Abstract
To obtain antibacterial properties for colored cotton fabrics, vat dyes were chosen to dye cotton because they can avoid serious discoloration during chlorination. In this study, we synthesized a reactive
Introduction
Cotton fabric is one of the most widely-used fabrics worldwide. However, it is susceptible to microorganism contamination, especially in hot and humid environments, which directly or indirectly affect the performance of the fabric.
1
It can provide a vehicle for the direct or indirect transmission of some microbes, which can have a negative effect on human health. Therefore, the application of an antibacterial finishing for cotton fabrics has drawn much attention from researchers.2–5 Antibacterial agents for textiles mainly include organic,6–8 inorganic,9,10 and natural antibacterial agents.11,12 In these applications, commercial antimicrobial agents have their limitations.
In this study, cotton fabrics were dyed with three vat dyes, and the dyed cotton fabrics were coated with a synthesized, reactive
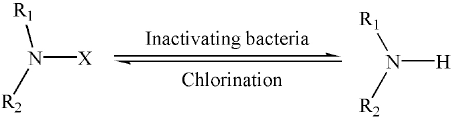
Biocidal and regeneration mechanism of
Experimental
Materials
Bleached cotton fabrics were provided from Zhejiang Guangdong Textile Dyeing Garment Co. Ltd. Cyanuric chloride (99%) was obtained from J & K Chemical Co. Ltd. 2,2,6,6-Tet-ramethyl-4-piperidinol was purchased from Jiaxing Sicheng Chemical Co. Ltd. C.I. Vat Brown BR, C.I. Vat Olive, and C.I. Vat Yellow 1 were provided from Yorkshire Chemical Co. Ltd. Other chemicals were purchased from Sinopharm Chemical Reagent Co. Ltd. and used without further purification.
Instruments
An Avance III 400 MHz digital nuclear magnetic resonance (NMR) spectrometer (Bruker AXS GmbH) was used to characterize the synthesized product (BTMPT). Fourier transform infrared (FTIR) spectra of cotton and coated cotton fabrics were recorded on a Nicolet Nexus spectrometer (Nicolet Instrument Corp.) using the attenuated total reflectance (ATR) method. Surface morphologies of uncoated and coated cotton fabrics were investigated using a SU1510 spectrometer (Hitachi). The color parameters of dyed cotton were measured on a Datacolor SF 600 spectrophotometer (Datacolor).
Preparation of BTMPT-Coated Dyed Cotton Fabrics
The cotton fabrics were dyed with three vat dyes (Vat Brown BR, Vat Olive, and Vat Yellow 1, Fig. 2a–c) through the leuco-dyeing process. Dyeing solution (vat dye 2% owf, NaOH (15 g/L), and Na2S2O4 (20 g/L) was prepared and heated to 60 °C for 10 min, followed by adding the cotton fabric. After 40 min, the cotton fabric was removed, evenly squeezed, and placed in the air for 10 min. The dyed cotton fabric was finished with water washing, soap washing using Na CO (2 g/L) and soap fakes (Sinopharm Chemical Reagent Co. Ltd., 2 g/L) with a liquor ratio of 1:30 at 95 °C for 10 min), and drying.
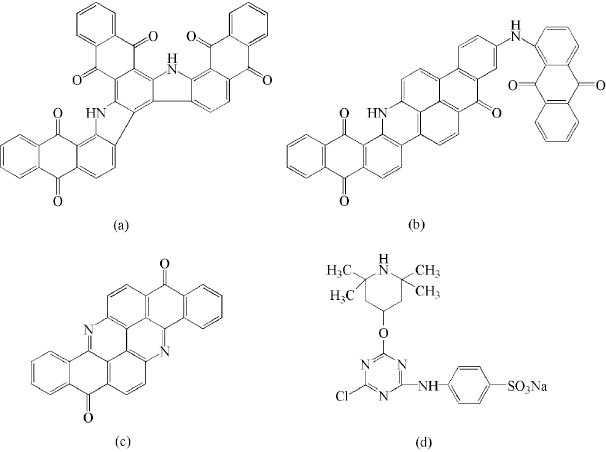
Structures of (a) Vat Brown BR, (b) Vat Olive, (c) Vat Yellow 1, and (d) BTMPT
BTMPT (Fig. 2d) was synthesized and coated onto the dyed cotton fabrics according to the literature procedure. 32
Chlorination and Titration
The pH of diluted commercial sodium hypochlorite solutions (2 g/L active chlorine) were adjusted to 7, 9, and 11 by dilute sulfuric acid solution, and the BTMPT-coated dyed cotton fabrics were immersed in these solutions for 3 h at ambient temperature. Then, the chlorinated cotton samples were washed thoroughly with distilled water and dried at 45 °C for 1 h to remove free chlorine residues on the surface.
The chlorine loadings on fabric samples were determined by iodometric titration. The samples were immersed in KI solution, and a few drops of 1% starch solution were added as indictors. After thorough soaking, the mixture was titrated with 0.001 N sodium thiosulfate solution. The titration ended when the color of the solution turned to colorless. The concentrations of active chlorine on the cotton samples were calculated according to Eq. 1.
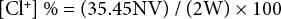
[Cl+] % is the wt% of oxidative chlorine on the sample, N and V are the normality (equiv/L) and volume (L) of the titrant sodium thiosulfate, respectively, and W is the weight of the cotton sample (g).
Color Measurement
The color parameters
The color differences
Antimicrobial Test
According to AATCC TM100-2004,
33
control and unchlorinated/chlorinated coated fabric samples were challenged with
Washing Stability Test
The durability and stability of BTMPT and chlorine on the coated cotton samples against standard washing cycles were evaluated according to AATCC TM61-2010. 35 Stainless steel canisters containing 0.15% AATCC detergent water solution (150 mL) and 50 stainless steel balls were placed in a Launder-Ometer (SDL Atlas) and rotated at 42 rpm and 49 °C. The coated cotton samples (2.54 × 5.08 cm) were subjected to 5, 10, 25, and 50 washing cycles. Each washing cycle of 45 min, was equivalent to five machine washings. All samples were washed with distilled water and then dried at ambient temperature. The chlorine loadings of half the samples were determined directly, and the other half of the samples were chlorinated again, and the chlorine loads were measured. The chlorine loadings on the samples were determined by the titration method discussed previously.
Breaking Strength Test
An electronic fabric strength tester, YG(B)026D-250 (Darong Textile Instrument Co. Ltd.), was used to evaluate the breaking strength of untreated cotton, dyed cotton, and unchlorinated and chlorinated dyed cotton fabrics according to the GB/T3923-2013 method. The measurement was carried out at ambient temperature. Tree replicates were prepared for each sample (5 × 20 cm), and the average value was recorded for analysis.
Results and Discussion
Characterization of BTMPT-Coated Dyed Cotton Fabrics
The FTIR-ATR spectra of Vat Brown BR-dyed cotton fabrics that were uncoated, those coated with BTMPT before chlorination, and those coated with BTMPT after chlorination are shown in Figs. 3a–c. respectively. New absorption peaks on the unchlorinated treated fabric appeared at 2846 and 1560 cm–1, which were attributed to methyl and benzene ring telescopic vibrations from BTMPT, providing evidence that BTMPT was coated onto the cotton fabrics. After chlorination, the above two peaks were shifted to 2852 and 1564 cm–1 respectively, due to conversion of N-H to N-Cl. The affinity of oxidative chlorine for the electron cloud density of the adjacent methyl and benzene rings led to the absorption peak shift to higher values.32,37
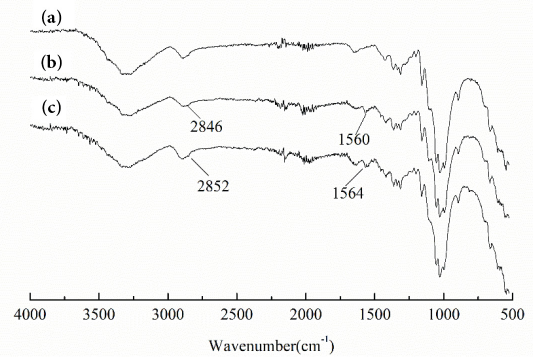
FTIR-ATR spectra of (a) dyed cotton, (b) dyed cotton-BTMPT, and (c) dyed cotton-BTMPT-Cl.
Fig. 4a–c shows the surface morphologies of uncoated and coated cotton fabrics. The surface of uncoated cotton was smooth, and the surface of the Vat Brown BR-dyed cotton fabric had some impurities. The surface of the Vat Brown BR-dyed cotton fabric coated with BTMPT became rough due to the bonding of BTMPT on the cotton surface.

SEM micrographs of (a) cotton, (b) dyed cotton, and (c) dyed cotton-BTMPT.
Effect of Chlorination pH on Chlorine Content and Color
In this study, Vat Brown BR, Vat Olive, and Vat Yellow 1, having good color fastness to chlorine bleaching, were used to dye cotton fabrics. Sodium hypochlorite solutions at various pH values were prepared to chlorinate the BTMPT-coated dyed cotton fabric. The color changes and chlorine loadings of the dyed cotton fabrics before and after chlorination were then investigated. With the decrease of pH of chlorination, the chlorine loadings on BTMPT-coated dyed cotton fabrics increased gradually (Fig. 5). When the pH of chlorination reached 11, the chlorine loadings on Vat Brown BR-dyed cotton fabric were 0.21%, the
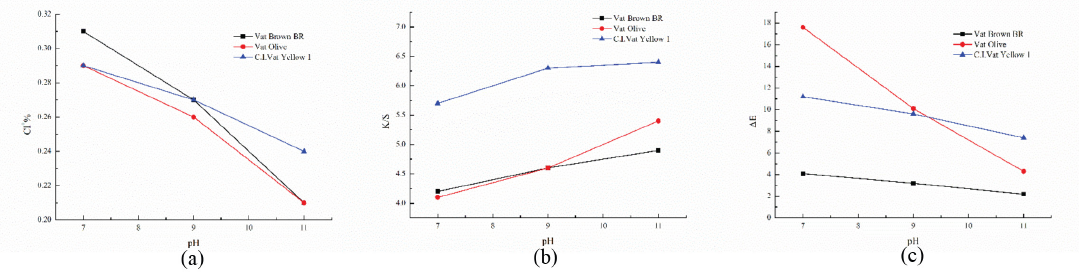
Effect of chlorination pH on the (a) chlorine loading, (b) K/S value, and (c) Δ
Antimicrobial Properties
The
Antibacterial activities of the Vat Brown BR-dyed cotton fabrics coated with BTMPT after chlorination with a chlorine load of 0.28% are shown in Table I. Fabrics were challenged with
Antibacterial Properties of Vat Brown BR Dyed Cotton Fabrics Coated with BTMPT
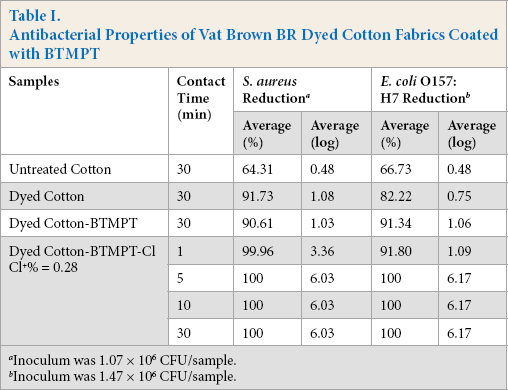
Inoculum was 1.07 × 106 CFU/sample.
Inoculum was 1.47 × 106 CFU/sample.
Washing Stability
Antibacterial fabrics coated with
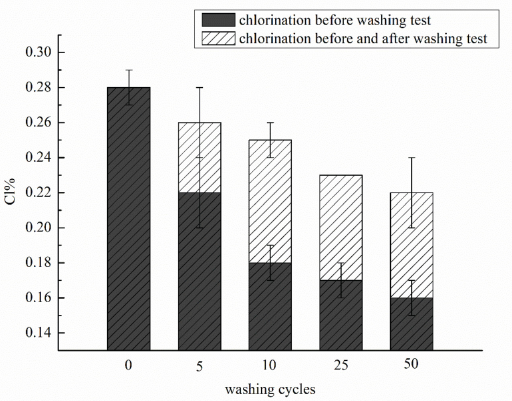
The washing stabilities of chlorine in dyed antibacterial cotton fabrics.
The chlorine loadings gradually decreased with the increase of washing cycles. The chlorine loadings on cotton fabric after 50 washing cycles remained 0.16%, which is sufficient to inactivate bacteria. 32 The loss of chlorine loadings can be recovered to at least 78.6% of initial content after re-chlorination.
Breaking Strength
The breaking strengths of the Vat Brown BR-dyed cotton fabrics before and after coating with BTMPT were tested and the results are shown in Fig. 7. The strength of cotton fabrics decreased after dyeing and antibacterial finishing, and was directly affected by process temperature, acidity, and alkalinity, among other factors. The effect of dyeing on the strength of cotton fabric was greater than that of antimicrobial finishing, most likely attributed to a greater effect on the fluidity of fibers in and between molecules when dyed. 42 The strength of the cotton fabric in warp and weft directions were 665 N and 304 N and decreased to 614 N and 288 N after dyeing, respectively. After coating with BTMPT, the strength of the dyed cotton fabric was further decreased slightly both in warp and weft to 599 N and 284 N, respectively. The strength reduction was small compared with untreated cotton, and the retention rates of cotton fabric in warp and weft were 90% and 93.4%, respectively. After chlorination, the strength of the antibacterial cotton fabric reduced further to a small degree, and the retention rates in warp and weft were 87.8% and 79.6%, respectively.
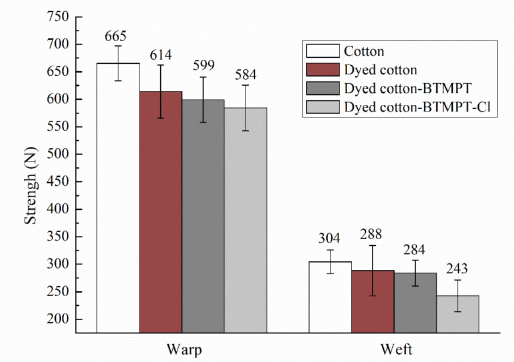
Breaking strengths of the dyed cotton fabrics coated with BTMPT.
Conclusion
Colored cotton fabrics with excellent antibacterial properties were obtained by Vat Brown BR dyeing followed by coating with the
The Vat Brown BR-dyed antibacterial cotton achieved more than 0.2% of chlorine loadings as well as a small color change of 2.2. The chlorinated BTMPT-coated Vat Brown BR-dyed cotton inactivated 100% of
Footnotes
Acknowledgements
This work was financially supported by the national first-class discipline program of Light Industry Technology and Engineering (LITE2018-21), the Fundamental Research Funds for the Central Universities (No. JUSRP51722B, No. JUSRP11806), the Project of Jiangsu Science and Technological Innovation Team, and 111 Projects (B17021).
