Abstract
Natural volatile antibacterial and anti-mycotoxin tea tree oil (TTO) with rice bran (RB) were used as a solid carrier for achieving a sustained release profile with high antimicrobial efficiency in polyethylene films. Acrylic acid (AAc) monomer was grafted onto a low-density polyethylene (LDPE) through melt blending using a Brabender Plasti-Corder. The low-density polyethylene-grafted acrylic acid (LDPE-g-AAc) was thoroughly characterized by attenuated total reflectance–Fourier transform infrared spectroscopy. LDPE and LDPE-g-AAc (80/20) were mixed with different contents of untreated RB and treated TTO/RB using melt blending to obtain sustainable composites, namely LDPE/LDPE-g-AAc/RB and LDPE/LDPE-g-AAc/TTO-RB, respectively. The effect of the addition of untreated and treated RB on the properties of biocomposites was assessed by using mechanical, barrier, and thermal properties. A prominent decrease in water vapor transmission rate occurred when adding 30 wt% of TTO/RB to LDPE/LDPE-g-AAc blend compared to virgin polymer. This decrease was due to the barrier effect of lignocellulosic material, particularly at high bio-filler content. The prepared biocomposites revealed good thermal stability when compared to virgin LDPE. Moreover, the biodegradability and antimicrobial properties of LDPE/LDPE-g-AAc/TTO-RB biofilms were enhanced with increasing TTO/RB contents from 10 phr to 30 phr due to the combination between LDPE-g-AAc and TTO. The obtained data revealed excellent possibility for using biopolymer grafted with antimicrobial TTO by adding RB for food packaging and biomedical purposes.
Keywords
Introduction
Polyolefin including low-density polyethylene (LDPE), high-density polyethylene, and polypropylene1,2 has actually paid great attention for scientists because of their use in a wide spectrum of engineering applications such as automobile, electrical cables, health-care purposes, food packaging, and other industrial applications. The major reasons are due to their availability from simple olefins monomer, easy processing respect to other polymers, excellent mechanical properties, as well as molding and recycling process. Furthermore, their resistance to ultraviolet irradiation and hydrophobicity nature could be a promising to increase their resistance to microorganism’s activity. Polyolefins are highly stable to the environmental effects, have long life, and can take several decades for the degradation. 3 These characters open opportunities for their use in geogrids for roadway purposes, especially in dessert region. Currently, polyolefin like LDPE is used as antibacterial and antifungal films for smart packaging when blended with modified clay and carvacrol oil. 4 Rozenblit et al. 5 have developed a new volatile antimicrobial oil–polyacrylic acid for prolonging shelf life of fresh foods. However, globally growing plastic consumption-based petroleum raises essential challenges regarding human health and environmental concerns, as these materials are dangerous due to either their production or their disposal in landfill locations and hence their marketability is limited. To avoid these drawbacks, lignocellulosic materials (i.e. agro-wastes) can be mixed with them to offer an adequate option for enhancing their properties or creating new composites with reasonable cost for end-use products. Currently, lignocellulosic materials have attracted a great attention of most researchers as “Green or Biomaterials” with much potential uses in a diversity of fields such as biodiesels,6,7 green energy,8,9 drug delivery, ceramic insulators, 10 and bio-composting purposes. 11 Among them, coffee grounds,12,13 kenaf fibers, 14 date stones, 15 rice husk,16,17 are designated as renewable or biomass materials. Nowadays, these biomaterials are used as reinforcing fillers with many different types of polymers or biopolymers to produce low density and economic sustainable biocomposites which lead to carbon dioxide emission reduction and fuel and environment saving especially when these kinds of materials are assembled in vehicle or aircraft component. 11 Nevertheless, their hydrophilicity limits their practical use as reinforcing material for hydrophobic polymers in industrial sectors. At present, numerous studies have been done on agro-wastes not only for enhancing their hydrophobicity in turns of improving the compatibility within the polymer matrices but also to reduce the risk related to their disposal in landfill zones. Among these literature, some focused mainly on coupling or compatibilizing agents addition to the polymer matrix,13–15,18,19 others on chemical treatment of agro-wastes by antibacterial resins, 20 or by torrefaction process, which is a thermal treatment ranging from 230°C to 300°C under nitrogen gas, with the intent of obtaining high hydrophobic material (i.e. minimize water uptake). 12 In this respect, Jagadish and Ray 21 have reported that the addition of natural pineapple leaves to epoxy resin, the mechanical properties were greatly enhanced. Vercher et al. 22 have studied the influence of natural fiber types like rice husk and pine wood sawdust on the properties of polyvinyl chloride and polyethylene, respectively, to produce wood–plastic composites. The obtained physico-mechanical results revealed that the wood–plastic composites can be potential candidates for outdoor decking applications rather than wood itself.
Nowadays, essential oils (EOs) extracted from plants such as moringa oil and tea tree oil (TTO) have paid much attention as a promising volatile antimicrobial compatibilizer for enhancing the interfacial adhesion between the bio-filler and the polymer, in addition to the antimicrobial behavior. Recent studies23–27 offer that EOs have been considered as antimicrobial agents when mixed with plastic or bioplastic for extending of shelf life foods. This is because of their healthful properties, stability, safety, as well as high antibacterial activity.28–31 With regard to the previous literature, the grafting of the branched LDPE by acrylic acid (LDPE-g-AAc) using melt-blending has not yet been performed to create a new component with different properties such as molecular weight, crystallinity, melting point, and thermal stability. In the present study, the prepared LDPE-g-AAc was characterized by an attenuated total reflectance–Fourier transform infrared (ATR-FTIR) spectrometer with the aim to make sure the process occurred. Afterward, LDPE was blended with the prepared LDPE-g-AAc to obtain LDPE/LDPE-g-AAc blend (80/20). The blend was then incorporated with various ratios (i.e. 10, 20, and 30 wt%) of low-cost untreated rice bran (RB) and it is treated by antimicrobial and anti-mycotoxin TTO. The main objective of this applied research was to estimate the mechanical properties, thermogravimetric analysis, barrier properties, biodegradability, and antimicrobial activity of the investigated biocomposites.
Experimental methods
Materials
LDPE food grade (density 0.93 g cm−3, melt flow index (190°C, 2.16 kg) 6.0 g/10 min, and softening point 87.4°C) was purchased from ExxonMobil Chemical (Kingdom of Saudi Arabia). Egyptian RB was cultivated and grinded using crushing machine to obtain RB powder with a particle size less than 250 µm. TTO with specific gravity ranging from 0.88 g mL−1 to 0.90 g mL−1 at ambient temperature was supplied by oil unit (National Research Centre, Giza, Egypt). It is composed of a mixture of antimicrobial 1.8 cineole and terpinen-4-ol compounds (Figure 1). Acrylic acid (AAc) monomer (purity 99%), Mohr’s salt, and benzoyl peroxide were purchased from Sigma–Aldrich (Germany). All other reagents were used without further purification. For the treatment procedure of RB-TTO, a homogenous RB with TTO essential oil (9:1 wt%) was prepared by using a mechanical stirrer at 40 r min−1 for 10 min. The obtained RB-TTO mixture was kept in a desiccator for mixing.
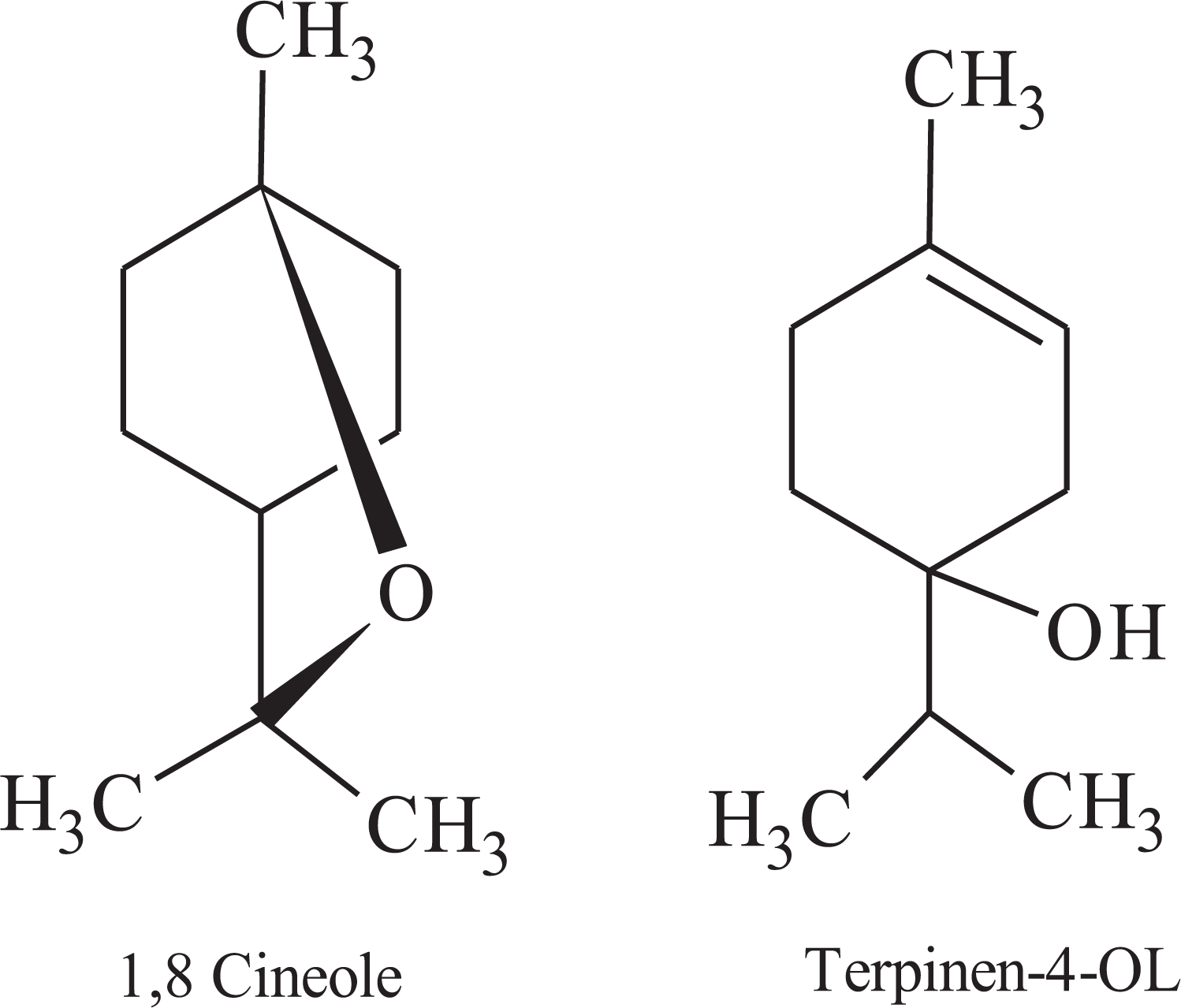
Chemical structure of TTO.
Methods
Grafting process of LDPE and AAc
The compounding strategy of grafting reaction of AAc onto LDPE was carried out using a Brabender Plasti-Corder Model PL 2100 (USA). Next, 80 g of LDPE was premixed in the mixing chamber of the compounder at 170°C with a gear speed of 80 r min−1 for 3 min. Then, 20 wt% of AAc was added and mixed with 4 g of Mohr’s salt and 1 g of benzoyl peroxide for additional 7 min for a total mixing time of 10 min. After extruding time, the mixture was quickly removed from the mixing chamber and cooled at ambient condition. The obtained LDPE-g-AAc was kept in a desiccator for characterization.
Identification of LDPE-g-AAc by FTIR spectroscopy
ATR-FTIR spectroscopy was conducted to clarify the chemical grafting between AAc and LDPE using a PerkinElmer 11650 FTIR spectrometer (Wellesley, Massachusetts, USA). The spectrum was measured in the range of 500–4000 cm−1 at a resolution of 4 cm−1 and 32 scans. The tested samples were prepared using a hydraulic press (Euro Press men, Mackey Bowley, UK) under a pressure of 120 kg cm−2 at 170°C for 30 s to obtain thin film of 200 µm in thickness.
Preparation of LDPE/ LDPE-g-AAc /RB and LDPE/PE-g-AAc/TTO-RB biocomposites
Prior to the melt blending, both LDPE and RB were dried in a laboratory oven at 65°C for 24 h. As presented in Table 1, LDPE and PE-g-AAc were mixed with untreated RB and treated TTO-RB at different ratios as follows: LDPE was preincorporated at 170°C for 3 min in a closed mixer as mentioned above, before adding LDPE-g-AAc, for 5 min, and then RB or TTO-RB filler was added for a total incorporating time of 10 min at a screw speed of 80 r min−1. For the LDPE and LDPE-g-AAc blends, LDPE was firstly mixed for 5 min before adding LDPE-g-AAc while retaining the previous conditions. The obtained mixes were molded to the desired forms for different tests and characterizations. They were then pressed in a molding set made up of 100 × 100 × 2 mm3 thick spacer located between two Teflon papers and two steel sheets, and each assembly was preheated for 5 min in a hydraulic press at 170°C, followed by compression under a pressure of 120 kg cm−2 for 2 min, and air cooling under a certain load until the assembled mold reached room temperature. The investigated mixes with RB and TTO-RB were labeled according to the ratio of RB and TTO-RB in the composite, as displayed in Table 1.
Sample designations and tensile properties for LDPE, LDPE-g-AAc, and their biocomposites.
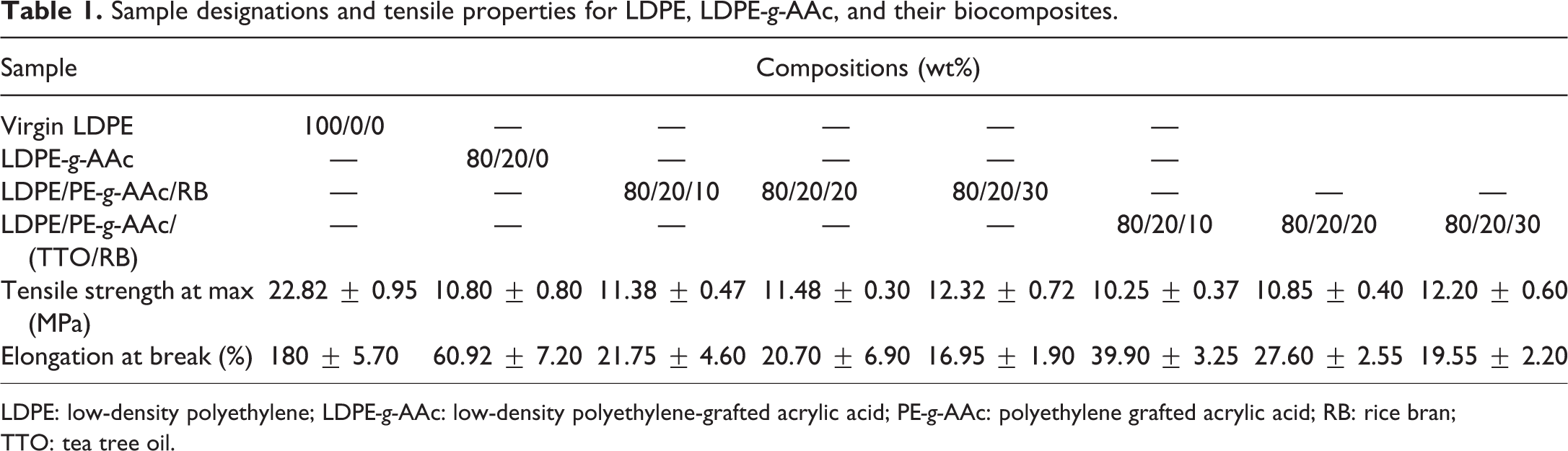
LDPE: low-density polyethylene; LDPE-g-AAc: low-density polyethylene-grafted acrylic acid; PE-g-AAc: polyethylene grafted acrylic acid; RB: rice bran; TTO: tea tree oil.
Mechanical properties
Tensile testing was performed by using a Zwick tensile testing machine (Model Z010, Germany), at a temperature of 23 ± 2°C. Stress–strain measurements were performed on dumbbell-shaped samples with an initial grip separation of 20 mm and a crosshead speed of 50 mm min−1 according to the ASTM D 638-14 standard. Five random specimens were cut from the compressed sheet and conditioned at 23 ± 2°C and 50 ± 5% relative humidity for not less than 40 h prior to the test. The obtained results are provided as the mean value and standard deviations were calculated.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was carried out under nitrogen atmosphere by means of a PerkinElmer TGA7 apparatus (PerkinElmer, Waltham, Massachusetts, USA). Gas flow of 30 mL min−1, heating rate of 10°C min−1, and a temperature range of 25–600°C were used for the analyses. Samples mass used for the experiments were about 10 mg, and the average of three replicates was recorded.
Permeability studies of the prepared biocomposites materials
Water vapor transmission rate (WVTR) and oxygen transmission rate (OTR) for virgin LDPE, LDPE-g-AAc, and their biocomposites were conducted using a Water Vapor Permeability Analyzer, Model W303-B according to ISO 9932-90 and ASTM D 3985-17 standards, respectively. This equipment is based on the cup method to measure smooth and uniform plastic sheets. Each specimen was cut from the sheet in ring shape of 50 mm and of 90 mm in diameter with 200 µm in thickness. The test was repeated three times, and the average values were recorded.
Biodegradability test
The biodegradability experiment of virgin LDPE, LDPE/LDPE-g-AAc blend, and their filled composites was performed in soil environment. The soil with botanical compost was taken from the field of agriculture, and then poured into a plastic pot (200 × 200 × 80 mm3) up to a height of around 70 mm. The investigated specimens (30 × 30 × 2 mm3) were weighed (Wi) and then buried in the pot to a depth of about 10 mm. Tap water was sprayed twice a day to maintain the moisture. The samples were carefully removed from the soil, cleaned, and dried in an oven at 60°C for 1 h. The specimens were reweighed (Wf) every 7 days for 30 days. The biodegradability percentage was calculated according to the procedure described in the previous article by Dalev et al. 32 as the following equation:
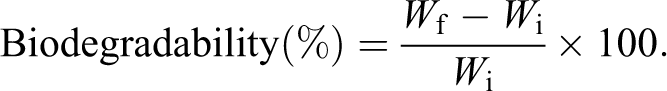
Evaluation of antimicrobial activity
The antibacterial activities of LDPE/LDPE-g-AAc/RB and LDPE/LDPE-g-AAc/(TTO/RB) composites were studied using petri dish agar method. It was employed to evaluate the antimicrobial activity of composites samples.33–36 The antimicrobial activities of 0.5 cm diameter LDPE disc (5 mm) saturated with about 1 mg sample and dried at room temperature under sterilized conditions. The composites samples discs were placed on agar plates seeded with test microbes and incubated for 24 h at the appropriate temperature of each test organism. Biocomposites samples were examined against four different microbial strains, that is, Staphylococcus aureus ATCC6538-P (G+ve bacteria), Escherichia coli (G−ve bacteria), Candida albicans ATCC 10231 (yeast), and Aspergillus niger NRRL A-326 (fungi). The bacterial and yeast test microbes were grown on a nutrient agar medium (DSMZ1) of the following components (g L−1): peptone (5.0), meat extract (3.0), agar (20.0), distilled water (1000.0 mL), and the pH to 7.0. On the other hand, the fungal test microbe was cultivated on Czapek-Dox medium (DSMZ130) of the following ingredients (g L−1): sucrose (30.00), NaNO3 (3.0), MgSO4 × 7 H2O (0.50), KCl (0.50), FeSO4 × 7 H2O (0.01), K2HPO4 (1.0), agar (18.0), distilled water (1000 mL), and the pH was adjusted to 7.2. The culture of each test microbe was diluted by distilled water (sterilized) to about 107–108 cells mL−1, then 1 mL of each was used to inoculate 1L-Erlenmeyer flask containing 250 mL of solidified agar media and finally poured in petri dishes (10 cm diameter containing 25 mL). Discs (5 mm diameter) were placed on the surface of the agar plates previously inoculated with the test microbe and incubated for 24 h for bacteria and yeast but for 48 h for fungus at 37°C and 30ºC, respectively.
Results and discussion
Characterization of LDPE-g-AAc
ATR-FTIR spectrum is a rapid technique used to study the chemical grafting between AAc and LDPE in the range of 500–4000 cm−1. The spectrum of virgin LDPE was also carried out for comparison. The results showed a good identification of the grafting process between LDPE and AAc by melt blending method as shown in Figure 2. It can be seen that the main peaks of AAc appeared at 979 cm−1, 1626 cm−1, and 1718 cm−1, which are assigned to the vibration bands of H in C=C–H, C=C, and C=O of carboxylic acid, respectively. 37 When LDPE and AAc are melt-mixed together, the characteristic peak of C=C is absent in case of LDPE-g-AAc with a reduction in the intensity of C=O band. This absence confirms that the chemical grafting between AAc monomer and LDPE polymer is achieved, as depicted in Figure 3. The facile grafting processes compared to carcinogenic solvent methods are very promising methods for preparing green polymer blends.
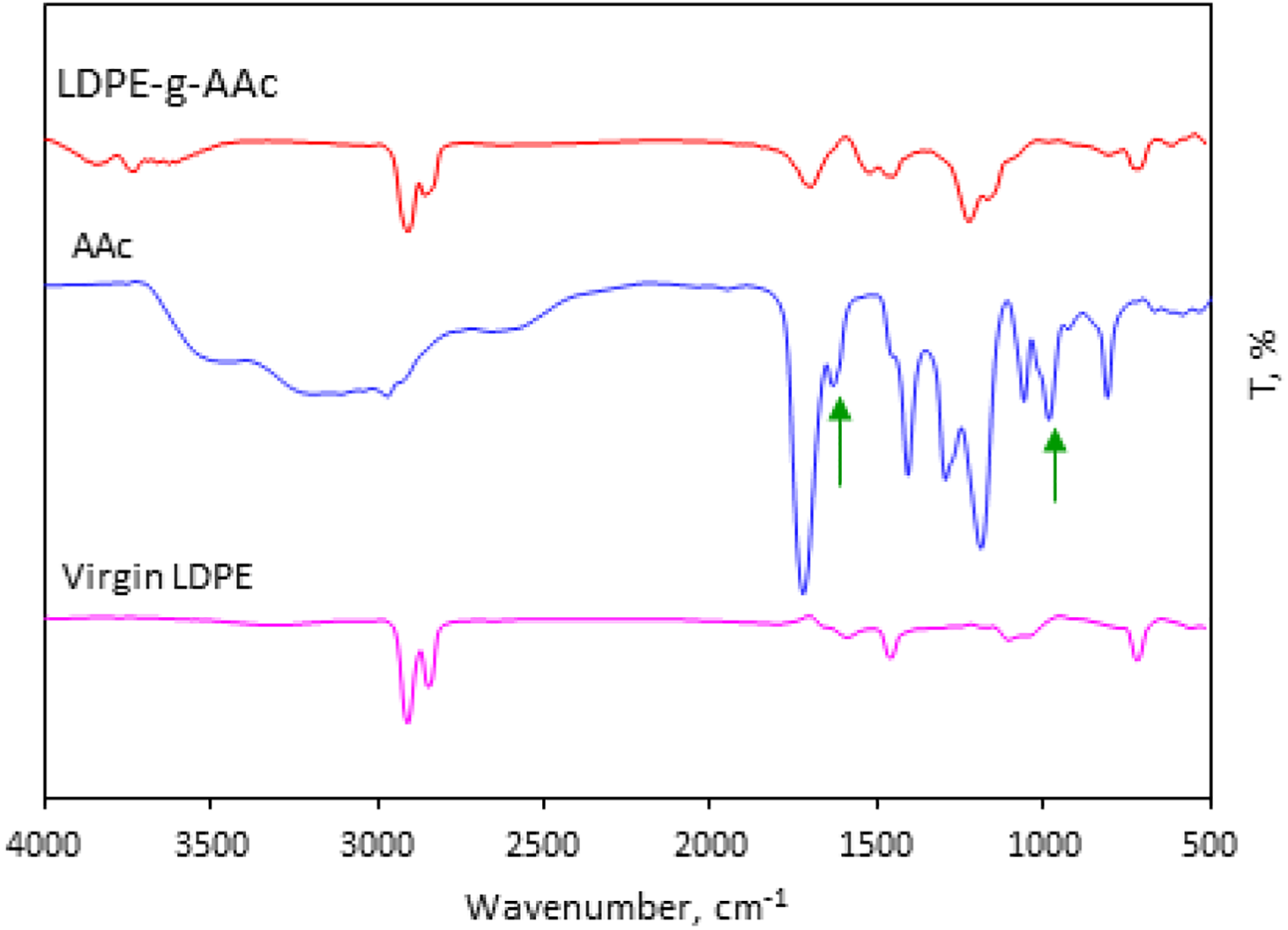
ATR-FTIR spectra of virgin LDPE, AAc, and LDPE-g-AAc.
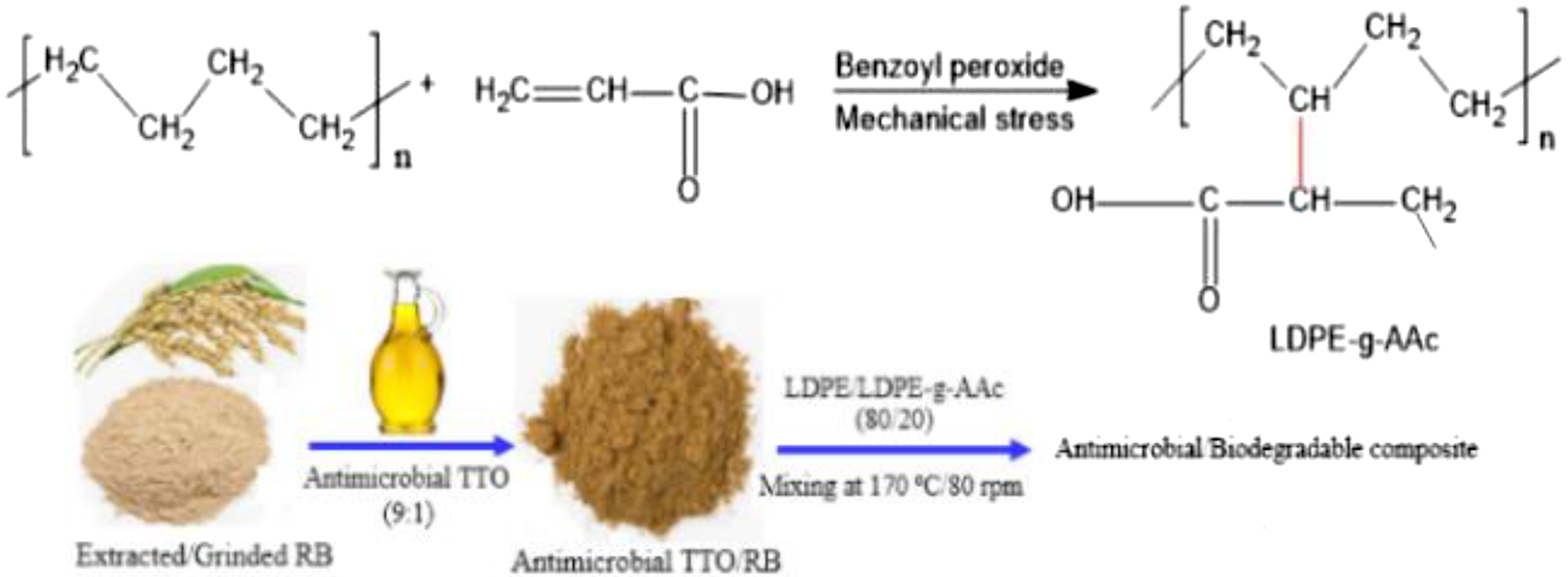
Grafting process of LDPE and AAc to obtain LDPE-g-AAc by melt blending and their processing with TTO/RB bio-filler.
Mechanical properties
Mechanical tests were conducted to explore the impact of TTO/RB addition on the tensile and hardness properties at ambient temperature compared with untreated RB. Typical tensile–strain curves for LDPE, LDPE-g-AAc and their filled composites containing different contents of untreated and treated RB by TTO oil (i.e. 10, 20, and 30 wt%) is shown in Figure 4(a) and the results are summarized in Table 1. The hardness Shore D is shown in Figure 4(b). Virgin LDPE is a perfect durable polymer, and both tensile strength and elongation at break was about 22.85 MPa and 180%, respectively. However, when the polymer mixed with 20 wt% of PE-g-AAc, both parameters remarkably decreased to approximately 10.80 MPa and 60.92%. This reduction is apparently due to the grafted AAc on LDPE, making them hydrophilic and leading to phase separation. This result agrees with the data reported elsewhere.38,39 Moreover, when untreated or treated TTO/RB with different contents inserted to LDPE/LDPE-g-AAc blend, no significant effect on the tensile strength is noticed within the experimental errors as shown in Figure 4(a). This decrease might be due to poor wettability between the components and to the fact that TTO oil amount is not enough to fulfill the compatibility, leading to poor interfacial bonding that can cause premature break of the composite, hence decreasing the tensile strength. Also, elongation at break for all samples based on TTO/RB decreased, but their values were still higher for LDPE/LDPE-g-AAc/RB composites, as demonstrated in Table 1. In contrast, the hardness value of LDPE/LDPE-g-AAc blend increased up to approimately 49.40 Shore D when compared to virgin polymer (i.e. 46.35 Shore D) as shown in Figure 4(b). Moreover, a regular increment in the hardness values was observed when adding different contents of RB or RB/TTO, ranging from 10 wt% to 30 wt % to the blend. This result is expected because of the strength of filler added. Also, it is observed that the hardness values for biocomposites based on treated RB by EO (TTO) are lower than those of LDPE/LDPE-g-AAc/RB composites. The reason may be due to the presence of EO which acts as a plasticizer, leading to an increase in the ductility of the investigated composites, thereby reducing the hardness of the material.
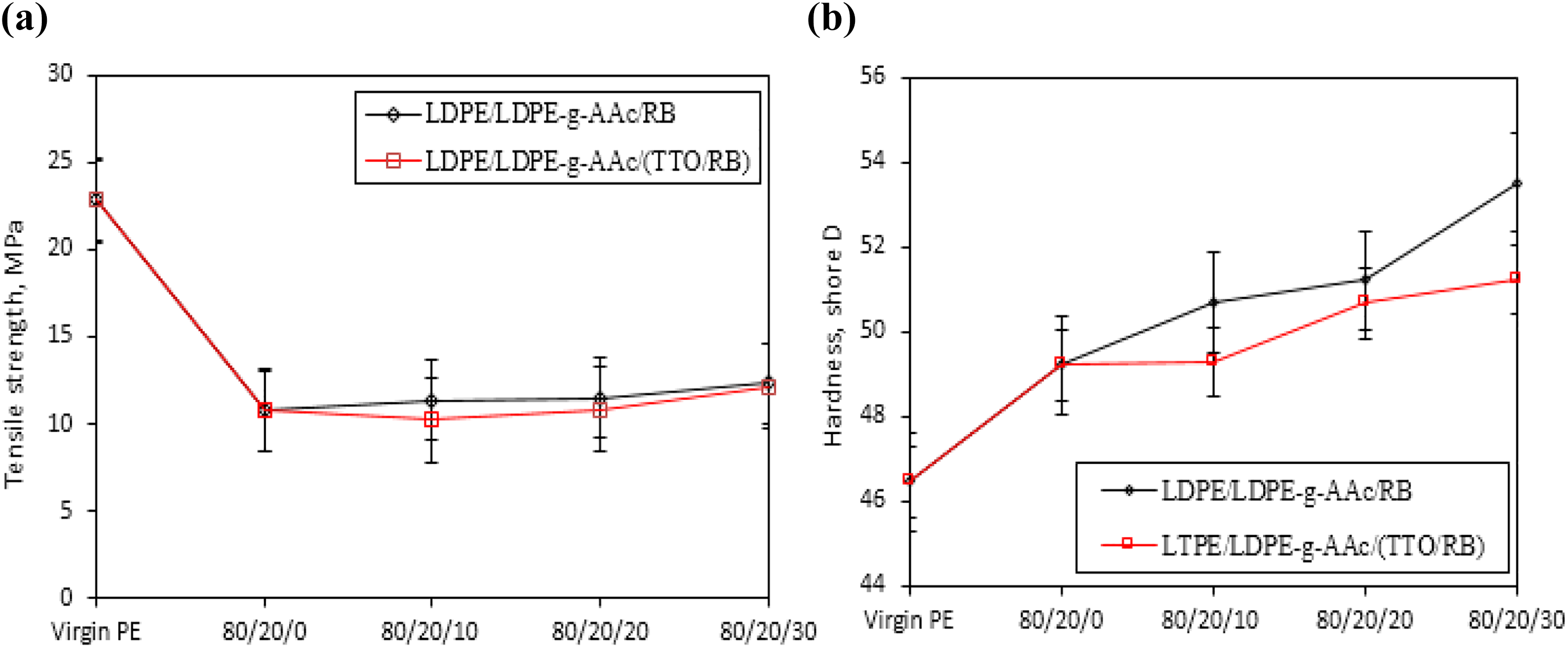
(a) Tensile strength and (b) hardness for LDPE/PE-g-AAc /RB and LDPE/PE-g-AAc/(TTO/RB) composites at different ratios.
Water vapor and oxygen barrier properties
WVTR and OTR were evaluated for virgin LDPE, LDPE-g-AAc and their charged films, and the results are presented in Table 2. Barrier properties are considered to be a vital test for packaging films. Hence, the packaging material shall be more protected from contaminates and the mycotoxin caused by microorganisms to prolong its shelf life. As can be seen in the table, virgin LDPE (100/0/0) demonstrated significantly low WVTR barrier property at approximately1.20 g m−2 24 h−1 compared to LDPE-g-AAc (80/20/0). This is because of the hydrophobicity of LDPE 40 and its crystallinity. 41 However, the latter showed excellent oxygen property at approximately 2.30 cm3 m−2 24 h−1 Pa−1 while that of the former was 57.91 cm3 m−2 24 h−1 Pa−1, due to the grafting of AAc which may hinder the oxygen gas to cross the material. Similar results have reported by Wang et al. 42 When increasing the untreated or treated RB contents up to 30 wt% to LDPE/LDPE-g-AAc blend, a drastic decrease in WVTR was noticed, that is, 1.76 and 0.73 g m−2 24 h−1, respectively, as compared to the polymer blend. This decrease is apparently due to the barrier effect of lignocellulosic material, especially at higher ratios, driving to the increasing of the wrinkles in the matrix. In addition, the chemical interaction between the hydroxyl groups in cellulose derivatives and the carboxylic group in grafted LDPE-g-AAc tends to form linkages. Nevertheless, a gradual increase in OTR values was observed when adding the untreated or treated RB reinforcing filler, as presented in Table 2. This may be due to the formation of the porosity in the polymer matrix by the filler addition. It can be also noticed that both WVTR and OTR values of biocomposites based on TTO/RB were mostly lower than those of the untreated RB.
Water vapor and oxygen permeability of LDPE/PE-g-AAc/RB and LDPE/PE-g-AAc/TTO-RB biocomposites.
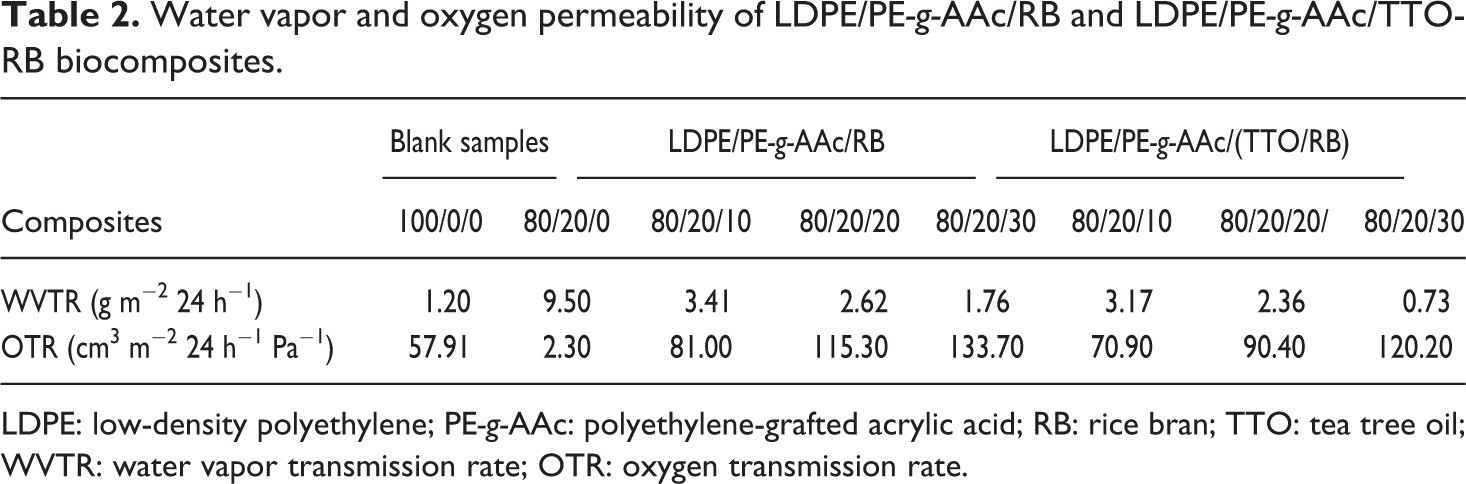
LDPE: low-density polyethylene; PE-g-AAc: polyethylene-grafted acrylic acid; RB: rice bran; TTO: tea tree oil; WVTR: water vapor transmission rate; OTR: oxygen transmission rate.
Thermogravimetric analysis
Figure 5(a) and (b) displayed TGA of LDPE, LDPE-g-AAc, and their reinforced composites and their corresponding data were recorded in Table 3. Iinitial represents the initial decomposition temperature whereas, Tfinal represents the final decomposition temperature. As shown in the figure, virgin LDPE and grafted LDPE-g-AAc started to decompose at approximately 233°C and 335°C, respectively, and almost no residue left beyond 500°C. On the other hand, T50 (i.e. temperature at 50 wt% of mass loss) of LDPE-g-AAc was 470°C and higher than that of virgin LDPE (∼414°C), indicating the superior thermal stability. This enhancement might be due to the chemical linkage formation during the grafting process, in addition to the antioxidant activity of AAc43,44 as revealed in Figure 3. However, Tfinal was 485°C and 498°C for virgin and its grafted polymer, respectively, which was corresponding to the carbonaceous materials, and was thermally degraded over a broad temperature range. From the curves, it can also be observed that after the addition of the untreated and treated RB, the thermal stabilities of some investigated specimens were decreased when compared to LDPE-g-AAc polymer, but they were still higher than that of virgin polymer. Upon adding 30 wt% of RB or treated RB to the LDPE and LDPE-g-AAc blend, the thermal stability and char residue at high temperature were evidently improved. Because, when the lignocellulosic material (RB) in the composite burns emitting gases, which acts as gas dilution in the gas phase, leading to a suppression of the flame. 15 Meanwhile, a char layer produced at the surface which in turn acts as an insulating barrier layer to oxygen and heat transfer to the condensed phase, inhibiting the flammability of the material. 45 Consequently, the char residue, gas species and crystallinity of the polymer played a significant role in the inhibition of polymer oxidation during the combustion.46,47
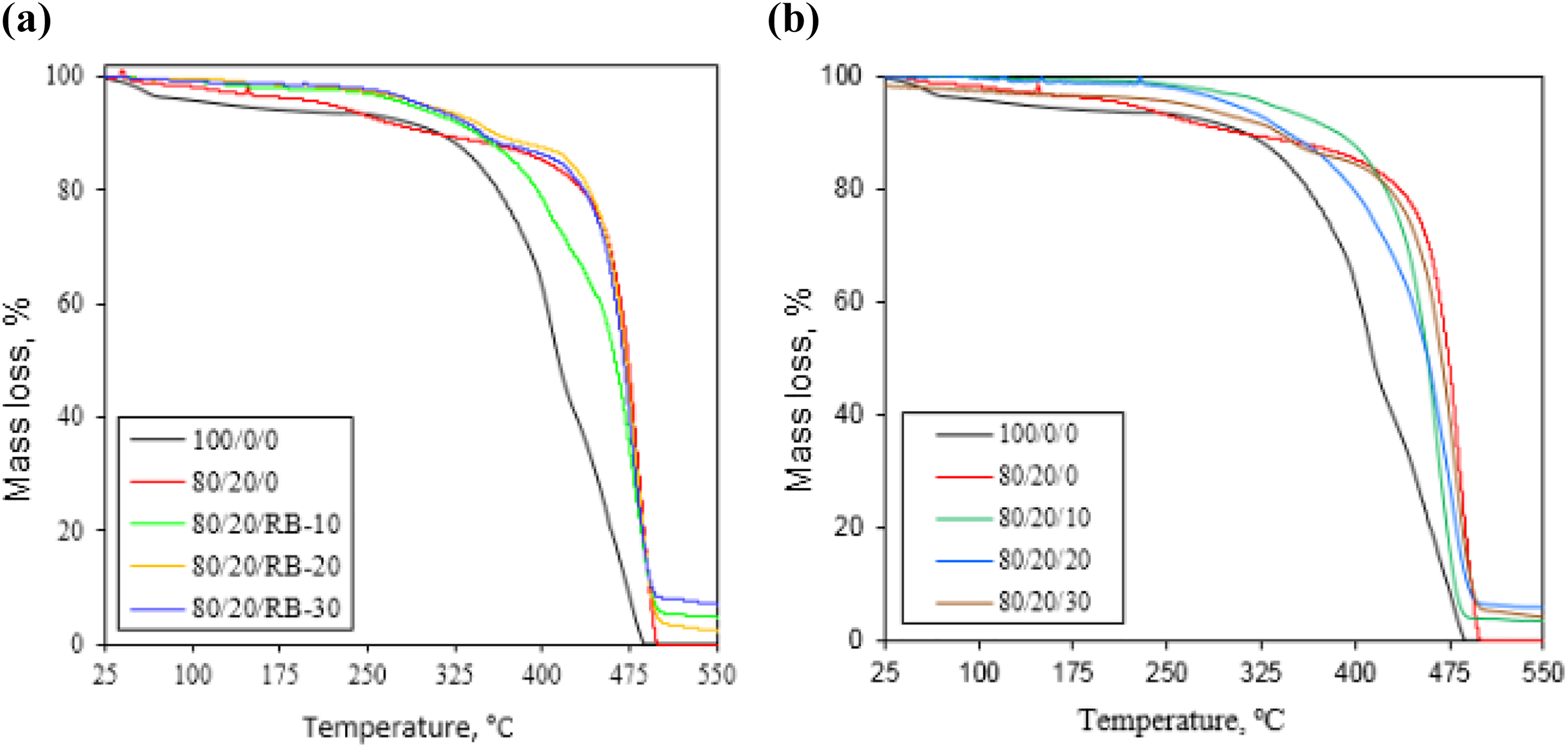
TGA thermograms for (a) the LDPE/PE-g-AAc/RB and (b) the LDPE/LDPE-g-AAc/TTO-RB biocomposites.
Thermogravimetric data of LDPE/LDPE-g-AAc/RB and LDPE/LDPE-g-AAc/TTO-RB biocomposites.
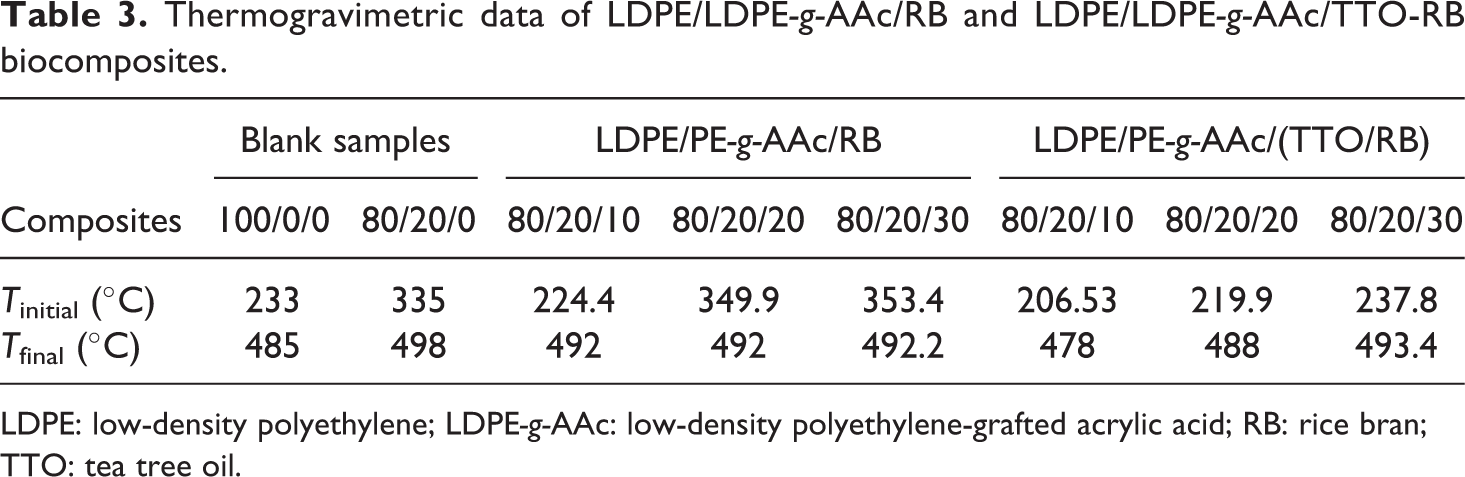
LDPE: low-density polyethylene; LDPE-g-AAc: low-density polyethylene-grafted acrylic acid; RB: rice bran; TTO: tea tree oil.
Biodegradability test
The biodegradability of virgin LDPE, LDPE-g-AAc blend and their charged composites was performed using wet soil with botanical compost as rich bacterial environment within 30 days, as shown in Figure 6(a) and (b). The figure shows that there is no biodegradation for virgin LDPE after 720 h, indicating its high stability to the environmental effects and long life for the degradation. However, the biodegradability percentage for grafted LDPE-g-AAc progressively increased with increasing burial time up to approximately 8.90% at 720 h. Evidently, the microorganisms in soil may attack the –COOH groups of AAc, causing polymer chain scission and reducing the molecular weight of the grafted LDPE-g-AAc. Consequently, –COOH groups of AAc convert into carbon dioxide and water, therefore increasing the biodegradability rate. 39 For comparison, specimens based on TTO/RB showed high biodegradation rate when increasing the filler content and burying time compared to LDPE/LDPE-g-AAc/RB specimens, especially at 30 wt% of filler content, as shown in Figure 6(a) and (b). The biodegradability percent for the former was 28.30% but was 13.40% for the latter (i.e. untreated RB) after 720 h. This improvement may be due to increase in carboxylic and hydroxyl groups in the material which help the accessibility to microorganism causing more chain session, thereby accelerating the degradation rate. These results are consistent with the data obtained by D. K. Mandal et al. 48 Thus, the faster degradation for LDPE blended with TTO-RB composites in soil depict them a new utilization in promising applications such as green packaging, biomedical purposes, and polyethylene film mulches. In addition, the biodegradable materials could keep going with new trends for preserving the environment when compared to petroleum-based polymers.
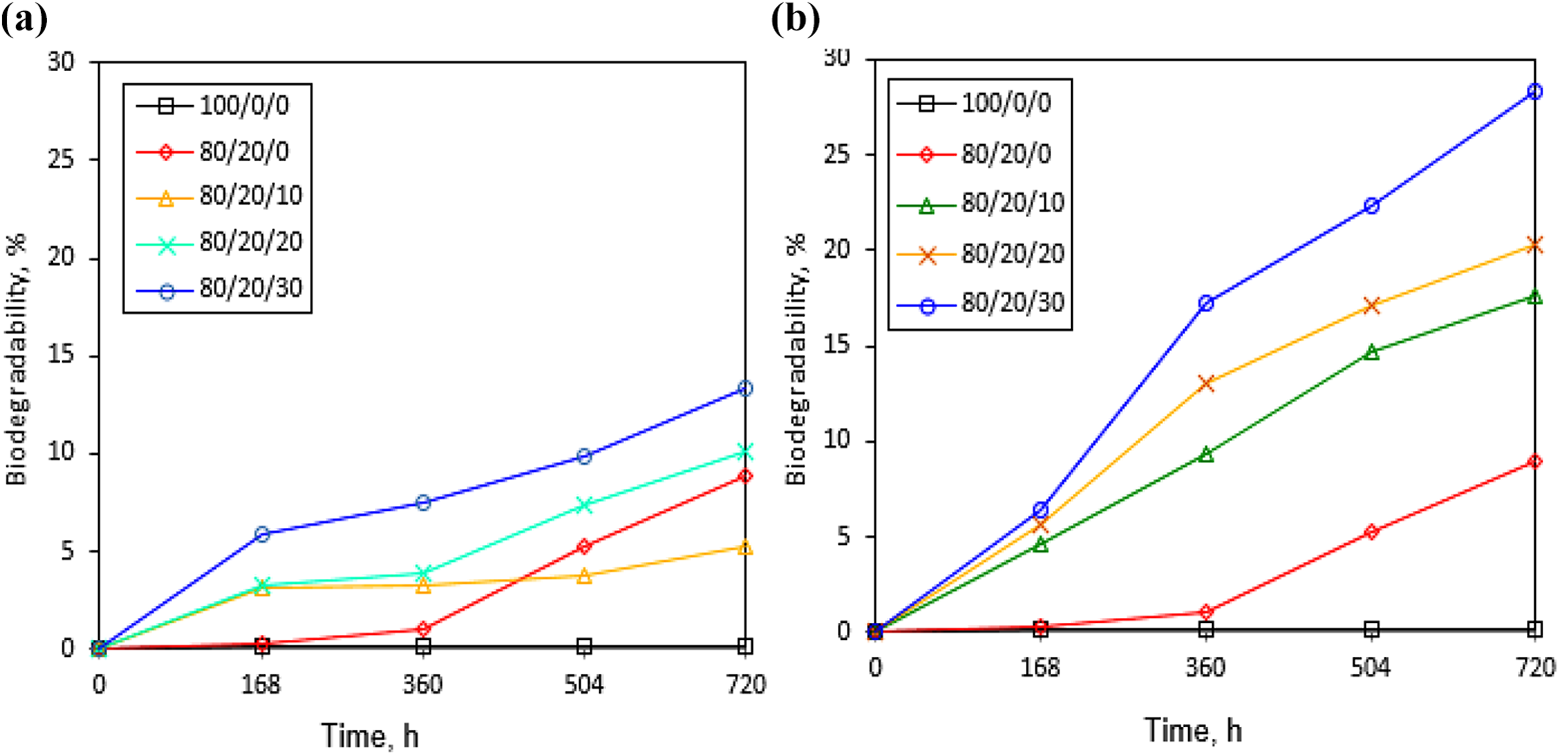
Biodegradability for (a) LDPE/PE-g-AAc /RB composites and (b) LDPE/PE-g-AAc/TTO-RB biocomposites.
Antimicrobial activity
Antimicrobial test for virgin LDPE, LDPE/LDPE-g-AAc blend and their biocomposites with variable ratios of untreated RB and treated RB by antimicrobial TTO was performed to assess these materials usages in sustainable packaging applications. The test was carried out against four different kinds of microbes: S. aureus (G+ve bacteria), E. coli (G−ve bacteria), C. albicans (yeast), and A. niger as mycotoxin (fungi). The results are reported in Figure 7 and Table 4, revealing that virgin LDPE, LDPE/LDPE-g-AAc and their biocomposites with untreated RB did not have any inhibition effects against all selected microbes in the experiment. Furthermore, their inhibition areas in the petri dish were almost zero for all investigated microbes. These data clearly proved that the bacterial activity of these biocomposites had not been affected by neither the grafting process nor the addition of RB biofiller. In contrast, halos of inhibition were noticed for LDPE/LDPE-g-AAc/RB biocomposites when TTO oil was added with different diameters ranging from 15 mm to 25 mm relying on the microbial type (Table 4). This enhancement is probably corresponded to the presence of hydroxyl groups in TTO which have a significant efficiency in killing microbes and inhibiting the growth of a wide range of bacteria (i.e. G+ve and G−ve bacteria), yeast, as well as fungi. Feyzioglu and Tornuk 27 have reported that the hydrophobicity of EO (TTO) is one of their key properties that enable them to break down the lipids in bacterial cell membrane, causing leakage of ions and other cell compounds, and therefore cell death eventually. These results are consistent with those obtained by Tian Xu et al. 49

Optical images of inhibition for LDPE/LDPE-g-AAc/RB and LDPE/LDPE-g-AAc/(TTO-RB) composites against Staphylococcus aureus, Escherichia coli, Candida albicans, and Aspergillus niger.
Antimicrobial activity for virgin LDPE, LDPE/LDPE-g-AAc and the prepared biocomposites with different ratios of TTO-RB against the four microbes.
LDPE: low-density polyethylene; LDPE-g-AAc: low-density polyethylene-grafted acrylic acid; RB: rice bran; TTO: tea tree oil.
Conclusions
In this work, AAc was successfully grafted onto the LDPE using a Brabender Plasti-Corder and their structure was confirmed by ATR-FTIR spectroscopy. The tensile properties showed that there was no significant effect on the tensile strength when adding either untreated RB or treated TTO/RB to the LDPE and its blend. On the other hand, a remarkable increase in the hardness values of filled samples was noticed with the increase of bio-filler content in the blend. Enhancement in WVTR barrier property was observed for biocomposite charged by 30 wt% TTO-RB (0.73 g m−2 24 h−1) compared to virgin LDPE (1.20 g m−2 24 h−1) and its blend (9.50 g m−2 24 h−1). However, LDPE/LDPE-g-AAc blends showed an excellent oxygen barrier by ∼2.30 cc m−2 24 h−1 Pa−1, indicating the prolonged shelf life of food could be achieved. The thermal stability of the filled biocomposites was achieved and the data showed quite shift in the TGA curves towards higher temperatures when untreated and treated RB were added, evidencing a suppression of the flame during the combustion. The biodegradability property of LDPE was clearly affected by the addition of LDPE-g-AAc and TTO. The inhibitory effect of LDPE/LDPE-g-AAc/(TTO-RB) biocomposites against S. aureus, A. niger, E. coli, and C. albicans were increased with increasing the contents of TTO-RB from 10 wt% to 30 wt%. Therefore, the LDPE/LDPE-g-AAc films based on antimicrobial EO TTO could be used for sustainable food packaging owing to their biodegradability and antimicrobial properties.
