Abstract
The quality of pot-spun viscose filament yarn (VFY) is adversely affected by yellowness due to cake-to-cake as well as inside-to-outside variations. Yellowness arises due to inefficient after treatment (AT) of cakes, which prevents effective removal of various impurities from the filaments. To identify the primary source of yellowness, viscose filaments from different AT stages were collected and analyzed to map the extent of total sulfur and metallic impurities in each of the process steps. Inductively coupled plasma-optical emission spectroscopy (ICP-OES), color spectrophotometry, and iodometric titration were used. Free sulfur content related best with the yellowness index, indicating the predominant effect of free sulfur on viscose filament yellowness. This enabled specific optimization of the AT process to reduce yellowness in viscose filaments.
Introduction
Continuous viscose filament yarns (VFY) are produced by three technologies: continuous spinning, pot spinning, and spool spinning. Freshly-spun VFY contain a large quantity of impurities and reaction byproducts owing to various chemical reactions in viscose dope making and regeneration.1,2 These impurities cause discoloration and degradation of the fibers, thus their efficient removal is necessary to achieve desired quality filaments. Although complete and uniform removal of impurities is desired, care must be taken to avoid fiber degradation due to severe chemical/mechanical treatments and repeated handling.
Unlike the continuous-spun yarn (CSY) process, pot-spun yarn (PSY) and spool-spun yarn (SSY) processes are divided process (i.e. the yarn is first collected as a cake or spool, and further treated and dried at different locations in the bulk state). Cake-to-cake as well as inside-to-outside differences in PSY cake properties lead to visually distinguishable color differences in the final cones, resulting in inferior quality products. 3 Cake-to-cake variations arise due to fluctuations in process parameters such as viscose properties, spin bath composition, and after-treatment (AT) conditions.3,4 Inside-to-outside cake variations are introduced due to changes in tension and shrinkage conditions of the yarn during pot spinning, which vary significantly, as the cake is formed layer-by-layer. 4 These inside-to-outside variations can only be reduced to a certain extent by making changes in spinning and drying conditions, but cannot be completely eliminated. 5
For PSY, water washing is the first stage of AT. Gritskov studied the effect of temperature of softened water and found no significant reduction in the impurity content at higher water temperature. 6 The effect of washing with distilled water, de-hardened water, and water treated with weakly acidic exchange resin on the degree of whiteness and impurity content was investigated. 7 Although washing with distilled water and de-hardened water gave marginal improvement in whiteness, the best results were obtained when yarn was washed with water treated with weakly acidic ion-exchange resin. Andriyuk et al. have studied the effect of iron (III) and aluminum (III) in softened water on tinting of viscose fibers and found slightly higher amounts of metals in tinted fibers. 8
To obtain high quality filaments, pot-spun cakes need to undergo efficient AT for effective removal of sulfur and metallic impurities. Cake-to-cake, as well as inside-to-outside, differences in yellowness are observed due to uneven removal of impurities. Various physical and chemical processes have been developed for the removal of sulfur and sulfurous compounds from VFY. Na2S, NaOH, Na2CO3, and Na2SO3 are the most commonly used de-sulfurization chemicals due to their ability to swell viscose filaments and react with sulfur and sulfurous compounds.
After desulfurization, bleaching of the VFY is carried out to improve whiteness by either solubilizing or removing the colored matter, or by altering or destroying their unsaturated or conjugated chromophoric sites. 9 Hydrogen peroxide, sodium hypochlorite, sodium chlorite, chlorine water, and so forth are commonly-used bleaching agents for viscose fibers.9,10 Apart from the active bleaching species concentration, bleaching operation effectiveness depends on the temperature and pH of the medium. 11 Under severe bleaching conditions or prolonged treatment time, oxidation of cellulose or a soft finish can take place, which is undesirable and must be avoided by careful control of bleaching. In the case of chlorine-based bleaching, an additional anti-chlorination step is required for residual chlorine removal to avoid yellowness and degradation of filaments. Soft finish application forms the last stage of AT process and provides the desired cohesion to the filaments for smooth processing in downstream operations.
For mapping the impurity levels across different AT stages, PSY samples were collected from different AT stages and analyzed for their yellowness index and sulfur and metallic impurity content. The mapping was used for identification of probable stages where the yellowness can be minimized. The study also identified the causes of yellowness generation in the filaments. The extent of inside-to-outside differences in color and physical properties of the PSY and the probable causes contributing to these differences were also studied.
Materials & Methods
Materials
Standard chemicals (AR grade) HCl (Loba Chemie, 35.4%), HNO3 (Finar, 69-71%), HF (Merck, 48%), and H2O2 (Emplura, 30%) were used for ICP-OES analysis. Other chemicals used for analysis were elemental sulfur (Merck), formaldehyde (SD Fine Chem, 37-41% w/v), glacial acetic acid, iodine solution (Lobal Chemie Reagents, 0.1 N), starch indicator (Merck, 10% concentration), and sodium sulfite (Na2SO3) solution (SD Fine Chem, 1.5% concentration). Millipore water was used for the preparation of all solutions.
Fiber Spinning and AT
Pot-spun viscose filaments (120 Denier/40 Filaments) used for this study were provided by Indian Rayon Industries Ltd. Post-spinning, the pot-spun cakes were subjected to an AT process comprised of the following stages: (1) water wash, (2) desulfurization, (3) bleaching, (4) chlorine removal (anti-chlor), and (5) soft-finish application on commercial lines, all at the same plant. The cakes were centrifuged after soft-finish application, dried in a hot air oven, and conditioned at 25 °C and 65% relative humidity (RH). To understand the effectiveness of each AT stage, a few cake samples were removed after the completion of each stage. Filaments from inner and outer layers of these cakes were also removed for further analysis.
Yellowness Index of Filaments
The yellowness index (
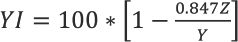
Determination of Total Sulfur and Metallic Impurities
Inductively coupled plasma-optical emission spectroscopy (ICP-OES, Spectro Arcos) was used to determine total sulfur and metallic impurities in the filaments. This method utilizes a high temperature ICP source to ionize the atoms, which are identified by their characteristic emission spectra and quantified by line intensity. Viscose filaments were dried at 80 °C for 2 h, and 1.0 g of it was taken in a special Teflon vessel. Filaments were digested by addition of HCl (4 mL), HNO3 (4 mL), HF (1 mL), and H2O2 (1 mL) and heating for 55 min at 220 °C in a special microwave oven. The resulting clear solution was cooled down and diluted to 25 mL by addition of Millipore water. Samples were injected into the ICP-OES analyzer to determine total sulfur and metallic impurity amounts.
Determination of Free Sulfur in Viscose Filaments
A method developed in our lab was used to determine the free sulfur content in filaments. A known amount of dried filament was taken in a conical flask along with sodium sulfite solution (1.5% w/v). The flask was then heated at ∼100 °C on a steam bath for ∼1 to 1.5 h using a reflux condenser. It was then cooled, and the solution filtered through Whatman filter paper. After adding the required amount of formaldehyde and acetic acid to the filtrate solution, it was titrated against 0.1 N iodine solution using starch as indicator. The endpoint was recorded when the clear solution turned blue. The value of I2 consumed was noted and used to calculate free sulfur content using Eq. 2.
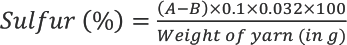
The following chemical reactions take place during the evaluation method adopted for free sulfur estimation. During boiling of the filaments, sodium sulfite extracts free sulfur out by reacting with it (Reaction 1) to form sodium thiosulfate, which is quantified by titration with iodine (Reaction 2).
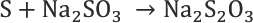

Results and Discussion
Variations in yellowness index values of cakes at different AT stages are graphically summarized in Fig. 1. Although, the yellowness index of the filaments decreased progressively through the AT process, they still adversely affected the quality of the filaments.
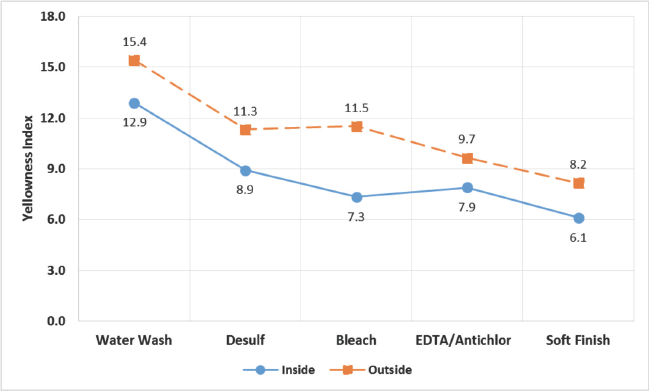
Yellowness index at different stages of AT process for inside and outside layers of yarn.
Various factors imparting yellowness to PSY are summarized in a cause and effect diagram (Fig. 2). Broadly, the discoloration could be due to material properties and/or process conditions. In terms of material properties, yellowness of filament depends on the yellowness index of pulp and its impurities. 13 Apart from those existing in pulp, certain impurities are also introduced during dope preparation due to side reactions between excess carbon disulfide (CS2) and NaOH, and regeneration in the spin bath. 2
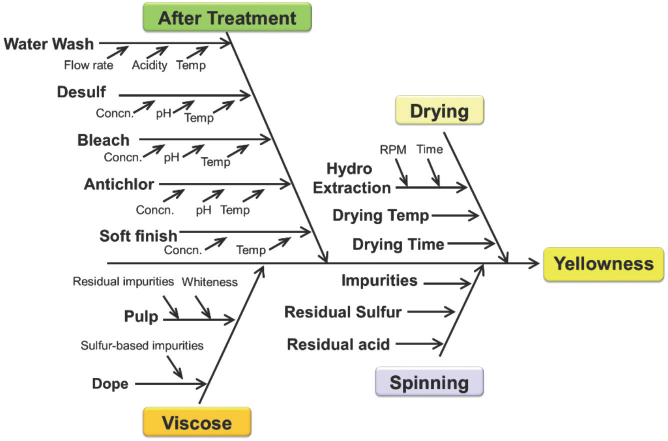
Cause and effect diagram for yellowness in viscose filaments.
Viscose preparation requires reaction of cellulose (Cell-OH) with alkali to form alkali cellulose (Cell-ONa), which reacts with CS2 to give cellulose xanthate. 14 Spinnable viscose solution is obtained by dissolving cellulose xanthate in dilute alkali solution. Viscose fibers are obtained by regeneration of cellulose xanthate to cellulose in a coagulation bath containing H2SO4, Na2SO4, and ZnSO4.
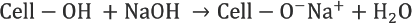
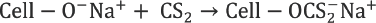
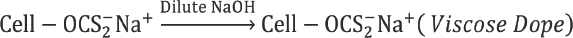
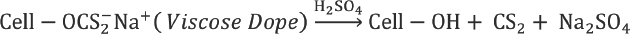
In a perfectly ideal, contamination-free process, sulfur added as CS2 (Reaction 4) forms CS2, metallic sulfides, and sulfates upon regeneration from sodium cellulose xanthate (Reaction 6). But in the actual process, various side reactions take place during viscose preparation and fiber spinning, resulting in formation of various sulfur compounds, which remain in the filament. 2 These reactions are represented schematically in Fig. 3. The excessive carbon disulfide and sodium hydroxide present in dope react to form sodium trithiocarbonate (Na2CS3), which further leads to various sulfur byproducts such as sodium sulfite (Na2SO3), sodium sulfide (Na2S), sodium sulfate (Na2SO4), and sodium thiosulfate (Na2S2O3). 2 Na2S is highly soluble in water and, therefore, most of the Na2CS3 is converted to it. Na2S reacts with O2 dissolved in water and forms Na2S2O3. Polysulfides (e.g., Na2S2) are also formed by the reaction of O2 with alkaline solution of Na2S. Some of the Na2S2 reacts with excess CS2 to yield sodium perthiocarbonate (Na2CS4).
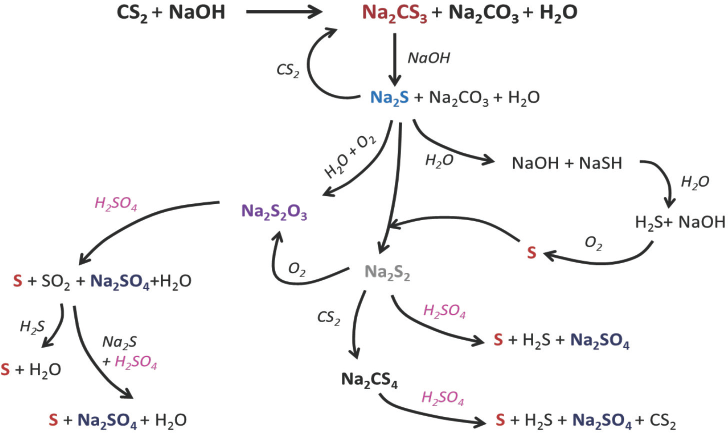
Schematic of reactions forming various sulfur compounds during viscose dope preparation and spinning.
Polysulfides, thiosulfate, and perthiocarbonate present in the dope react with H2SO4 from the spin bath during regeneration and form elemental sulfur (S), which remains trapped in the filaments. During spinning, apart from the formation of impurities due to the action of the spin bath, filaments can also pick up impurities present in the spin bath. 5
AT process conditions dictate the extent of removal of various impurities from filaments, which in turn affect the yellowness. During drying of the cakes, excessive temperature and longer duration can cause oxidation of soft finishes and/or cellulose and cause yellowness.
As residual impurities in viscose filaments impart yellowness, thorough analysis of filaments after each AT stage was carried out to map their extent of removal. Fig. 4 summarizes variation in total sulfur content of filaments at various AT stages. Total sulfur was comprised of sulfates, sulfides, polysulfides, and elemental sulfur present in the filament. 2
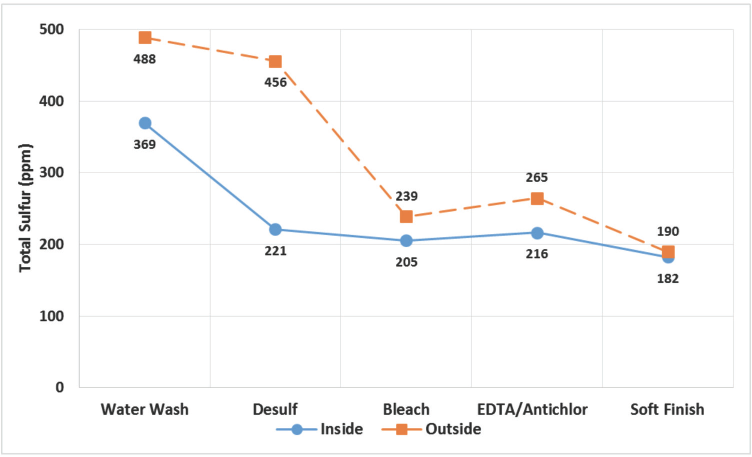
Total sulfur content at different stages of AT process for inside and outside portions of the cakes.
Total sulfur content decreased drastically in the early stages of AT (i.e., desulfurization and bleaching), while removal was gradual towards the later stages. But this total sulfur reduction did not fully explain the reduction in yellowness in viscose filaments through all the AT stages. At the end of the AT process, filaments from inside and outside had comparable total sulfur content, but there were significant differences in their yellowness (Figs. 1 and 4). This indicated that the yellowness difference was not directly correlated with total sulfur content. Similar observations were made by Butkova et al. for CSY yarn, where they found the same total sulfur content in filaments from dark and light bobbin layers. 5
Outside filaments had greater amounts of total sulfur than inside, but this difference narrowed towards the end of the process. Although, the differences in total sulfur were minimized, there were still significant differences in yellowness of the cakes after application of the soft finish. The total sulfur content of the filament did not relate very well to the actual yellowness difference; therefore, the free sulfur content of these filaments was measured.
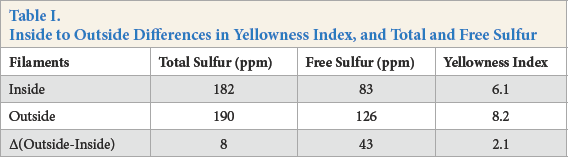
Fig. 5 shows the variation in free sulfur content of filaments with progressing AT stages. Similar to total sulfur content, free sulfur content also decreased drastically in the early stages of AT. But unlike total sulfur, where the differences were minimized towards the end of the process, there were significant differences in free sulfur content towards the end of the AT process (Table I). This is represented graphically in Fig. 6. This indicated that the free sulfur content correlated better with yellowness of the viscose filaments.
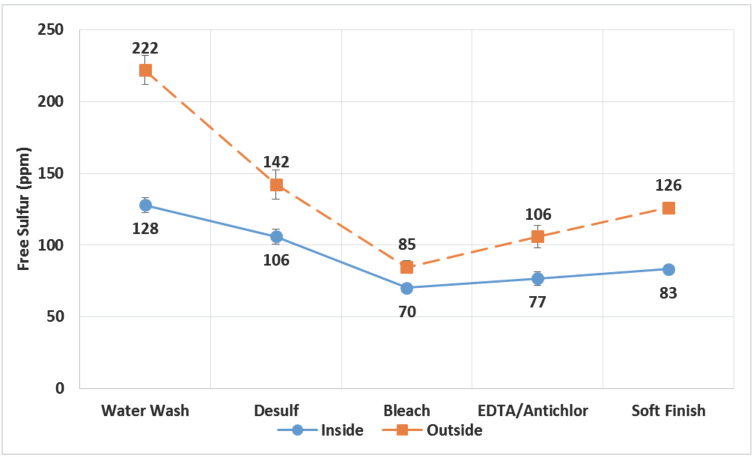
Variation in free sulfur content of filaments at different steps of AT process for inside and outside portions of the cakes.
Inside to Outside Differences in Yellowness Index, and Total and Free Sulfur
Metallic impurities, such as iron (Fe), aluminum (Al), manganese (Mn), zinc (Zn), and lead (Pb) present in the filaments, were also analyzed after each AT stage by ICP-OES. Fe content did not vary significantly across AT stages and was found to be between 26-47 ppm. Other metallic impurities were present in insignificant amounts. Fe salt content of more than 7 ppm have been reported to impart a greyish yellow color to viscose filaments. 5 Although, the Fe contents measured here were greater than those reported in literature, they did not change significantly throughout the process and could not be related to inside-to-outside differences in yellowness.
During the pot-spinning process, tension and shrinkage conditions of the yarn vary significantly as the cake is formed layer-by-layer. 4 As the yarn toward the inner layers shrinks (relaxes) without any tension, while yarn toward the outside layers shrinks on a hard base (shrinkage over the adjacent inner layer), yarn in the inner layers is more relaxed than those that toward outer layers. Because of these differences in relaxation, during AT, inner layers swell to a significantly greater extent than outer layers and offer greater resistance to outward flow of treatment liquor, resulting in inside-to-outside differences in free sulfur content and yellowness (Figs. 1 and 5). It appears that efforts to overcome the inside-outside differences in yellowness would require changing the way the PSY pot is built as well as optimizing design and parameters in the AT process.
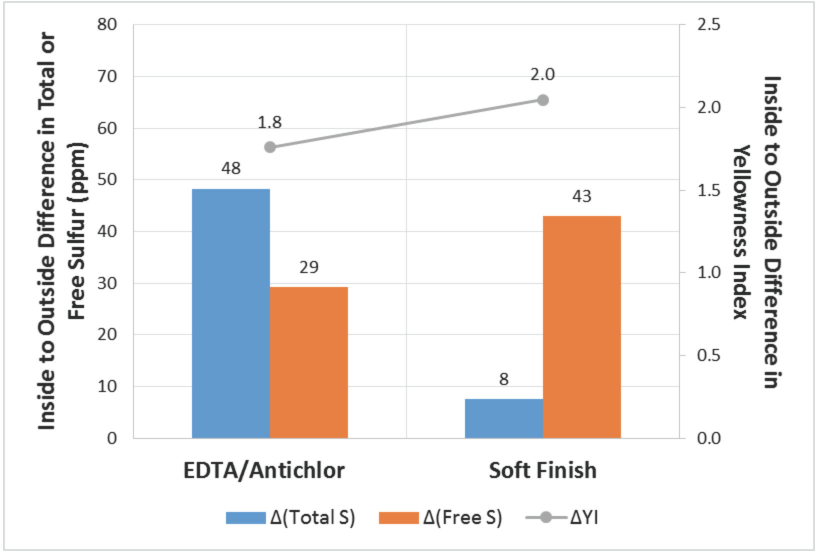
Differences in total sulfur, free sulfur, and yellowness index towards the end of AT process.
Conclusion
Mapping of the yellowness index at various steps of the PSY AT indicates a specific pattern of progressive decrease in yellowness. In addition, the yellowness index of samples from inside and outside of the cake also show significant differences: the outside samples always showing a higher yellowness index. Evaluation of corresponding samples for total sulfur, free sulfur, and metal impurities indicate that there was a better correlation between free sulfur content and yellowness. Therefore, any effort to reduce yellowness should focus on removing the free sulfur efficiently in the AT process. On the other hand, the inside to outside variation in yellowness of the cakes would require addressing aspects of both cake formation and design/process optimization of the AT process.
