Abstract
Polyurethane foams encompass a wide range of materials which, depending on the foam structure, chemical composition, and presence of flame retardants, can be very combustible or fire resistant. The first commercial flame retardants for polyurethane foams were developed 60 years ago with a focus on fire retardant efficiency. These legacy flame retardants are relatively small volatile molecules that can migrate from the foam to the environment. Now the flame retardant industry has been changing with a focus on reactive or polymeric flame retardants. This article gives a short overview of these new commercial flame retardants.
Fire hazard of polyurethane foams
Polyurethane (PU) foams encompass a wide range of foamed materials with very different properties, starting from low-density open cell flexible and rigid foams all the way to high-density isocyanurate closed cell foams. From the point of view of response to flame, PU foams are considered to be thermally thick materials. This means that the heat applied to the foam doesn’t dissipate deeply, but stays on the surface layer. The surface reaches high temperature quickly and therefore PU foams are easy to ignite. Because of the thin walls of the cells and the availability of oxygen, PU foams, especially open cell foams, burn very fast and produce large amounts of heat in a short period of time. According to National Fire Protection Association (NFPA) statistics for 2016–2020, 1 upholstered furniture were the items first ignited in 1% of home structure fires, but those fires were responsible for 15% of civilian fire deaths and 5% of home fire injuries. In the same period, mattresses and bedding were the items first ignited in 2% of home structure fires, but those fires were responsible for 12% of civilian fire deaths and 9% of home fire injuries. A further analysis by NFPA 2 shows that the actual contribution of upholstered furniture to home fire deaths and injuries is much larger than that because fires involving fire spread to upholstered furniture from other items, when the upholstered furniture is not the first item ignited, represent the second leading fire scenario for fire deaths. Furniture and mattress fires become large quickly and can spread beyond the room of origin. Typical upholstered chairs or sofas will show a maximum heat release rate of at least 4–5 MW and produce 200–500 MJ of total heat.3,4 Figure 1 illustrates an experimental burn of two full-sized couches in a small room setup. The results are highly dependent on the exact bed assembly and the size of the mattress; when fully involved in the fire, that bed gave 1.8–6 MW peak heat release rate and the temperature in the room got close to 1000°C. 5 In other examples, mattresses have been shown to give the maximum rates of heat release of over 5 MW.6,7

Experimental burn of two couches.
Paradoxically, although PU foams are easy to ignite, it is also easy to extinguish the fire when the flame is still small. This relates to the same inherent property of the PU foam of being thermally thick material. Because the heat cannot penetrate into the depth of the foam the heated layer where the foam decomposes and produces combustible gases is shallow. Burning foam shows significant turbulence and fluctuation of the flame (Figure 2), which is an indication of the instability of combustion. Such small flames can be extinguished by small changes in the fuel supply or by decreasing the heat by means of incomplete combustion. Flame retardants (FRs) added to the PU foams are specifically designed to extinguish small accidental fires. FRs provide a variety of fire extinguishing mechanisms, which will be discussed later on.

Flame retarded foam (left) and untreated foam (right) in vertical ignition resistance test.
“No ignition—no fire” is the best strategy to mitigate the fire hazard of PU foams. However, if the ignition source is too large it can overcome the first line of defense—ignition resistance. In this case, FRs can provide the second line of defense, which decreases the heat release rate (the key property governing the intensity of a fire8,9) and the rate of fire growth and limits fire spread. This strategy is required to be used for rigid PU (and polyisocyanurate) foam insulation materials. Foam plastic insulation materials used in construction are required by US codes (International Building Code (IBC) 10 and International Residential Code (IRC) 11 ) to meet a very severe fire test (NFPA 286, room-corner test 12 ) or be covered by a thermal barrier and also meet a somewhat less severe fire test (ASTM E84, Steiner tunnel, 13 Class B) in order to be permitted for use in habitable locations (such as rooms in houses). When foam plastic insulation is used as parts of exterior wall systems (in facades) or of roofing systems, they are also required to comply with severe fire tests and be protected. Interestingly, recent full-scale tests of roofing assemblies with photovoltaic (PV) panels performed by the Italian National Association of Rigid Polyurethane Foam (ANPE) showed that in the event of panels fire the damage to the undelaying wood decking is smaller for polyisocyanurate (PIR) insulation compared with mineral wool insulation. 14 Even more severe requirements exist for foam plastic insulation used in concealed areas such as plenums. The proper FR treatment of the foam will decrease the probability of fires spreading quickly throughout buildings. When foam plastic insulation is used in cavity walls, if the insulation foam does not completely fill the wall cavity, that is another mechanism for the flame to spread rapidly upward within the concealed cavity behind the walls. 15
Traditional FRs in PU foams
Weil and colleagues16,17 published two reviews and a book chapter 18 on flame retardancy of polyurethane foams. Traditionally, aliphatic chlorophosphates, aliphatic phosphates, and aliphatic phosphonates were used or are currently being used as FRs for PU foams (Table 1). General observation from the long history of use of these FRs is that the efficiency is somehow related to the phosphorus or phosphorus plus chlorine content. However, it seems that the volatility of phosphate esters also plays an important role, probably as important as the content of phosphorus and chlorine in the molecule. Although many of the early FRs were selected on a trial and error basis, there is clear indication that the gas phase action is needed for PU foams. The mechanism and chemistries of scavenging of active free radicals in the flame by halogen and phosphorus species have been well documented.19,20
List of traditional flame retardants used (some discontinued) in PU foams.

*Isoteniscope, ASTM D2879.
Researchers at Brooklyn Polytechnic University (now NYU)21,22 studied in detail the mechanism of FR action of tris-(2-dichloroisopropyl phosphate (TDCP) in flexible PU foams. Interestingly, they found that the flexible foam extinguishes quickly if it is ignited from the bottom up because of massive volatilization of TDCP into the flame. Very little flame scorched molten material was observed on the surface of the extinguished foam. This points to the limited supply of combustible gases and explains instability of combustion of the foam. It takes a longer time to extinguish the foam when it is ignited from the top, because of the pool of molten decomposing foam (mostly polyol) that is formed and stabilizes the flame. The flame still extinguishes when the critical concentration of the FR is built up in the condensed phase and in the gas via evaporation and decomposition.
Different from flexible foams, rigid foams have a closed cell structure that prevents flash volatilization of the FR, but still the FR easily goes in the gas phase when cells open due to pyrolysis. The ignition of the rigid foams is an interesting phenomenon, because the flame flashes over the surface and then quickly retreats. If the heat flux to the surface is not sufficient the flame can extinguish. The foam may reignite again if the heating is continued. Since the rigid foam is more densely cross-linked compared with the flexible foam, it doesn’t melt away but undergoes charring. In such a scenario the best strategy to flame retard foam is trying to leverage both the gas phase and condensed phase modes of action. This is achieved by combining phosphate ester FRs and reactive bromine-based FRs. In the technical literature, there is a common perception that brominated FRs provide gas phase action (true), but their important condensed phase action is often overlooked.23–25 Commercial brominated FRs for rigid foams are typically diols or monohydric alcohols. They become part of the PU network and cannot volatilize as a molecule, but instead, bromine radicals are produced in the condensed phase. The bromine radicals need to find hydrogen in order to form stable HBr. Therefore, bromine attacks PU aliphatic chains and this leads to more crosslinks, unsaturations, and aromatization, which are all precursors of the char. 26
Isocyanurate foams are made with significant 2.0–3.5 times excess of isocyanate over polyol. Excess of isocyanate forms an isocyanurate cross-linked network. Since the isocyanurate groups are rich in nitrogen and they are thermally more stable than the urethane groups, the isocyanurate foam is intrinsically more fire resistant than rigid spray PU foams and typically do not require help with brominated FRs.
Although high volatility alkyl phosphates or chloroalkyl phosphates were correct choices for good FR efficiency, after a few decades in use they started showing other problems. FRs initially dissolved in the polymer (foam walls) will eventually migrate to the surface and slowly start volatilizing. This is especially true for open cell flexible and rigid foams. This may not lead to significant deterioration of the FR performance but volatile FRs can be detected in houses and eventually in the environment. US EPA (Environmental Protection Agency) performed detailed studies27–29 on migration of Tris-(2-chloroethyl phosphate) (TCEP), Tris-(2-chloroisopropyl phosphate) (TCPP), and TDCP from rigid foams covered with dust in stainless steel chambers and measured the dynamics of sorption on FRs on the chamber walls and different construction materials and other items found in typical homes. It was shown that volatilization depends on the vapor pressure of FRs (Table 1), but the dust doesn’t play a significant role as a vapor sink.
Some of the FRs not associated with hazards to humans or the environment at the time of the introduction to the market later on after additional studies showed problems. For example, the most efficient FRs used in rigid PU foams, dimethyl methylphosphonate (DMMP), and tris-(2-chloroethyl phosphate) (TCEP) (Table 1) were found to exhibit some degree of mutagenicity and were removed from the market. Because of high volatility, DMMP and TCEP were never used in the flexible foams. TDCP (Table 1), with significantly lower volatility, was traditionally used in flexible PU foams to pass the California (TB-117) open flame standard for upholstered furniture as well as in automotive foams to pass MVSS 302. Although the vapor pressure of TDCP is very low, it can still migrate from the foam by thermal or photo-initiated decomposition mechanisms. 30 TDCP was also listed in Proposition 65 as a suspected carcinogen in California and requires special labeling. Since 2013, California doesn’t require furniture to comply with the open flame test and therefore TDCP is not used in furniture anymore. It is probably a good decision to remove TDCP from its use in furniture, but lowering Californian regulation on the fire safety of upholstered furniture was a move in the wrong direction, especially in view of the new FR alternatives being available and developed in the market, as discussed in the following section.
Polymeric and reactive FRs
Because of the issues related to exposure to high volatility FRs in foams, it seems the logical solution would be the use of nonvolatile and relatively heavy molecules such as polymeric FRs. The potential of such polymeric molecules to migrate from the foam is very low. In addition, due to their large size, they are not likely to penetrate the cell membranes of living organisms and thus, are not expected to be bioavailable or possess intrinsic toxicity. An even more attractive idea is using FRs with alcohol or isocyanate functionalities that will participate in the foaming process and become a part of the PU network. These FRs no longer exist as chemical species once reacted into the polymer backbone, making it impossible to leach, bloom, or escape from the polymer into the environment.
These reactive FRs are not new ideas and were discussed in the technical literature already in the 1970s.31,32 Then, why this was not practiced? One of the main problems is losing the gas phase FR mode of action, which is very important for PU foams. Polymeric molecules are heavy and obviously will not volatilize as easily as the FRs listed in Table 1. Reactive FRs built into the PU polymer chains will not volatilize before chains break and may not deliver FR species to the flame fast enough to achieve critical concentration.
There are also other problems associated with the process of producing the foam. Polymeric or oligomeric FRs have high viscosity and this adds challenges with pumping and mixing them. Keeping low viscosity is especially important for rigid spray foams applied on-site and often outdoor at low temperature. Reactive FRs pose problems with the production of the foam as well. A typical reactive FR would be a short chain polyol. It might not be problematic using short chain polyols in rigid foams or in PIR foams, but it is a significant challenge in flexible foams where the short chain polyols tend to close cells and cause shrinkage.
Nevertheless, there are few successful examples of commercially available reactive and polymeric FRs. One of the earliest reactive FR for rigid PU foams is diethyl N,N bis(2-hydroxyethyl) aminomethylphosphonate (Fyrol® 6, Figure 3). It is made by reacting diethylphosphite, diethanol amine, and paraformaldehyde. It is a short chain diol with the pendant phosphonate group. P-CH2-N is a relatively weak link, and it breaks during combustion releasing diethyl phosphite, which can easily volatilize to the gas phase. Thus diethyl N,N bis(2-hydroxyethyl) aminomethylphosphonate enables permanency of the FR in the foam during service time, but provides essential gas phase mode of action in the case of fire. Because it is the short chain diol, diethyl N,N bis(2-hydroxyethyl) aminomethylphosphonate cannot be used in flexible foams.
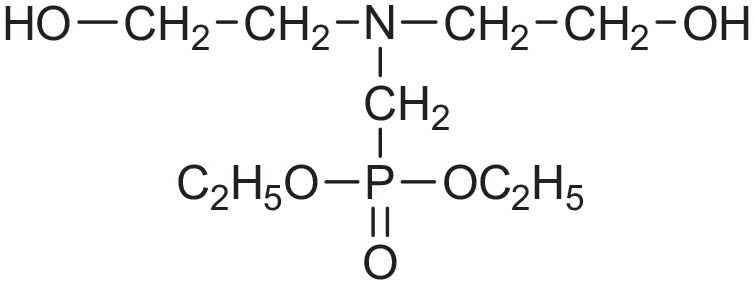
Diethyl N.N bis(2-hydroxyethyl) aminomethylphosphonate.
Another old phosphorus containing diol available in the market for rigid foams is Amgard® 82 (formerly known as Vircol® 82). It is the reaction product of dibutyl acid pyrophosphate and propylene oxide. More recently, two phosphorus-based diols Exolit® OP 550 and OP 560 for flexible PU foams were introduced to the market in Europe and the United States.
Several brominated diols (PHT-4 Diol®, Saytex® RB-79, and Ixol®) as well as a monoalcohol (FR-513) are available for use in the rigid PU foams. Since brominated reactive FRs are very viscous liquids or solids they are typically dissolved in TCPP. The bromine radicals can split from the brominated FR regardless of whether the FR is situated in the main chain or as the pendant group, and therefore design of reactive brominated FRs is less challenging.
One successful example of a polymeric FR is poly(ethyl ethylene phosphate) (Fyrol PNX and Fyrol PNX-LE, Figure 4). It is especially favored by the automotive industry where low emissions (VOC) and fogging are critical. Although the average molecular weight of PNX is about 900 Dalton, it is not bioaccumulative and it shows very high efficiency in flexible PU foams. It has a high phosphorus content of 19% that is partially responsible for the high efficiency. It is also believed that PNX can depolymerize upon heating perhaps, initiated by the attack of polyol OH groups that results in producing cyclic 1,2-ethylene ethyl phosphate (Figure 4). This volatile cyclic phosphate provides gas phase action. Thus, PNX can be considered as a “smart” FR that sits dormant during normal use, but the flame triggers its action. PNX still has a high viscosity, however; due to very high efficiency it is used in the foam at low concentrations and therefore high viscosity can be tolerated. Based on data available so far, this polymer is not expected to be toxic.
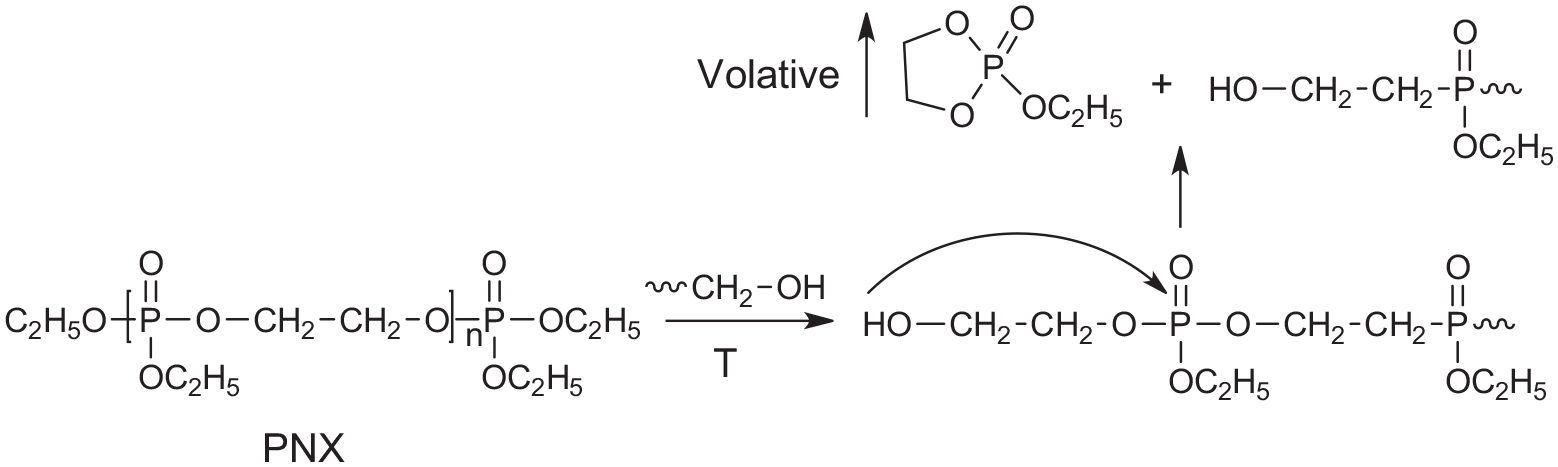
Mechanism of depolymerization of poly(ethyl ethylene phosphate).
New developments
As discussed above, the first generation of FRs for PU foams was developed with the maximum FR efficiency in mind. This was achieved with additive-type nonreactive FRs. Other unintended consequences of using relatively mobile chemicals in the high surface application (foam) were not taken into consideration at the time. To be fair, at the time there were no sensitive analytical methods available to monitor the chemicals in the environment at very low concentrations. During the past years, it has become clear that new sustainable FRs for foams should be reactive, or polymeric, or solid inorganics (not desirable for the PU industry, traditionally using only liquids).
Recently, two reactive sustainable FRs were introduced to the market: VeriQuelTM R100, presented at the CPI conference in 201433 and VeriQuel F100, presented at the CPI conference in 2016. 34 VeriQuel R100 is a reactive monofunctional, an all phosphorus-based FR specifically designed for PIR foams as a single FR and it is also used in combination with reactive brominated FRs in the PUR (polyurethane) spray foam. VOC and solvent extraction experiments confirmed the complete incorporation of VeriQuel R100 into the PU network. The efficiency of VeriQuel R100 is similar to TCPP and it can replace TCPP on a one-to-one basis in most rigid PU foam applications. For example, both TCPP and VeriQuel R100 can achieve Class I in ASTM E-84 tunnel test at 15 parts loading in the PIR form of 270 isocyanate index. 33 Being halogen-free VeriQuel R100 also shows lower smoke generation. Based on the data available so far, the product is not expected to be toxic to human health or the environment.
VeriQuel F100 is also monofunctional and an all phosphorus-based FR, but it is designed for flexible PU foams. VeriQuel F100 is highly efficient and requires 1.5–2.5 times lower loading compared with TDCP. For example, 7 parts of TDCP is required in 29 kg/m3 polyester polyol flexible PU foam to pass automotive MVSS 304 test, whereas only three parts of VeriQuel F100 are needed to pass this test. 34 Because of such low loading the essential physical properties of the foam are fully preserved. As measured in common automotive industry emission tests DIN 75201 and VDA-277, VeriQuel F100-based foam shows FOG and VOC numbers similar to non-FR foam and significantly lower than TDCP. Toxicity studies performed on this product came out favorably.
It seems counterintuitive that new developments are focusing on monofunctional FRs, which are in essence chain-terminators in the foaming process and may affect the physical properties of the foam. However, due to the high efficiency and low concentration, it doesn’t happen. In contrast, monofunctional FRs give obvious advantages because they are always positioned as pendant groups attached to the PU network and therefore, provide life service permanence, but at the same time labile enough to split from the polymer in the case of fire and provide very desirable gas phase action. Another benefit of monofunctional FR is the relative forgiveness in the foaming process. Being monofunctional alcohols, they don’t tighten foam as much as difunctional and multifunctional FRs and can be formulated with minimal adjustments.
Conclusion
And finally, a few remarks about the cost and long-term benefits of polymeric and reactive FRs. There is a common notion that any new more sophisticated FR product introduced to the market is likely to be more expensive than the old one being replaced. However, we always need to look at the cost-performance in use. As discussed above, proper design of polymeric and reactive FRs can significantly cut the use dosage and as a result cost in use becomes similar or sometimes even more favorable than traditional FRs. Combining safer toxicology and the environmental profile of reactive and polymeric FRs with lower use levels brings their safety margins even higher. Although there are no studies available on the potential loss of FR performance of traditional FRs over long service life, it is clear that reactive and polymeric FRs by design should not have this problem.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
