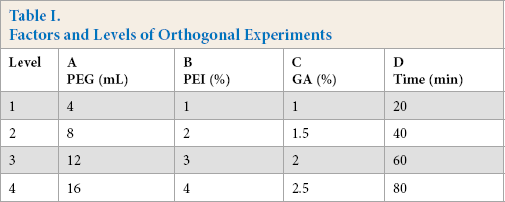
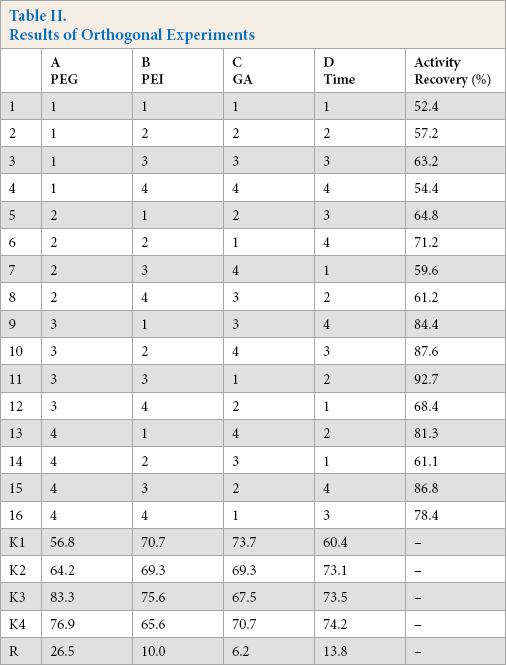
The preparation process of CLCAs was optimized using a four-factor and fourth-level orthogonal experiment. The settling agent (PEG), cross-linking agent (GA), PEI, and cross-linking time were taken as test factors. The level table of L16(44) orthogonal factors are shown in Table I, and the results are given in Table II. The results showed that the optimum preparation conditions of CLCAs were as follows: PEG (12 mL), PEI (3%), GA (1%), and cross-linking time (40 min). Under these conditions, the retention rate of enzyme activity was 92.7%.
Factors and Levels of Orthogonal Experiments
Results of Orthogonal Experiments
Thermal Stability of CLCAs
The activity profiles of free-cellulase and CLCAs at different temperatures are shown in Fig. 1. The original maximum enzyme activity was set to 100% as a reference. The results showed that CLCAs exhibited better stability compared with the free enzyme between 40 °C and 70 °C. After 1 h of incubation at 60 °C, the free enzyme retained 81% residual activity, while CLCAs retained 93% of its initial activity. At 70 °C, both enzyme activities decreased more rapidly compared with that at 60 °C, and the free cellulase decreased more quickly than CLCAs. The increased thermal stability may be due to the crosslinking and aggregation of cellulase, which by increasing the rigidity of the structure prevented conformational changes.
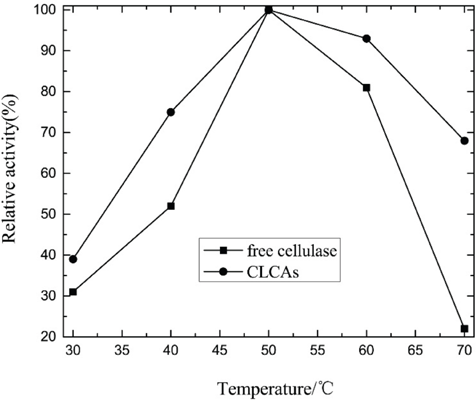
Thermal stability of CLCAs.
pH Stability of CLCAs
The effect of pH variations in the range of 4.0 to 8.0 on the activity of free-cellulase and CLCAs was investigated. As shown in Fig. 2, CLCAs revealed broader enzyme activity compared with free cellulase. The broader enzyme activity of CLCAs perhaps resulted from the silica coating on the surface of cross-linking enzyme aggregates, which inhibited the degradation of cellulase. Moreover, the covalent bond formation among enzymes and PEI might also reduce the conformational flexibility, which stabilized the conformation of the enzyme's active sites.
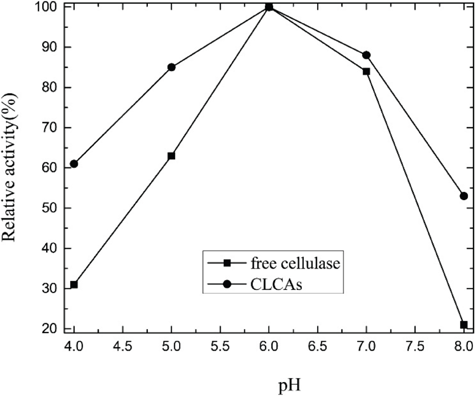
pH stability of CLCAs.
Reusability of CLCAs
The reusability of immobilized enzymes is important for industrial applications. The recyclability of CLCAs is shown in Fig. 3. The relative enzyme activity of CLCAs was 71% after six uses and 51% after ten uses. The CLCAs showed great reusability. The observed loss of activity can probably be attributed to mechanical losses that inevitably occur on pipetting the supernatant from the CLCAs.
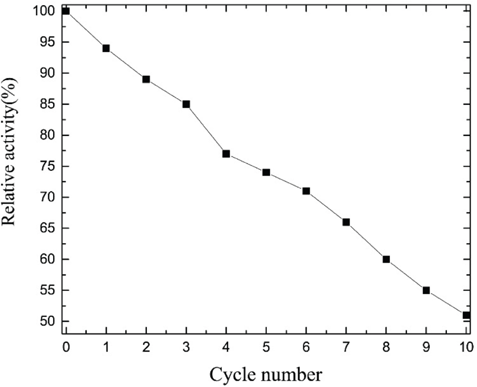
Reusability of CLCAs.
Surface Morphology of CLCAs
The surface morphologies of free cellulase and CLCAs were characterized by scanning electron microscopy (SEM). The results were shown in Fig. 4. It can be seen that free cellulase (as a powder prepared by spray drying the liquid enzyme) was spherical with good mono-dispersity. The size of free cellulase was not uniform. The aggregates of crosslinked cellulase were irregular flocculent in shape. There was no obvious agglomeration phenomenon.

SEM images (a) free cellulase and (b) CLCAs.
Particle Size of CLCAs
Particle size distribution was measured using a Malvern laser Zetasizer analyzer. The number and volume average size distribution of CLCAs are shown in Fig. 5. The mean size of CLCAs was 27.5 d.μm and the distributing index (PDI) was 0.307.
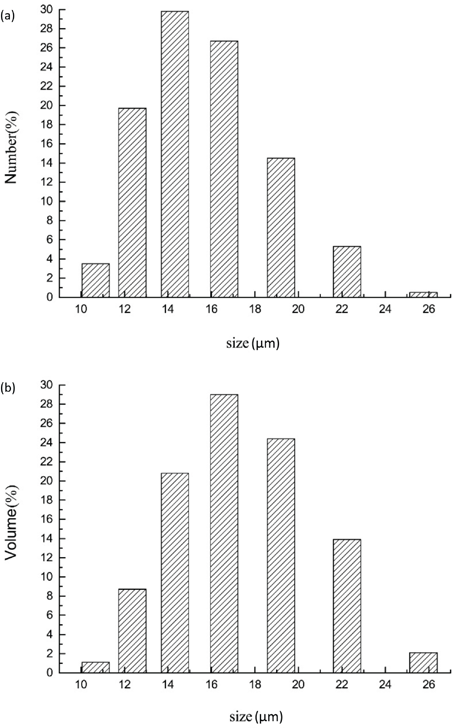
Particle size distribution graphs. (a) Number statistics graph and (b) volume statistics graph.
Fabric Hand
Softness Properties
Softness is a comprehensive reflection of physical and mechanical properties of the fabric. It can be evaluated by shear stiffness, bending stiffness, and drape coefficient of the fabric. Table III shows the softness of the untreated and treated fabrics.
Shearing Rigidity, Bending Rigidity and Drape Coefficient of the Fabrics
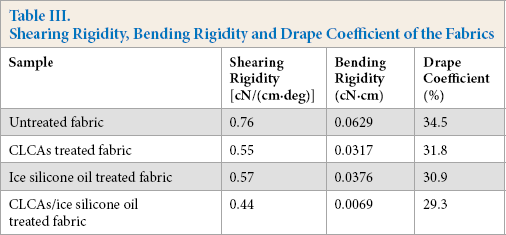
The shear stiffness, bending stiffness, and drape coefficient of the fabrics decreased after CLCAs and ice silicone oil treatment. Shear and bending properties, and drape coefficient, of fabrics are important indexes to determine the flexibility and handle of the fabrics. The smaller the values, the better the softness. CLCAs can catalyze the degradation of cellulose, which can remove the hairiness on the surface of cotton fabric. The surface of CLCAs-treated fabric became smooth and soft. After finishing with ice silicone oil, the friction resistance between yarns of the fabric decreased, and the fabric was prone to bending deformation. Therefore, the softness of cotton fabric can be improved by finishing with CLCAs and ice silicone oil.
Smoothness Properties
The dynamic friction coefficient, average deviation of friction coefficient, and surface roughness of the fabrics were measured using a Kawabata KES-FB4 fabric surface friction analyzer. The test results are shown in Table IV.
Smoothness of the Fabrics
The dynamic friction coefficient, average deviation of friction coefficient, and surface roughness of the cotton fabric decreased after CLCAs and ice silicone oil treatment. The results indicated that the smoothness of the treated fabrics increased. CLCAs treatment resulted in a decrease in fabric bending stiffness. The bending resistance of the fibers was reduced and the relative motion occurred easily. A lubricating layer was formed on the fiber surfaces by ice silicone oil treatment, which reduced the dynamic friction coefficient, average deviation of friction coefficient, and surface roughness. Therefore, the smoothness of the treated fabrics increased.
Human Hand Panel
In addition to the instrumental-based determinations of hand properties, a human hand panel comprised of 10 college students was used to evaluate the softness and smoothness of the untreated fabric and CLCAs/ice silicone oil treated fabrics. The results, available from the author upon request, confirmed those of the instrumental-based study that treatment improved both the softness and smoothness of the fabric.
FTIR Analysis
The FTIR spectra for the original and cool cotton fabrics are presented in Fig. 6. Besides the characteristic absorptions of the original sample, two new absorption peaks appeared in the cool cotton fabric. The absorption peak at 1501 cm−1 was the Si-O-C characteristic peak. The absorption peak at 1259 cm−1 was the bending vibration peak of Si-CH. The appearance of new absorption peaks at 1501 and 1259 cm−1 suggested that a silicone oil group had been introduced into the molecular structure of the cool cotton fabric. The silicone oil groups improved the smoothness and softness of the cotton fabrics.
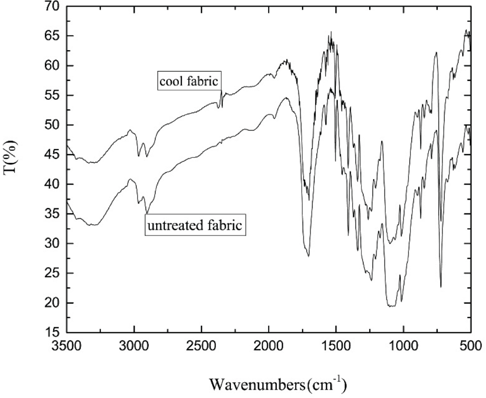
FTIR spectra of the samples.
TGA
The thermal stability of the fabrics was estimated by using TGA, and the results are shown in Fig. 7. The initial decomposition temperature of the untreated fabric was about 256.1 °C. The mass loss percentage was 98.90% due to the thermal decomposition of the fabric. The initial decomposition temperature of cool cotton fabric was 247.7 °C and the mass loss percentage was 99.56%. The CLCAs and ice silicone oil had a certain effect on the TG curves of the cotton fabrics. Generally speaking, the thermal stability of the cool cotton remained good.
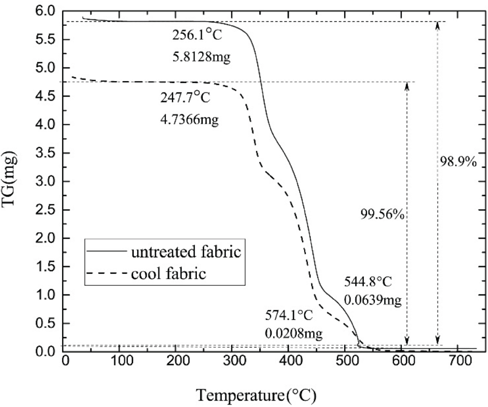
TGA curves of the samples.
Heat and Humidity Transitivity
The capillary effect, moisture permeability, air permeability, moisture resistance, and thermal resistance are important indexes for evaluating fabric comfort. Table V shows the values of these properties for cotton fabric before and after CLCAs and ice silicone oil treatment.
Heat and Humidity Transitivity of the Fabrics
The capillary effect, moisture permeability, and air permeability of the cotton fabrics increased after CLCAs and ice silicone oil treatment. The hairiness on the surface of CLCAs-treated fabric was removed due to the degradation of cellulose. The fabric became soft and fluffy because of the increase in inter-fiber pores. Therefore, the wettability, moisture permeability, and air permeability of the treated fabrics significantly improved. Moreover, many fine channels were formed inside the fibers after CLCAs and ice silicone oil treatment, which also enhanced these fabric properties.
Thermal resistance reflects the capacity of heat transfer between media. The smaller the thermal resistance of the fabric, the better the heat dissipation performance. Wet resistance is the ability of fabric to prevent water vapor from passing through. The thermal resistance and moisture resistance of the untreated fabric were 0.0285 m2 °C/W and 4.852 Pa m2/ W, respectively. After CLCAs and ice silicone oil treatment, the thermal resistance and moisture resistance of the cotton fabric reduced to 0.0194 m2 °C/W and 4.691 Pa m2/ W, respectively. The results showed that the heat dissipation and sweat conductive properties of cool cotton fabric were superior to that of ordinary cotton fabrics.
Conclusion
Cellulase was immobilized by a crosslinked enzyme aggregation technique and characterized through a series of stability tests. The immobilized cellulase (CLCAs) showed better pH tolerance and thermal stabilities than the free enzyme. The relative enzyme activity of CLCAs is 71% after six uses. The CLCAs showed great reusability, which is an important factor for industrial use of enzymes.
Cotton fabric was treated with CLCAs and ice silicone oil to obtain a cool feeling fabric. FTIR analysis indicated that the silicone oil group was introduced on the treated fabric surface. Thermal stability of the treated fabric remained good. The softness, smoothness, heat dissipation, and sweat conductive properties of cool cotton fabric were superior to those of the ordinary fabric.