Abstract
Cotton fabric (CF) was coated with a new multifunctional flame-retardant system consisting of melamine phosphate (MP) and nickel (II) dimethylglyoxime (NDMG). The new coating was applied to enhance the flame retardancy, antibacterial activity, thermal stability, color strength, UV protection, and electrical properties of CF. The weight percentages of NDMG and MP that led to the maximum improvement in CF properties were identified. According to vertical and horizontal flammability tests, coating the fabric with 29%MP/1%NDMG prevented flame propagation in the samples. Moreover, it increased the limiting oxygen index (LOI) of CF from 17.9% to 36.9%. Thermogravimetric analysis (TGA) data presented that the MP/NDMG coating enhanced the char residue at 750°C. Antimicrobial investigations indicated that the coating 29%MP/1%NDMG reduced the growth of
Keywords
Introduction
Cotton fabrics (CFs) are natural biomacromolecules and have outstanding properties such as biodegradability, good hydrophilicity, dyeability, air permeability, and renewability. Therefore, CFs are exceedingly utilized in the home textiles and clothing industries.1–3 For decades, electrical properties have been the subject of study in textile materials.
4
Particularly, the dielectric properties of CFs have attracted researchers’ interest because cotton is the most manufactured natural polymer in the world and is used in a variety of textile-based electronic devices.5,6 Unfortunately, CF is a highly flammable material with a LOI of 16% - 18%, and it fails to prevent flame propagation in horizontal and vertical flammability tests according to ISO 3795, ASTM D6413, and ISO 11,925-2 standard test methods.1–3,7 To enhance the flame retardancy of CF, flame retardant materials are applied to the fabric using an after-finish method.
8
Phosphorus-nitrogen flame retardants are widely studied to replace halogenated flame retardants that have bad side effects on humans and the environment. The role of phosphorus as a flame retardant appears in the condensed phase, where it is converted at high temperatures to phosphorus-rich compounds. During the pyrolysis of CF, the phosphorus-rich compounds prevent the formation of levoglucosan and flammable volatile compounds and extremely increase the rate of dehydration of cotton cellulose into a char layer.9–14 On the other hand, nitrogen-containing compounds realize flame retardancy in the gas phase. During combustion, the nitrogen-containing compounds degrade and produce non-flammable gases in the combustion zone. These non-flammable gases spread over the surface of CFs and form a preventive layer that forbids the penetration of flammable gases into the combustion zone.15–17 Melamine phosphate (MP) is a P/N flame retardant, and it can be prepared by the reaction between phosphoric acid and melamine in the presence of methanol as a medium.
17
MP is widely used with other flame-retardant materials, such as pentaerythritol, to enhance the flame retardancy of polymers and textiles. Makhlouf et al.
3
used MP with nano-chitosan to improve the flame retardancy of CF. Polyvinyl alcohol (PVA) was used in the coating formulations as a binder. The interesting results in this study are the ability of PVA/MP/nano-chitosan coatings to improve the LOI value of cotton fabric from 17.2% to 57.9%. And the values of after-flame time (
Nickel (II) dimethylglyoxime (NDMG) is a metal chelate complex. It can be prepared through the reaction between nickel (II) chloride and dimethylglyoxime. The final product has a pink color, but when the pH of the solution is increased to 8 pH units, the colour becomes red.18–20 It was reported that the NDMG complex showed certain antibacterial activity against
Herein, CF was coated with NDMG, MP, and MP/NDMG. PVA (2 wt%) was used as a binder in coating formulations. The flammability, thermal stability, antimicrobial activity, UV protection, color fastness, mechanical, durability, and electrical properties of the coated fabrics were studied. The flame retardancy mechanism of MP/NDMG was suggested.
Experimental work
Materials
CF (100%) with a surface density of 235 g/m2 was acquired from Texmar Company, Egypt. PVA was purchased from SD. Fine-Chem Limited (SDFCL), India. PVA has a molecular weight of 85,000 – 24,000 g/mol, and the degree of hydrolysis is 86%–89%. Melamine (99%), phosphoric acid (85%), and methanol (99%) were obtained from Alfa Aesar Company, Germany. Dimethylglyoxime (≥97%), nickel (II) chloride hexahydrate (≥98%), and sodium hydroxide pellets (≥98%) were supplied by Sigma-Aldrich Company, Germany. Deionized water was obtained from a select fusion water purification unit that was purchased from Purite Limited Company, UK.
Synthesis of NDMG
NDMG was prepared through the reaction between nickel (II) chloride hexahydrate and dimethylglyoxime. In a 1 L glass beaker, 29.7 g of nickel (II) chloride hexahydrate was dissolved in 500 mL of deionized water using a magnetic stirrer at room temperature. Also, in a 2 L glass beaker, 29.3 g of dimethylglyoxime were dispersed in 1 L of deionized water at room temperature for 2 h. Then, nickel (II) chloride solution was added dropwise to the dimethylglyoxime dispersion, and the solution color became pink. At the same time, a 1 M sodium hydroxide solution was prepared. Sodium hydroxide solution was added dropwise to the NDMG solution with continuous stirring until the pH of the solution reached 10 pH units and the color of the solution became completely red. The solution was filtered using a vacuum pump, and the product was washed many times with deionized water to eliminate the surplus sodium hydroxide. The product was dried at 45 °C in an oven for 3 h and pulverized to a fine powder.
Synthesis of MP
MP was synthesized by the reaction between 50 g of phosphoric acid and 30 g of melamine in the presence of 400 mL of methanol as a medium. The reaction was fulfilled in a round-bottom flask with a condenser. The flask was placed on a hot plate (at 120°C) with a magnetic stirrer, and the reaction continued for 4 h. The final product was nominated, washed with 600 mL of methanol, dried at ambient temperature for 96 h, and crushed to a smooth powder.
Preparation of flame-retardant coating and its application on CF

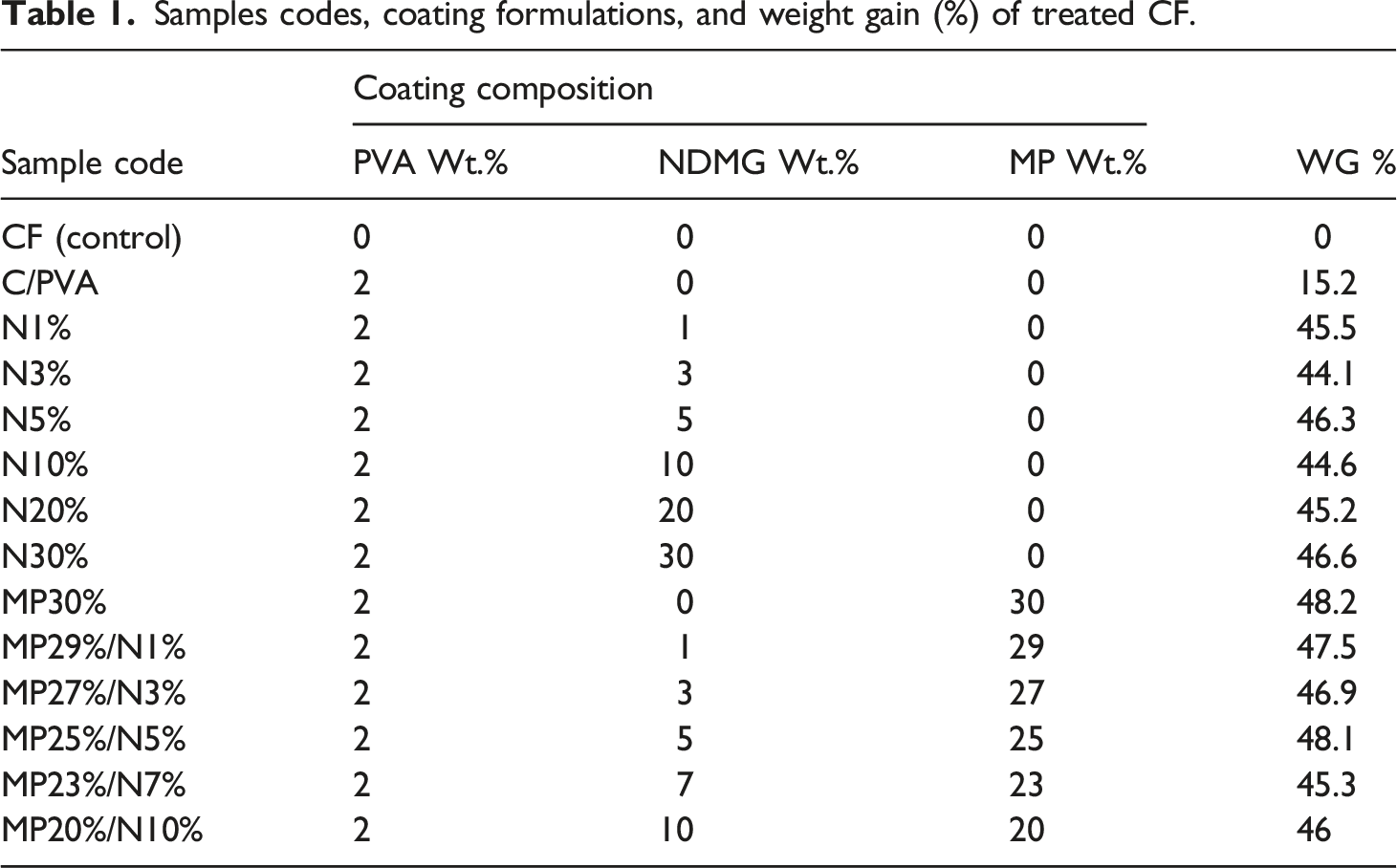
Samples codes, coating formulations, and weight gain (%) of treated CF.
Characterization
FTIR spectra were obtained in the optical range 4000-400 cm−1 using a Nicolet 380 spectrophotometer. XRD analysis was implemented using an Empyrean diffractometer from Panalytical Co., the Netherlands. The diffraction angle range was between 2θ = 4° and 2θ = 60°, and the scanning rate was 0.013°/s. pH measurements were performed with a Jenway 3510 bench pH/mV meter. Before measuring the pH value of the solution, the instrument was calibrated with 4, 7, and 10 standard buffer solutions. The pH values were measured at 25 °C. The UL94 flame chamber (purchased from Fire Testing Technology, FTT Ltd, UK) was used to perform vertical and horizontal flame spread tests. The rate of burning was determined using the ISO 3795 (1989) standard test method. The vertical flame spread test was carried out following ASTM D 6413 (2016). The samples’ dimensions were 25.5 cm × 9 cm. The results reported here are the averages of three measured samples. The LOI values were measured by an oxygen index instrument according to ISO 4589–2 (2017). The samples' dimensions were 15 cm × 5 cm. Thermogravimetric analysis (TGA) measurements were performed from room temperature up to 750°C under a nitrogen atmosphere using the Shimadzu DTG60 instrument. The heating rate was regulated at 10°C/min, the nitrogen flow rate was adjusted at 30 mL/min, and the weights of the samples were between 7 and 8 mg. SEM pictures were obtained by FEI-Quanta 250 FEG-SEM, Holland. Energy-dispersive X-ray spectroscopy (EDX), coupled with SEM, was used to conduct the elemental analysis.
Color-matching spectrophotometer (model Colour Eye 3100-SDL England) was used to measure colour strength (K/S) in accordance with AATCC 110-2000. K/S values were calculated from reflectance data using Kubelka-Munk equation (2).
Where K/S is the ratio of absorption and scattering coefficients and R is the reflectance at the wavelength of maximum absorbance of the dye, 650–700 nm for red dyes. The ultraviolet protection factor (UPF) was measured by a UV–3101 Spectrophotometer purchased from Shimadzu Company, Japan. Antibacterial activity investigations against G +ve and G -ve bacteria were conducted according to AATCC 147-2004 and the results were expressed using Zone inhibition (in mm). Four-terminal HP 8144 A precision RLC metre was used to measure the dielectric constant (



where
Results and discussion
Characterization of NDMG and MP
The final product of NDMG was characterized by FTIR, SEM, EDX, and XRD. The FTIR spectrum of NDMG is presented in Figure 1(a), and it shows characteristic bands at 1242 and 1105 cm−1, which are referred to symmetrical and asymmetrical stretching vibrational modes of N–O. The band at 3438 cm−1 is attributed to the OH group. The intense absorption band at 1565 cm−1 is attributed to the C=N group. The bands at 517 and 424 cm−1 are assigned to the Ni-N bond. The bands at 1368 and 986 cm−1 are due to the N–OH bond bending vibrational mode and the N–O symmetric stretching vibrational mode, respectively. The band at 748 cm−1 refers to the C=N-O group. The absorption bands at 1790 and 2924 cm−1 refer to intramolecular hydrogen bonding and the C-H group in the NDMG complex. These results confirm the formation of NDMG, and they are in accordance with literature data.18–20 (a) FTIR and (b) XRD of NDMG and MP, (c),(d) SEM photographs of NDMG and MP.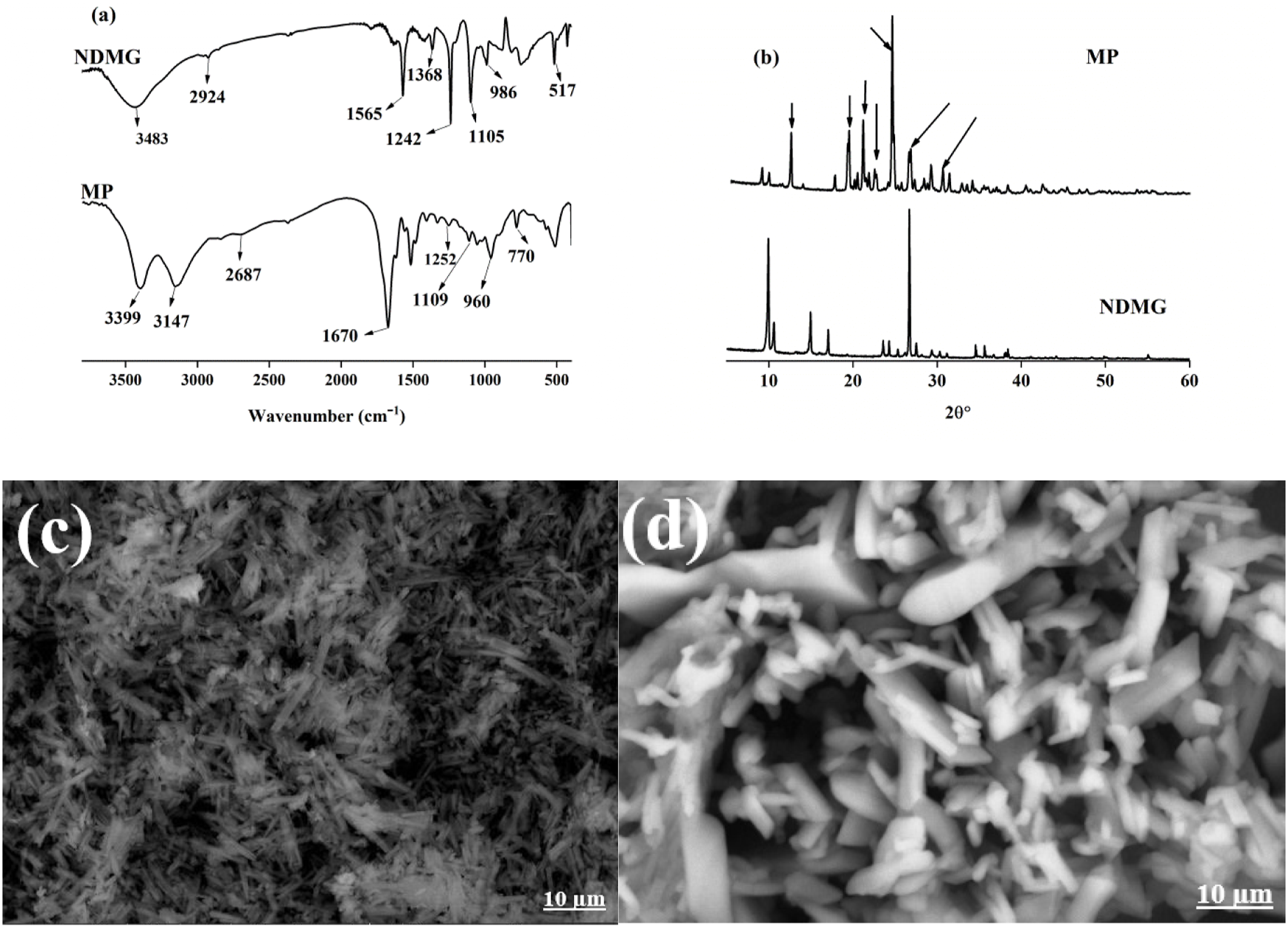
XRD analysis of NDMG in Figure 1(b) shows a diffraction peak with high intensity at 2θ = 9.9° due to the (110) plane, and it confirms the synthesis of a highly crystalline structure. 20 The XRD peaks at the 2θ values of 10.6°, 14.9°, 17.1°, 23.6°, 24.2°, 25.3°, 26.7°, 27.5°, 28.1°, 29.2°, 32.61°, 34.5°, 35.6°, 38.4°, 43.67°, and 46.08° can be referred to as the (200), (211), (130), (002), (510), (112), (022), (521), (240), (710), (150), and (242) planes, respectively, and they indicate the formation of NDMG with good purity. 20
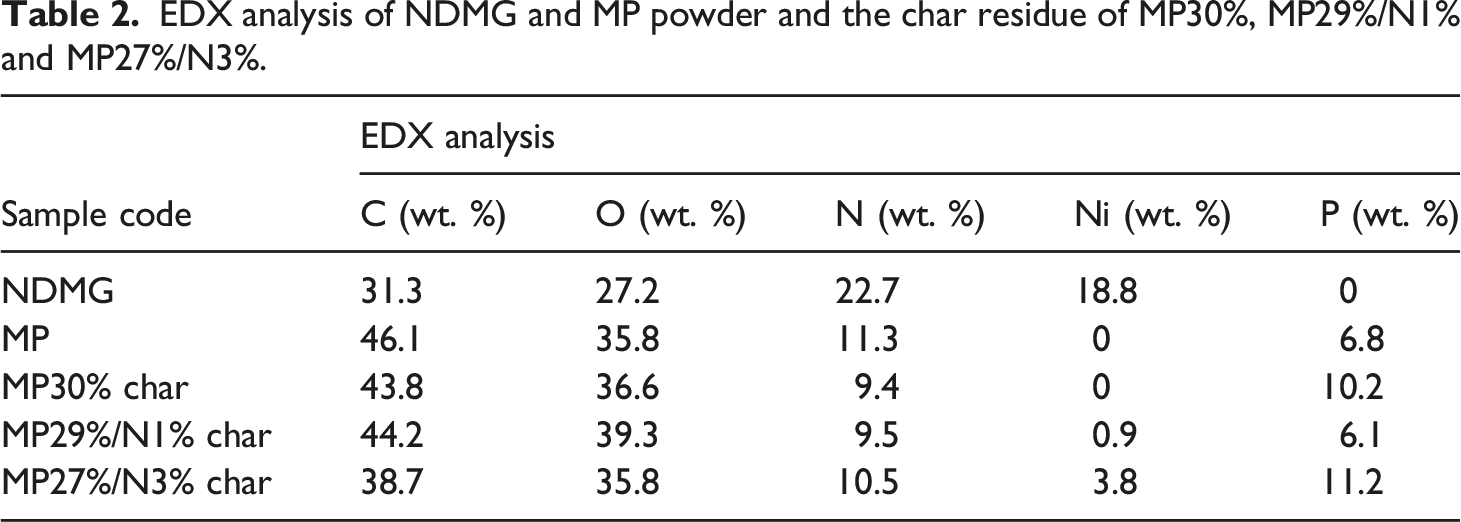
EDX analysis of NDMG and MP powder and the char residue of MP30%, MP29%/N1% and MP27%/N3%.
Morphology of control and treated CFs
SEM is used to study the surface morphology of control and treated samples. The SEM image of the control sample in Figure 2(a) shows that the surface is smooth and crimped. SEM images of the samples N30% and MP29%/N1%, Figure 2(b), (c), manifest that the flame-retardant substances are present on the surface and between fibres. Also, it is clearly seen that NDMG, in the form of a nanofiber-like structure with various lengths, is located on the surface and between fibres. The adhesion of flame-retardant coatings on CF surfaces is attributed to that PVA can form hydrogen bonds with CF and the selected flame retardants.2,3,26 SEM images of control (a), N30% (b), and MP29%/N1% (c).
TGA data of control and treated CFs
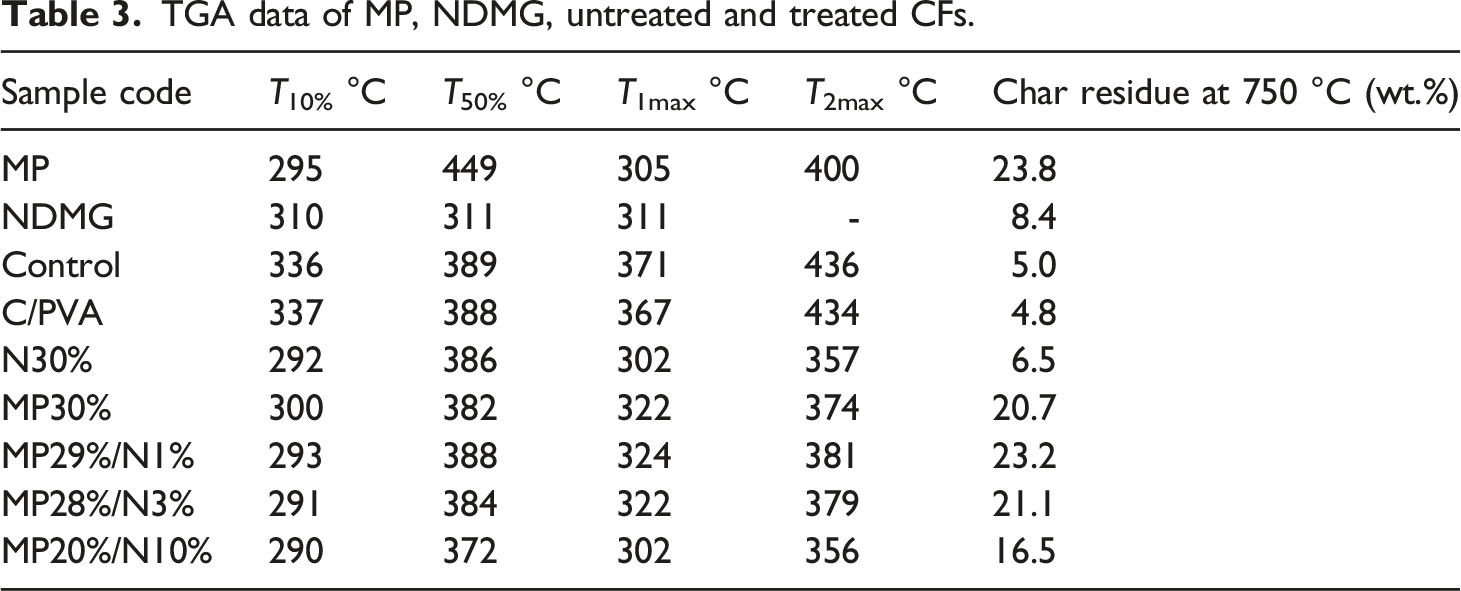
TGA data of MP, NDMG, untreated and treated CFs.
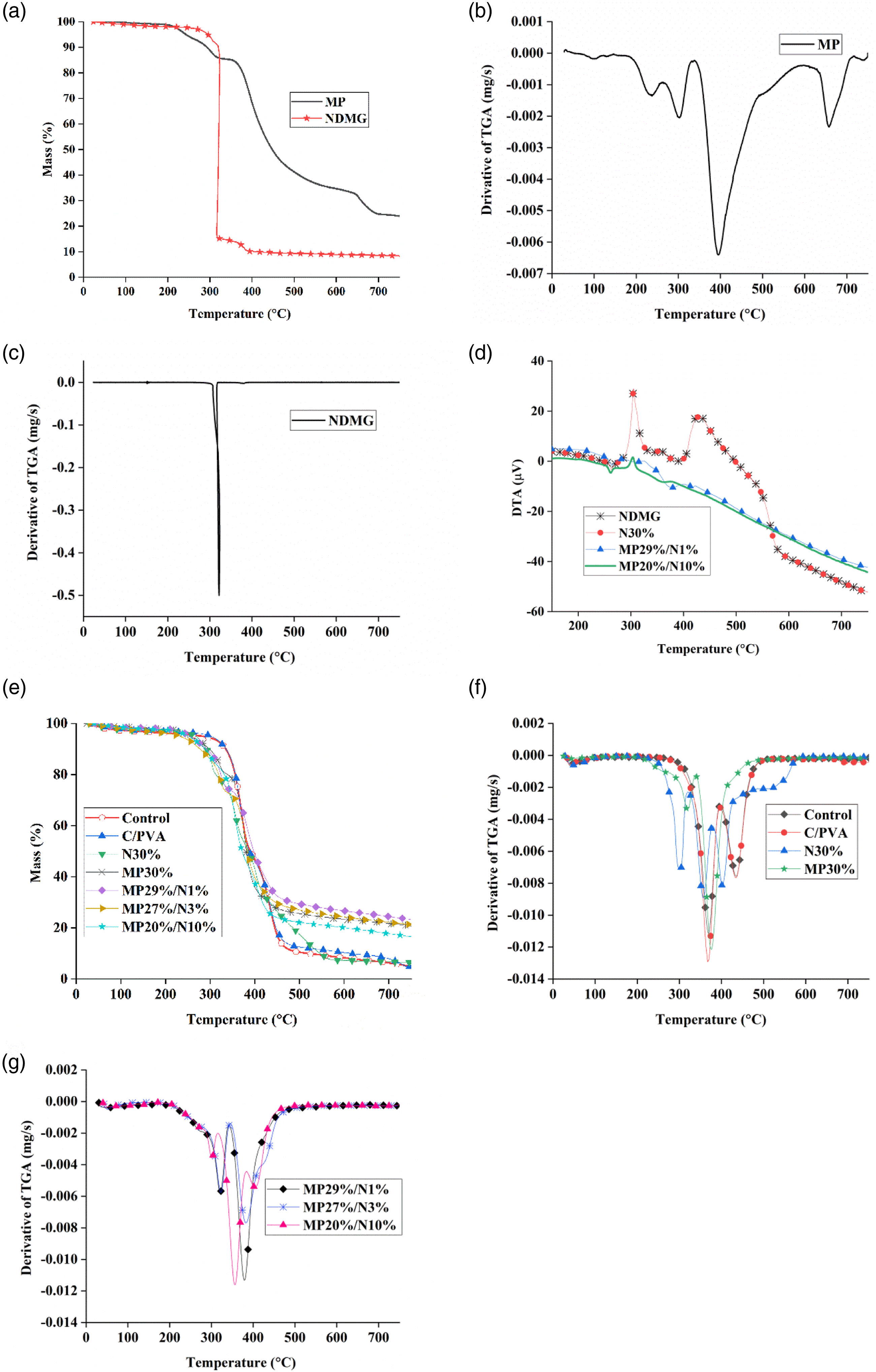
(a), (e) TGA of MP, NDMG, control and treated samples, (b, c, f, g) derivative of TGA of NDMG, MP, control, and treated samples, (d) DTA of NDMG, N30%, MP29%/N1%, and MP20%/N10%.
The control sample attains
Flammability properties
Horizontal and vertical flammability data
Results of horizontal and vertical flammability tests, and LOI.
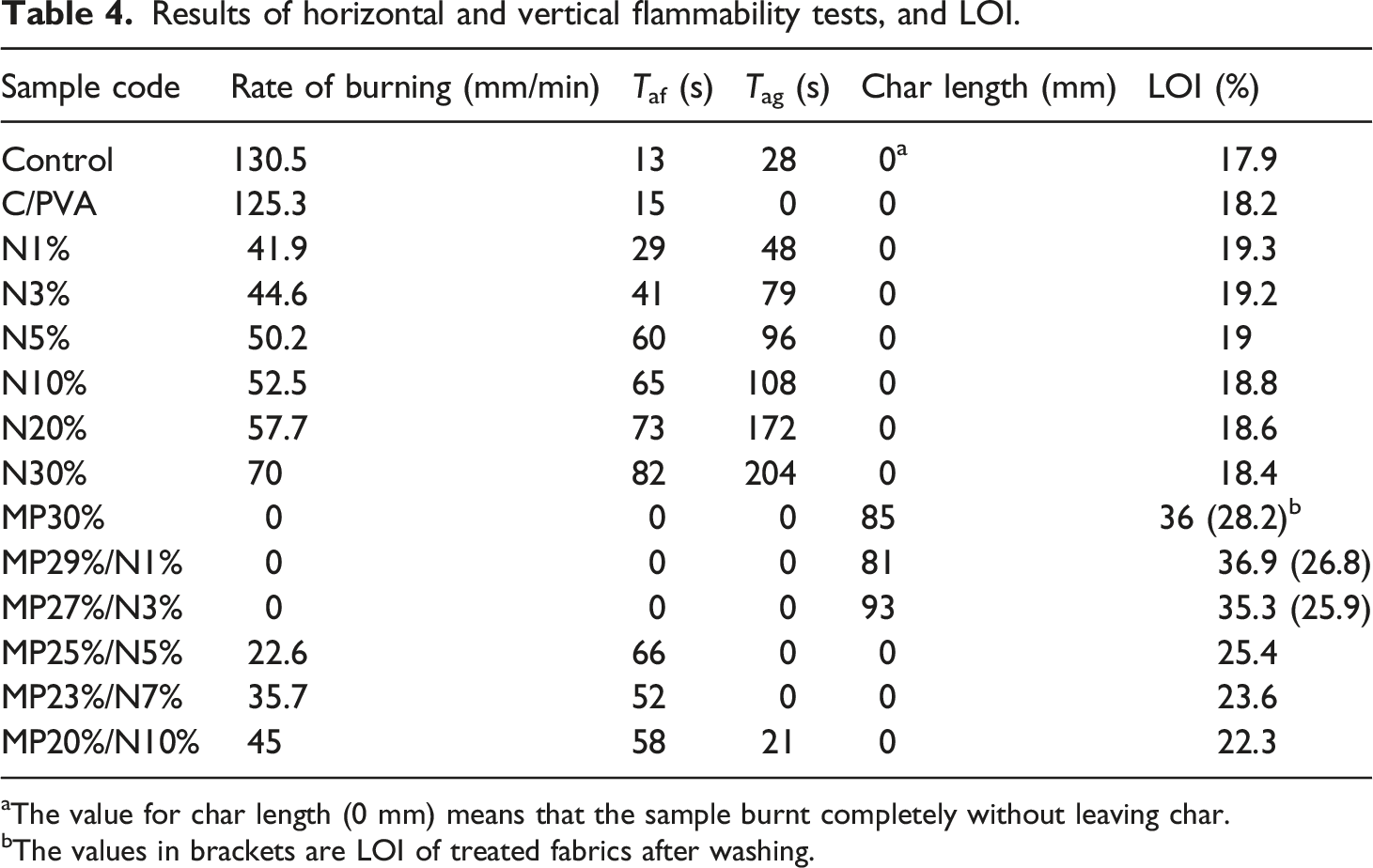
aThe value for char length (0 mm) means that the sample burnt completely without leaving char.
bThe values in brackets are LOI of treated fabrics after washing.
Vertical flammability test data following ASTM D 6413 (2016) is presented in Table 4. The control sample ignites at the lower end as soon as it meets the flame, and it burns completely with Digital photographs of (a) control, (b) N20%, (c) N30%, (d) MP30%, (e) MP29%/N1%, (f) MP27%/N3%, (g) MP25%/N5%, (h) MP23%/N7%, and (i) MP20%/N10% after vertical flammability test.
LOI data
The LOI data for the control and coated samples are shown in Table 4. Control and C/PVA samples have LOI 17.9% and 18.3%, respectively. PVA/NDMG coating shows a little enhancement in LOI value, where the LOI of CF increases by 1.4%, 1.3%, 1.1%, 0.9%, 0.7%, and 0.5% in N1%, N3%, N5%, N10%, N20%, and N30%, respectively. It is obvious from Table 4 that as the loading level of NDMG grows up in coating composition, the enhancement in LOI decreases. The addition of PVA/MP to CF (MP30% sample) enhances the LOI of CF to 36%. ∆ LOI% is utilized to express the improvement in LOI values, and it is the difference between the LOI% of the treated sample and the LOI% of the control. ∆ LOI% attains 18.1% in the MP30% sample. Furthermore, the LOI of MP29%/N1% is 36.9%, and Δ LOI reaches 19%. This indicates that the addition of 1 wt% NDMG to the PVA/MP coating can cause more enhancement in the flame retardancy of the control sample compared with the PVA/MP coating alone. The LOI values of MP27%/N3%, MP25%/N5%, MP23%/N7%, and MP20%/N10% samples are increased by 16.7%, 7.5%, 5.7%, and 4.4%, respectively, relative to the control sample. However, the LOI values drops in MP27%/N3%, MP25%/N5%, MP23%/N7%, and MP20%/N10% samples in comparison with the LOI of MP30% and MP29%/N1% samples. This indicates that as the loading level of NDMG in the coating’s formulation increases, LOI values decrease, as shown in Table 4. This agrees with the data obtained from vertical and horizontal burning rate data. The reasons for this behavior are discussed in section (Flame retardant mechanism). The LOI and vertical flammability test data indicate that the optimum concentration of the NDMG in a PVA/MP/NDMG coating is 1 wt% (MP29%/N1% sample), where the LOI value is enhanced, and the char length (after the vertical flammability test) is decreased compared with the MP30% sample.
To evaluate the durability of the flame-retarded CFs, MP30%, MP29%/N1%, and MP27%/N3% were washed with deionized water containing 2 g/L detergent at room temperature. The samples, after the washing process, were dried in an oven at 60 °C for 2 h and tested by an oxygen index instrument. The LOI results are presented in brackets in Table 4. The LOI values of MP30%, MP29%/N1%, and MP27%/N3% are 28.2%, 26.8%, and 25.9%, respectively, after the washing process. These results indicate that MP30%, MP29%/N1%, and MP27%/N3% samples still have good flame retardancy action after washing.
Flame retardancy mechanism
SEM images, EDX, and FTIR analysis were used to study the surface morphology, elemental composition, and functional groups of char residue. Figure 5(a), (a1), (b), (b1), (c), (c1)) shows the SEM images of MP30%, MP29%/N1%, and MP27%/N3% samples after the vertical flammability test, and the EDX analysis is presented in Table 2. SEM images of the MP30 sample, Figure 5(a), (a1)), show the formation of a char layer with a porous structure, and the fibers of CF remained after the test, which means that MP succeeded in conferring good protection for the fabric against fire. This agrees with the digital photograph in Figure 5(a) for the MP30 sample after the vertical flammability test. EDX analysis of MP30% in Table 2 shows that the char contained C (43.8.4%), N (9.4.8%), P (10.8%), and O (36.6%) atoms. FTIR analysis in Figure 5(d) displays an absorption peak at 3422 cm−1 (for NH2 and OH of adsorbed moisture); 2958 and 2923 cm−1 (for C-H of CH2 and CH3); and 2859 cm−1 (for C-H of alkane); 1730 cm−1 for (C=O); 1650 and 1504 cm−1 (for C=C aromatic); 1411 and 717 cm−1 (for C-N); 782 cm−1 (for P-O-C); 1245 cm−1 (for P=O); 1088 cm−1 (for P-N); 1015 cm−1 (for P-O-P); 782 cm−1 (for C-H aromatic); and 480 cm−1 (for O-P-O).2,3,17 According to SEM, EDX, and FTIR analysis of the char residue of the MP30% sample, it is expected that MP coating decomposed to give NH3, CO2 and water vapor, which diluted the oxygen concentration in the combustion zone in the gas phase. In the condensed phase, MP was converted to polyphosphoric acid, which crosslinked with the OH of the fabric to form a coherent and compact char layer. The decomposition of cellulosic fabric coated with MP led to the formation of char, which contained a polyaromatic structure with P-N, C-N, C=O, C-H, P-O-P, and P-O-C groups. This char was able to prevent the flame from spreading in the vertical flammability test. ((a, a1), (b, b1) and (c, c1)) are SEM images of MP30%, MP29%/N1%, and MP27%/N3% char residue after vertical flammability test, and (d) FTIR analysis of char residue of MP30% and MP29%/N1% samples.
SEM images in Figure 5(b), (b1)) of the MP29%/N1% sample present the formation of an intact, cellular, and dense char layer. In addition, the char succeeded in saving cotton fibers from fire. Elemental analysis in Table 2 indicates that the char contained C (44.2%), P (6.1%), N (9.5%), O (39.3%), and Ni (0.9%). The SEM images of the MP27%/N3% sample in Figure 5(c), (c1)) display that the char has a cellular structure with small bubbles on the surface. But the images also indicate the presence of certain cracks on the char layer. The elemental analysis of the char displays that the char residue contained C (38.7%), N (10.5%), P (11.2%), Ni (3.8%), and O (35.8%) atoms (Table 2). FTIR analysis of the MP27%/N3% sample in Figure 5(d) displays an absorption peak at 3425 cm−1 (for NH2 and OH of adsorbed moisture); 2944 cm−1 (for C-H of CH2 and CH3); 1725 cm−1 for ( C=O); 1605 cm−1 (for C=C aromatic); 1407 cm−1 (for C-N); 872 cm−1 (for P-O-C); 1245 cm−1 (for P=O); 1091 cm−1 (for P-N); 1009 cm−1 (for P-O-P); 721 cm−1 (for P-O-P); and 480 cm−1 (for O-P-O and Ni-O).17,20 It is clear in Table 4 that the addition of NDMG at 1 wt% to MP enhances the flame retardancy of CFs. At concentrations > 1%, the enhancement in flame retardancy decreases, and an antagonism effect appears. This behavior was confirmed by flammability and thermal analysis measurements. The synergistic effect between MP and NDMG in the MP29%/N1% sample is due to that the NDMG decomposes during combustion and produces NH3, H2O, and CO2 in the gas phase. In addition, MP degradation leads to the production of NH3 and H2O. These non-flammable gases dilute the oxygen concentration in the combustion zone. Moreover, they assist in the intumescence of the char layer and the formation of cellular structure and bubbles on the surface of CF. In the condensed phase, Ni2+ react with polyphosphate groups to form nickel phosphate (NiP) with terminal P-OH groups. These terminal groups interact with the (OH) groups of CF to form phosphate ester. Moreover, Ni2+ may be act as a catalyst for melamine polyphosphate (which is produced from MP with increasing temperature) to crosslink and set up a small number of bridges between two phosphate groups. This stabilizes the structure of melamine polyphosphate and makes more phosphorus available for phosphorylation of CF.30–36 Therefore, in the sample MP29%/N1%, the addition of NDMG to MP leads to an enhancement in char residue in TGA data, as shown in Table 3. In contrast, the higher concentrations of NDMG (>1%) lead to the formation of higher concentrations of NiP in the condensed phase. The higher concentrations of NiP make melamine polyphosphate chains crosslinked by many salt bridges, and the individual chains become rigid and their mobility greatly restricted. This is equivalent to taking a part of MP from the system, and this leads to a decrease in the flame retardancy of coated fabrics.30,31 Therefore, the char residue in TGA data in Table 3 decreased to 21.9% and 16.3% as in samples MP27%/N3% and MP20%/N10%, respectively.
Mechanical and physical properties
The mechanical and physical properties, namely tensile strength, air permeability, UPF, and K/S, of control and treated samples are presented in Figure 6. It is clear in Figure 6(a) that there is a slight decrease in the tensile strength values of the processed cotton relative to the control sample. The decline in tensile strength can be attributed to the diffusion of NDMG and MP into the microstructure of the CF. This leads to the rigidity of the fibrillar structure of cotton and hence a decrease in the tensile strength of the fabric.29,37 (a) Tensile strength, (b) air permeability, (c) UPF value, and (d) K/S values of control and treated samples.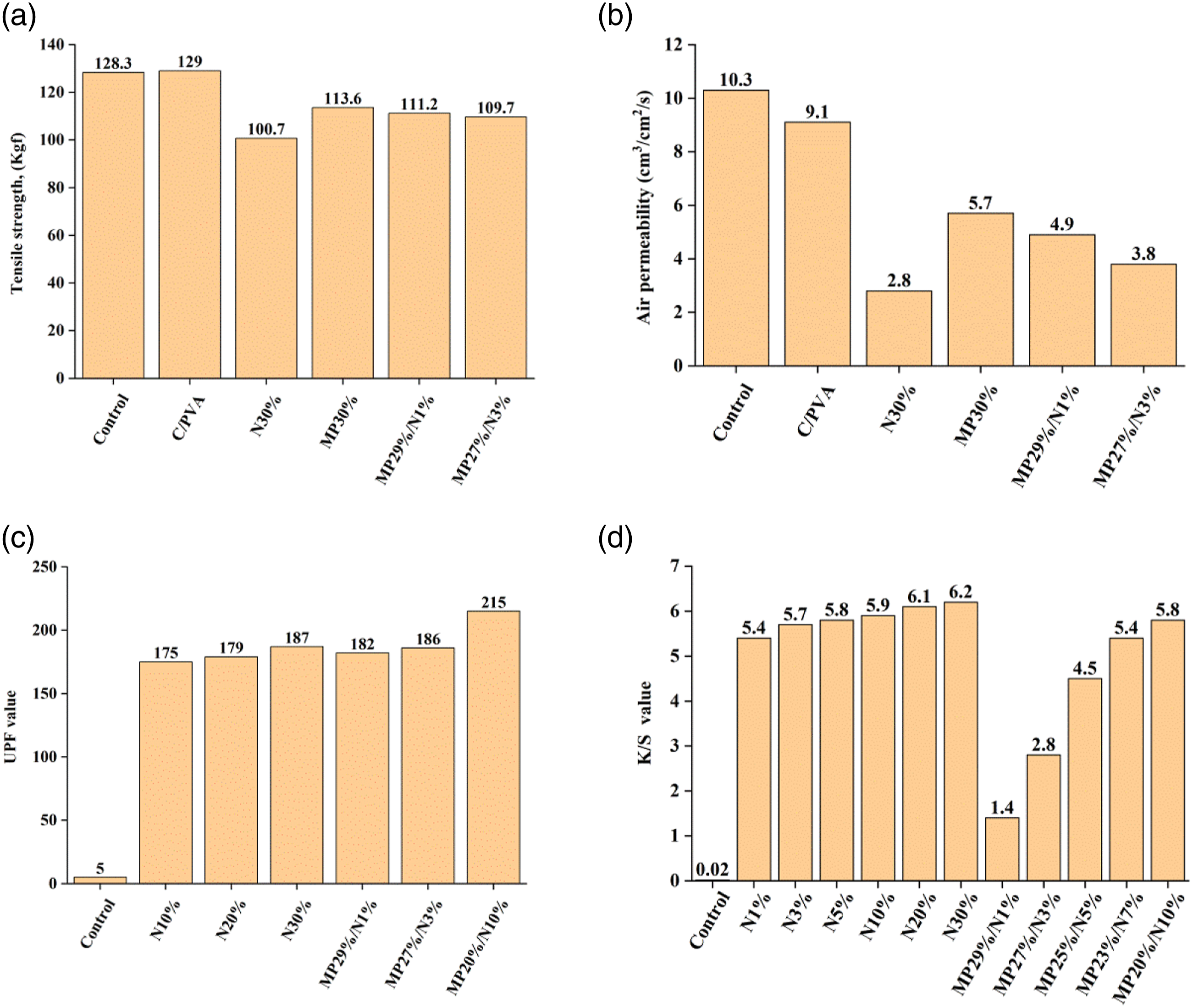
The air permeability data of control and coated fabrics are shown in Figure 6(b). It is clearly seen that the air permeability of cotton is decreasing with the addition of coatings. This can be attributed to the decline in the air space between fibres after the coating process. 38 The maximum reduction in air permeability value is observed in the N30% sample. Furthermore, MP29%/N1% and MP27%/N3% show a decrease in air permeability compared to MP30%. This means that the addition of NDMG to the coating formulation has a negative effect on air permeability due to its ability to diffuse in the microstructure of the CF and reduce the air spaces between fibres.
UV protection
The results of the ultraviolet protection factor (UPF) of treated samples are presented in Figure 6(c). The Australian Standard for Sun Protective Clothing (AS 4399:2020) presents three UPF classifications based on the magnitude of solar UVR blocked. These classifications are minimum, good, and excellent protection and are based on UPF value. Minimum and good protections are attained when the UPF value reaches 15 and 30, respectively. Excellent protection is achieved when the UPF value is 50 or higher. According to the data in Figure 6(c), the coated samples have excellent UV protection, where the UPF value is ≥175. The addition of NDMG greatly enhances the UPF value of CF, which can be attributed to the strong absorption properties of NDMG in the UV region. 39
Colour strength
The value of K/S is usually utilized to point out the amount of dye content in the coloured fabrics. The increase in the K/S value indicates greater colour intensity, and consequently, a higher amount of dye is taken up by the fabric. The results of K/S are presented in Figure 6(d). The K/S value of the control sample is 0.02 and is increased by the addition of NDMG to coating formulations. The data in Figure 6(d) indicate that all the NDMG-coated samples (N1%, N3%, N5%, N10%, N20%, and N30%) have close K/S values (5.4 – 6.2). The higher K/S values for these samples, compared to the control sample, are attributed to the presence of NDMG with a red colour and PVA, which supports the residence of NDMG on the surface and between the fibres of the fabric. It can also be seen in Figure 6(d) that the increase in NDMG concentration in the coating formulations does not show a significant change in the K/S value. This may be attributed to the absence of chemical bonds between NDMG and CF. The results in Figure 6(d) show that MP has a great effect on colour strength. The K/S values of the samples coated with MP/NDMG are reduced by the increment in MP concentration in the coating formulation. For instance, the K/S value of MP20%/N10% is 5.8, while the MP29%/N1% sample has a K/S value of 1.4. This behaviour may be attributed to the presence of free acidic OH groups in MP. When MP is added to the PVA/NDMG system, the pH of the solution declines from 7.5 pH unit (where the metal complex, NDMG, has a red colour) to 4.3 pH unit (where the metal complex has a pink colour). As a result, as the concentration of MP decreases (and thus the free acidic OH groups decline) and the concentration of NDMG increases in the coating formulations, K/S values begin to rise again, as shown in Figure 6(d). The K/S values of MP27%/N 3%, MP25%/N 5%, MP23%/N 7%, and MP20%/N10% are 2.8, 4.5, 5.4, and 5.8, respectively.
Antimicrobial activity
CF can confer suitable conditions for the growth of microorganisms on its surface. The hydrophilic properties of CF enable it to preserve oxygen, nutrients, and moisture, which are necessary for growing different kinds of bacteria. Therefore, it is substantial to improve the antibacterial activity of the fabric. It has been reported that dimethylglyoxime alone doesn’t have antibacterial action against G +ve and G –ve bacteria on its own and that the metal chelate (NDMG) has only minor antibacterial action.
18
The antimicrobial activities of control, C/PVA, MP30%, MP29%/N1, MP27%/N3%, and N30% against G +ve and G –ve bacteria are presented in Figure 7(a), (b), and the results are expressed by inhibition zone diameter (IZD) in mm. Figure 7(a) clearly shows that control, C/PVA, and N30% samples have no significant antibacterial action against G +ve and G –ve bacteria. Bacteria were unable to grow on the surface of the sample MP30%, as shown in Figure 7(b). Coating the CF with MP/NDMG, as in the samples MP29%/N1% and MP27%/N3%, enhances its antibacterial activity. MP29%/N1% sample shows IZDs of 13 and 16 mm for G +ve and G –ve bacteria, respectively, while MP27%/N3% sample presents IZDs of 12 and 14 mm. The higher activity of MP/NDMG coating against G +ve and G –ve bacteria may be attributed to the existence of (O=P-OH) groups in MP, which reduce the polarity of nickel ions mainly due to the sharing of their positive charge with the donor groups. This behavior enhances the lipophilic nature of the nickel atoms, supports their permeation via the lipid layer of the microorganism more efficiently, and hence destroys G +ve and G –ve bacteria more forcefully.
40
In conclusion, NDMG alone and MP alone were not able to make a great improvement in the antibacterial properties of CF. In contrast, the addition of NDMG/MP coatings to CF can improve its antibacterial and flame-retardant properties. (a) Antimicrobial activity of control (1), C/PVA (2), N30% (3) against G +ve and (g)ve bacteria, and (b) antimicrobial activity of MP30% (4), MP29%/N1% (5) and MP27%/N3% (6) against G +ve and (g)ve bacteria.
Electrical properties
The internal responses of dielectric materials to an alternating electric field are quantified by their dielectric properties. The dielectric constant (ε`), the dielectric loss (ε``), and AC resistivity ( (a-f): Dependence of the dielectric constant (ε`), dielectric loss (ε``), and AC resistivity (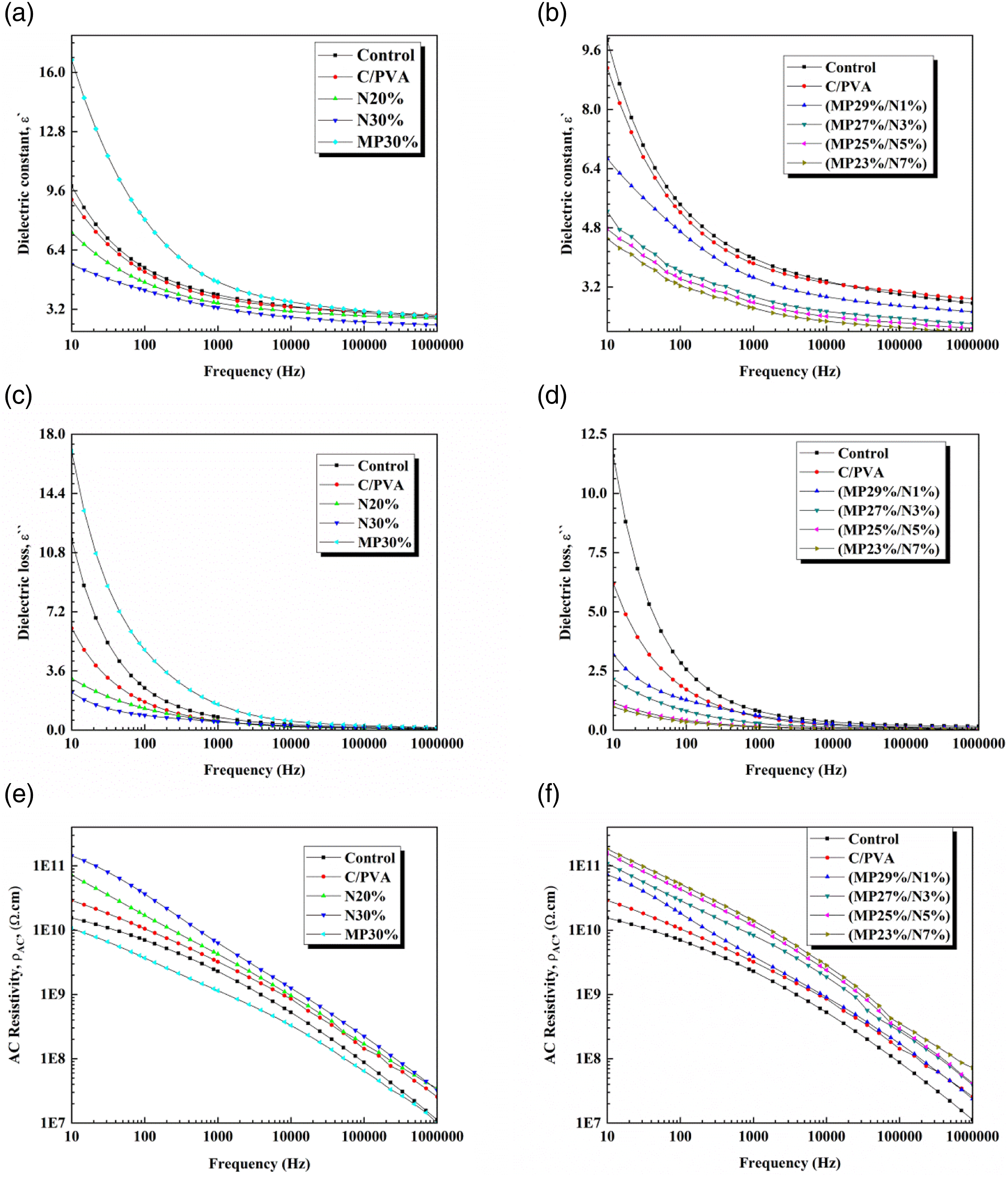
Conclusions
CF was coated with NDMG, MP, and MP/NDMG to enhance its thermal, antibacterial, and flame retardancy properties. TGA data indicated that coating the fabric with 30% MP and 29% MP/1% NDMG enhanced the char residue at 750°C to be 20.9% and 23%, respectively. The addition of higher concentrations of NDMG (>1 wt%) in coating formulations led to a decrease in the char, which remained at 750°C. The rate of burning data showed that NDMG can only slow down the flame spread in CF. Meanwhile, MP and MP/NDMG can inhibit the flame spread in the fabric, as was clear from the results of the MP30%, MP29%/N1%, and MP27%/N3% samples. Furthermore, the addition of NDMG at a concentration above 3 wt% in the coating system decreased the flame retardancy properties compared with the MP30% and MP29%/N1% samples. The LOI value of the control sample was enhanced by adding MP and MP/NDMG coatings, and the maximum enhancements were 36% and 36.9%, respectively, as observed in MP30% and MP29%/N1% samples. Cotton fabric`s antibacterial activity against G +ve and G –ve bacteria was enhanced by adding MP/NDMG coating. UPF data indicated that the coated samples presented excellent UV shielding, and the UPF values of the coated samples were higher than 175. The colour strength (K/S) values of coated samples were in the range of 1.4 – 6.2. The addition of MP and MP/NDMG to the fabric’s surface significantly reduced its tensile strength. The dielectric characteristics of the CF were improved by adding NDMG and MP. The treated sample (MP23%/N7%) exhibited the best value of Ac electrical resistivity (1.40E + 10 Ω cm at 1000 Hz), and it had a low dielectric constant of 2.6 at 1000 Hz.
Footnotes
Authors’ contribution
Aksam Abdelkhalik: Conceptualization of the work; collecting, analysis and interpretation of data, and preparing the draft of the work, and approved the version to be published. Ghada Makhlouf: Design of the work, carrying out experimental work, preparing the draft of the work and revised the manuscript, and approved the version to be published. Heba Ameen: Design of the work, carrying out the experimental work, preparing the draft of the work and revised the manuscript, and approved the version to be published. Abear Abdullah El-Gamal: Conceptualization of the work, performing the experimental work, preparing the draft of the work, revised the manuscript, and approved the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
