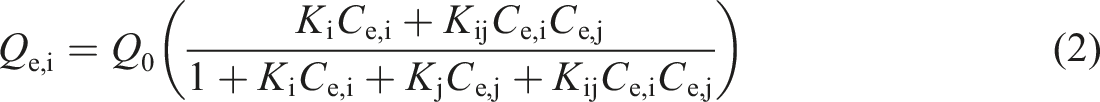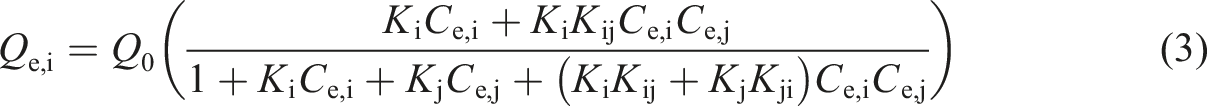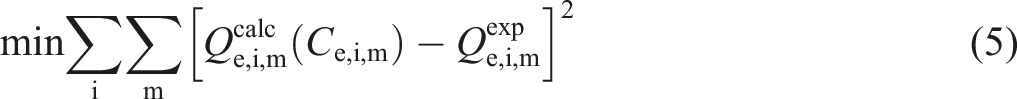Abstract
This present work describes the competitive coordination of iron (III) and copper (II) ions with amidoximated polyacrylonitrile nanofiber and the catalytic performance of the resulting complex (Fe-Cu-AO-n-PAN). The coordination results showed that the increase of the initial concentration of metal ions was beneficial to the increase of the coordination amount. There were both competition and synergistic effects between the two metal ions. But AO-n-PAN was more inclined to coordinate with Fe3+ ions. The promotion effect of Cu2+ ions on iron coordination due to weak positive electric property and small ion radius increased with its initial concentration in the solution. The Langmuir-Freundlich isotherm model among of four selected isotherm models for binary system showed the best fit to the co-coordination reaction between AO-n-PAN and Fe3+-Cu2+ binary solution. Fe-Cu-AO-n-PAN as heterogeneous Fenton catalyst displayed improved catalytic performance than mono-metal complexes due to its better dye adsorption and the synergistic effect between Cu2+ and Fe3+ ions during degradation process, and both the alkali-resistant and the reusability of it were improved at the same time.
Keywords
Introduction
In recent years, while the textile industry plays an important role in the economy and people’s livelihood, it also produces a large amount of organic wastewater, which causes serious environmental pollution and affects human health [1]. Therefore, it is urgent to find a simple and effective method to solve the pollution problem caused by printing and dyeing wastewater. Fenton reaction as an advanced oxidation technology can effectively degrade organic pollutants by activating H2O2 to produce active species with strong oxidizing properties (mainly hydroxyl radicals, ·OH) [2,3]. Especially compared with the homogeneous Fenton catalysts, the heterogeneous Fenton catalysts fixing iron species on various carriers can catalyze the oxidative degradation of organic pollutants even completely mineralize them into small non-toxic molecules such as CO2 and water, avoiding the recontamination caused by iron sludge and working in wide pH range [4,5].
However, with the increase of pH, the catalytic performance of heterogeneous Fenton catalysts is generally reduced. Especially in an alkaline environment insoluble precipitation will be formed between iron ions and hydroxide ions resulting in the catalytic performance significantly reducing due to the active center’s amount decrease. Thus, alkali-resistant treatment, such as the introduction of alkali-resistant active centers, is very necessary. In addition, the catalytic activity of the heterogeneous Fenton catalysts with iron as active species needs to be further improved because of the slow redox cycle rate of Fe2+/Fe3+. To solve this kind of problem, some researchers used the method of adding electron-rich materials to improve the conversion rate of oxidation state/reduction state of the catalytic center, thus enhancing the catalytic activity [6]. Such as Fe@Fe2O3 core-shell nanowires, in which the molecular oxygen activation process could generate superoxide radicals to assist iron core for the reduction of ferric ions to accelerate the Fe2+/Fe3+ cycles [7]. Additionally, ascorbic acid [8] and tetrapolyphosphate [9] could be added to guarantee enough soluble Fe2+ ions for Fenton reaction via the complexation with iron or ferrous ions or via some other additional effects for example single-electron molecular oxygen reduction pathway. To obtain a high proportion of Fe2+/Fe3+ concentration, zero-valent molybdenum catalytic Fenton-like system were explored [10]. Moreover, carbon materials, such as graphitized mesoporous carbon [11] and hydrothermal carbon [12], can be used as not only carriers but also electron donors to accelerate the Fe3+/Fe2+ redox cycle. In addition to electron-rich materials, metal sulfides (MoS2, WS2, Cr2S3, CoS2, PbS, or ZnS) also can be served as co-catalysts for Fenton or Fenton-like technology [13,14]. Both surface metallic active sites and reducing sulfur species contributed to Fe2+ regeneration, thus increasing the accumulation of ·OH oxidants in the system, and the efficiencies varied with the properties of metal sulfides surface. The ultimate goal of these redox cycle rate of Fe2+/Fe3+ improving strategies is to produce more free radicals for oxidative degradation. Previous studies have reported that some polyvalent metals (e.g., Co, Mn, and Cu) can promote the decomposition of H2O2 generating peroxy radical (HO2·) and ·OH via a Haber-Weiss-like mechanism during the Fenton reaction [6]. Thus, copper species have been used as a co-catalyst for iron species in the Fenton reaction to promote the oxidative degradation rate of organic pollutants [15,16]. Therefore, the introduction of co-catalyst in heterogeneous Fenton catalyst with iron as active species is necessary.
Besides the catalytic activity of iron species, the carrier of heterogeneous Fenton catalysts is another important factor affecting the catalytic performance. Compared with inorganic porous materials, organic membrane-like or block carriers, nanofibers prepared by electrospinning or similar nonwoven fabrication technique possess large specific surface area and high porosity. These structure advantages can facilitate the rapid mass transfer of dye molecules in nanofibers, forming local “concentration effect” and enhancing the effective contact between dye molecules and active sites, thus accelerating the catalytic degradation rate [17]. Furthermore, the fibrous carrier allows the catalysts to be easily cut into a suitable shape for application and recycling. In our previous studies, the Fe3+ ion was used to form fibrous metal complexes through coordination bonds with polymer nanofiber carriers containing ligands. The unsaturated coordination of iron ions caused by steric resistance and electrostatic repulsion of metal center makes them have the ability to catalyze the degradation of H2O2 generating ·OH. So the iron-containing nanofiber metal complex can be used as heterogeneous Fenton catalyst showing better catalytic activity than that with commercial fiber as a carrier [18]. Moreover, Fe and Cu ions can coordinate with amidoximated polyacrylonitrile (PAN) to form complex and bimetallic complex with catalytic performance [19]. It is expected to effectively improve the efficiency of heterogeneous Fenton catalyst by a binary metal catalyst system with nanofiber as carrier, that is, Fe and Cu were co-coordinated with modified PAN nanofibers to combine the large specific surface area size effect of nanofibers and the promotion of metal ion cocatalyst. However, the reaction kinetics of co-coordination between two metal ions and amidoximated PAN fiber and the catalytic performance of bimetal complex with nanofiber as the carrier has not been studied.
In our work, the amidoximated PAN nanofiber Fe-Cu bimetal complex was prepared via the co-coordination reaction between Fe-Cu binary solution and amidoximated PAN nanofiber. In order to study the reaction kinetics during the co-coordination reaction, a variety of binary adsorption models were used to fit the data, so as to deeply understand the co-coordination reaction between Fe-Cu binary metal ion solution and amidoximated PAN nanofibers. Further, the obtained bimetallic complex was used as a heterogeneous Fenton catalyst in the simulated wastewater treatment process of azo reactive dyes to investigate its catalytic activity.
Experimental
Materials and reagents
Commercially PAN yarns (Shanghai Sanli Wool Factory) were used as the polymer material of nanofibers, in which the content of acrylonitrile monomer was about 86.96%. Analytical reagents N,N-dimethylformamide (DMF), acetone, hydroxylamine hydrochloride (NH2OH·HCl), sodium hydroxide (NaOH), ferric chloride (FeCl3·6H2O), copper sulfate (CuSO4·5H2O), 30% hydrogen peroxide (H2O2), ethanol, and spectral grade potassium bromide powder (KBr) were used as received (Tianjin Kermel Chemical Reagent Co, Ltd). An azo dye, Reactive Red 195 (abbr. RR 195) was used without purification (Shanghai Macklin Biochemical Technology Co, Ltd).
Preparation of amidoximated PAN nanofiber
The original PAN nanofiber (n-PAN, average diameter was 489 nm) was prepared with homemade multi-pin electrospinning apparatus (Scheme 1). The optimized process parameters were 16 wt.% PAN/DMF spinning solution, 15 kV work voltage, 10 cm work distance between the multi-pin and the receptor, and 1.5 mL h−1 feed rate of the spinning solution. The receptor was covered with aluminum foil and grounded. The obtaining n-PAN was removed from the aluminum foil and placed in a vacuum oven at 60°C for 24 h to remove the residual solvent, and then modified with NH2OH·HCl according to the literature [18]. In detail, dried n-PAN was immersed into 0.40 mol L−1 hydroxylamine hydrochloride solution with the mass to volume ratio of 1 g:150 mL under pH 5.5–6 adjusting by NaOH solution. The amidoximated treatment was carried out at 70°C and stirring for 2 h and then the nanofiber was separated and washed with deionized water for 3–5 times until neutral to obtain amidoximated n-PAN (denoted as AO-n-PAN). Then, it was dried in a vacuum oven at 60°C to constant weight for reserve use, and the percentage of nitrile group converted to amidoxime group (CP%) in n-PAN was calculated to be 25.7% [20,21]. Schematic diagram of the multi-pin electrospinning apparatus
The co-coordination between AO-n-PAN and Fe3+-Cu2+ binary solution
The complex of AO-n-PAN and Fe3+-Cu2+ (denoted as Fe-Cu-AO-n-PAN) was prepared by immersing 1.0 g dried AO-n-PAN into 100 mL Fe3+-Cu2+ binary solution with certain concentrations at 50°C and stirring for a special time. During the analysis of the influence of metal species on each other, the initial concentrations of Fe3+ ions were 18.6, 39.3, 57.8, 80.4, and 100.8 mmol L−1, and Cu2+ ions were 20.1, 41.1, 59.9, 80.1, and 100.3 mmol L−1, respectively. After the complex forming, Fe-Cu-AO-n-PAN was washed with deionized water for 3–5 times and dried in a vacuum oven at 60°C before further treatment. The concentrations of two metal ions in the solution were measured by Vista-MPX ICP (Varian, USA) to calculate the uptake of metal ions (QM). It was worth noting that the measurement of the concentration of metal ions in the reaction endpoints should be carried out by combining the residual liquid and the washing liquid. Complexes of AO-n-PAN and single metal ion (Fe3+ or Cu2+) were prepared with the same process for control, denoted as Fe-AO-n-PAN and Cu-AO-n-PAN, respectively.
Binary isotherms
According to our previous work [22], both Langmuir isotherm and Freundlich isotherm had high R2 values when fitting the data from the coordination between single metal ions and AO-N-PAN, but Langmuir isotherm was relatively more suitable. However, the adsorption process of Fe3+-Cu2+ binary solution by AO-n-PAN cannot be described by Langmuir or Freundlich isotherm commonly used in single metal ion systems due to the addition of competitive ions. Thus, to further understand the adsorption process of two metal ions on AO-n-PAN during the co-coordination reaction, four binary isotherms [23] (competitive Langmuir, uncompetitive Langmuir, partial competitive Langmuir, and Langmuir-Freundlich isotherm) were, respectively, used to analyze the data of the coordination reaction.
Competitive Langmuir isotherm
Competitive Langmuir isothermal model is based on the Langmuir hypothesis. In a binary metal ions system, two cations compete for adsorption sites with each other, and they were adsorbed to different adsorption sites, respectively. In our work, the two cations are Fe3+ and Cu2+ ions, the adsorbent is AO-n-PAN, and competitive Langmuir isotherm can be expressed by equation (1) [23].
Uncompetitive Langmuir isotherm
Uncompetitive Langmuir isotherm is another variant of the Langmuir equation. In a binary metal ions system, one kind of metal ion can occupy not only a single adsorption site, but also the same adsorption site simultaneously with another kind of metal ion. The equilibrium equation can be expressed by equation (2) [23].
Partial competitive Langmuir isotherm
In the partial competitive Langmuir isotherm, one kind of metal ion not only occupies a single adsorption site but also coordinates with an adsorption site occupied by another kind of metal ion. The equilibrium equation can be expressed by equation (3) [23].
Langmuir-Freundlich isotherm
The Langmuir-Freundlich isotherm (equation (4) [23]) is constructed by introducing a power exponent on the basis of the Langmuir isothermal adsorption equation. Initially, it was an empirical formula for the adsorption of multiple gases [24]. The model can be applied to the adsorption process of multiple component liquid by using the equilibrium concentration of each component instead of the partial pressure of the gas.
Data fitting
The data fitting procedure is the determination of the best set of parameters in relation to the adherence of isotherm predictions to experimental data. Different objective functions can be used in the parameter estimation, for example, sum of the squares of the errors, sum of the absolute errors, and average relative error, and in our work the minimization of the sum of the squares of the errors was selected to estimate the parameters, which is expressed by equation (5) [23].
Characterization of Fe-Cu-AO-n-PAN
To study the effect of modification and coordination on the morphology of the nanofibrous membrane, the morphologies of n-PAN, AO-n-PAN, and the fiber metal complexes were observed via an S-4800 field emission scanning electron microscope (FE-SEM, Hitachi, Japan) under the condition of 10 kV voltage. In addition, an APOLLO XL cold-field characteristic X-ray energy spectrometer (EDAX) combined with SEM was used to determine the surface elements and their contents of complexes. A Tensor 37 Fourier transform infrared spectrometer (Bruker, Germany) was used to investigate the chemical structure changes of n-PAN during the amidoximated modification and coordination reaction with metal ions. A total reflection method (ATR) was carried out during the scanning in the range of 4000-500 cm−1 with a resolution of 4 cm−1 for 32 times.
The catalytic activity of Fe-Cu-AO-n-PAN
Fe-Cu-AO-n-PAN was used as the heterogeneous Fenton catalyst for the degradation of RR 195. Typically, 0.2 g of dried complex was immersed into 50 mL of a mixed solution of RR 195 and H2O2 at pH = 6 (unless otherwise specified), in which the initial concentration of RR 195 was 0.05 mmol L−1 and H2O2 3 mmol L−1. Then, under visible light irradiation (400 W high-pressure mercury lamp, Foshan Osram Illumination Co, China, and the visible light intensities were 9.165 mW cm−2 (400–1000 nm) and 0.465 mW cm−2 (365 nm)), the absorbance value of RR 195 solution at the maximum absorption wavelength (522 nm) was measured by a UV-2400 Shimadzu spectrophotometer at a certain time interval during the reaction process, and the corresponding decolorization rate (D%) was calculated. And the RR 195 solutions treated by complexes were measured by a 1020 A total organic carbon analyzer (OI Analytical, USA) to determine the degree of dye mineralization, TOC removal rate (TOC%), which was calculated according to equation (6).
Results and Discussion
The co-coordination between AO-n-PAN and Fe3+-Cu2+ binary system
Effect of Cu2+ ions concentration on the coordination of Fe3+ ions
Figure 1(a) gives the non-linearized adsorption isotherm curves of Fe3+ ions in the presence of Cu2+ ions with different concentrations. Figure 1(a) shows that the content of Fe3+ ions (Qe,Fe) on Fe-Cu-AO-n-PAN at equilibrium increased with the increase of Ce,Fe regardless of the concentration of Cu2+ ions, indicating that high Ce,Fe value was conducive to the coordination of Fe3+ ions onto the ligand sites. Figure 1(b) shows that the selectivity of AO-n-PAN for Fe3+ ions (Qe,Fe) increased with the increase of the initial concentration of Cu2+ ions implying that the addition of Cu2+ ions was beneficial to the coordination reaction between Fe3+ ions and AO-n-PAN. Whilst the content of Cu2+ ions (Qe,Cu) on Fe-Cu-AO-n-PAN decreased with the increase of Fe3+ ion concentration. The possible reasons can be explained as follows. Both the N and O atoms of amidoxime group participated in the coordination reaction as two dentate ligands simultaneously, and the saturation coordination number of Fe3+ ion was 6. Therefore, three adjacent amidoxime groups in AO-n-PAN can form a saturated complex with Fe3+ ions, and at this time, all empty orbitals of Fe3+ ions were occupied resulting in no catalytic activity. However, the large chelated ring formed with the amidoxime groups and Fe3+ ion displayed enhanced steric hindrance, and there was electrostatic repulsion between the adjacent central Fe3+ ions. These reasons led to the unsaturated coordination of Fe3+ ion, which also was the fundamental reason for its catalytic performance. When Fe3+ ions cannot occupy more adjacent amidoxime groups on the same molecular chain due to the steric hindrance and electrostatic repulsion, Cu2+ ions could be “intercalated” between the already occupied amidoxime groups due to its small ionic radius and weak positive electricity. On the other hand, sulfate groups (SO42−) were also introduced into the system as adding copper salt. The electronegative Cl− and SO42− in the system were easy to approach the coordinated electropositive metal ions, and both of them can provide electrons for metal ions and act as ligands forming complex. Compared with the relatively poor stability of the coordination bond between Cl− ion and Fe3+ ion, SO42- can be used as a bidentate ligand similar to “bridging bond unit”[26]. One side was connected with coordinated Fe3+ or Cu2+ ions, and the other side coordinated with free Fe3+ ions in the solution. Furthermore, the electronegativity of SO42− partially weakened the positivity of the metal ion, improving the coordination ability of Fe3+ ions with amidoxime groups. Thus, the Qe,Fe value was indirectly increased because of the increase of Cu2+ ions concentration. Even though the bridging effect of Cu2+ and sulfate groups promoted the coordination reaction of Fe3+ ions, the strong coordination ability of Fe3+ ions enabled them to occupy most of the coordination sites quickly, thus inhibiting the coordination reaction of Cu2+ ions with relatively low Qe,Cu value (Figure 1(b)) and the decreased adsorption percentage of metal ions (AdFe%) due to the existence of competitive ions (Figure 1(c)). In the presence of increasing Cu2+ ions concentrations non-linearized adsorption isotherms of Fe3+ ions (a), Qe,M (b), and Ad% (c) of metal ions at different initial Fe3+ ions concentrations.
Effect of Fe3+ ions concentration on the coordination of Cu2+ ions
In the processes of co-coordination reaction between AO-n-PAN and Fe3+-Cu2+ binary metal ions solution with different concentrations, the non-linearized adsorption isotherm curves of Cu2+ ions in the presence of different concentrations of Fe3+ ions are shown in Figure 2(a). As shown in Figure 2(a), the content of Cu2+ ions (Qe,Cu) on Fe-Cu-AO-n-PAN at equilibrium increased with the increase of Ce,Cu regardless of the concentration of Fe3+ ions, indicating that high Ce,Cu value was conducive to the coordination of Cu2+ ions onto the ligand sites, which is similar to the changing trend of Fe3+ ions. However, as shown in Figure 2(b) and (c), the Qe,Cu and AdCu% values decreased with increasing the initial concentration of Fe3+ ions, and the inhibition effect increased with increasing the initial concentration of Fe3+ ions. The possible reasons included two aspects had been described in section “Effect of Cu2+ ions concentration on the coordination of Fe3+ ions”. On the one hand, Fe3+ ions with strong coordination ability can compete with Cu2+ ions for coordination sites. On the other hand, even though the increase of the initial concentration of Cu2+ ions and the bridging effect of sulfate groups would enhance the chance of Cu2+ ions contacting the coordination site, the coordinated Fe3+ ion as the central ion showed strong positive electricity, hindering the accessibility of Cu2+ ions to ligand sites due to the electrostatic repulsion between Fe3+ and Cu2+ ions. In the presence of increasing Fe3+ ions concentrations non-linearized adsorption isotherms of Cu2+ ions (a), Qe,M (b), and Ad% (c) of metal ions at different initial Cu2+ ions concentrations.
In conclusion, AO-n-PAN would coordinate with Fe3+ ions in priority and had a higher coordination amount. There were mutual competition and synergistic interaction between the two kinds of metal ions. In detail, the addition of Cu2+ ions was beneficial to the increase of the coordination amount of Fe3+ ions, while the latter had a strong competitive effect on the former, which is consistent with the coordination reaction between single metal ions and AO-n-PAN, that is, Fe3+ ions were more prone to coordinate with AO-n-PAN than Cu2+ ions [22].
Data fitting
Isotherm models for binary sorption of Fe3+ ions (i = 1) and Cu2+ ions (i = 2).
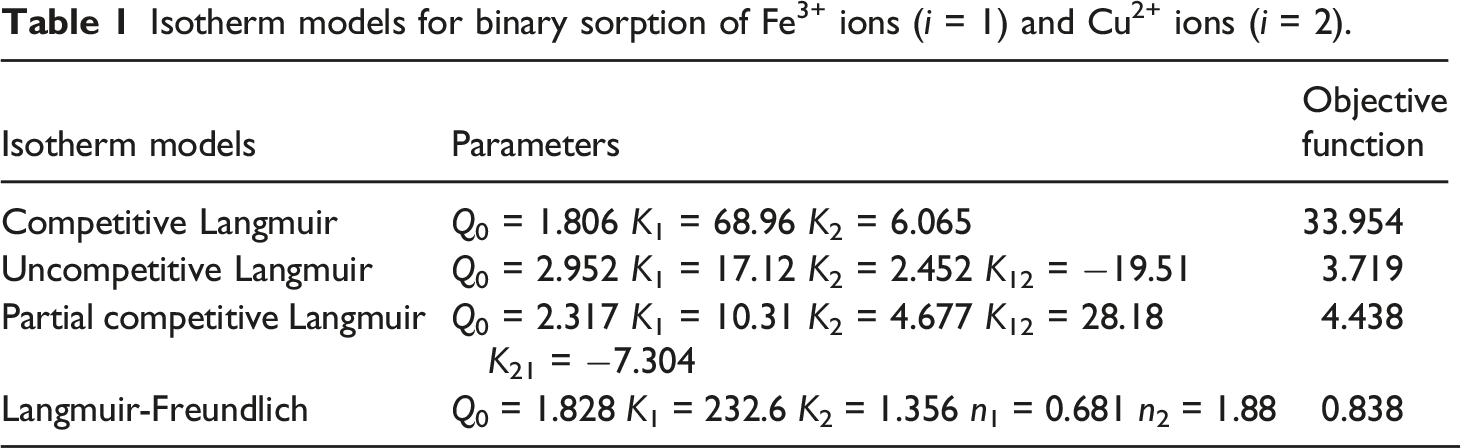

Predict and experimental data for Fe3+ ions and Cu2+ ions in a binary system by Langmuir-Freundlich isotherm.
Characterization of Fe-Cu-AO-n-PANs
The complexes of AO-n-PAN and Fe3+-Cu2+ binary metal ions with the initial concentration ratios (Fe3+:Cu2+ = 2:1, 1:1 and 1:2) were denoted as Fe-Cu-AO-n-PAN1, Fe-Cu-AO-n-PAN2 and Fe-Cu-AO-n-PAN3, respectively. The morphologies of n-PAN, AO-n-PAN, and Fe-Cu-AO-n-PANs are given in Figure 4. As can be seen from the SEM images, the nanofibers in the unmodified n-PAN (Figure 4(a)) were straight and distributed randomly and uniformly throughout the SEM image. After the amidoximated modification treatment, the nanofibers became bent due to swelling and chemical reaction, and the diameter of the fibers slightly increased. However, the surface of the nanofibers became rough after the co-coordination reaction, indicating the successfully anchoring of metal ions on the surface of the nanofibers without significant influence on the morphology of the nanofibers. It is worth noting that the surface roughness of the nanofiber decreased with the decrease of the Fe3+ ions proportion in the initial solution. The reason may be due to the decrease of the Fe3+ ions concentration resulting in the decrease of the total metal content on the nanofiber surface. SEM images of n-PAN (a), AO-n-PAN (b), Fe-Cu-AO-n-PAN1 (Q0 = 1.51 mmol g−1, c), Fe-Cu-AO-n-PAN2 (Q0 = 1.29 mmol g−1, d), Fe-Cu-AO-n-PAN3 (Q0 = 1.10 mmol g−1, e), and EDAX spectrum of Fe-Cu-AO-n-PAN2 (f).
The surface chemical element composition of Fe-Cu-AO-n-PAN2 was characterized by EDAX during the morphology observation (Figure 4(f)). As shown in the spectrum, both Fe and Cu elements were detected besides C, N, and O elements in Fe-Cu-AO-n-PAN, identifying the fixation of Fe3+ and Cu2+ ions on the nanofiber surface. It should be noted that the initial concentration of the two metal ions in the Fe3+-Cu2+ binary metal ions solution was the same, while the molar ratio of Fe3+ to Cu2+ (nFe:nCu) in Fe-Cu-AO-n-PAN2 (calculated from the data in the top right insert in Figure 4(f)) was about 5, indicating that Fe3+ ions were more likely to coordinate with AO-n-PAN than Cu2+ ions. This result was in good agreement with isotherm fitting analysis.
In order to further determine the co-coordination reaction between Fe3+-Cu2+ binary metal ions and AO-n-PAN, FTIR was used to characterize Fe-Cu-AO-n-PANs. Their FTIR spectra are shown in Figure 5. As shown in the spectrum of n-PAN, absorption bands appeared at 2928, 2240, 1736, 1454, 1360, 1240, and 1033 cm−1, which corresponded to the stretching vibration of CH2, C≡N, C=O (carboxyl group in the second and third monomer), the bending deformation vibration of CH2, swing vibrations of CH2 and CH, and C-O, respectively[27–29]. After the amidoximated modification, the intensity of the absorption band attributed to C≡N (2240 cm−1) was significantly weakened, and at the same time, new absorption bands appeared at 3700–3000, 1649, 1103, and 1933 cm−1 which were attributed to the stretching vibration of -OH and C=N, C-N in amidoxime groups, and the deformation vibration of -OH, respectively [30]. These results identify the occurrence of the amidoximated modification reaction. Further after coordination reaction with metal ions, these absorption bands shifted slightly. The absorption band at 1649 cm−1 shifted to 1670 cm−1, as well as the absorption band at 1103 cm−1 to 1110 cm−1, and the absorption band at 933 cm−1 to 940 cm−1. This was because the coordination of the Fe3+ and Cu2+ ions with the N and O atoms of -NH2 and -OH in amidoxime groups, leading to the electrons transferred from N and O atoms to metal ions. As a result, the polarity and bonding electron cloud density of N-H and O-H bands changed and the vibration of these bands was blocked, eventually leading to the absorption band shift. This was consistent with the results in the literature [30,31]. FTIR spectra of n-PAN, AO-n-PAN, and Fe-Cu-AO-n-PANs.
Catalytic performance of Fe-Cu-AO-n-PAN
Effect of the initial concentration ratio of Fe3+ and Cu2+ ions
The catalytic performance of Fe-Cu-AO-n-PAN2 was characterized by catalyzing the oxidative degradation of RR 195 with H2O2, taking Fe-AO-n-PAN and Cu-AO-n-PAN as controls. The D% values of RR 195 solution were shown in Figure 6. The data in Figure 6(a) show that the D% values of the RR 195 solution in the presence of three complexes gradually increased with the extension of time, indicating the catalytic performances of three complexes. And the catalytic activity of Fe-AO-n-PAN was higher than that of Cu-AO-n-PAN under the same conditions identifying the good catalytic property of the former. In addition, Figure 6(a) also shows that the D% values in the presence of Fe-Cu-AO-n-PAN2 were higher than those in the presence of two mono-metal complexes. This was due to the synergistic effect between Fe3+ and Cu2+ ions, which significantly improves its catalytic activity [32]. On the other hand, the high catalytic activity of Fe-Cu-AO-n-PAN2 may also due to its good adsorption for dyes. Degradation of RR 195 with H2O2 catalyzed by different complexes (a) and their corresponding adsorption properties.
H2O2 was not added in the adsorption experiments compared with the degradation experiments, thus there only existed adsorption effect. It can be seen from Figure 6(b) that the D% values of the dye solution gradually increased with the extension of time, indicating that all the three fibrous metal complexes had an adsorption effect on RR 195. Moreover, Fe-AO-n-PAN had a better adsorption quantity for RR 195 than that of Cu-AO-n-PAN. The reason was that there was a higher electrostatic attraction capacity between Fe3+ ions and the electronegative sulfonic acid group due to the higher positive electrical property of Fe3+ ions than that of Cu2+ ions. It is worth noting that Fe-Cu-AO-n-PAN2 had a better adsorption quantity for RR 195 than those of the mono-metal complex. This was because the higher coordination field stabilization of the d orbital of Cu2+ ions enabled them to form Cu-organic complexes with dye molecules that can be used as ligands. Therefore, the introduction of Cu2+ ions was conducive to the improvement of the affinity of the bimetal complexes to dye molecules. Finally, the good adsorption of Fe-Cu-AO-n-PAN2 was conducive to the contact between dye molecules and active radicals, thus contributing to the improvement of its catalytic performance.
By comparing TOC% values, it can be found that the degrees of oxidative degradation of RR 195 by Fe-Cu-AO-n-PAN2/H2O2 was higher (68.8%) than those of in the presence of mono-metal complexes (Fe-AO-n-PAN: 60.5%, Cu-AO-n-PAN: 31.4%) and positively correlated with D% values, indicating that Fe-Cu-AO-n-PAN2 showed higher catalytic activity and can generate more active radicals to degrade dye molecules and even mineralize them.
Effect of pH values
Fe-Cu-AO-n-PAN2 (Q0 = 1.29 mmol g−1) was immersed into RR 195 and H2O2 mixed solution with different pH values and carried out for 120 min under the condition of light radiation to investigate the effect of pH value on the catalytic activity of Fe-Cu-AO-n-PAN2. The D% values of RR 195 solution changed with time are shown in Figure 7. Figure 7(a) shows that D% values of RR 195 at different pH values gradually increased with the extension of reaction time in the presence of Fe-Cu-AO-n-PAN2, which meant that Fe-Cu-AO-n-PAN2 can play a catalyst role in the oxidative degradation of RR 195 at a pH range of 3–9. It’s worth noting that the D% values of RR 195 under acidic and neutral conditions were significantly higher than those under alkaline conditions. This may be because metal ions, especially Fe3+ ions, interacted with hydroxide in an alkaline environment to form an insoluble precipitate and occupied the active site on the surface of the complex, thus impeding the generation of ·OH, finally slowing down the oxidative degradation of dyes [33]. Another possible reason was that there were relatively few H+ ions in an alkaline environment, which was not conducive to ·OH production [34]. Figure 7(b) shows that the D% values at 60 min (D60%) of RR 195 in Fe-Cu-AO-n-PAN2 were higher than those in the presence of the other two mono-metal complexes under acidic or neutral conditions. This was due to the synergistic effect between Fe and Cu complex centers: on the one hand, the addition of Cu2+ ions was conducive to the redox cycles of Fe3+/Fe2+ and Cu2+/Cu+, accelerating the decomposition of H2O2 to produce more ·OH, thus improving the catalytic activity of the complex [19]; on the other hand, the metal-organic complexes formed between dye molecule and the catalyst metal center promoted the adsorption of the dye molecules [34], facilitating the oxidative degradation of RR 195 via ·OH in the system. It’s worth noting that the D60% value of Fe-Cu-AO-n-PAN2 was slightly lower than that of Cu-AO-n-PAN under alkaline conditions, which may be due to the occupation of active sites by insoluble precipitates formed by a large proportion of Fe3+ ions in the complex, inhibiting the generation of free radicals. Degradation of RR 195 with H2O2 catalyzed by Fe-Cu-AO-n-PAN2 at different pH levels (a); comparison of RR 195 degradation with H2O2 catalyzed by different complexes (b).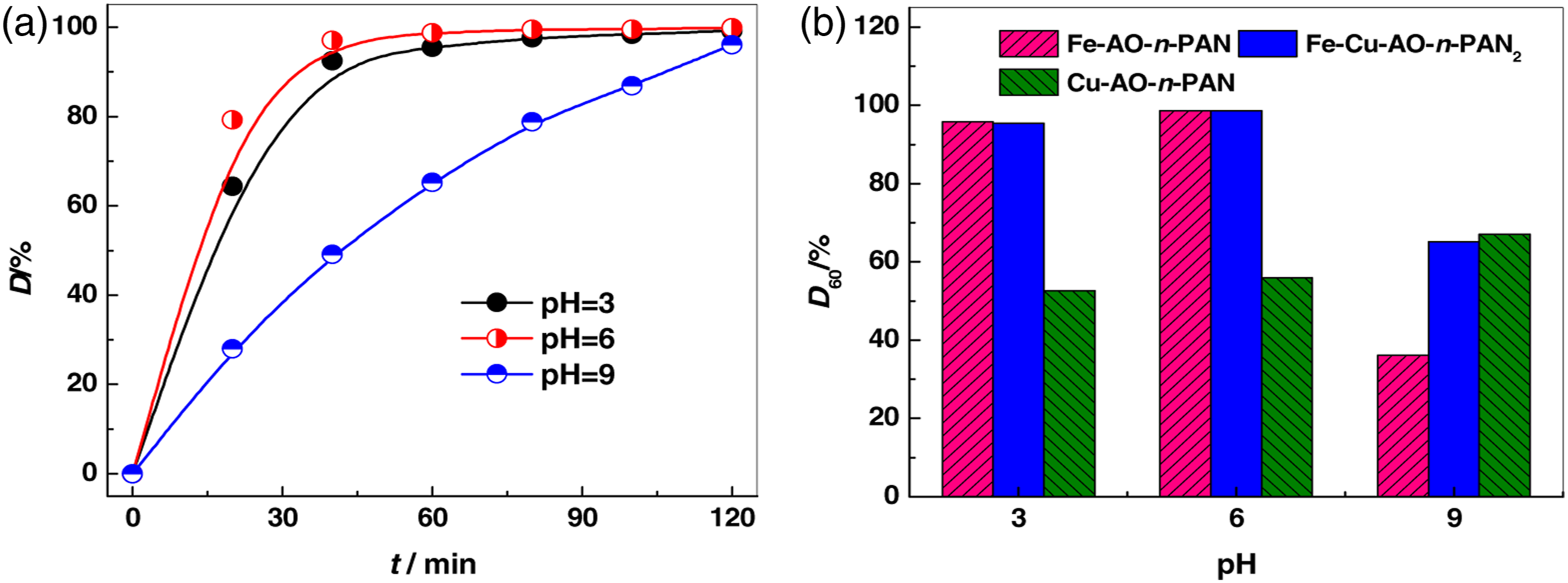
Reusability
The reusability of Fe-AO-n-PAN (Q0 = 1.11 mmol g−1), Cu-AO-n-PAN (Q0 = 1.09 mmol g−1), and Fe-Cu-AO-n-PAN2 (Q0 = 1.29 mmol g−1) were characterized by thoroughly washing the complexes with a deionized water-ethanol mixed solution (v:v = 1:1) and dried at 50°C, and followed the degradation process of fresh RR 195 solution with the washed catalyst from the previous runs. These processes were repeated four times and the results are shown in Figure 8. It can be seen that in the presence of the three complexes, the D% values of RR 195 increased with the prolongation of the reaction time during the five runs, indicating that the three complexes had high removal efficiency for RR 195. However, the D% value of RR 195 treated with Fe-Cu-AO-n-PAN2 did not significantly decrease after five runs, which was still about 90% of the first run. The D% value of RR 195 treated with Fe-AO-n-PAN was also still about 90% of the first run after three cycles, but about 70% after five cycles. Whilst the D% value of RR 195 treated with Cu-AO-n-PAN after five cycles was only 37% of its first run. The loss of catalytic performance may be due to the residual intermediate products on the surface of the complex, which hindered the contact between the complex and the dye molecules. Especially, in the presence of Cu-AO-n-PAN, the degree of oxidative degradation of the dye was low, and the complex intermediate composition resulted in poor reusability due to the blockage of the pores in the complex. However, the introduction of Cu2+ ions in Fe-Cu-AO-n-PAN2 improved the catalytic activity of the complex enabling the dye molecules to be effectively mineralized into smaller molecules, which alleviated the blockage of the pore structure in the complex and thus showing high reusability. The dye degradation recycles in the presence of complexes.
Conclusions
Fe-Cu-AO-n-PANs were prepared by the co-coordination reaction between AO-n-PAN and Fe3+-Cu2+ binary metal ions solution. The increase of the initial concentration of metal ions was beneficial to the increase of the coordination amount. The addition of Cu2+ ions was beneficial to the coordination of Fe3+ ions, and it indirectly increased the coordination amount of Fe3+ ions due to its weak positive electric property and small ion radius acting as a bridge. The promotion effect increased with the increase of the initial concentration of Cu2+ ions in the solution. However, in the same concentration of a co-coordination solution, AO-n-PAN was more inclined to coordinate with Fe3+ ions. They rapidly occupied coordination sites and increased the electrostatic repulsion between coordination center atoms, thus inhibiting the coordination reaction of Cu2+ ions and this effect even severely with the increase of the initial concentration of Fe3+ ions. Langmuir-Freundlich isotherm can well describe the co-coordination reaction between AO-n-PAN and the mixed solution of Fe3+-Cu2+ ions. During the coordination, the coordination reaction rate of Fe3+ ion was much higher than that of Cu2+ ion, and there were both competition and synergism effects between the two metal ions. The coordination between AO-n-PAN with Fe3+-Cu2+ binary metal ions solution forming Fe-Cu-AO-n-PANs had no significant effect on the nanofiber morphology. And the resulting Fe-Cu-AO-n-PAN displayed better catalytic degradation performance than those of mono-metal complexes. The addition of Cu2+ ions was beneficial to improve the dye adsorption of the complex. Combined with the synergistic effect between Cu2+ and Fe3+ ions, the catalytic degradation rate of Fe-Cu-AO-n-PAN was faster, and both the tolerance to alkaline environment and the reusability were improved at the same time.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanxi Province Science Foundation for Youths (201901D211089, 201901D211095, 201901D211097, 201901D211099), Scientific and Technological Innovation Programs of Higher Education Institutions in Shanxi (2019L0202, 2019L0205, 2019L0229, 2019L0327), National Natural Science Foundation of China (21908156), Young Researcher Grant Programs by Kunshan Association for Science and Technology.