Abstract
Spider silk is a promising biomaterial, with a wide range of potential applications in the medical, military, and commercial sectors. In the past decade, researchers have been developing recombinant spider silk (RSS) proteins with the help of advanced genetic engineering techniques, including gene design, cloning, and protein expression in different foreign hosts. The extraordinary strength and extensibility of natural spider silk fiber are constituted from the tandemly repeated amino acid sequences and antiparallel crystalline β-sheet segments formed by polyalanine motifs. Additionally, mechanical properties are also influenced by extrusion and drawing of fibers in the natural spinning process. To date, the wet spinning process was found to be a commercially successful method for spinning recombinant silks with properties comparable to their natural counterparts.
Introduction
Spider silk displays mechanical properties five and three times stronger than steel and Kevlar, respectively.1,2 It is also biocompatible and biodegradable, which makes it an attractive material for the biomedical field. Spider silk is used to produce a variety of materials like films,3,4 fibers,5-7 coatings, 8 gels, 9 foams, 10 microspheres,11-14 and foam and microspheres.
Different methods are followed to produce these materials. Due to their outstanding mechanical properties and biocompatibility, many applications in various industries are foreseen.
In general, natural silks can be harvested from live spiders, but their cannibalistic nature and low yield make it difficult to farm them like silkworms. 15 As a result, many efforts were made to produce recombinant spidroins in bulk quantities. The use of genetic engineering to prepare recombinant spider silk (RSS) has been a most effective strategy so far. Spidroins have been successfully expressed in different organisms, including yeasts, 16 bacteria, 17 plants, 18 insect cells, 19 and the milk of transgenic goats20,21 using recombinant technology. Based on previous studies on recombinant spidroins, several factors are necessary to produce silk-like fibers, including the protein sequence, secondary structure, protein solubility, and spinning conditions. This brief review will discuss the mechanical properties and solvent systems of artificial spider silk fibers in accordance with the most recent and successful methods.
Recombinant Spider Silk Production
Several methods to synthesize recombinant spidroin have been reported, the most popular and widely-used host is the Gram-negative bacterium Escherichia coli. It provides a controlled, efficient, and cost-effective system for large scale production. Since eukaryotic organisms can secrete different proteins in the extracellular medium, hosts like Pichia pastoris (methylotrophic yeast) have been used to express larger RSS proteins. 16 Mammalian cells are yet another host system to express the RSS protein. The spider silk protein was expressed in bovine mammary epithelial alveolar (MAC-T) cells and baby hamster kidney (BHK) cells. 22 These systems produced soluble proteins of 60-140 kDa. In addition to above-mentioned methods, multicellular systems like transgenic plants and animals were investigated to produce RSS. Mammary glands of transgenic mice 23 and goats 21 were genetically modified to secrete silk protein. The most effective multicellular system for producing spider silk was the silkworm. Interestingly, silkworms not only secrete protein, but can also produce silk fibers. A chimeric silkworm and spider silk protein with gene encoding the native Nephila clavipes MaSp1 protein was expressed in Bombyx mori. 24 Recombinant silk fiber spun by the transgenic worm exhibited mechanical properties tougher the natural silkworm silk.25,26
Spiders use a controlled protein denaturing mechanism, along with shear forces, to spin silk fibers. This polymeric flow induced crystallization mechanism is still a mystery; it was shown to be 1000 times more efficient than artificial processes. 27 The artificial fiber production process involves different approaches, including wet spinning, dry spinning, and electrospinning methods. Notwithstanding, the wet spinning method is extensively used to spin RSS fibers. This method uses a coagulation bath containing methanol, isopropanol, and ethanol. The fibers produced using wet spinning are brittle and exhibit poor mechanical properties, requiring an additional stretching process. 28 Post-spinning processes improve the mechanical performance by influenc-ing the protein secondary structure, increasing molecular orientation, and reducing the fiber diameter. The wet-spun fibers are usually subjected to a post-spinning process by immersion in coagulant, post drawing at different ratios, and steam annealing.29-31
Two commonly-used strategies to produce recombinant spidroins are 1) expressing parts of the native sequence and 2) expressing engineered consensus repeats and chimeric sequences. Table I shows the summarized data of artificial fibers produced using different strategies and mechanical properties (average values) from 2010 to 2017. Interestingly, the latest studies show that the focus of this research area has been shifted towards the production of native-like aqueous spinning dopes. It is well known that the concentration of native proteins in spider glands are stored in a range between 30 and 50% (w/v).32,43 Jones et al. first reported the production of aqueous spinning dope, but the concentration used to spin fibers was limited to 12%. 20 Andersson et al. successfully reported a chimeric RSS protein engineered with N- and C-terminal domains where the dope concentration reached up to 50%. 32 This is the highest concentration of aqueous dope produced to date. Heidebrecht et al. reported a high-molecular weight recombinant spidroin of 286 kDa using SUMO (small ubiquitin modifier) fusion technology. 34 Lin et al. reported a dimeric protein with a molecular weight of 378 kDa by linking the C-terminal domain via disulfide bonding. 35 The fibers produced by Xia et al. (284.9 kDa) reported a high strength of 508 ± 108 MPa, but exhibited low extensibility limited to 15 ± 5%. 17 To improve the mechanical properties, the fibers were stretched to 500% of its original length after spinning. The fibers produced by Heide-brecht et al. were stretched to 600% of its original length after spinning, displaying excellent extensibility and toughness of 110 ± 25% and 183 ± 33 MJ/m3, respectively. 34 Despite having a higher molecular weight than the protein produced by Xia et al., 17 the fiber displayed a lower strength of 370 ± 59 MPa. This may be due to variations in the amino acid sequence and the post-processing technique that followed.
Processing Parameters and Fiber Properties of RSS Fibers
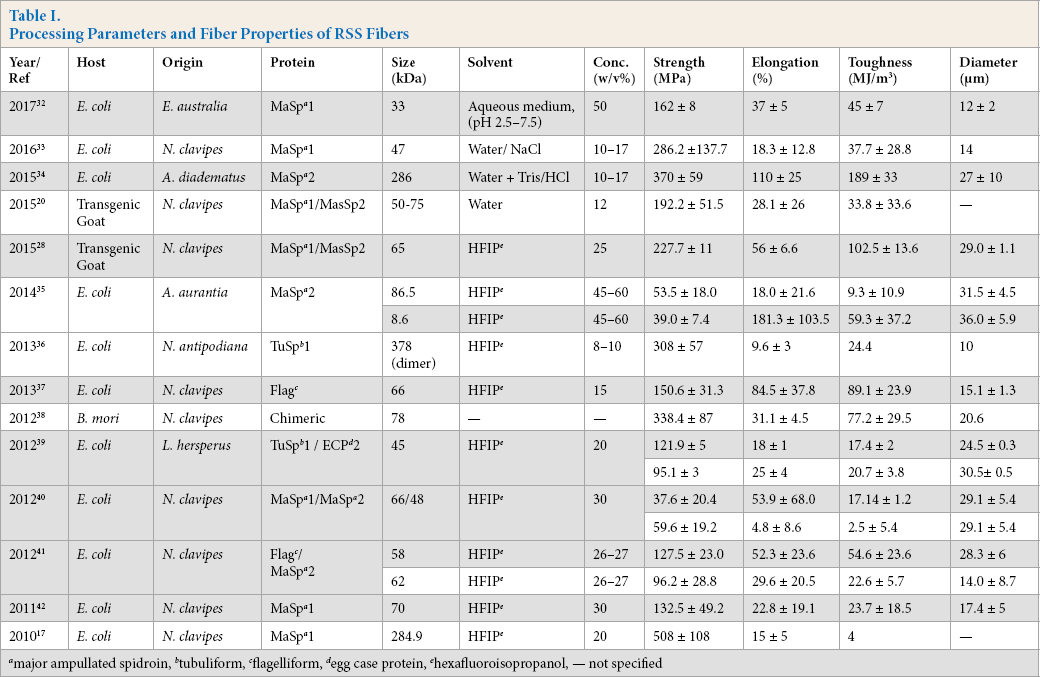
major ampullated spidroin,
tubuliform,
flagelliform,
egg case protein,
hexafluoroisopropanol, — not specified
Unstretched fibers produced by Andersson et al. in a bio-mimetic wet-spinning setup showed excellent toughness of 45 MJ/m3. 32 This is the highest value so far for reported unstretched RSS fibers. Nevertheless, fibers with the highest elongation of 181.3% were reported by Albertson et al. 36 After spinning, the fibers were stretched 4 times in an 85% isopropanol/water bath at 60 °C. The water in the coagulant acts as a plasticizer, leading to better chain orientation and optimal molecular confirmation, improving the mechanical properties. Although the production of recombinant spider silk through wet spinning gave improvements in toughness and extensibility comparable to natural fibers, the strength and stiffness of the recombinant silk need further improvement.17, 20, 28, 32-42 The ability of the wet-spinning method to produce stronger and tougher recombinant spider silk-like native fibers has reached its limits. The fibers produced had either high strength and stiffness or high elongation and toughness.
Application of Recombinant Spider Silk Fibers
Fibers produced using recombinant spider silk proteins are used for a wide range of applications such as tissue engi-neering, 44 fiber optics,45,46 and high-performance products. 47 Recombinant spider silk fibers are one of the new generation of biomaterials due to their excellent biocompatibility and mechanical properties. 48 Sterilized RSS fiber meshes (4RepCT) produced using the self-assembly process showed excellent mechanical stability and allowed the growth of fibroblasts. RSS fibers modified with cell binding RGE, RGD, IKVAV peptides displayed enhanced cell attachment proper-ties, 49 and attachment of antibiotics produced fibers with antibacterial properties. 50 Johansson et al. evaluated 4RepCT RSS self-assembled fiber meshes for formation of pancreatic islets; their performance were compared with films and foams. 51 However, the results showed that foams were able to enhance the cell attachment, insulin production, and the amount of intracellular calcium. 51
In general, silk fibers are strong biocompatible optical materials with tunable biodegradability. 45 Their excellent mechanical properties and optical clarity make them suitable for optical biomaterial applications. Qiao et al. reported the use of RSS fibers as implantable waveguides with low-loss light guiding properties. 46 Interestingly, the results revealed that RSS fibers were able to guide light with low optical loss comparable to regenerated silk fibroin. RSS showed an optical loss of 0.8 ± 0.1 dB/cm in air and 1.9 ± 0.3 dB/cm in rat muscles compared with regenerated silk fibroin that has displayed 1.7 ± 0.1 dB/cm in air and 3.1 ± 0.4 dB/cm in rat muscles respectively.
In recent years, RSS fibers successfully emerged in the commercial market as various end products suitable for high-performance applications. 52 Spiber, a Japanese-based company, along with apparel makers Goldwin and The North Face, were first to introduce a windproof jacket made of recombinant spider silk. Adidas, in cooperation with AMSilk of Germany, produced biodegradable sport shoes made of synthetic spider silk fibers commercially marketed as Biosteel. Moreover, this high-performance category product was primarily produced using renewable resources that may lay foundation for a large-volume opportunity in the future. Kraig Biocraft Laboratories (USA) genetically modified silk worms with spider DNA to produce a stronger and more flexible silk thread called Dragon Silk. Dragon Silk is expected to open up the high-performance textile market with a wide range of applications due to their scalability potential and less expensive production cost.
Conclusions
Although many researchers were successful in the artificial production of spider silk fibers, many challenges remain to duplicate all properties of natural spider silks. The most important challenge is the preparation of protein spinning dope. The constituents of the artificial spinning dope show completely different molecular weights, solubilities, and fiber mechanical properties compared to the native dope. Second, the fiber formation process of recombinant spider silk protein occurs during precipitation of the protein in the coagulant. Unlike the natural spider silk, the stress-induced phase separation process is absent in the artificial spinning process, which results in anisotropic molecular arrangements; for native dragline silk, isotropic molecular arrangement is observed. Third, the ability of the spider to produce thinner fibers supports improved mechanical properties. But from overall analysis, it is understood that along with morphology (diameter and fiber surface) of the fiber, the hierarchical structure, micro/nano fibrils, and control of ordered and unordered regions play an important role in determining the mechanical properties of the fiber. There-fore, it is important to understand the mechanism behind natural spider silk production in defining the mechanical properties of spider silk. Advancing the technology to match such challenges and developing new systems to produce fibers is an interesting area for future research.
Footnotes
Acknowledgement
This work was supported by the National Natural Science Foundation of China (51373147), Science and Technology Planning Project of Guangdong Province (2016A050503013) and the Research Grants Council, University Grants Committee (PolyU 5158/13E).
