Abstract
In this study, the relative effect of aftertreatments with two fixing agents (A and B) on the washfastness of C.I. Sulfur Black 1 dyed cotton fabric was evaluated using ISO 105-C09. The surface chemistry of untreated and aftertreated sulfur dyed cotton fabrics were studied by using X-Ray Photoelectron Spectroscopy (XPS). The N(1s) XPS spectrum of fixing agent A aftertreated fabric showed the concentration of uncharged and quaternary nitrogen species, while fixing agent B portrayed only uncharged nitrogen. Fixing agent A improved the washfastness of the dyed fabric to a greater extent than fixing agent B, owing to the presence of the quaternary nitrogen species. Improvement in the resistance to oxidation of the dye during domestic laundering was observed.
Introduction
Sulfur dyes are a major class of textile dyes that are applied to cellulosic textiles and its blends. 1 They offer an economical method of achieving dyeings of good color strength and acceptable fastness on cellulosic substrates. 2 They have good washfastness and moderate to good lightfastness in heavy shades. 3 However, the main limitations associated with this class of dye are the lack of availability of bright colors and fading on exposure to laundering with detergent and perborates/peroxides formulations. To meet the growing requirements of customers regarding improved washfastness, garment retailers are in turn demanding their suppliers achieve these properties. 4
Aftertreatments with cationic fixing agents for improving the washfastness of sulfur dyeings were explored by Burkin shaw. 4–7 Many dye fixing agents are cationic, and are based on various quaternized or otherwise cationically-charged organic nitrogen compounds. Cationic fixatives are available under various trade names from several suppliers.
Cationization of cellulose involves the introduction of amine groups or quaternary ammonium groups via its hydroxyl groups. It was reported by Gränacher that cationized cellulose having ether linkages could be synthesized by reacting the hydroxyl groups with other compounds. 8
Studies were carried out to recognize the impact of cationic fixing agents on the improved fastness of solubilized sulfur, leuco sulfur, and sulfur dyeings by wash tests such as ISO 1O5-CO6/C2, 5 ISO 4, 7 and Marks and Spencer C46. 4 The introduction of cationic sites was carried out as pretreatments 9 and afertreatments for leuco and oxidized sulfur dyeings.4–7,10 In addition to exhaust application, pad-dry and pad-fash cure methods of cationization also improve the washfastness of leuco sulfur dyeings. 5
An attempt to improve the washfastness of sulfur dyes was made by Burkinshaw using a commercial cationic fixing agent. Pretreatments and aftertreatments of cotton fabric with this fixing agent resulted in sulfur dyeings with superior washfastness and imparted substantial enhancement in color strength. The reaction of this fixing agent with the nucleophilic thiol group of the reduced sulfur dye led to the formation of a large molecular size, sulfur dye-fixing agent complex that retained low diffusional power within the fiber.7,9
In this study, the relative effect of aftertreatments with two cationic fixing agents on the washfastness of Sulfur Black 1 dyed cotton fabric using ISO 105-C09 has been assessed. The differences in the chemical nature of the two fixatives have been studied through surface chemical analysis of the fabrics using the XPS method. The names of the proprietary fixing agents have been replaced with fixing agent A (FA) and fixing agent B (FB).
The proprietary reactive cationic FA of unknown structure, is believed to form an extensively polymerized and insoluble network of fixative and dye, probably due to the reaction between tertiary amine and cationic groups present in stoichiometric proportions. The cationic polymer, possibly containing a polyamine backbone, is a fiber-reactive wet fastness improver, free of formaldehyde. Reactive groups are formed as a result of reaction with epichlorohydrin to produce a reactive cationic chlorohydrin derivative. 11
The FB structure is particularly designed to interact with direct dyes containing a number of different sulfonate groups, but also to complex with the copper atom in the dye molecule resulting in a more stable interaction between dye and fixative. 11
The surface chemistry of the outer surface of C.I. Sulfur Black 1 dyed cotton fabrics aftertreated with the stated fixing agents has been investigated for the first time. The surface sensitive X-ray photoelectron spectroscopic (XPS) technique was used to probe the nature of the outer 3–10 nm of the cotton's surface to characterize the surface functionalities, establish the elemental composition, and assess the durability of the surface sulfur dye for different aftertreatments. This spectroscopic technique helped in fully understanding the nature of the fiber surface interface of untreated and aftertreated cotton fabric on exposure to a chemically aggressive bleaching medium, and the improvements produced as a result of the aftertreatments.
Experimental
Materials
A plain woven, bleached, 100% cotton fabric (103 g/m2 and EPI × PPI of 77 × 66) was used throughout the study. The fabric was generously provided by Phoenix Calico Ltd. Diresul Leuco Sulfur Black 1 dye and Diresul Reducing Agent D (Clariant) were used. The ECE non-phosphate detergent and tetraacetylethylenediamine (TAED, 92% active) were purchased from the Society of Dyers and Colourists (SDC). Sodium perborate tetrahydrate (97% active), hydrogen peroxide, and sodium bicarbonate (laboratory grade) were purchased from Aldrich Chemicals Ltd. Two commercial cationic fixing agents (FA and FB) were used.
Methods and Procedures
Dyeing Procedure
The cotton fabric was introduced into a dye bath containing 5% owf Diresul Black RDT liquid (Diresul Leuco Sulfur Black 1), 25 g/L of sodium chloride, 13 mL/L of sodium hydroxide (1335 g/L), and 9 g/L of Diresul Reducing Agent D with a liquor to goods ratio (LR) of 10:1. The dye bath was raised to 98 °C at a gradient of 4 °C/min and maintained at the boil for 60 min. The temperature was then reduced to 50 °C, as shown in Fig. 1.
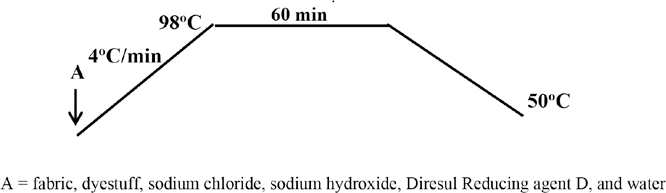
Sulfur dyeing method.
The dyed fabrics were washed thoroughly with cold water to remove any unfixed surface dye. The fabric was then oxidized with 5 g/L of hydrogen peroxide and 1 g/L of soda ash at 40–45 °C for 15 min. It was then finally soaped with 1 g/L of non-ionic detergent at the boil for 20 min and then rinsed with warm and cold water. The LR used for oxidation and soaping was 10:1.
FA Aftertreatment
Sulfur dyed fabric was introduced into a liquor containing 6% owf FA at 20 °C. The LR was 10:1 and a Mathis Labomat was used for the exhaust application of the finishing liquor. The liquor temperature was increased to 40 °C at a gradient of 4 °C/min. Then, 2 mL/L of caustic soda was added to maintain an alkaline pH and the process further continued for 30 min. The sample was then removed, rinsed in warm and cold water, and finally air dried. The application method is shown in Fig. 2.
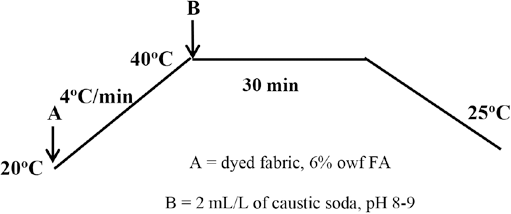
Application of FA.
FB Aftertreatment
Sulfur dyed fabric was introduced into a finishing bath containing 4% owf FB and 5 g/L of Glauber's salt. The LR was 10:1 and a Mathis Labomat was used for the exhaust application of the finish. The temperature of the liquor was increased to 60 °C at a gradient of 4 °C/min and maintained at this temperature for 20 min. The sample was then removed, rinsed in warm and cold water and finally air dried. The application procedure is shown in Fig. 3.
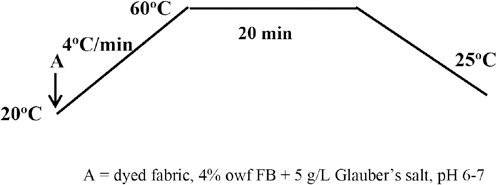
Application of FB.
Color Analysis
The colorimetric data of the dyed and aftertreated cotton fabrics were measured using a Datacolor Spectroflash 600 spectrophotometer, with a 10° standard observer and D65 illuminant, and were the average of four measurements. The color strength (K/S) was evaluated using the Kubelka–Munk equation (Eq. 1).
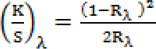
K is the absorption coefficient, S is the scatter coefficient, and R is the reflectance expressed as a fractional value at wavelength of maximum absorption λ.
The percent color loss of the untreated and aftertreated dyed fabrics after laundering were determined by Eq. 2.
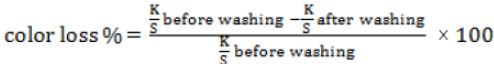
ISO 105-C09 Testing
The dyed fabric sample measuring 10 × 5 cm was weighed and an LR of 1:100 was used. It was introduced into a liquor containing 10 g/L of ECE non-phosphate detergent, 1.8 g/L of TAED, and 12 g/L of sodium perborate at 25 °C, followed by raising the temperature to 60 °C at 2 °C/min, and then maintaining the bath at 60 °C for 30 min. The specimens were then removed, rinsed, and air dried.
XPS Analysis
XPS analysis was performed using a Kratos Axis system spectrometer. The fabric samples (5 × 5 mm) were cut from the center of the specimen and attached to the sample holder using a double-sided tape of the same size. The samples were irradiated with monochromatic Al Kα X-rays (1486.6 eV) with a power of 150 W. The wide-scan spectra were recorded with a pass energy of 160 eV, from which the surface composition (C, O, S, and N) was determined. High resolution C(ls), N(ls), and S(2p) spectra were recorded with a pass energy of 40 eV and binding energy (BE) values were calculated relative to the C (1s) photoelectron peak at 285.0 eV. Charge compensation for the samples was achieved using a 4-7 eV beam at a flood current of ∼0.1 mA, with an electrically-grounded 90% transmission nickel mesh screen. All samples were analyzed in duplicate using CASA XPS software (Casa Software Ltd.).
Results and Discussion
Surface Chemical Analysis before Laundering
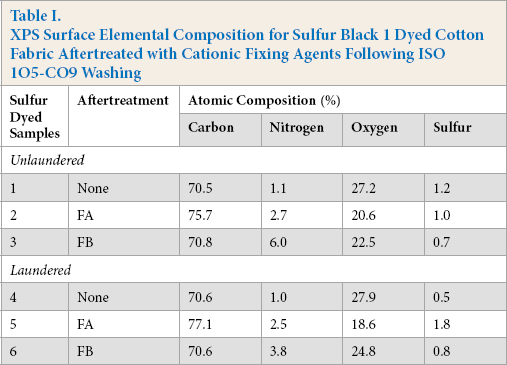
The XPS survey spectrum of sulfur black dyed fabric shows the major photoelectron peaks located at BE values of 531, 400, 285, and 164 eV corresponding to the O(1s), N(1s), C(ls), and S(2p) signal intensities, respectively. The percent atomic concentrations of these elements present on the surface of unlaundered and laundered dyeings are presented in Table I.
XPS Surface Elemental Composition for Sulfur Black 1 Dyed Cotton Fabric Aftertreated with Cationic Fixing Agents Following ISO 105-C09 Washing
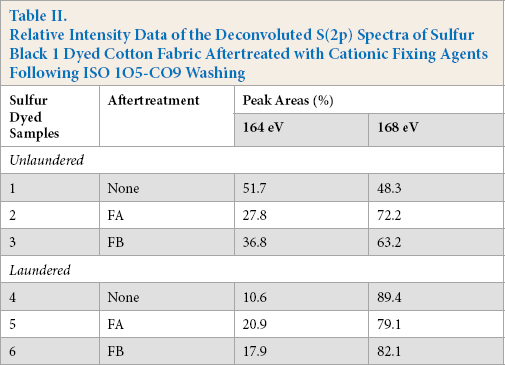
Examination of the high resolution S(2p) spectrum of untreated fabric showed broad sulfur signals with well-separated peak structures at 164 eV (unoxidized, S2+ form) and 168 eV (oxidized, S6+ form) BE values (Fig. 4). A peak at 168 eV contributing around 48.3% (Table II) of the total sulfur content represents the re-oxidation of the alkali reduced sulfur dye thiols to reform the insoluble disulfide bond-based sulfur dye resulting in the over-oxidized S6+ species. 12
Relative Intensity Data of the Deconvoluted S(2p) Spectra of Sulfur Black 1 Dyed Cotton Fabric Aftertreated with Cationic Fixing Agents Following ISO 105-C09 Washing
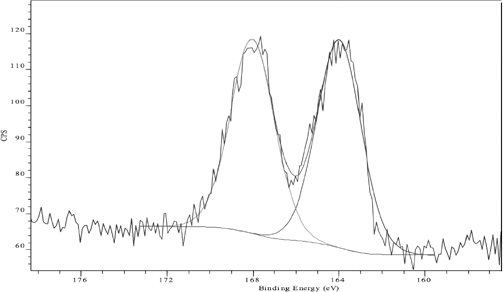
S(2p) XP spectrum of untreated, unlaundered Sulfur Black 1 dyed cotton fabric.
Aftertreatment with FA reduced the amount of the lower BE spectral component at 164.0 eV (51.7% to 27.8%) as shown in Fig. 5. In addition, significant reduction of the surface sulfur concentration (1.2% to 1.0% atomic sulfur, Table I) was observed, probably due to the removal of surface dye. Similar behavior could also be seen for FB aftertreated sample, showing the reduction in energy spectral component at 164.0 eV (51.7% to 36.8%) in Fig. 6, as well as a surface sulfur concentration reduction (1.2% to 0.7% atomic sulfur, Table I).
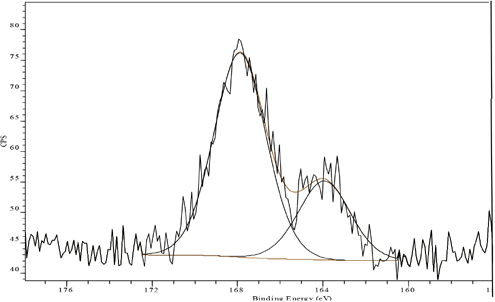
S(2p) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FA and unlaundered.
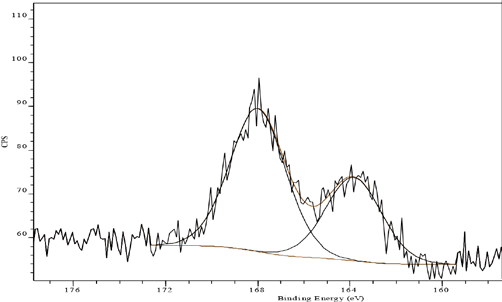
S(2p) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FB and unlaundered.
Relative amounts of carbon with different numbers of oxygen bonds were estimated from the high resolution C(1s) carbon spectra, the summary of which is presented in Table III. The carbon compound classification was based on the number of oxygen bonds attached to the carbon atoms: the peaks at 285.0 eV (C1), 286.6 eV (C2), 288.1 eV (C3), and 289.5 eV (C4) are due to carbons with 0, 1, 2, or 3 bonds to oxygen respectively. 13 For sulfur dyed fabric aftertreated with FA (sample 2), deconvolution analysis showed an increase of oxygen-based functional groups that are C2, C3, and C4, compared to untreated dyed fabric (sample 1).
Relative Intensity Data of the Deconvoluted C(1s) Spectra for Sulfur Black 1 Dyed Cotton Fabric Aftertreated with Cationic Fixing Agents Following ISO 1O5-CO9 Washing
These results imply that the surface of the sulfur dyed cotton fabric became partially oxidized by fixative exposure, resulting in the reduction of the non-cellulosic cotton component (C–C/C–H bonds) by 31%, and a significant increase in the oxidized species (C2 by 7%, and in C3 and C4, by 43%). Analysis of the FB aftertreated fabric (sample 3) showed that these oxidized species increased to a greater extent: 14% for C2 and >100% for C3 and C4, as compared to untreated, dyed fabric.
In a typical N(1s) XPS spectrum, nitrogen bound to carbon in primary, secondary, or tertiary amines or amides occurs at BE values of 399-400 eV, whereas for cationic quaternary ammonium nitrogen species, the peak is shifted to a higher BE value of 401.5-402.5 eV. 14 Examination of the N(1s) XPS spectrum of FA aftertreated sample indicated the presence of a higher BE signal at 402 eV, which was assigned to the cationic nitrogen in the fixing agent, as well as an uncharged component at 400 eV (Fig. 7), indicating that FA had a high content of charged nitrogen. On the other hand, FB aftertreated fabric represented only the uncharged N(1s) surface intensity (Fig. 8). In addition, dyeings aftertreated with either of the two fixing agents showed increased atomic nitrogen content (1.1% to 2.7% and 6.0% for samples 2 and 3, respectively). This increase is a reflection of the greater availability of fixative molecules to react with the cellulose hydroxyl anions or anionic dye molecules.
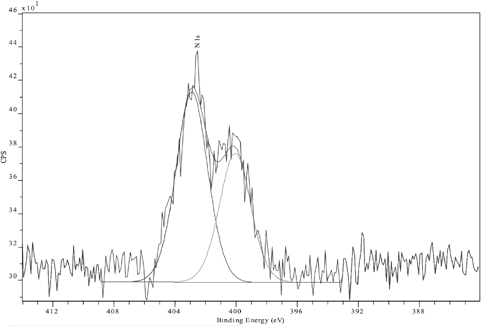
N(1s) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FA and unlaundered.
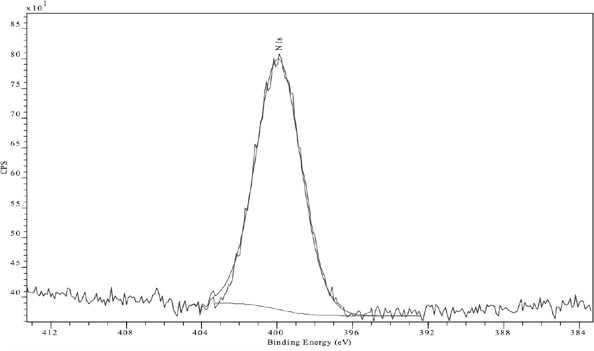
N(1s) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FB and unlaundered.
Surface Chemical Analysis after Laundering
The purpose of aftertreatment of sulfur dyed fabric with cationic fixing agent was to reduce the color loss during washing and hence extend the lifetime of the garment.
Examination of the S(2p) spectrum of untreated laundered sulfur dyed cotton (Fig. 9) demonstrated a significant increase in over-oxidized sulfur species. In addition, concomitant reduction of the surface sulfur concentration (from 1.2% to 0.5% atomic sulfur, Table I) was observed, which was presumably due to oxidation of the dye disulfide bonds leading to the formation of water-soluble sulfonated short chain dye fragments that were lost into solution. 12 Also, the surface oxygen content clearly increased, indicating exposure of the cotton to a highly-oxidative environment produced by aggressive TAED-catalyzed perborate treatment (sample 4, Table I).
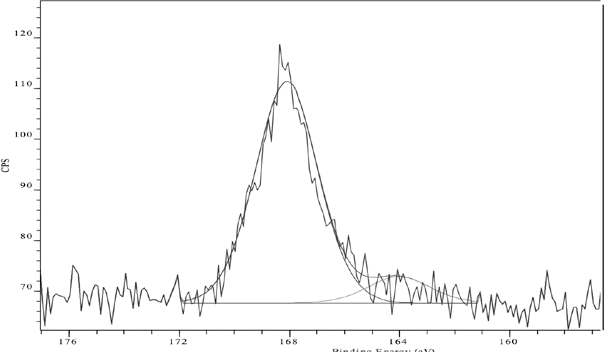
S(2p) XP spectrum of untreated and laundered Sulfur Black 1 dyed cotton fabric.
In contrast, the S(2p) XP spectrum of FA aftertreated and laundered fabric mirrored a slight (10%) increase in the level of over-oxidized species at 168 eV (Fig. 10, Table II), presumably owing to the formation of an extensively polymerized and insoluble network of fixative and dye. Similar behavior could also be observed for FB aftertreated laundered fabric; however, the increase was much greater (30%) than that for FA, indicating its lower resistance to oxidation in perborate-based washing (Fig. 11, Table II).
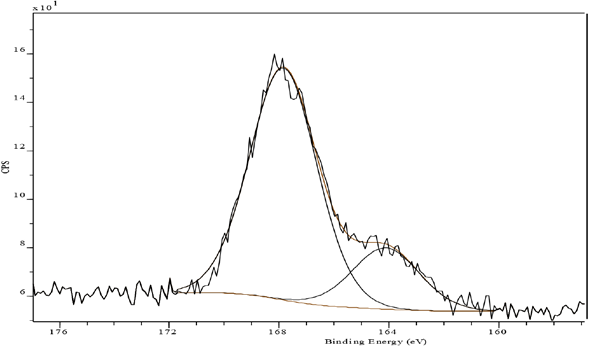
S(2p) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FA and laundered.
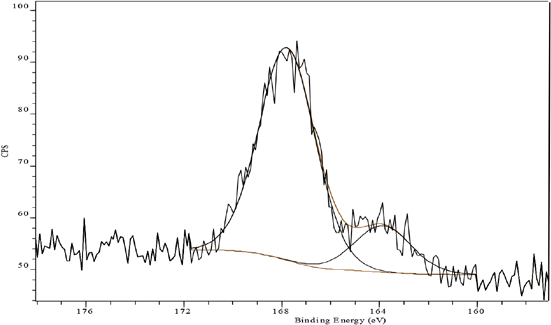
S(2p) XP spectrum of Sulfur Black 1 dyed fabric aftertreated with FB and laundered.
Surprisingly, FA and FB aftertreated and laundered fabrics showed increased atomic sulfur concentrations than their unlaundered counterparts (Table I), which possibly suggests that, due to aftertreatments, the sulfur-containing compounds in the subsurface may be exposed and are retained even after laundering. It can be postulated that the subsurface of aftertreated sample had a lower level of oxidation, showing an increased concentration of sulfur, than the unlaundered samples.
Table III shows the relative intensity data of the deconvoluted C(1s) spectra for unlaundered and laundered fabrics. Examination of untreated laundered fabric (sample 4) indicated a significant rise in oxidized species intensities at 286.6 eV (24%), 288.0 eV (>100%) and 289.0 eV (>100%). Presumably, this change was due to a loss in surface hydrocarbon residue and some oxidation of the cellulosic backbone. The effect of comparable laundering of FA aftertreated fabric (sample 5) was to reduce signal intensity of the C3 and C4 components (by 23% and 55%, respectively) indicating the presence of a far less oxidative and degradative environment. Similarly, FB laundered fabric (sample 6) exhibited a decrease in C3 and C4 components by 36% and 42%, respectively, reflecting decreased oxidized functionalities.
The effect of laundering was to reduce the surface nitrogen intensity, but for aftertreated samples a significant amount of nitrogen was still observed (Table I). This decrease was more pronounced for FB aftertreated fabric (37%), indicating the relatively poor stability of the finish on the sulfur dyed fabric under perborate-based washing conditions. However, the fabric aftertreated with FA preserved more nitrogen content after washing and there was a loss of only 7%. This shows that the effect of washing was to remove the uncharged nitrogen species from the finished fabric due to the alkaline cleaning conditions and the oxidative nature of the detergent formulation. As the amount of reduction in surface nitrogen concentration was less in FA aftertreated fabric than FB, the charged nitrogen species were more substantive to the fiber surface and durable to washing than the uncharged component.
Influence of Aftertreatment on Fabric Color
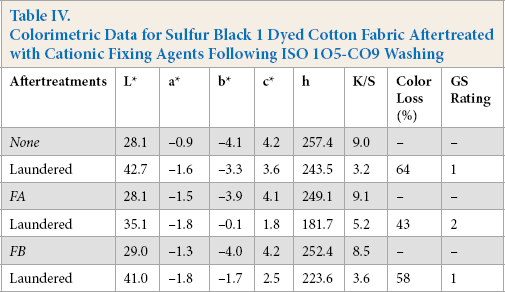
The K/S value of the dyed fabric is assumed to be directly proportional to the amount of dye present on the fabric. The color coordinates, K/S values, and the percent color loss data of untreated and aftertreated sulfur dyed cotton fabrics is presented in Table IV. The difference in K/S values of untreated and aftertreated samples before laundering was small, showing that the aftertreatment had little effect on the color strength of the dyeings. The untreated fabric underwent significant fading after laundering amounting to a color loss of 64%. In contrast, aftertreatments on sulfur dyed fabric gave noteworthy improvements in minimizing the percentage color loss, with a reduction to 43% and 58% for FA and FB, respectively. These results were in good agreement with the XPS data.
Colorimetric Data for Sulfur Black 1 Dyed Cotton Fabric Aftertreated with Cationic Fixing Agents Following ISO 1O5-CO9 Washing
Mechanism
Cationic polymers have the ability to develop strong electrostatic bonds between the positive charge on the fixative and sulfonate groups on the dye. In this case, the bond energy is of the order of 550–1000 kJ/mol, compared with 15–60 kJ/mol for normal hydrogen bonding between dye and fabric. 2 There is a potential for the formation of a large molecular size dye-cationic agent complex, with low aqueous solubility, arising from the electrostatic forces of attraction between the anionic groups in the dye and the polycations. 1 For this to occur, the dye must contain anionic groups similar to the sulfonated groups in direct dyes. In spite of the fact that little is known about the exact chemistry of the sulfur dyes, it has been proposed that some sulfur dyes may contain sulfonic acid groups. 11 Hence, the large surface area may lead to maximizing the cumulative effect of weak intermolecular forces, and also, physical entrapment.
Similarly, considering that the dyed fabric itself contains carboxylic acid groups and that these groups are further generated as a result of hydrogen peroxide oxidation in the final stage of dyeing, improved adsorption of the dye onto the fabric might may be due to the ion-ion interactions between the cationic fixing agent and the anionic carboxylic acid groups on the substrate.1,12
Conclusions
The surface-sensitive XPS analysis has demonstrated that laundering the sulfur dyed cotton fabric using the ISO 105-CO9 protocol resulted in significant surface oxidation and surface dye loss due to the activated bleaching environment.
The effect of aftertreatment was to reduce the surface dye loss by increasing the resistance of the dye to oxidation. It was apparent that FA and FB were effective in not only reducing the visual color fade, but that this protective effect was similarly reflected in surface sulfur concentration and S(2p) spectra. The intensity of oxidized carbon species also decreased as a result of aftertreatments with the cationic fixing agents as compared to the untreated fabric. Charged nitrogen species in FA were found to be essential elements for improving the washfastness of the sulfur black dyed fabric against the oxidative action of the detergent. The uncharged nitrogen of FB was less protective, probably due to a combination of poor attraction for the dye and alkaline degradation.
It can be presumed that there are either electrostatic forces of attraction between the dye sulfonate functionalities and polymer cationic charges, Van der Waals’ forces, or hydrophobic bonding between the dye molecule and uncharged and charged nitrogen species that may increase dye insolubilization, leading to the observed improvements in dye washfastness.
Footnotes
Acknowledgments
The author would like to acknowledge the financial support of a Ph.D. scholarship from NED University of Engineering and Technology, Karachi, Pakistan through the Higher Education Commission to carry out this study at the School of Materials, The University of Manchester, Manchester, UK.
