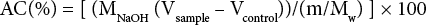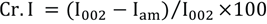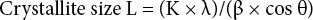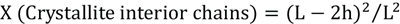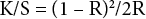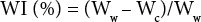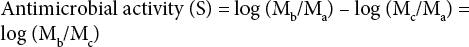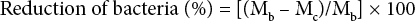Abstract
Cationic cotton was prepared with Girard's Reagent T through interconnecting dialdehyde linkages using glyoxal or glutaraldehyde. The presence of Girard's Reagent T in the treated cotton cellulose was confirmed by various analytical methods. Enhanced dyeability of cationized cotton was confirmed by K/S value evaluation after dyeing with acid and reactive dyes. Crosslinking was corroborated by wrinkle recovery angle measurements. The results verified different reactivities of the two dialdehydes toward cellulose and Girard's Reagent T; glyoxal was a more effective interconnecting linker than glutaraldehyde. Therefore, simultaneous treatment with dialdehyde and Girard's Reagent T provided cationic cotton, resulting in enhanced dyeability, hydrophilicity, antibacterial properties, and resilience. This process was rapid and efficient, and used non-formaldehyde reagents.
Introduction
Cotton is an important fiber in the clothing market.1,2 Its popularity is mainly due to its high degree of hydrophilicity along with low static properties and good comfort characteristics provided by the presence of hydroxyl groups.1,2 Cotton also exhibits some adverse properties such as low wrinkle resistance and low affinity toward anionic dyes, thereby requiring large amounts of salt during dyeing.1–3
Since cotton is a semi-crystalline polymer, hydrogen bonds in amorphous areas can be readily broken and reformed, appearing as wrinkles during end-use applications. 4 This problem can be minimized by introducing more permanent crosslinks between chains that could snap the fibers back to their original dimensions.
The most common commercial crosslinker for cotton is dimethyloldihydroxyethylene urea (DMDHEU). Use of DMDHEU has several drawbacks, such as formaldehyde release, reduction in strength and dyeability, and yellowing.4,51,2,3,4–butanetetracarboxylic acid (BTCA) was studied for many years as a replacement for DMDHEU, but it requires very high curing temperatures (180 °C) and is quite expensive, limiting its wide application. 6 In other studies, dialdehydes such as glyoxal (GO) and glutaraldehyde (GA) have been proposed as alternative non-formaldehyde crosslinkers.4,5 They have several advantages over BTCA, such as reactions at lower curing temperatures and lower cost.
Girard's Reagent T (GT, (carboxymethyl)trimethylammo-nium chloride hydrazide)) is a cationic compound containing a reactive amine functional group. It is known to react very rapidly with aldehydes even at room temperature, and is used for qualitative and quantitative analysis of aldehyde groups. 7 It has not been classified as a toxic material in the United States or the European Union and is believed to be relatively safe. However, since no LD50 or LC50 values are available in Material Safety Data Sheets (MSDS), its toxicological effect must be evaluated in more detail for further practical application. Girard's Reagent T has also been used to introduce cationic charges in various materials such as proteins and oligosaccha-rides.7,8 However, our literature survey indicates that GT has been little used in cellulose cationization.
Cationization of cotton generally improves anionic dye affinity3,5 and antimicrobial properties. 5 Improving affinity for conventional anionic dyes is very important, since the amount of salts needed during dyeing could thereby be reduced, with environmental benefits. Since GT had no functional group to react with cotton cellulose, an interconnecting linkage must be used during the treatment.
Therefore, the objective of this current study was to attach GT to cotton using an interconnecting linkage. A dialde-hyde was used as the linkage because of its eco-friendly properties (as a non-formaldehyde agent) and its ready reactivity with both cellulose hydroxyl and amine groups in GT. The presence of GT in cotton was directly or indirectly measured by Fourier transform infrared spectroscopy (FTIR), surface zeta potential measurement, dye affinity, and elemental analysis. Other characteristics of the treated cotton fabric were also investigated by water imbibition (WI), scanning electron microscopy (SEM), thermo-gravimetric analysis (TGA), X-ray diffraction (XRD), and antimicrobial activity studies.
Experimental
Materials
Cotton fabrics obtained from FITI Korea (KS K0905, WxF 16.5 × 14 tex) with a warp density of 35 threads/cm and a fill density of 31 threads/cm were used throughout the study. The specimen was cut into 17 × 22 cm (WxF) pieces weighing ∼4 g each. Chemicals used were glyoxal (GO, 40% solution) and glutaraldehyde (GA, 25% solution) from Daejung Chemicals, aluminum sulfate catalyst from Junsei, Triton X-100 from Ducksan Chemicals, and Girard's Reagent T (GT) from Sigma Aldrich Korea.
Pad-Dry-Cure
About 4 g of cotton fabric was immersed in a padding bath for 5 min at a 40:1 liquor-to-fabric (LR) ratio. Two dips-two nips padding was used with a two-bowl vertical laboratory padder. Tree different molar ratios of dialdehyde (GO or GA) and GT were used (1:0.5, 1:1, and 1:2). The treated fabrics were dried at 100 °C for 5 min in a forced oven (VOS-201SD, Eyela Inc.) and cured at 135 °C for 5 min in a curing oven (VOS-301SD), unless otherwise noted. The treated cotton fabric was then rinsed several times with deionized water and air dried.
Content Analyses
Aldehyde contents (AC) in the treated cotton fabrics, both dialdehyde only and dialdehyde + GT treated, were evaluated by titration. 9 The treated cotton fabric was immersed at pH 4 for 2.5 h in 0.25 N hydroxylamine hydrochloride solution containing methyl orange indicator. The reaction produced a cellulose polyoxime (Cell-(CH=N-OH)n) along with releasing one HCl equivalent for each formyl residue, which was titrated with 0.1 M NaOH solution to pH 4 until the red-to-yellow end point. 9
MNaOH was the molecular weight of NaOH (40.00 g/mol), Vsample was the amount of 0.1 M NaOH solution in the treated cotton (L), Vcontrol was amount of 0.1 M NaOH solution in untreated cotton (L), m was the weight of the specimen (0.1 g), and Mw was the molecular weight of the repeating unit of (C6H8O5)n in dialdehyde cellulose (160.12 g/mol).
Moreover, a CHNS/O elemental analyzer (SmartScope Inc.) was used with a Flash 200 thermal conductivity detector (TCD) to detect the presence of GT within cotton fabric. About 200 mg of the specimen was evaluated for 10 min at the 100-ppm level with an accuracy of 0.1∼0.3%.
Instrumental Analyses
Treated cotton fabrics were also analyzed by FTIR (Vertex 70, Bruker) with an attenuated total reflection (ATR) attachment at 64 scans and 4 cm– 1 resolution. Thermal characteristics were measured by thermogravimetric analysis (TGA, TG/ DTA 6200, Seiko) under a nitrogen atmosphere (50 mL/min) with aluminum pan in the 25 to 700 °C range at a 10 °C/min rate. The residue was obtained at 400 °C. 1 , 6 Scanning electron microscopy (SEM, CX-100S, Coxem Inc.) was used to analyze cotton surface characteristics. The crystalline structure of cotton cellulose was evaluated by X-ray diffraction (XRD, D2 Phaser, Bruker) using a Co-Kα (λ = 0.1524 nm) source operating at 30 kV and 10 mA. The sample was scanned at 2°/min with a 0.02 increment and 2θ range of 10° to 40°. Crystalline index (Cr.I) was calculated using Eq. 2. 10
I002 is the height of the crystalline peak and Iam is the height of the amorphous shoulder. Crystallite size L of reflection of planes was also calculated from the Scherrer equation (Eq. 3).
K is the Scherrer constant of value (0.94), λ is the X-ray wavelength (0.1542 nm), β is the half-height width of the diffraction band, and θ is the Bragg angle corresponding to the planes. Furthermore, the surface chains occupied a layer ∼0.57 nm thick so the proportion of crystallite interior chains (X) was calculated using Eq. 4. 10
L is the crystallite size for the reflection of plane and h is the layer thickness of the surface chain (0.57 nm). In addition, the surface zeta potential was measured by a zeta potentiometer (BI-870, Brookhaven Inc.) to estimate extent of cationic charge after treatment.
Dyeability
Treated and untreated cotton fabrics were dyed with C.I. Acid Red 1 at 3% owf and a fabric-to-liquor ratio (LR) of 1:50 and adjusted to pH 2∼3 using formic acid. The amount of dye and dyebath pH were selected according to previous studies. 1 , 4 Dyeing was carried out in an infrared (IR) dyeing machine (DL-6000, Daelim Starlet Inc.) Dyeing was started at 40 °C, the temperature was increased to 90 °C for 25 min, maintained at 90 °C for 30 min, and then decreased to 60 °C at a rotation speed of 40 rpm/min. C.I. Reactive Blue 19 was also used at 3% owf and 1:20 LR. Dyeing with or without salt or alkali was used to evaluate the effect of these additives during reactive dyeing. Dyeing with alkali and/or salt was started at 30 °C, increased to 60 °C for 20 min, maintained at 60 °C for 30 min, and kept at 60 °C for another 45 min after adding alkali (20 g/L) and/or salt (40 g/L) at 40 rpm/min. 11 Structures of both dyes are shown in Scheme 1.
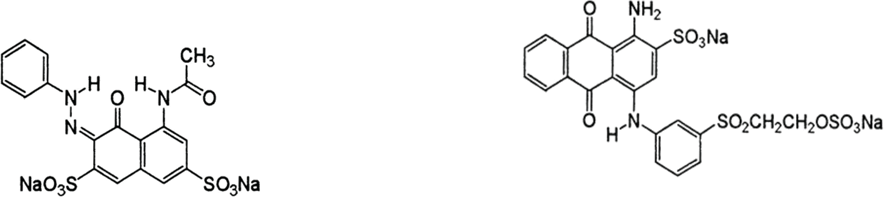
Chemical structures of C.I. Acid Red 1 and C.I. Reactive Blue 19.
Reflectance of the fabrics were measured using a Color i7 colorimeter (CM-512M3, X-rite Inc.) and K/S values were calculated by reflectance using the Kubelka-Munk equation (Eq. 5). 5
K is the absorbance coefficient, S is the diffraction coefficient, and R is the reflectance. Laundering colorfastness was measured according to AATCC Test Method (TM) 61-2010, Colorfastness to Laundering. 12 To test the resilience of the treated fabrics, a Monsanto wrinkle recovery angle (WRA) tester was used. The WRA value is an indirect way to determine crosslinking within cotton cellulose and was expressed in degrees (w+f). 5
Water Imbibition
Hydrophilic characteristics of the cotton fabrics were measured by water imbibition (WI). Approximately 0.05 g of specimen was immersed in distilled water for 2 h, weighed (Ww), and centrifuged at 9000 rpm (MF-300 Centrifuge, Hanil Inc.) for 30 min at 20 °C, followed by weighing (Wc). The WI value was calculated using Eq. 6. 5
Mechanical Strength
Mechanical strength of the fabrics was measured using a Universal Testing Machine (H10KS, Hounsfield Inc.). The size of fabric specimen was 2.5 cm (width) and 15 cm (length). The breaking distance was 7 cm at a rate of 300 mm/min. Five replications were tested to obtain the average value. 1
Antimicrobial Activity
Antimicrobial activity of the cotton fabrics was evaluated for two microorganisms—Staphylococcus aureus (ATCC 6538) and Klebsiella pneumoniae (ATCC 4352)—according to the Korean standard KS K 0693. A detailed procedure was given in our previous study. 5 The reduction of bacteria on the fabrics was calculated according to Eqs. 7 and 8.
Ma is the average number of bacteria in a reference sample immediately after inoculation with test bacteria, Mb is the average number of bacteria in a reference sample after incubation for 24 h, and Mc is the average number of bacteria in an antimicrobial sample after incubation for 24 h. If the value of Ma was lower than that of Mb, the measurement of the sample was confirmed to fail by natural bacterial reduction due to the external environment. 5
Results and Discussion
GO versus GA Linkage
GO and GA are both dialdehydes, but the former is a smaller molecule and can be present in various forms (e.g., as monomer, dimer, or trimer) including formation of cyclic compound in aqueous solution (Scheme 2). 13 GA is larger, with three additional methylene groups, and is mainly present as monomer in aqueous solution. 14 Furthermore, GA has dehydrated, hemihydrated, or cyclic hemiacetal forms depending on the temperature. 14
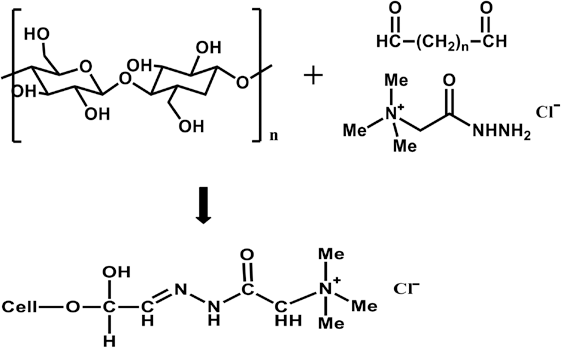
Cationic cellulose preparation with Girard's Reagent T through dialdehyde interconnecting linkage (n = 1 for glyoxal and n = 3 for glutaraldehyde).
The FTIR spectrum of GO did not show a carbonyl peak, whereas the aldehyde carbonyl peak appeared in the GA spectrum at ∼1720 cm–1 (Fig. 1). This suggests that GO was mainly present in various hydrated forms, while GA was a mixed state of carbonyl and hydrated forms, as observed in the previous study. 5 These structural differences between GO and GA (methylene groups and hydrated forms) could influence their reaction with cellulose, in the GT reaction with the cotton cellulose-dialdehyde conjugate, and consequently, in the final characteristics of the treated substrates.
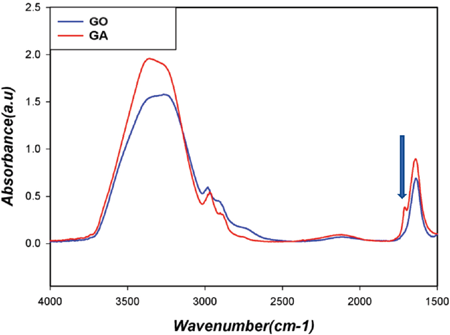
FTIR spectra of aqueous glyoxal (GO) and glutaraldehyde (GA) solutions.
Aldehyde Content
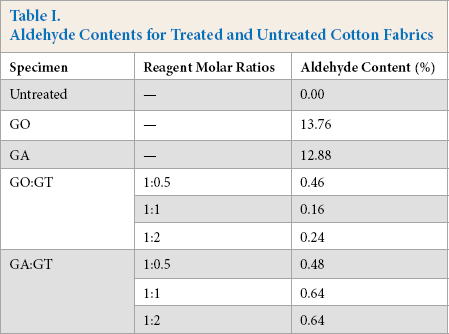
Treatment of cotton with dialdehydes GO or GA considerably increased aldehyde content (AC) values as shown in Table I. The presence of a single-sided reaction (anchoring) of dialdehyde with cellulose and cleavage of hemiacetal linkage at pH 4 were possible reasons for high AC values. On the other hand, simultaneous dialdehyde and GT treatment resulted in very low AC values, indicating high stability of imine linkages between aldehyde and amine at pH 4. Furthermore, the addition of GT substantially improved the stability of hemiacetal linkages, due to the presence of imine and quaternary ammonium groups, capturing acidic protons, and consequently, minimizing acidic cleavage of hemiacetals into aldehyde. These effects would result in low AC values.
Aldehyde Contents for Treated and Untreated Cotton Fabrics
FTIR Analyses
FTIR was used to investigate fixation of GT onto cotton cellulose through the dialdehyde linkage. GT and dialdehyde react to form an imine via nucleophilic addition (Scheme 2). The medium intensity imine absorption peak generally occurs at around 1690∼1640 cm–1. 7 The intensity of the imine peak, however, was even weaker due to the adjacent amide group in GT. Since the amide carbonyl group also appears in this region, the overlap made separation of these two peaks very difficult. These imine and amide carbonyl absorption peaks in the dialdehyde and GT-treated cotton fabrics appeared, but with low intensity (Figs. 2 and 3).
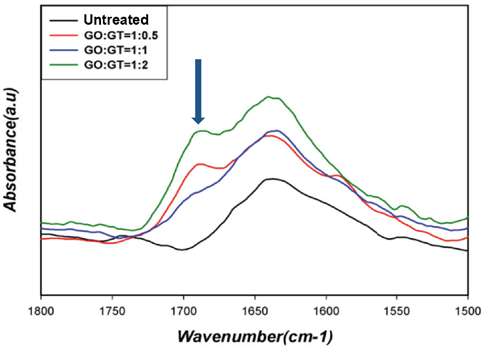
FTIR spectra of cotton fabrics treated with GO and GT at various molar ratios.
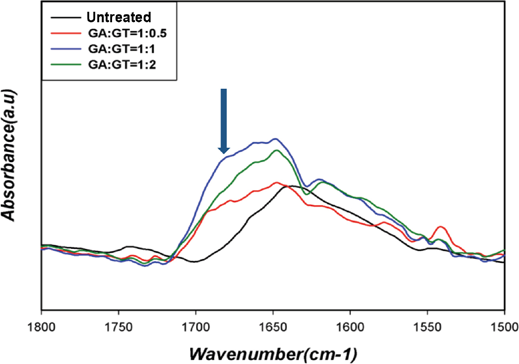
FTIR spectra of cotton fabrics treated with GA and GT at various molar ratios.
Surface Zeta Potential and Elemental Analysis
Surface zeta potential evaluation for the cotton specimens treated with dialdehyde and GT gave all positive values (27.4, 27.6, 62.31, and 63.80) for untreated specimens, and those treated only with GO, GO, and GT (1:1), and GA and GT (1:1), respectively. The increase in positive values confirmed the effect of cationic GT on the treated cotton surface, regardless of dialdehyde type. In addition, these results show that dialdehyde effectively formed a linkage between cellulose and GT. Furthermore, use of GA gave slightly greater zeta potential values, suggesting a more favorable reaction of GA with GT than with cotton cellulose.
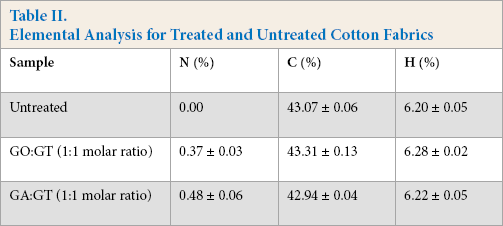
Preferential reaction of GA with GT over reaction with cotton cellulose was also corroborated by elemental analyses (Table II). The results revealed that nitrogen amounts increased considerably after treatment with GT regardless of dialdehyde type. The greater amount of nitrogen in the GA-treated cotton again confirmed preferred reaction of GA with GT. Therefore, this result confirmed that GT was readily fixed onto cotton through dialdehyde linkages.
Elemental Analysis for Treated and Untreated Cotton Fabrics
Dye Affinity
To investigate effect of simultaneous cationization and cross-linking on dyeability, K/S values of the treated cotton fabrics were assessed after dyeing with C.I. Acid Red 1 (Fig. 4). The treated cotton fabrics showed no visual yellowing effects before dyeing. Among three different molar ratios, a 1:1 ratio of GA and GT resulted in the greatest improvement in K/S values of the treated cottons, while K/S values for GO- and GT-treated cotton were quite low at all molar ratios.
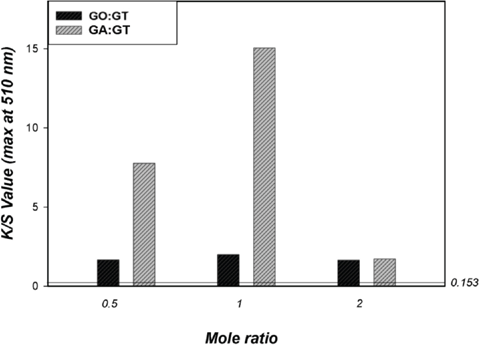
K/S values for cotton fabrics dyed with C.I. Acid Red 1 (untreated cotton = 0.153).
To obtain high affinity towards anionic acid dyes, the treated cotton fabrics must have a high content of cationic GT linked to it. A relatively low pH (2∼3) was used in the dyebath to ensure maximum ionic interaction between the anionic dye and the cationic cotton. GT must also be linked to the cotton cellulose, otherwise, GT would be washed away during the rinsing process and the fabric would lose its cationic properties.
To ensure GT is properly linked to cotton cellulose, two competing reactions must be considered: the reactions of dialdehyde with cotton cellulose and with GT. Based on various experimental results, we believe that GO reacted with cotton better, whereas GA reacted better with GT at certain concentrations. This was substantiated by the greater K/S values observed at low molar ratios of GA and GT. However, a high GT content could hinder the reaction of GA and cotton, and consequently, impair effective GT linkage to cotton. On the other hand, the reaction of GO and cotton was generally hindered by the presence of basic GT, resulting in fewer cationic GT linkages to cotton, and consequently, low K/S values after anionic acid dye dyeing.
Curing Time Effect on Dye Affinity
As shown in Fig. 5, K/S values of the treated cotton fabrics increased with increased curing time, but only with GA- and GT-treated cotton. An extended curing time considerably increased fixation of GT onto cotton. Even though K/S values for the cotton fabrics treated with GO and GT at all molar ratios were still greater than that of untreated cotton (0.15), the increase was much less than those of GA- and GT-treated cotton. The reaction of GO with GT was limited even after long curing times. Colorfastness of the acid-dyed cotton fabric improved from 1–2 for untreated cotton to 3–4 for GO- and GT-treated fabrics, or to 2–3 for GA- and GT-treated fabrics. The presence of the cationic group on cotton improved both dye affinity and dye fixation of an unconventional dye (acid dye) for dying cotton. An acid dye with a large chromophore would further enhance colorfastness on the cationized cotton.
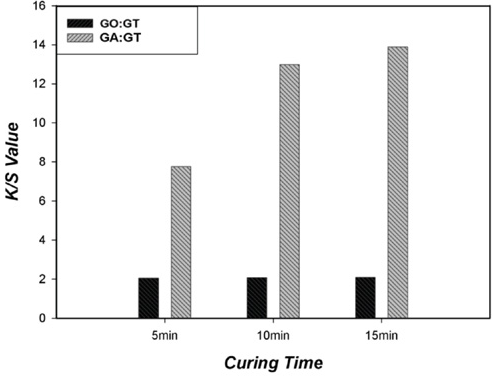
Effect of curing time for treated cotton fabric (molar ratio = 1:1).
Table III shows the K/S values of the cotton fabrics dyed with C.I. Reactive Blue 19. This dye contains an ethyl sulfonyl group, which is converted into an activated vinyl group by addition of alkali. Thus, in the absence of alkali, this dye behaves like an acid dye because contains one sulfonate group and one sulfate group (Scheme 1). Addition of salt could assist dye sorption into the fiber interior, while alkali was needed for dye fixation. For this reason, dye affinity to the treated cotton fabric was measured under four different dyeing conditions: with no additives, alkali only, salt only, and both additives.
K/S Values of Treated and Untreated Cotton Fabrics Dyed with C.I. Reactive Blue 19 under Various Conditions
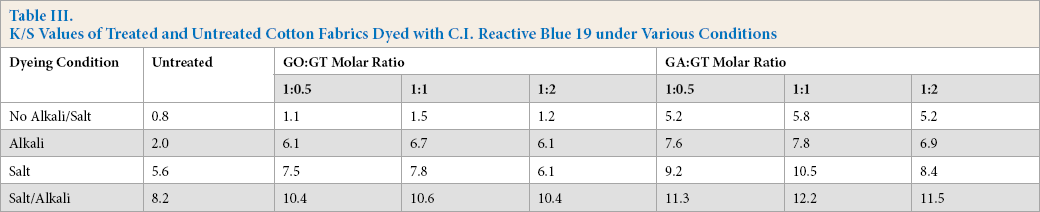
For untreated cotton, the salt effect gave better K/S values than the alkali effect. Without both additives, no dye fixation occurred on untreated cotton, but K/S values were considerably improved by cationization with GA and GT. This again confirmed that GA was more suitable than GO for linking GT to cotton. Furthermore, addition of alkali or salt to the dyeing process improved both untreated and cationized cotton fabric K/S values. It must be noted that improvements in K/S values were always greater with cotton fabrics treated with GA and GT, regardless of the additives used. Cotton cationization could be performed in the absence of additives for the cotton fabrics treated with GA and GT. Therefore, cotton cationization cotton could eliminate, or at least considerably reduce, the need for additives such as salt and alkali in reactive dyeing. Small amounts of additives however could be added to improve dye fixation on cationized cotton.
WRA
Fig. 6 shows WRA values of untreated cotton fabric and cotton fabrics treated by dialdehyde and GT at different molar ratios.
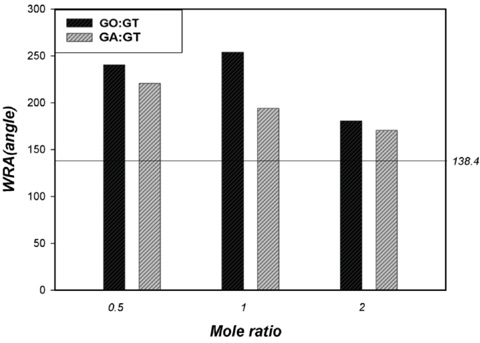
WRA for treated cotton fabrics (untreated cotton = 138.4°).
The results revealed greater WRA values for the treated cotton fabrics compared to the untreated cotton fabric, regardless of the dialdehyde type and GT molar ratio. GO-treated cotton fabrics demonstrated greater WRA values than corresponding GA-treated cotton fabrics. Since the WRA value is an indirect measurement of crosslinking, GO was preferred over GA as a cotton cellulose crosslinking agent, as well being an effective linking agent between cotton cellulose and GT.
Morphology and Crystalline Structure
As shown in Fig. 7, SEM micrographs showed that the morphology of the cotton fabrics treated by dialdehyde and GT appeared similar to that of untreated cotton fabric. No surface deposition was observed. In addition, no significant difference was found in XRD spectra of the GO-treated cotton fabric from that of the untreated cotton fabric, regardless of the GO:GT molar ratio (Fig. 8). The GA-treated cotton fabric also showed the exact same trend (not shown). It appears that the cellulose I structure was preserved during the treatment. 10 In addition, the crystallinity index (Cr.I), crystallite size (L), and crystallite interior chains (X) values were all in the range of 84.3 ± 2, 6 ± 0.5, and 0.65 ± 0.02, respectively, regardless of the dialdehyde type and molar ratio with GT. These values were practically the same as that of untreated cotton, again attesting that the treatment mainly occurred in amorphous regions.

SEM images of cotton fabrics treated with GO or GA, and GT. (a) untreated cotton, (b) GO:GT = 1:1 at 700×, (c) GO:GT = 1:1 at 1500×, (d) GA:GT = 1:1 at 700×, and (e) GA:GT = 1:1 at 1500×.
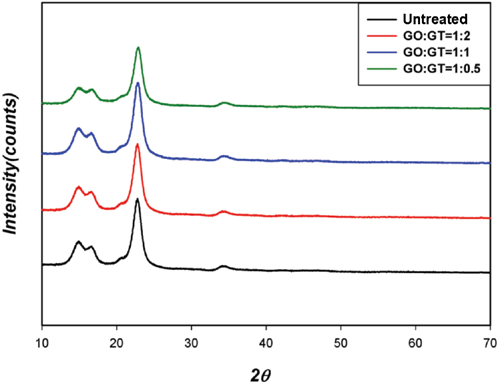
XRD spectra of cotton fabrics treated with GO and GT.
Water Imbibition
Crosslinking and cationization treatment could significantly influence the hydrophilicity of treated cotton. Therefore, water imbibition (WI) was evaluated as shown in Fig. 9. Two opposing factors could influence cotton hydrophilicity in this study: a decrease from crosslinking and an increase from the presence of cationic GT. The greater WI value of the treated cotton fabrics, when compared to untreated cotton fabric, indicated that the presence of cationic GT had the greater effect. Cotton fabrics treated with GA and GT gave greater WI values than those treated by GO and GT, regardless of molar ratios. This was due to the high content of GT in the GA-treated cotton and the longer GA chain crosslinks, providing more spaces between cellulose chains.
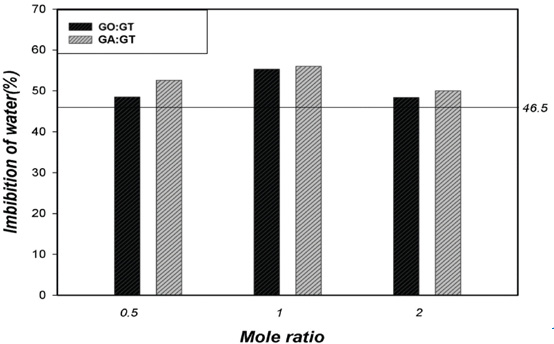
WI values for treated cotton fabrics (untreated cotton = 46.5%).
Thermal and Mechanical Characteristics
Previous studies indicated that greater crosslinking generally gave greater amounts of residue in TGA.1,6 As illustrated in Fig. 10, the formation of greater residue amounts (at 400 °C) for all treated cotton fabrics than that found for untreated cotton fabric, indicated improved thermal stability due to crosslinking cotton cellulose using dialdehyde. In addition, greater amounts of residue for GO-treated cotton fabric than for GA-treated cotton fabric verified the preferential reaction of GO toward cotton fabric over GT. Fig. 11 showed that mechanical properties of the treated cotton fabrics decreased slightly after treatment.
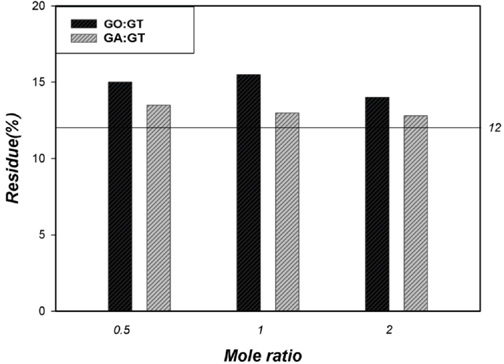
TGA residues at 400 °C for treated cotton fabrics (untreated cotton = 12%).
Antimicrobial Activity
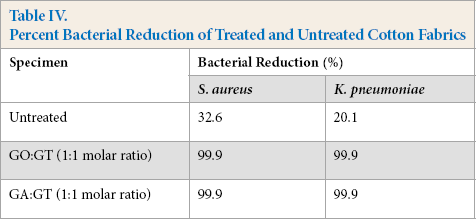
As shown in Table IV, cotton fabrics treated with dialde-hyde and GT showed 99.9% of bacterial reduction against Gram-positive S. aureus and Gram-negative K. pneumonia bacteria, regardless of dialdehyde type. GT, a quaternary ammonium compound, may break anionic bacterial cell walls, causing cell content leakage and death of the bacteria.15–19 Furthermore, GA itself is known to be biocidal. 18
Percent Bacterial Reduction of Treated and Untreated Cotton Fabrics
Conclusions
Cationic cotton was prepared by linking Girard's Reagent T (GT) to cotton cellulose using dialdehydes, such as glyoxal (GO) and glutaraldehyde (GA), as linkers. Various evaluation methods, including FTIR analysis, K/S values after dyeing with acid and reactive dyes, surface zeta potential, AC analysis, elemental analysis, WRA, WI, and residue amounts by TGA were carried out. The results obtained confirmed that GO was more favorable in reacting with cotton cellulose, whereas GA was more efficient in reacting with GT. Therefore, use of GO gave greater WRA and residue values in TGA due to greater cellulose crosslinking and use of GA provided greater K/S values, positive zeta potential, nitrogen content, and WI due to higher GT contents.
SEM and X-ray diffraction studies indicated that treatment did not change the surface morphology and crystalline structure of cellulose and most of the dialdehyde and GT reaction occurred in the amorphous cotton regions. The strength of the treated cotton fabric decreased slightly, however.
Therefore, linking GT to cotton cellulose through a dial-dehyde linkage produced cationized cotton with greater dye affinity, hydrophilicity, WRA values, and antimicrobial properties. This was a rapid, efficient, and eco-friendly process using a non-formaldehyde reagent.
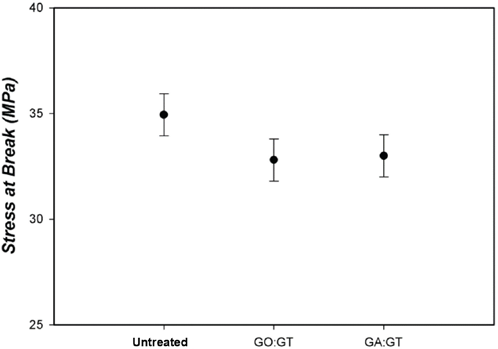
Stress at break for treated cotton fabrics.
Footnotes
Acknowledgment
This work was supported by the BK21 plus Program through NRF grant funded by the Ministry of Education (No. 31Z20150313339) in the Republic of Korea.