Abstract
Spider silk is renowned for its exceptional mechanical properties, and compatibility and biodegradability for various functional applications, such as drug delivery. By a facile HFIP (1,1,1,3,3,3-hexafluoro-2-propanol)-on-Oil method, microspheres and microcapsules were assembled from a recombinant spider eggcase silk protein. Doxorubicin (Dox) can be loaded into microspheres or microcapsules before or after their preparation, while maintaining morphological integrity. The isoelectric point of the silk proteins was 4.8, which affected the drug loading of silk spheres for different pH media. For enzyme-assisted drug delivery, Dox-loaded silk spheres effectively released the drug under enzymatic degradation by addition of proteinase K.
Introduction
Although accelerated development of drug delivery systems has been witnessed in recent years, strategies to fulfill the promising potential of smart biomacromolecular delivery remains challenging. Of various natural polymers, silk fibroin has been widely explored due to its bottom-up control, from composition to sequence, and from structure to properties. 1 In addition to fibers, silk proteins can be produced in morphologies as diverse as spheres, capsules, foams, films, and gels. 2
Drug delivery systems function as drug reservoirs, or to alter the pharmacokinetics and biodistribution of the associated drugs, which are chemically bound or physically entrapped. In comparison to other silk forms, silk microspheres and microcapsules are more extensively investigated as vehicles for cargo storage due to their ability to release active ingredients. Microspheres, being solid and containing a uniform matrix, present greater potential to disperse beneficial agents homogeneously throughout the matrix. 3 Nevertheless, microspheres are often limited to conditions where a controlled release is required under triggers (e.g., light, thermal, biological, chemical, electrical, and magnetic stimuli). 4 Microcapsules, having a shell wall and a liquid core, offer versatility for encapsulating large molecules, while allowing small ones to permeate freely. Encapsulation is used for various purposes, allowing for controlled and/or targeted release of the encapsulated drug. 5
Given that a wide range of pH values and enzymes exist in the human body's environment, the ability to be highly sensitive to particular pH or enzymatic conditions is of importance with regard to a defined delivery system.6,7 A variety of approaches have been developed to prepare micro-spheres and microcapsules responsive to a specific pH or enzyme trigger. Of equal importance in choosing a method is how to avoid negative impacts on the morphology integrity and biological potency when these systems are loaded with bioactive materials.
In comparison with well-known dragline silk, greater attention is now being paid to eggcase silk due to its unique properties. TuSp1, short for tubuliform spider silk, is the major component of eggcase silk. 8 Recently, we prepared engineered TuSp1 (eTuSp1) from a black widow spider, Latrodectus mactans, 9 by recombinant gene technology. Spider silk is widely explored as inspirations for modern materials.10-11 TuSp 1 protein is spun into silk fibers to build up several centimeters of capsules (eggcases) for offspring protection, while eTuSp1 is a direct fabrication in morphology of spheres/capsules for drug delivery.
Herein, we present a novel and facile procedure to spontaneously assemble eTuSp1 microspheres/microcapsules by an HFIP (1,1,1,3,3,3-hexafuoro-2-propanol)-on-Oil method with potential for drug delivery. We define facile as being 1) rapid formation, 2) easy fabrication, and 3) energy-saving and cost-effective. Prior to assembly, lyophilized eTuSp1 proteins were dissolved in HFIP. This organic solution of eTuSp1 was added to silicon oil, accompanied by the evaporation of HFIP. Upon contact with the oil, large droplets of eTuSp1 solution are split into numerous smaller droplets, mainly due to the low surface tension of HFIP and oil. However, the transition from droplets to solid spheres/capsules is actually induced by solvent evaporation. 12
Experimental
Materials
The eggcase silk protein, from a black widow spider, was genetically engineered by using a single conserved repeat unit with an His6 tag, as previously reported by our group. 13 Briefly, during cloning, the polymerase chain reaction (PCR) with seven forward and seven reverse oligonucleotides were assembled into the target gene to amplify the cDNA, which contained 161 amino acid residues (GenBank: AAY28947.1). All of the materials used for these preparations were purchased from Sigma Aldrich. Ten, the cDNA fragment was subjected to gel extraction, ligation with plasmid vector (pUC18-SN), and transformation with Escherichia coli. Finally, the modified plasmid was verified by DNA sequence analysis. To induce the protein, 50 μg/mL of ampicillin was added at the mid-log growing phase, together with 2 mL of isopropyl-β-D-thiogalactoside (IPTG) at 37 °C for 2 h. Subsequently, the bacterial cells were lysed for 20 min in an ice bath under soni-cation. After centrifugation at 6000 g for 5 min, the supernatant was tested with Coomassie blue staining and immunoblotting. The target protein was obtained by purification using Ni-NTA agarose resin. The resultant protein had a molecular weight of 16 KDa. The lyophilized protein can be dissolved in HFIP for required concentrations. Accompanied by the evaporation of HFIP, eTuSp1 microspheres/microcapsules were assembled by adding 3 µL of spidroin solution to 8 µL of silicon oil without or with negative pressure (negative pressure provided by a fume hood allowed for rapid evaporation), respectively (Fig. 1). For potential drug delivery application, silk spheres/capsules were loaded with a model drug-doxorubicin (Sigma-Aldrich) as an anti-tumor drug.
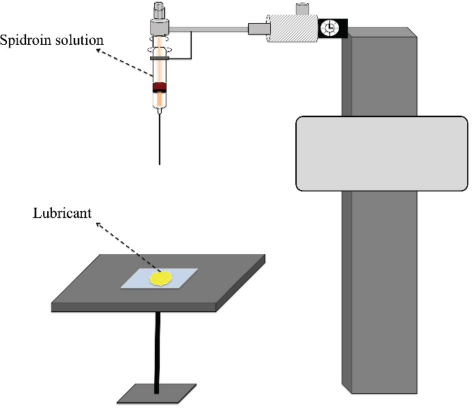
Preparation process of silk spheres/capsules in a lubricant substrate.
Drug-Loading Process
These spider silk microspheres can either be pre-loaded or post-loaded with a drug. In the pre-loading method, the silk proteins were mixed with the drug in HFIP and the spheres were subequently formed using the HFIP-on-Oil method. This method is easily conducted and convenient. However, the loading efficiency often depends on the amphiphilicity of the protein and the target drug.
In the post-loading method, the spheres were prepared first and then incubated with the drug medium. This method is good at precisely controlling the loading percentage and efficiency. A series of factors, such as the pH and the molecular weight of the drug, are very critical for this approach.
After preparation, silk microspheres were loaded with doxorubicin (Dox) in 10 mM phosphate buffer at pH 7. This loading process was conducted at ambient temperature with gentle agitation for 1 h. After centrifugation at 10,000 g for 30 min, Dox-loaded silk spheres were prepared with the w/w ratio (%) of Dox to spheres as 20%. The Dox concentration was spectrophotometrically measured at a wavelength of 490 nm and calculated using a Dox standard calibration curve. To demonstrate the effect of electrostatic interaction on the loading process, silk spheres were labeled with Sypro (Thermo Fisher Scientific) first and then exposed to Dox solution at pH 7 and pH 3 for 10 min. To improve structural stability, the Dox-loaded spheres were treated with methanol (90%) solution to induce the transition of secondary structures.
Drug-Release Process
For enzyme-triggered drug release, the enzyme digestion of Dox-loaded silk spheres were performed at 37 °C with 20 μg/ mL of proteinase K (Sigma Aldrich) and 0.05% sodium dodecyl sulfate (SDS). The enzymatic degradation was observed by the aid of a confocal laser scanning microscope (CLSM).
Characterization
Scanning Electron Microscopy
A JEOL JSM-6335F scanning electron microscope (SEM) was used to determine the dimension and surface morphology of silk spheres and capsules. The solutions of microspheres and microcapsules were pipetted onto a conductive silicon wafter and air dried, subsequently coating with platinum/palladium for 90 s by ion sputtering.
CLSM
Confocal images of silk spheres and capsules were performed with a Leica TCS SP8 system. Under the excitation of different lasers, the fluorescent spheres/capsules were observed.
UV-Visible Spectroscopy
Ultraviolet and visible (UV-Vis) absorbance was measured using an Ultrospec 2100 Pro Spectrophotometer (Amersham Biosciences). The maximum absorbance wavelength of the Dox was 490 nm. A standard concentration curve of Dox was made from 0.25 to 100 μg/mL.
Zeta Potential
The zeta-potential of the same concentrations of Dox-loaded silk spheres were determined in different pH media with a Zetasizer Nano ZS (Malvern Instruments) instrument at 37 °C.
Results and Discussion
The SEM image of silk spheres/capsules is shown in Figs. 2A and B, respectively. The microspheres were observed to have compact surfaces free of large defects. While under negative pressure, microcapsules were obtained with porous micro-structures, predominantly attributed to the rapid evaporation of HFIP. The average size of microcapsules was much larger than that of microspheres at the same protein concentration of 5 wt%.
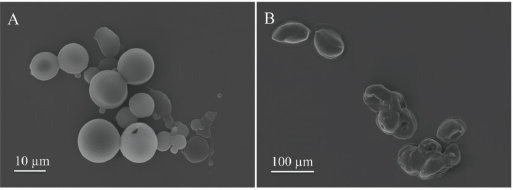
SEM image of silk spheres (A) and capsules (B) at the same protein concentration of 5 wt%.
One of the most straightforward and effective strategies to channel active ingredients into the silk-based delivery system is by directly dissolving or suspending them into the silk protein solution. As depicted in Figs. 3A and B, the Dox can be uniformly pre-loaded to silk spheres and capsules, respectively, without affecting their morphologies.

Confocal image of Dox-loaded silk spheres (A) and capsules (B).
The net/surface charge of silk spheres was investigated by using zeta-potential analysis (Fig. 4). The isoelectric point of silk spheres was ∼4.8. When the environmental pH was below 4.8, the silk spheres were positively charged. However, the silk spheres were oppositely charged in media with a pH above 4.8. To further develop the loading behavior of silk spheres in drug delivery systems, the labeled spheres were exposed to the Dox solution at two different pH conditions (Fig. 5). After 10 min of incubation, the positive Dox was thoroughly loaded inside and on the surface of the negative spheres at pH 7 by electrostatic interaction. Whereas, Dox could not be loaded into the spheres at a pH of 3.
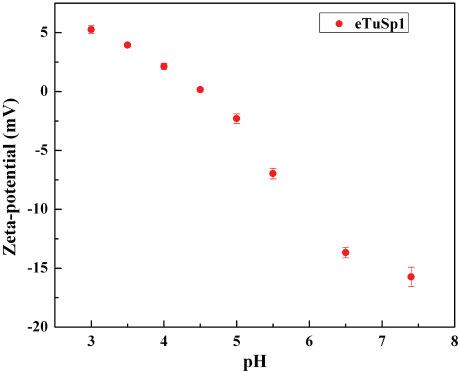
Zeta-potential of silk spheres in different pH media.

The loading behavior of silk spheres in the Dox solution under different pH conditions.
To further demonstrate the potential of silk spheres/capsules as carriers for drug release applications, Dox-loaded silk spheres with a 20% payload were investigated in the enzyme-assisted delivery system (Fig. 6). Proteinase K was the typical proteolytic enzyme used to trigger drug release. As shown in Fig. 6, the collapse of Dox-loaded silk spheres corresponded to the loss of fluorescence after addition of proteinase K. The drug release lasted 7 min after addition of 20 μg/mL of proteinase K. As expected, the original silk spheres without methanol treatment would degrade faster under the same concentration, mainly attributed to the α-helix-rich secondary structure. Therefore, it is feasible to regulate the release of Dox-loaded silk spheres in a controlled manner by enzyme and methanol treatment.
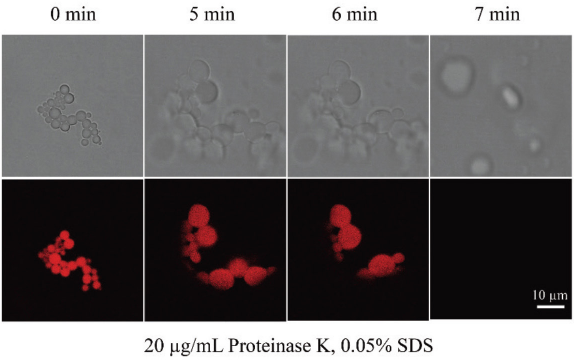
Enzyme-assisted release of Dox-loaded silk spheres by addition of proteinase K.
Conclusion
Microspheres, from a recombinant spider eggcase silk protein, were rapidly prepared with a smooth structure in a lubricant-based system. Under a negative pressure, microcapsules were obtained instead with a porous structure. Using a preloading method, the Dox was uniformly dispersed in the matrix of microspheres and microcapsules, meanwhile, their morphologies remained intact. The isoelec-tric point of microspheres (∼4.8) was determined as a means to regulate the charge of spheres in different pH media. Significant differences were observed in the Dox loading process at pH 7 and 3 due to electrostatic interactions. For enzyme-assisted drug delivery, silk spheres loaded with 20% Dox effectively released the drug within 7 min upon exposure to proteinase K.
Footnotes
Acknowledgement
This work was supported by the Research Grants Council, University Grants Committee (PolyU 5158/13E) and National Natural Science Foundation of China (51373147).
