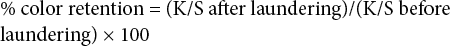Abstract
Sulfur dyed cellulosic goods exhibit limited wash fastness against oxidative laundering; particularly to washing powder formulations containing activated bleaches. This study presents a novel two-bath exhaust finishing process to improve the wash fastness of Sulfur Black 1 dyed cotton fabrics reduced with two different reducing agents—sodium sulfide and a commercial glucose-based Diresul reducing agent D. The finishing process involves sequential aftertreatments with a cationic fixative and synthetic tanning agent (syntan). A Box-Behnken design with three variables: concentrations of cationic fixative and syntan, and application temperature of syntan (at three different levels) was studied to understand the effects of these variables on the color retention of the dyed cotton fabrics. Color retention of the sulfur dyeings was improved using the newly developed finishing process.
Introduction
Sulfur dyes are widely used on cellulosic fibers and their blends with polyester, nylon, and acrylic because of their low cost, availability of dark shades, good wet fastness and moderate to good light fastness.1,2 They are still useful for producing shades of black, brown, navy, olive, and green color in medium to heavy depths.
Sulfur dyes are insoluble in water and lack affinity for cellulosic fibers. They require a complex dyeing procedure involving reduction and oxidation mechanisms. Firstly, these dyes have to be reduced or vatted before application to convert them into a water soluble leuco enolate form, which has substantivity towards cellulose. Finally, they are re-oxidized to convert them into their insoluble form inside the fiber. 3 When these dyes are subjected to an oxidative perborate-based laundering environment, the dye disulfide bonds are oxidized and form water-soluble sulfonated short chain dye fragments, which are lost into the washing solution. 4
In spite of having reasonably good properties and wide applications, sulfur dyes are gradually losing their significance and getting partially replaced with reactive dyes due to the former's low resistance to oxidative laundering and the considerable decrease in latter's price. 1 Another major reason for their continuing unpopularity is the environmental hazard created by the use of the conventional reducing agent, sodium sulfide; which is highly toxic for marine life and sewage systems. In this context, a substantial body of work has been undertaken on the use of reducing sugars for the dyeing of cellulose with sulfur dyes so as to replace this hazardous component by more eco-friendly alternatives.5-7
Furthermore, cellulosic goods dyed with sulfur dyes are particularly susceptible to perborate bleach-containing washing powders, thus exhibiting limited wash fastness against oxidative bleaching. To improve the wet fastness of sulfur dyed cotton fabric, a considerable amount of work involving the exhaust and continuous afertreatments with several cationic fixatives have been explored by Burkinshaw.8-12 However, the resistance of sulfur dyed cellulosic substrates against the action of oxidative bleaching formulations, including bleach activators has not been taken much into consideration.
In the past, investigations have been made on sequential afertreatments with metal salts, tannin/tannic acid, polymeric cationic agents, and syntan. The fastness properties of various dyes and substrates had been taken into consideration.13-17 Syntans are chiefly comprised of condensates of formaldehyde with phenolsulfonic or naphtholsulfonic acid. Some of them contain polar groups (carbonamide, sulfonamide, and ureides), and they can be divided into several types such as: phenolic, thiophenolic, and dihydroxy diphenyl sulfone. 18
Recently, studies involving the improvement of wash fastness of Sulfur Black 1 dyed cotton fabric with sequential aftertreatments using a cationic fixative (Tinofix ECO)/cationic reactant (choline chloride) followed by tannin (Bayprotect Cl) against the oxidative bleaching action of detergent/ perborate/bleach activator formulations (ISO 105 C09) have been reported.19-21 It was found that the extent of wash fastness improvement imparted by the cation-tannin system was greater than that given by a cationic fixative alone. This study is based on developing a cation-syntan aftertreatment method for Sulfur Black 1 dyed cotton fabrics (reduced with two reducing agents—sodium sulfide and Diresul reducing agent D) that makes it resistant to the aggressive home laundering processes involving the bleach activator tetraacetylethylenediamine (TAED).
Experimental
Materials
A plain woven, bleached, 100% cotton fabric having GSM 103 and EPI × PPI of 77 × 66 was used throughout the study. The fabric was generously provided by Phoenix Calico Limited. Diresul Black RDT-LS LIQ 200 (C.I. Sulfur Black 1) dye and Diresul RAD (reducing agent D) manufactured by Clariant were used. The oxidation bath was comprised of hydrogen peroxide and sodium bicarbonate (laboratory grade), both purchased from Aldrich Chemicals Ltd., UK. The ECE non-phosphate detergent and TAED (92% active) were purchased from the Society of Dyers and Colourists (SDC) Enterprises, Bradford, UK. Sodium perborate tetra-hydrate (97% active, laboratory grade) was purchased from Aldrich Chemicals Ltd., UK. Cationic fixative agent (Tinofix ECO, Solophenyl, CIBA Geigy) and syntan (Nylofixan PSA, Clariant) were also purchased.
Dyeing with Diresul RAD
All dyeings were carried out in sealed stainless steel dye pots of 1000 cm3 capacity, housed in a Mathis Labomat laboratory dyeing machine. Dyeing of cotton with Diresul RAD was carried out by the method prescribed by Clariant, shown in Fig. 1. The cotton fabric was introduced into a dye bath containing 5% omf of Sulfur Black 1 dye liquid, 25 g/L of sodium chloride, 13 mL/L of sodium hydroxide (67 °Tw), 9 g/L of Diresul RAD with a liquor to goods ratio (LR) of 10:1. The dye bath was raised to 98 °C at a gradient of 4 °C/min and maintained at this temperature for 60 min. The solution was then cooled to 50 °C.
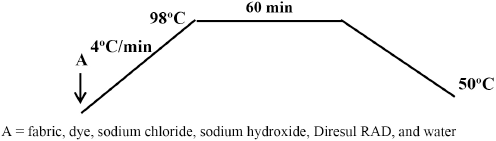
Sulfur dyeing method for cotton using Diresul RAD.
Dyeings were washed thoroughly with cold water to remove any unfixed dye. The dyed fabric was then oxidized with 5 g/L of hydrogen peroxide and 1 g/L of soda ash at 40-45 °C for 15 min. It was then finally soaped with 1 g/L of non-ionic detergent at the boil for 20 min and then rinsed with warm and cold water. The LR used for oxidation and soaping was 10:1.
Dyeing with Sodium Sulfide
Dyeing of cotton fabric with sodium sulfide was carried out with 5% omf of dye, 5 g/L of sodium sulfide, 5 g/L of sodium carbonate, and 15 g/L of sodium chloride with an LR of 10:1 as shown in Fig. 2.
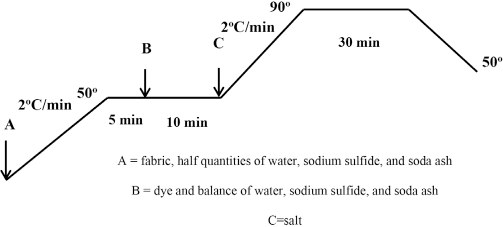
Sulfur dyeing method for cotton using sodium sulfide.
The fabric was first heated to 40-50 °C in the presence of half the quantities of water, sodium sulfide, and soda ash. The liquor was kept at this temperature for 5 min and then the dye and the balance of water, sodium sulfide, and soda ash were added. The dye bath was further kept for 10 min at the same temperature and then salt was added. The temperature was then increased to 90 °C at a gradient of 2 °C/min and treated for 30 min. The solution was cooled to 50 °C, and the fabric was then washed thoroughly with cold water to remove any unfixed surface dye. Rinsing and oxidation were done in the same way as described in the previous section.
Aftertreatment with Tinofix ECO (Cationic Fixative)
Sulfur dyed fabric was aftertreated with a liquor containing x% omf of Tinofix ECO in a Mathis Labomat with an LR (liquor to goods ratio) of 10:1. The pH of the liquor was 6-7 (as prescribed by the manufacturer). The fabric was treated at 40 °C for 30 min, rinsed in warm and cold water, and finally air dried. The method of application is represented in Fig. 3.

Cation aftertreatment.
Aftertreatment with Nylofixan PSA (Syntan)
Nylofixan PSA was used to treat Sulfur Black 1 dyed cotton fabric aftertreated with the above said cationic fixative. Cat-ionic aftertreated cotton was aftertreated with liquor containing x% omf of syntan, having a pH of 4 maintained with acetic acid. The samples, along with the liquor, were treated at 40, 60, and 80 °C for 30 min with an LR of 10:1. The finished fabric was finally rinsed with warm and cold water, and air dried. The application method is shown in Fig. 4.

Syntan aftertreatment.
Determination of Wash Fastness (ISO 105 C09)
A dyed fabric sample measuring 10 × 5 cm2 was weighed and an LR of 1:100 was used. It was introduced in a liquor containing 10 g/L of ECE non-phosphate detergent, 1.8 g/L of TAED low temperature bleach activator, with or without 12 g/L of sodium perborate, at 25 °C, and raising the temperature to 60 °C at 2 °C/min, and maintaining at 60 °C for 30 min. The specimens were then removed, rinsed, and air dried.
Color Retention Measurement
The colorimetric data for the dyed and aftertreated cotton fabrics were measured using a Datacolor Spectroflash 600 spectrophotometer, with a 10° standard observer and D65 illuminant, and were the average of four measurements.
The percent color retention of the untreated and aftertreated dyed fabrics after laundering were determined using K/S (color strength) with Eq. 1.
Methodology and Design of Experiment
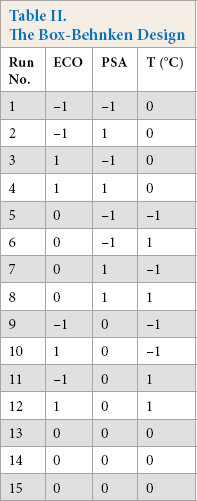
Response surface methodology is an empirical modeling technique devoted to the evaluation of the relationship of a set of controlled experimental factors and observed results. It requires a prior knowledge of the process to achieve a statistical model. Basically, this optimization process involves three major steps: performing the statistically designed experiments, estimating the coefficients in a mathematical model, and predicting the response and checking the adequacy of the model. Significant variables, like concentrations of cationic fixative and syntan, and application temperature of syntan, were chosen as the critical variables and designated as X1, X2, and X3, respectively. The low, middle, and high levels of each variable were designated as -, 0, and + respectively, and given in Table I. The actual design of experiment is given in Table II. Computation was carried out using multiple regression analysis using the least-square method with the help of the software design expert.
Experimental Design

The Box-Behnken Design
The quadratic (second degree) polynomial equation (Eq. 2) was used to develop the mathematical relationship of color retention on the chosen variables.
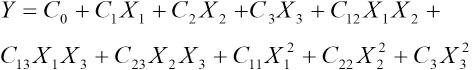
Y is the predicted color retention, C0 is a constant, C1, C2, and C3 are linear coefficients, C12, C13, and C23 are cross product coefficients, and C11, C22, and C33 are quadratic coefficients.
A multiple regression analysis was done to obtain the coefficients and the equations can be used to predict the color retention. The degree of experiments chosen for this study was Box-Behnken, a fractional factorial design for three independent variables. In the model given in Eq. 2, interactions higher than first order were neglected. This design is preferred because relatively few experimental combinations of the variables were adequate to estimate potentially complex response functions.
A total of 15 experiments were necessary to estimate the 10 coefficients of the model. Using multiple linear regression analysis, the set of coefficient for color retention of the dyed fabric was calculated.
Results and Discussion
The experiments were designed and conducted according to the Box-Behnken model. The process parameters were optimized to enhance the color retention of the dyed fabric during laundering. The regression equations obtained after analysis of variance gave the color retention of dye as a function of different concentrations of Tinofix ECO and Nylofixan PSA, and temperature. All terms regardless of their significance are included in Eqs. 3 and 4.
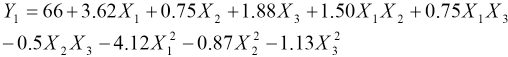
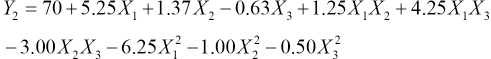
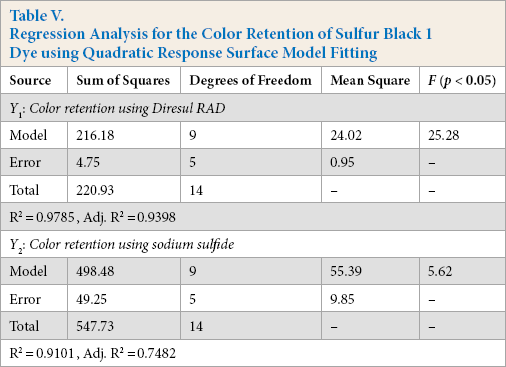
Y1 and Y2 are predicted responses for color retention using Diresul RAD and sodium sulfide, respectively. The square regression for Y1 and Y2 was significant at the level of 97% and 91%, respectively. The color retention of Sulfur Black 1 dyed fabric from the model at each experimental point is summarized in Table III. The coefficients of Eq. 2 are calculated using design expert and their values are listed in Table IV. The summary of the analysis of variance (ANOVA) is shown in Table V.
Experimental Values for Color Retention a
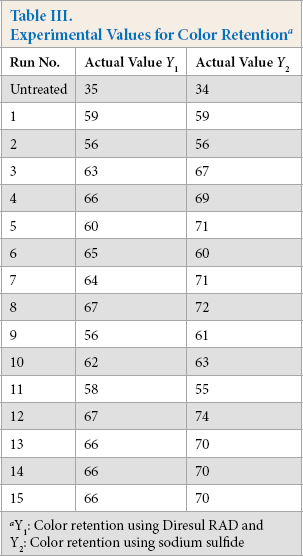
Y1: Color retention using Diresul RAD and Y2: Color retention using sodium sulfide
Coefficients of the Model a
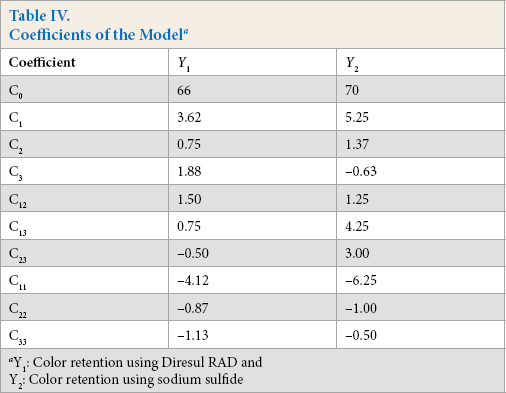
Y1: Color retention using Diresul RAD and Y2: Color retention using sodium sulfide
Regression Analysis for the Color Retention of Sulfur Black 1 Dye using Quadratic Response Surface Model Fitting
Optimization of Tinofix ECO and Nylofixan PSA Concentrations
A contour was plotted representing the maximum color retention versus concentration of Tinofix ECO and Nylo-fixan PSA. Here the color retention increased with increased Tinofix ECO and Nylofixan PSA concentrations, reaching a maximum value of 66 and 70 between 5% to 8% of Tinofix ECO and 7% to 10% of Nylofixan PSA using Diresul RAD and sodium sulfide, respectively, as shown in Figs. 5a and b. The optimization level of color retention was at 6% of Tinofix ECO and 7% of Nylofixan PSA.
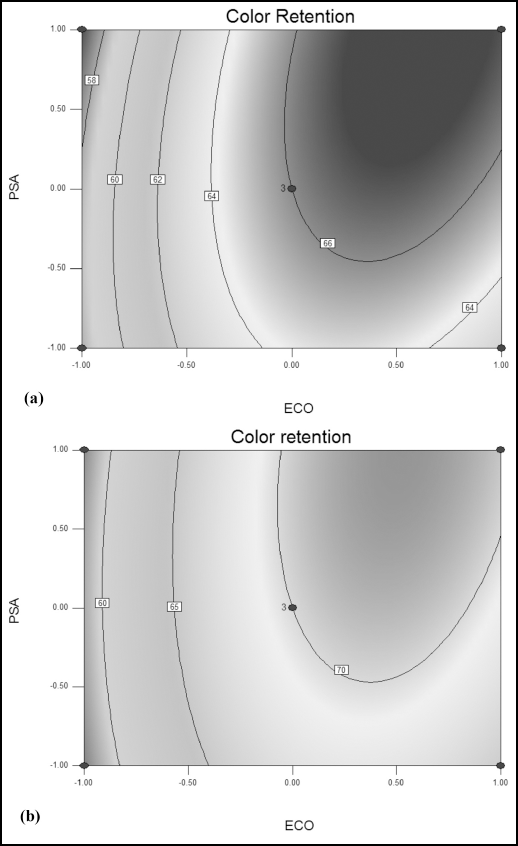
Contour plot representing color retention versus Tinofix ECO and Nylofixan PSA using (a) Diresul RAD and (b) sodium sulfide.
The cationic fixative probably has the potential for the formation of a large molecular size dye-cationic agent complex with low aqueous solubility arising from the electrostatic forces of attraction between the possibly present anionic groups in the dye and the polycations. 9
The increase in color retention was presumably due to the formation of large molecular size complexes between the cationic polymer and anionic syntan, which inhibited the dissolution and desorption of dye molecules and protects the dye against the ISO 105 C09 washing regime, based on modern detergent formulation. The increase in the concentration of cationic fixative provided a greater number of sites available for syntan aggregates to react and form complexes.
However, color retention as a result of aftertreatments was relatively higher when sodium sulfide was used as a reducing agent. This may be due to the comparatively smaller molecular size of the untreated dyeing produced with sodium sulfide, which gave a lower wash fastness as shown in Table III. Aftertreatments improved the color retention to a greater extent owing to the formation of comparatively larger complexes than for those dyeings that were reduced with Diresul RAD. This phenomenon was explained by Blackburn and Harvey, according to which the optimum concentration and redox potential of reducing sugars caused the dye to produce the best wash fastness, as the dye molecule was either too large or fragmented above or below that value. It is proposed that upon oxidation during laundering, the smaller dye molecules would be likely to display lower levels of wash fastness in relation to larger fragments, because the larger crystallites of the dye molecules form in situ when larger reduced dye molecules reform into the polymeric parent structure. 5
It is a limitation of the sulfur dye that it is oxidized when exposed to the bleaching environment of perborate-based laundering regimes. This action is even more aggressive if a bleach activator such as TAED is present in the laundering solution. The oxidized dye is the precursor to loss of dye from the fiber. The smaller dye fragments produced would lead to lower wash fastness as compared to the larger ones, as they would easily get desorbed during laundering.
Optimization of Tinofix ECO Concentration and Nylofixan PSA Application Temperature
The contour plot in Figs. 6a and b represents the maximum color retention versus Tinofix ECO concentration and Nylofixan PSA application temperature. Here, the color retention increased with increased Tinofix ECO concentration, reaching a maximum value of 66 and 70 between 5% to 8% of Tinofix ECO. However, using sodium sulfide as a reducing agent, the temperature of Nylofixan PSA can be fixed at a lower level of 40 °C. The optimal color retention was achieved with 6% of Tinofix ECO (at 60 °C) and 5% of Tinofix ECO (at 40 °C) for Diresul RAD and sodium sulfide, respectively.
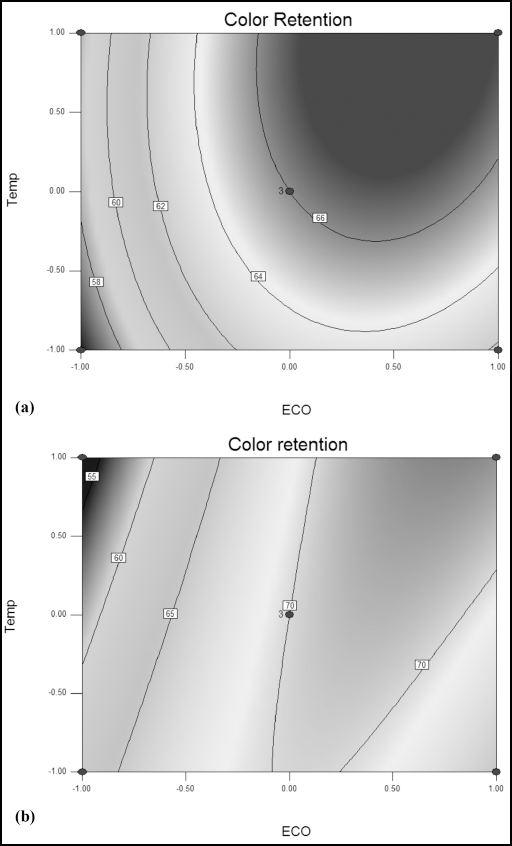
Contour plot representing color retention versus Tinofix ECO and temperature using (a) Diresul RAD and (b) sodium sulfide.
It was observed that as the temperature of Nylofixan PSA was raised, the color retention increased using Diresul RAD. Such an effect is probably due to the greater extent of fiber swelling with increasing application temperature and the higher uptake of the syntan at greater Tinofix ECO concentrations. This can be attributed to the higher kinetic energy of the syntan molecules and their consequent greater interaction with the cationic fixative, resulting in the higher diffusional power of the large-sized complexes within the substrate.
This trend was reversed in the case of sodium sulfide; the color retention decreased with increased temperature. This may be due to dye desorption through increased kinetic energy at elevated temperature as the dyeings produced by sodium sulfide are assumed to be smaller in size as compared to RAD reduced dyeings, hence the cation-syntan complexes would also be comparatively smaller in size, and would consequently be desorbed easily at elevated temperatures.
Optimization of Nylofixan PSA Concentration and Application Temperature
Color retention against the concentration and application temperature of Nylofixan PSA is represented by contour plots in Figs. 7a and b. For Diresul RAD, on increasing the concentration from 6% to 8% and temperature from 40 to 60 °C there was a concomitant increase in color retention. However, this trend was not observed beyond the aforementioned levels of concentration and temperature, illustrating the attainment of optimum parameters at a temperature of 60 °C and a concentration of 8% (Fig. 7a).
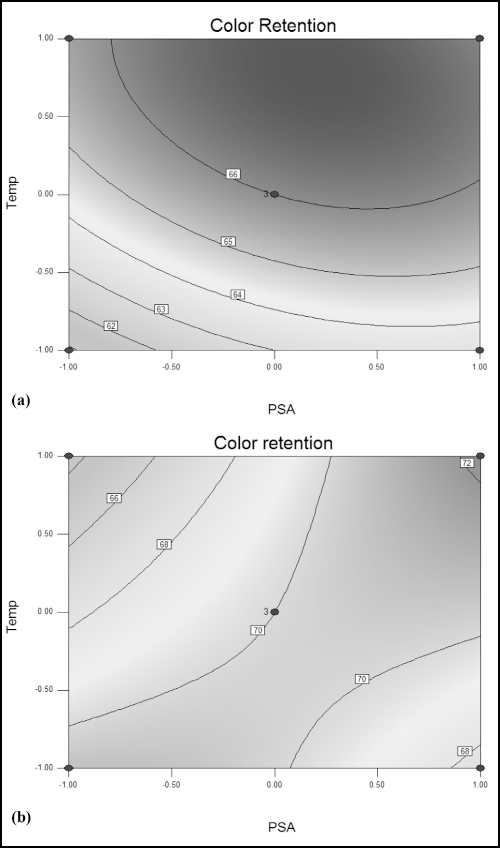
Contour plot representing color retention versus Nylofixan PSA and temperature using (a) Diresul RAD and (b) sodium sulfide.
When sodium sulfide was used, this trend was reversed. The color retention decreased with the increased temperature and concentration of PSA, reaching a maximum of 70 between 40 to 50 °C and 6% to 8% of PSA as shown in Fig. 7b. Hence, the optimized parameters of color retention for sodium sulfide were 40 °C and 6% of PSA. This may possibly be due to desorption of the dye through increased kinetic energy as discussed previously.
Conclusions
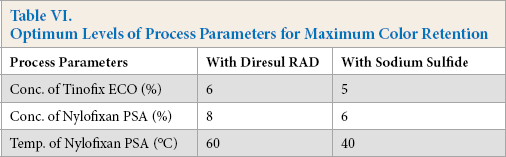
Application of the response surface method to the color retention of Sulfur Black 1 dyed cotton fabric gave an empirical correlation between the process parameters. An optimum combination of significant parameters was obtained for both of the reducing agents considered. The optimum levels of process parameters are given in Table VI. It can be noted that with sodium sulfide, relatively lower ECO and PSA concentrations, and temperature, were required for maximum color retention. The possible mechanism involved in increased color retention as a result of sequential aftertreatments was the formation of a large molecular size and low aqueous solubility complex between the cationic compound and the anionic syntan within the dyed fabric which would reduce the propensity of the dye to be desorbed from the fiber during laundering.
Optimum Levels of Process Parameters for Maximum Color Retention