Abstract
Sesame dye was successfully extracted from food products freely available in the market by a simple boiling method. The extraction temperature and time were 100 °C and 138.3 min, respectively, and the concentration of sesame dye extract was 425 g/L. The sesame dye extract, when used for dyeing nylon fabric, followed the Beer-Lambert law. The optimal conditions for dyeing with sesame dye extract were extracted concentration = 100 g/L, pH = 4, and dyeing temperature = 100 °C. The sesame dye extract was used for dyeing pre-mordanted nylon fabric and good colorfastness results were achieved.
Introduction
Dyeing of textile material with natural dyes has a long history. Although natural dyes have many advantages when compared with synthetic dyes in terms of health, safety, and ecology, since they are non-carcinogenic and biodegradable,1-3 their application has been limited mainly to natural fibers such as cotton, wool and silk.4-6 Natural dyes have seldom been used for dyeing synthetic fibers. In this study, sesame foodstuff was used to obtain dye extract for dyeing nylon fiber. Using foodstuff to obtain dye extract offers several advantages that include the extract being non-toxic, harmless, environment-friendly, and pollution free.7-9 In addition, it can help reduce food wastage caused by expiration. This study explores the use of foodstuffs for synthetic fiber dyeing.
Generally, a mordant is required for dyeing textile substrates with natural dyes.6-9 Metal ions in the mordant can serve as electron acceptors for electron donors to form coordinated bonds with dye molecules. 10 This leads to improved dye uptake and retention, which results in greater depth of shade and colorfastness properties. 10 In this study, a sesame dyeing solution was extracted by a simple boiling method and a pre-mordanting dyeing method was used (i.e., nylon fabric was treated with mordant before dyeing). Four mordants were used for pretreating the nylon fabric before dyeing with sesame dye extract. The conditions for extracting sesame dye from foodstuff were identified and the optimal dyeing conditions were determined. Based on the optimal dyeing conditions, dyeing behavior was investigated in terms of exhaustion properties, color yield, and colorfastness.
Experimental
Material
Ready-to-dye 100% nylon fabric (Teijin DuPont Nylon Ltd.) was used (warp: 20 tex, weft: 25 tex, fabric density: 55 ends per cm and 35 picks per cm, fabric weight: 0.08 g/cm2). The fabric was first washed with 1 g/L nonionic detergent (Lenetol B, Cheung Wah Ho Dyestuffs Co. Ltd.), with a liquor-to-goods ratio (LR) of 50:1 at 60 °C for 15 min, and then rinsed with deionized water for another 15 min to remove oil and impurities. The fabric was dried at 50 °C in an oven and finally conditioned under standard environment of 65 ± 2% relative humidity (RH) and 21 ± 1 °C for 24 h prior to all experiments.
Pre-Mordanting
Dyeing with natural dyes may be carried out with one of four methods: without mordanting, pre-mordanting, simultaneous mordanting, and post-mordanting. In this study, the pre-mordanting method was used. Pre-mordanting was conducted with four different mordants (reagent grade from Sigma Aldrich; used as received with purity (> 98%): (a) aluminum potassium sulfate (Al2K2(SO4)4·12H2O, Alum), (b) copper sulfate (CuSO4·5H2O), (c) ferrous sulfate (FeSO4·7H2O), and (d) stannous chloride (SnCl2·2H2O). In pre-mordanting, 10% owf mordant was applied at 25 °C with an LR of 50:1. After running the bath for 15 min, the temperature was raised at the rate of 1 °C/min until 80 °C and was kept there for 30 min. The fabric was then taken out, squeezed, and finally air dried without washing to make it ready for subsequent dyeing.
Extraction of Sesame Dye
Sesame (in seed form and brown color) was used as the natural dye source and was obtained from the local market. To find the optimal extraction conditions for the natural dye, two extraction parameters, extracted concentration and extraction time, were studied. During optimization, when one parameter was varied, the other parameter was kept constant. To obtain the best extraction results, the extraction temperature used was 100 °C. After extraction, the residual sesame was removed by filtration and the extract in solution form was used directly for dyeing.
Extract Concentration
Varying concentrations of sesame extract were prepared by directly boiling 5, 10, 20, 50, 70, 100, 200, and 500 g of sesame in 1 L deionized water at 100 °C for 2 h in individual beakers using a thermostatic shaking bath. The extracted dyeing solution was cooled to room temperature (RT, 25 °C) and filtered. The absorbance of the extracted dyeing solution was then measured using a UV-Vis spectrophotometer (UH5300, Hitachi) and absorbance (A) against concentration was plotted. The extracted dyeing solutions were used directly for dyeing and their concentrations are termed “extracted concentration” in this study. For example, when extracting 5 g of sesame in 1 L of water, the extracted concentration is defined as 5 g/L.
Extraction Time
Dye extraction was conducted for different periods of time (i.e., 10, 20, 30, 50, 60, and 180 min), but the sesame concentration and extraction temperature were kept constant at 100 g/L (100 g material in 1 L deionized water) and 100 °C, respectively. The extracted dyeing solution was cooled to RT and filtered. The absorbance of the extracted dyeing solution was measured by the UV-Vis spectrophotometer and the absorbance against extraction time was plotted.
Preparation of Calibration Curve
A stock solution of sesame extract (50 g/L) was prepared to verify whether the Beer-Lambert law was obeyed and absorbance over the visible spectrum (400-700 nm) was measured. Based on the absorbance curve, the λmax was obtained. The absorbance at various concentrations (i.e., 10, 30, 50, 70, and 100 g/L of stock solution) was measured at the λmax wavelength and a calibration curve was prepared.
Optimal Dyeing Conditions
Optimal dyeing conditions were obtained based on color yield in the fabric and the amount of dye remaining in the dyebath. Ferrous sulfate pre-mordant (2.0 g) with nylon fabric was dyed with an LR of 50:1 using a thermostatic shaking bath according to the dyeing conditions in Table I. The dyeing profile is shown in Fig. 1. In addition, a control dyeing of nylon fabric without mordanting was conducted, with extracted concentration = 100 g/L, pH = 4, and dyeing temperature = 100 °C.
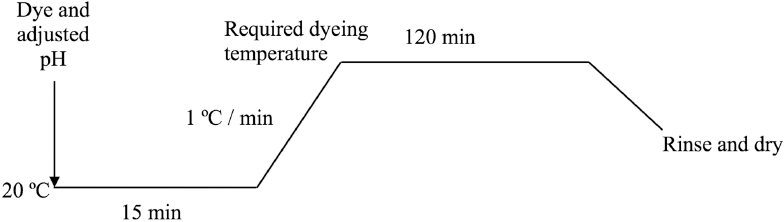
Dyeing profile.
Dyeing Conditions
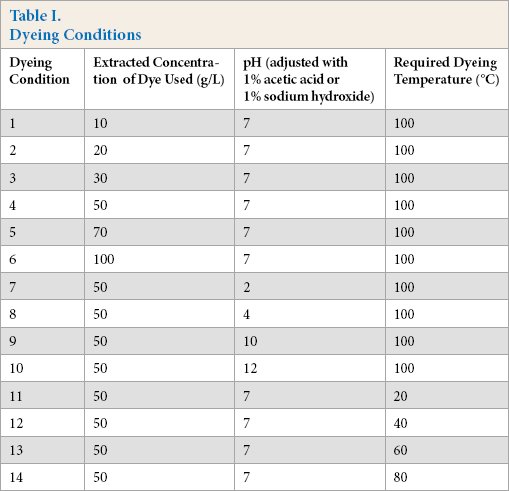
After dyeing, the dyed fabric was removed from the dyebath and squeezed gently to remove excess dyeing solution from the fabric. Ten the fabric was rinsed thoroughly in running deionized water until there was no color bleeding. The fabric was completely dried at 50 °C in an oven, and finally conditioned under standard conditions (65 ± 2% RH and 21 ± 1 °C for 24 h) prior to color measurement. The dyeing solution was cooled to 25 °C before measuring the absorbance.
After obtaining the optimal conditions, dyeing of nylon fabric was carried out and dye exhaustion was determined at the prescribed time. In this study, dye concentration in the dyebath was determined at 0, 15, 30, 45, 60, 90, and 120 min. Concentration of dye was measured at λmax using the UV-Vis spectrophotometer based on the calibration curve prepared previously. Eq. 1 was used for calculating the percent dye exhaustion at different dyeing times.
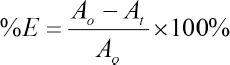
%E is the percent dye exhaustion at time t, Ao is absorbance of dye solution at 0 min, and At is absorbance of dye solution at time t (i.e., 0, 15, 30, 45, 60, 90, and 120 min).
Based on the %E values, an exhaustion curve of %E versus time was obtained and exhaustion at the equilibrium (%Em), defined as a state in which the rate of dye exhaustion is equal to the rate of dye desorption so that %E will no longer increase, was obtained.
Color Measurement
The dyed fabric was conditioned (21 ± 1 °C and 65 ± 2% RH) before color yield measurement with a Macbeth Color Eye 7000A spectrophotometer (D65 illuminant and 10° observer) from 400 to 700 nm. The fabric was folded twice for ensuring opacity and color yield was measured twice. The measured results were then averaged. The color yield is expressed as a K/S value calculated using Eq. 2. The K/S value at λmax was measured; the higher the K/S value, the better the color yield.
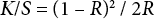
K is the absorption coefficient (depending on concentration of colorant), S is the scattering coefficient (caused by the dyed substrate), and R is the reflectance of the colored sample.
Meanwhile, CIE L*, a*, and b* values were measured and ΔE (total color difference) was obtained.
Fastness Evaluation
Colorfastness to light, washing, and rubbing was evaluated in accordance with ISO 105 B02 (Colour fastness to artificial light: Xenon arc fading lamp test), ISO 105 C01 (Colour fastness to washing), and ISO 105 X12 (Colour fastness to rubbing) respectively.
Results and Discussion
Optimal Conditions of Extraction of Sesame Dye
Optimal values of two extraction variables (i.e., extraction time and extracted concentration) were used to investigate the optimal condition for extraction of sesame dye. Relationships between absorbance and extraction time and extracted concentration are shown in Figs. 2 and 3, respectively. The absorbance curves in Figs. 2 and 3 were represented by a polynomial equation. With the use of differentiation, the maximum point of the absorbance curve was obtained.
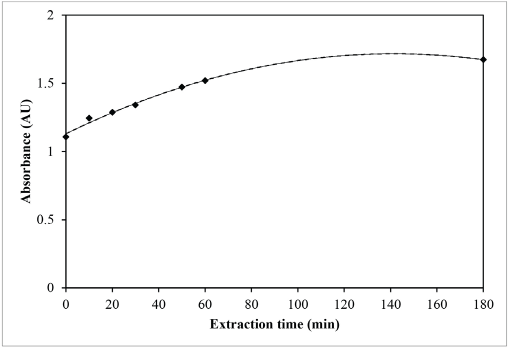
Relationship between absorbance and extraction time of sesame extract.
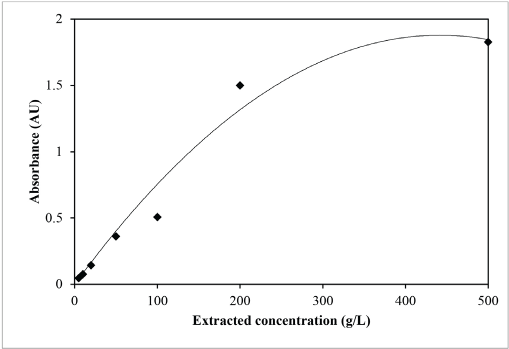
Relationship between absorbance and concentration of sesame in the extract.
For absorbance vs. extraction time, the representing equation of the absorbance curve was y = -3 × 10−5x 2 + 0.0083x + 1.1305 (Fig. 2). For absorbance vs. extracted concentration, the representing equation of the absorbance curve was y = -1 × 10−5x 2 + 0.0085x (Fig. 3).
By setting dy/dx = 0, the maximum extraction time and extracted concentration was calculated.
For the maximum extraction time (dy/dx = 0), -3 × 10−5(2) x + 0.0083 = 0, and therefore x = 138.3 min.
For the maximum concentration of the extracted dye (dy/dx = 0), -1 × 10−5(2)x + 0.0085 = 0, and therefore x = 425.0 g/L.
As a result, the optimal extraction conditions of sesame dye were extraction time = 138.3 minutes, extracted concentration = 425 g/L, and extraction temperature = 100 °C.
Determination of λmax and Calibration Curve
Fig. 4 shows the absorbance curve (absorbance vs. wavelength) of the sesame dye extract at 50 g/L. The λmax refers to the highest value in the absorbance curve. Based on Fig. 4, the λmax of sesame dye extract was 400 nm. The calibration curve of the sesame dye extract is shown in Fig. 5. The correlation between absorbance and dye extract concentration was 0.9979 (R 2 = 0.997) indicating that the dye extract followed the Beer-Lambert law.
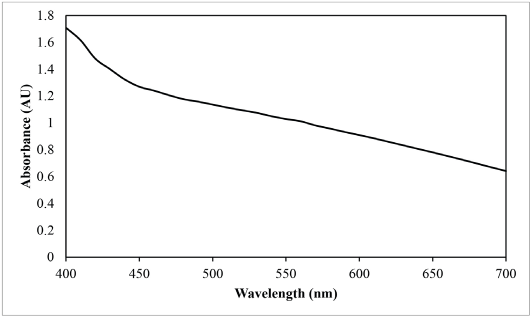
Absorbance curve of sesame extract.
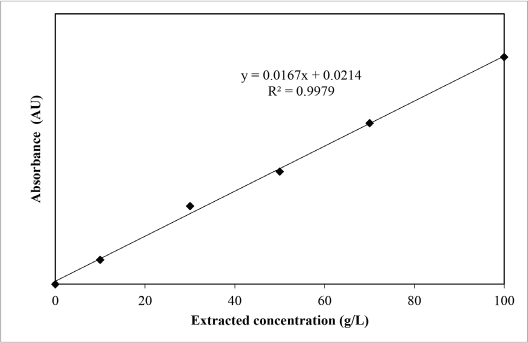
Calibration curve of sesame extract.
Optimal Dyeing Conditions
Optimal values of three dyeing variables (i.e., dye concentration, pH, and dyeing temperature) were used for investigating the optimal dyeing conditions for sesame dyeing of nylon fabric. According to the Kubelka-Munk theory, the K/S value is correlated to the reflectance factor (i.e., Eq. 2). 11
Extracted Dye Concentration
According to Fig. 6, shapes of the reflectance curve at different concentrations were similar (i.e., no chromaticity shift happens when the concentration of the extracted sesame dyeing solution increases). Generally, the amount of dye molecules absorbed by the fabric increases as the concentration of the extracted sesame dyeing solution increases. As shown in Fig. 7, the K/S value of dyed nylon fabric using sesame extract at a concentration of 100 g/L was the highest. As a result, at the extracted sesame concentration of 100 g/L, the color yield was found to be the highest and the percent reflectance was the lowest. Based on the results in Fig. 6, the extraction conditions may not have any adverse effect on the shade of the extracted dyeing solution.
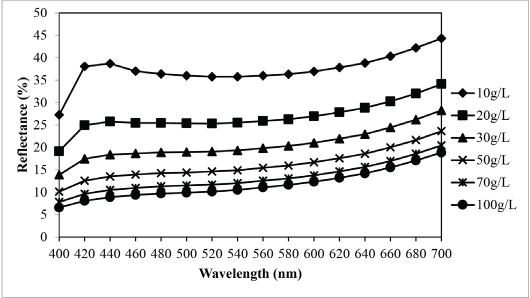
Relationship between concentration of the extract and reflectance of sesame dyed nylon fabric.
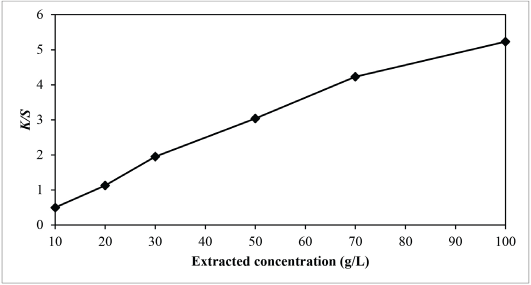
Relationship between extract concentration and K/S values of sesame dyed nylon.
pH Effects
Figs. 8 and 9 show reflectance curves and K/S values of extracted sesame dyed nylon fabric, with the maximum K/S value at pH 4. With the increase in pH values from 6 to 10, the K/S value decreased accordingly, which shows that it was better to dye nylon fabric in an acidic medium with the sesame dye extract. Since nylon has an amino group that can become cationic in nature under acidic conditions, it can be dyed with natural dyes under acidic pH conditions similar to acid dyes. 12 At an acidic pH during dyeing, a large amount of hydrogen ions are present in the dyebath, which can be absorbed by nylon fiber to make it positively charged. The positively charged nylon can attract more dye anions to increase the dye-fiber combination. 13 Thus, the higher uptake of sesame extract under acidic conditions could be due to structural and chemical features of the nylon fiber and sesame extract. It was postulated that during dyeing under acidic conditions, the electrostatic forces between the positively-charged amino groups in the nylon fiber and the sesame extract dye molecules should make an important contribution in increasing the dye absorption.
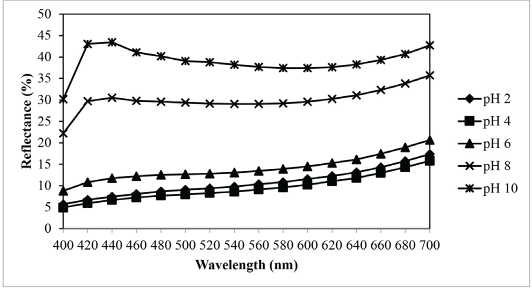
Relationship between pH and reflectance of sesame dyed nylon.
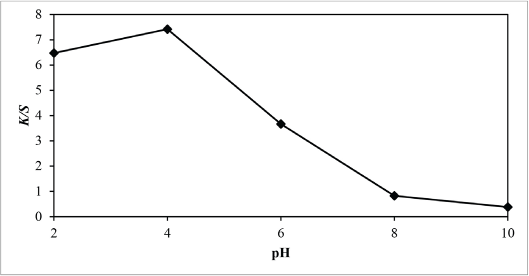
Relationship between pH and K/S values of sesame dyed nylon.
Based on the results of Fig. 8, there were no significant changes in the shape of the reflectance curves at different pHs. Thus, the pH value may not affect the final shade of the dyed fabric.
Dyeing Temperature
According to Fig. 10, shapes of the reflectance curves at different dyeing temperatures were similar (i.e., no color shifts occurred when the temperature of the dyeing solution was increased). The results reveal that the dyeing temperature did not have an adverse effect on the final shade of the dyed fabric. From Fig. 10, the curve of the percentage reflectance at 100 °C was the least, and from Fig. 11, the color yield of the nylon fabric at 100 °C was the greatest. Therefore, the best dyeing temperature was 100 °C. In summary, the optimal dyeing conditions to dye nylon fabric with sesame dye extract as determined in this study were dye extract concentration = 100 g/L, pH = 4, and dyeing temperature = 100 °C.
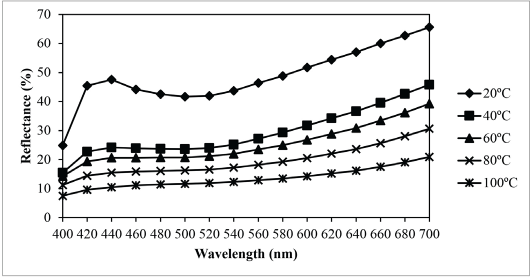
Relationship between temperature and reflectance of sesame dyed nylon.
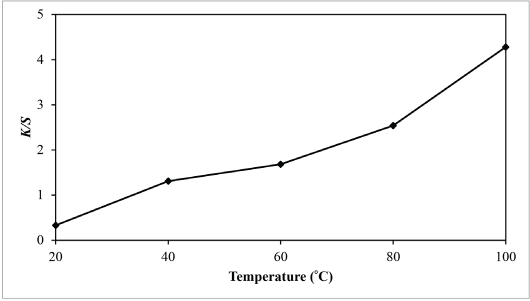
Relationship between temperature and K/S values of sesame dyed nylon.
Exhaustion Dyeing
The exhaustion curve (Fig. 12) shows %E at equilibrium (%Em) and time of half-dyeing (t1/2) were 23.06% and 25.0 min, respectively.
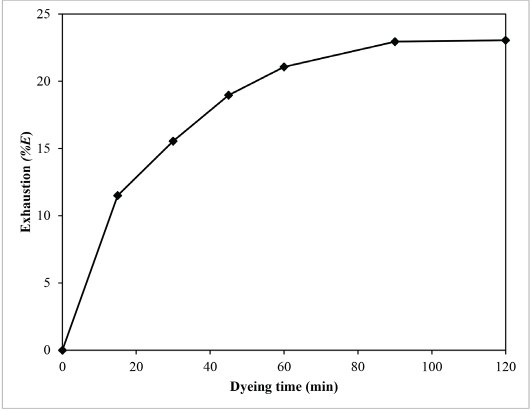
Exhaustion curve of sesame dyeing.
The exhaustion curve provided important information on the rate of dyeing, which can be expressed by the time of half-dyeing (t1/2). Too rapid or too slow dyeing rates are undesirable because the former may cause unevenness, whereas the latter may damage the fiber due to the prolonged contact with hot dye liquor.
Mordanting
Mordants are considered an important part of the natural dyeing process for most natural dyes. Four mordants were used for pre-mordanting, aluminum potassium sulfate, copper sulfate, ferrous sulfate, and stannous chloride. With the use of different mordants, shades of the nylon fabric may change even though the same dyeing solution is used. Meanwhile, color fastness of the natural dye dyed fabric may also change.
Color Change
Variations in color of fabrics dyed with the sesame extract, but with different mordants, were observed from the percentage reflectance curve, K/S values and CIE L*, a*, and b* values.
Fig. 13 shows that reflectance curves of sesame dyed nylon fabrics were similar. This means that the mordants tested did not change the λmax of these dyeing solutions. In the case of sesame dye, the K/S value of fabric pre-mordanted with ferrous sulfate was the best (Fig. 14), followed by aluminum potassium sulfate and stannous chloride. The K/S values of these three mordanted fabrics were higher than the control fabric. However, the K/S value of copper sulfate mordanted fabric was less than that of the control fabric. Table II summarizes the CIE color values and color differences of different dyed nylon fabric samples.
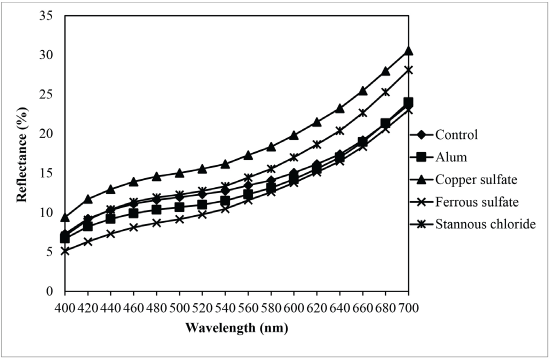
Reflectance of sesame dyed nylon with different mordants.
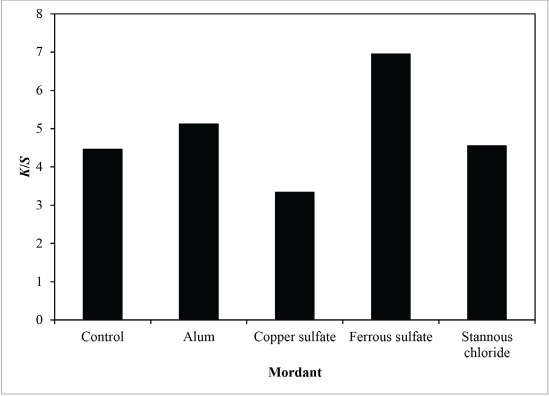
K/S of sesame dyed nylon with different mordants (measured at λmax).
Effect of Mordants on CIE Values for Dyeing of Nylon with Sesame Dye
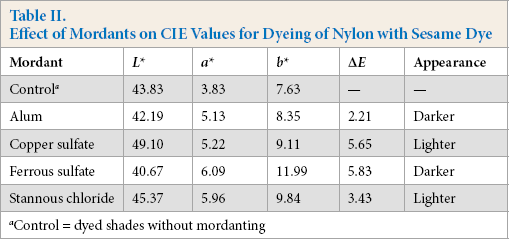
Control = dyed shades without mordanting
The ferrous sulfate mordant contributed the best result and the highest K/S value. It provided the darkest shade when dyeing nylon fabric with sesame dye.
Table II shows the CIE L*, a*, and b* and ΔE* values of nylon fabric dyed with different mordants for pre-mordanting. The pre-mordanting dyeing of nylon gave darker shades and was reflected by smaller L* values and higher ΔE* values. Pre-mordanting with ferrous sulfate provided the greatest differences in ΔE* as compared to the control, which was mainly attributed to lower values of L*.
From the dyeing results of sesame extract and nylon fabric in this study, it was revealed that the chemical structure of the natural dye from sesame extract was like synthetic dyes, which were able to form complexes with metals. The dye-metal complex is a stable form, which enhances dye absorption. As a result, with the pre-mordanting process, good dye uptake and final shade build-up were achieved.10,12 Since natural dyes can form metal complexes with different colors, different metal salts used in the pre-mordanting process were used for obtaining different final color shades of the dyed fabrics.
Colorfastness
When dyed fabric is subjected to particular end use conditions, such as exposure to light, washing, and crocking, its color may change (i.e. color change) and some color may transfer to neighboring materials (i.e. staining). Light-fastness is an important property a dyed fabric needs for long-term use, especially in outdoor textile material. 13
As shown in Table III, pre-mordanting of sesame dyed fabrics gave improvements in lightfastness, indicating that the mordants had a positive effect on lightfastness. Copper sulfate and ferrous sulfate gave better lightfastness results than other mordants. Generally, a stronger dye-fiber bond results in a better light stability of the absorbed dye in the fiber and hence light would have less influence on dye stability. This promotes the improvement of lightfastness. In this study, the increase in lightfastness after pre-mordanting with ferrous sulfate and copper sulfate may be due to the formation of strong and stable dye-metal-fiber bonds.10,14
Effect of Mordant on Fastness Properties with Sesame Dye Solution
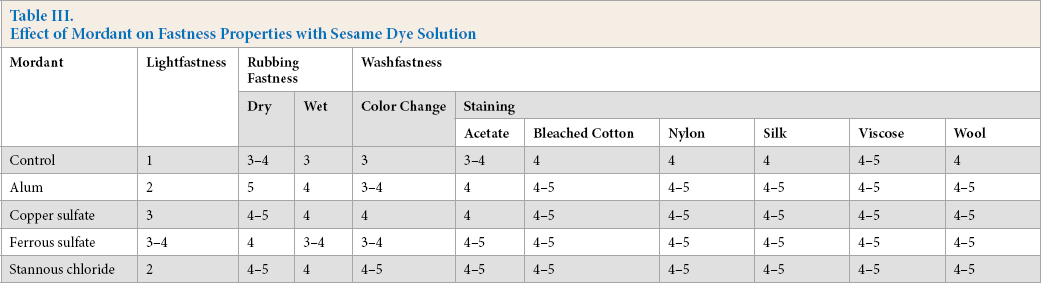
Table III shows that the washfastness of sesame dyed fabrics was good to excellent. Washfastness of dyes is influenced by the amount of dye absorbed in the fiber and the state of the dye inside the fiber. The amount of dye absorbed in the fiber is a function of the geometry of the dye molecule (i.e., molecular size and molecular weight) and the molecular chain arrangement of the fiber (e.g. linearity of fiber molecular chain). Trough the interaction between dye and fiber, the dye-fiber forces tend to make dye molecules remain inside the fiber. For the dyeing of pre-mordanted nylon fabric with sesame natural dye, dye-metal-fiber bonding may occur, which can help retain the sesame natural dye inside the fiber (i.e., it cannot be removed easily during washing).15,16
Table III also shows that dry and wet rubbing fastness of sesame extract dyed nylon fabric improved when using mordants because of the formation of dye-metal-fiber bonding to retain the sesame dye inside the fiber.
Conclusions
In this study, sesame dye extract was obtained by boiling in water. The sesame dye extract obeyed the Beer-Lambert law and was used for dyeing pre-mordanted nylon fabric. Optimal conditions for dyeing with sesame dye extract were extracted concentration = 100 g/L, pH = 4, and dyeing temperature = 100 °C. Depending on the mordants used, good dyeing results were obtained to different extents when compared with control dyeing results. Ferrous sulfate mordant produced the highest color yield. The sesame dye extract dyed nylon fabric achieved good colorfastness results due to dye-metal-fiber bonding.
Footnotes
Acknowledgement
The authors would like to thank the Hong Kong Polytechnic University for the financial support for this work.
