Abstract
The present study investigates the efficacy of chitosan and glyoxal-crosslinked chitosan (Chi-Gly) as reinforcing agents for enhancing the mechanical, physical, and functional properties of recycled paper. The research aims to evaluate the comparative performance of these two forms of chitosan in imparting strength characteristics and improved functionality to recycled paper substrates. Chitosan and Chi-Gly solutions prepared via glyoxal crosslinking were incorporated into recycled pulp suspensions at varying dosages. Laboratory handsheets were fabricated, and their properties were systematically evaluated through mechanical testing, morphological analysis, wettability measurements, and antibacterial assays. The results demonstrated significant improvements in the tensile index, burst index, and bending resistance for the chitosan and Chi-Gly treated papers, with the Chi-Gly exhibiting superior reinforcement. Notably, the Chi-Gly treated paper exhibited a higher wet tensile index and lower water absorption capacity than the control. SEM analysis revealed a denser, more cohesive fiber network facilitated by chitosan and Chi-Gly, aiding the reinforcement. The treated papers exhibited reduced hydrophobicity and pronounced antibacterial activity against E. coli (a gram-negative bacterium) and S. aureus (a gram-positive bacterium), with the Chi-Gly treatment outperforming chitosan. Notably, the treatments improved the functional properties without negatively impacting optical brightness. The findings highlight the synergistic effects of glyoxal crosslinking on chitosan’s reinforcing ability and the potential of these biopolymers as sustainable and multifunctional additives for the recycled paper industry.
Introduction
Cellulose, a primary raw material employed in the manufacturing of paper and paperboard, generates an estimated 400 million tons of waste paper annually. 1 A significant portion of this waste paper is either disposed of in landfills or incinerated, raising substantial environmental concerns and resulting in the squandering of valuable cellulose resources. 2 The recycling of fibers for papermaking presents an opportunity for energy conservation and pollution mitigation. 3 However, the hornification process, which occurs during recycling, diminishes the mechanical properties of paper due to reduced fiber binding capacity and impaired fiber swelling ability.4–6 Additionally, the inherent hydrophilicity of cellulosic paper may restrict its range of applications. 7
Comparison of wet and dry tensile indices for various paper additives.
Chitosan, a biodegradable and biocompatible polymer derived from the deacetylation of chitin, has gained significant attention due to its beneficial properties, such as low toxicity, antioxidant and antibacterial effects. 20 Owing to these properties, chitosan finds applications in various fields, including food, chemical, textile, medicine, functional materials, and agriculture.21,22 In recent years, research has focused on utilizing chitosan as a papermaking additive to enhance the wet and dry strength of paper.23–26 For instance, a study by Gaafar et al. 27 investigated a chitosan Schiff base, 4-(2-Hydroxyaniline)pent-3-en-2-one chitosan (2-HyA-CS), and its ZnO nanocomposite (2-HyA-CS/ZnO). The modified chitosan and its nanocomposite demonstrated superior adsorption efficiency for removing Remazol Brilliant Blue R dye from aqueous solutions compared to pure chitosan. Moreover, the 2-HyA-CS/ZnO nanocomposite exhibited enhanced antimicrobial activity against both Gram-positive and Gram-negative bacteria, as well as a yeast strain. The study also reported promising cytotoxic effects of the nanocomposite against human cancer cell lines. These findings highlight the potential of chitosan derivatives and nanocomposites in expanding the functional properties and applications of chitosan-based materials.
Bacterial and viral infections, such as those caused by coronaviruses and noroviruses, pose a constant threat to human health and healthcare systems. Consequently, efforts to control these pathogens are essential. Developing durable antibacterial and antiviral materials is imperative to safeguard human well-being and ensure the safety of healthcare workers. 28 Chitosan, a biopolymer possessing antioxidant and antibacterial properties, can be utilized alone or in combination with other polymers.29,30
Recently, glyoxal has garnered attention as a promising cross-linking agent due to its desirable properties, including excellent water solubility, cost-effectiveness, and versatile functionality. Furthermore, glyoxal can form an acetal between its aldehyde and the hydroxyl groups of the glucosamine units in chitosan. This dual functionality and distinct property of glyoxal can play a crucial role in binding to chitosan via its carbonyl and/or hydroxyl groups in the backbone, resulting in the combination of more chitosan macromolecules, thereby increasing the molecular weight and improving the physicochemical properties of chitosan.31
Chitosan’s structure resembles that of cellulose, enabling strong bonding with fibers and providing dry and wet strength in papermaking.32,33 The chemical interactions between chitosan and cellulosic fibers, as well as the modifications introduced by glyoxal crosslinking, play a crucial role in enhancing the properties of recycled paper.34,35 Chitosan is a polycationic biopolymer, and its structure resembles that of cellulose. This structural similarity allows for strong interactions with the cellulosic fibers in paper. The proposed mechanisms include: a. Ionic interactions: The protonated amino groups (-NH3+) of chitosan can form ionic bonds with the negatively charged groups (e.g., carboxyl groups) present on the fiber surfaces.
36
b. Hydrogen bonding: The hydroxyl groups (-OH) of chitosan can form hydrogen bonds with the hydroxyl groups of cellulose in the fibers, creating strong intermolecular interactions. c. Covalent bonding: The amino groups (-NH2) of chitosan can react with the aldehyde groups (-CHO) present in the oxidized cellulose fibers, forming covalent imine (-C=N-) bonds.
29
These interactions between chitosan and the cellulosic fibers promote better fiber-fiber bonding, resulting in improved mechanical properties, such as tensile and burst strength. Glyoxal, a dialdehyde compound, acts as a crosslinking agent for chitosan, leading to the formation of chemically crosslinked chitosan (Chi-Gly). The proposed reaction mechanism involves
30
: a. Acetal formation: The aldehyde groups (-CHO) of glyoxal can react with the hydroxyl groups (-OH) of the glucosamine units in chitosan, forming acetal linkages (-CH(OR)2). b. Schiff base formation: The aldehyde groups of glyoxal can also react with the amino groups (-NH2) of chitosan, forming imine (-C=N-) or Schiff base linkages.
The objective of this study is to elucidate the efficacy of physically crosslinked chitosan (pure chitosan) and chemically crosslinked chitosan (with glyoxal) as reinforcing agents for enhancing the wet and dry strength properties of recycled paper. The investigation aims to evaluate the comparative performance of these two forms of chitosan in imparting strength characteristics to recycled paper substrates under wet and dry conditions. By examining the reinforcing effects of physical and chemical crosslinking mechanisms, this research provides insights into the potential application of chitosan as a sustainable and effective strength additive for the recycled paper industry. The study seeks to determine whether the chemical crosslinking of chitosan with glyoxal offers superior reinforcement capabilities compared to physically crosslinked pure chitosan when incorporated into recycled paper. Ultimately, the findings will contribute to developing eco-friendly and high-performance papermaking additives derived from renewable biopolymers.
Materials and methods
Raw materials and chemicals
Characteristics of mixed office waste recycled pulp.

Preparation of chitosan and glyoxal-crosslinked chitosan solutions
The synthesis of chemically crosslinked chitosan was carried out through a sequential process. Figure 1 presents a schematic overview of the experimental procedure employed in this study. Firstly, 1.0 g of chitosan powder was added to 100 mL of a 1% (v/v) acetic acid solution. This mixture was subjected to magnetic stirring for 1 h at an elevated temperature of 60°C to ensure complete dissolution of the chitosan biopolymer. Acetic acid as a solvent facilitated the protonation of the amino groups present in the chitosan backbone, rendering it soluble in the aqueous medium. Subsequently, an aqueous glyoxal solution, a dialdehyde compound, was introduced into the chitosan solution at varying concentrations of 0.2, 0.4, and 0.6% based on the dry mass of chitosan. These different concentrations were employed to investigate the effect of the glyoxal crosslinker on the properties of the resulting chemically crosslinked chitosan. After adding the glyoxal solution, the mixture was subjected to further magnetic stirring for 10 min at the same temperature of 60°C. This step was crucial to facilitate the cross-linking reaction between the amino groups of chitosan and the aldehyde groups of glyoxal. The elevated temperature promoted the formation of covalent imine bonds (-C=N-) between the two reactants, leading to the cross-linking of the chitosan macromolecules. The resulting chemically crosslinked chitosan solution was allowed to cool to room temperature before being utilized as a reinforcing additive in the papermaking process. The crosslinking reaction with glyoxal was expected to enhance the mechanical properties, including wet and dry strength, of the recycled paper substrates. Schematic illustration of the experimental procedure for chitosan modification and paper treatment.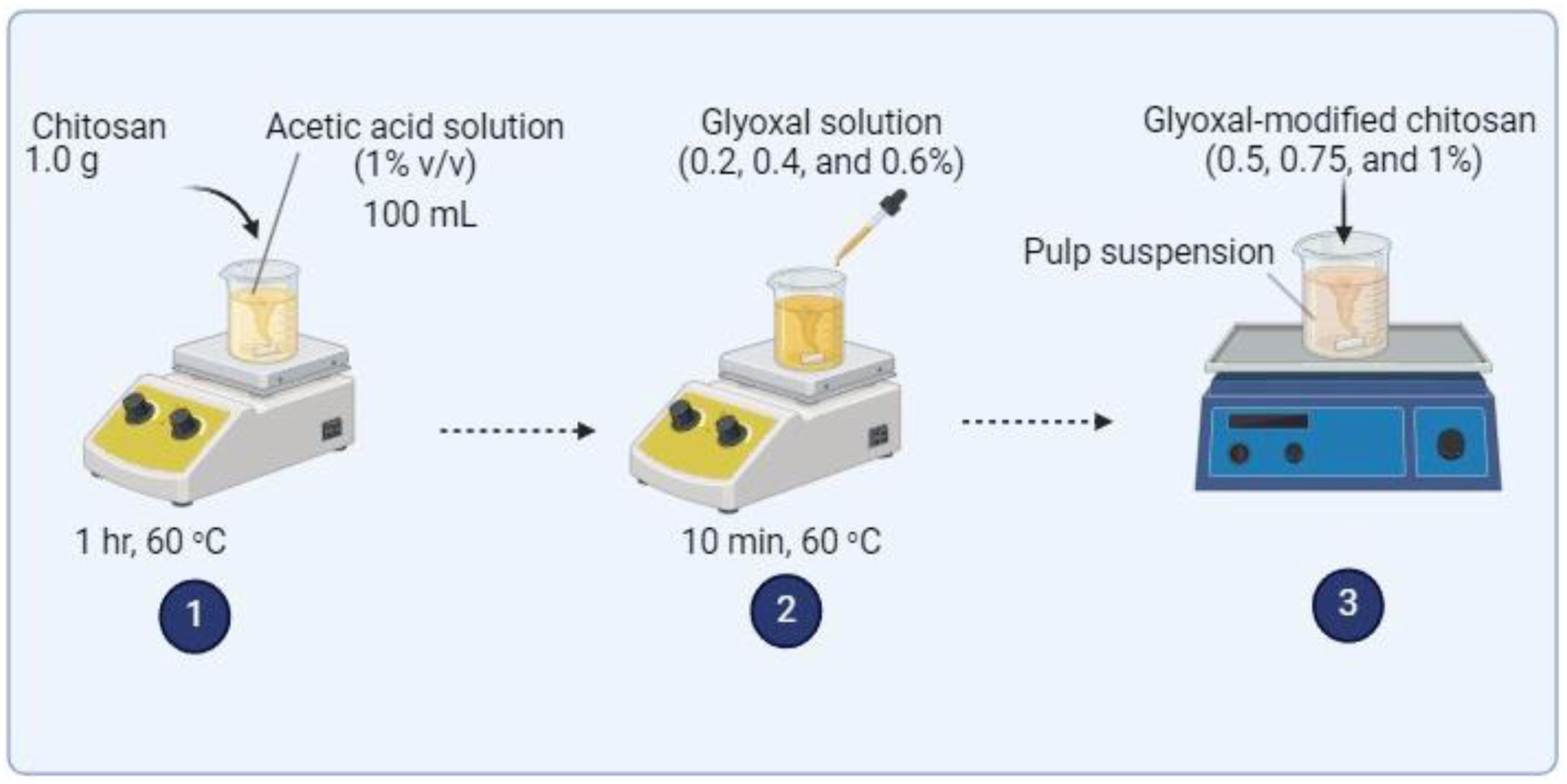
Handsheet preparation and treatment
After the preparation of chitosan and Chi-Gly solutions, they were introduced into the disintegrated pulp suspension of MOW recycled pulp at three dosage levels: 0.5%, 0.75%, and 1%, based on the oven-dried mass of the pulp, following the TAPPI T 200 sp-01 standard. 37 Subsequently, laboratory handsheets (159 mm × 159 mm) with a target basis weight of 80 ± 1 g/m2 were fabricated using a laboratory handsheet maker (FORMAX) following the TAPPI T 205 sp-02 standard. 38 The prepared handsheets were dried using a laboratory rotating dryer at 70°C.
Fourier transform infrared spectroscopy analysis
The infrared spectra of the unmodified chitosan and glyoxal-modified chitosan samples were recorded using a PerkinElmer-100 Spectrum FTIR spectrometer (PerkinElmer, USA). The spectra were obtained in the spectral range of 4000 to 400 cm−1 by employing the potassium bromide (KBr) disc technique. This analytical method facilitated examining the characteristic functional groups and the investigation of potential chemical modifications induced by the glyoxal crosslinking reaction with chitosan.
Scanning electron microscopy
The morphological evaluation of the paper structure was conducted using a Scanning Electron Microscope (SEM, Hitachi, model SU3500, Tokyo, Japan) operated at an acceleration voltage of 20 kV. To prepare the samples, cut patterns of the laboratory handsheet strips were affixed onto sample stages using conductive double-sided adhesive tapes. Subsequently, the samples were coated with a thin layer of gold to prevent electrical charging during the observation.
Contact angle
The wettability of the paper surfaces was characterized by measuring the contact angle of deionized water droplets using the sessile drop technique. An Optical Contact Angle instrument (OCA, Model OCA30\_WT300e, DataPhysics Instruments GmbH, USA) was employed. A 20 μL deionized water droplet was gently deposited onto the surface of both chitosan-treated and untreated paper samples using a micro-syringe. The evolution of the droplet shape was continuously recorded via video capture over time. Subsequently, the acquired video footage was analyzed using the instrument’s image analysis software to determine the dynamic contact angle, droplet diameter, and height at specified time intervals.
Water absorption
The water absorption capacity of the produced paper samples was evaluated in accordance with the ISO 12625-8 standard. This method involves measuring the mass of deionized water absorbed per unit mass of the test piece (75 × 75 mm) under specified conditions and expressing the result as a percentage of the dry paper mass. Furthermore, the time required (in seconds) for complete absorption of deionized water by the paper samples was recorded for different test pieces. This analysis provided insights into the water absorption behavior and kinetics of the paper samples, which can have implications for their end-use applications and performance under various environmental conditions.
Physical and mechanical properties testing
To evaluate the physical, mechanical, and antibacterial properties, the handsheets were conditioned for 24 h at 23 ± 1°C and 50 ± 2% relative humidity following the TAPPI T 402 sp-08 standard. 39 The target properties were measured in accordance with the respective TAPPI standards: pulp water retention value: Um-256 40 ; paper thickness: T 411 Om-10 41 ; burst index: T 403 om-02 42 ; tensile index: T 494 om-01 43 ; wet tensile strength: T456 om-03 44 ; bending resistance (stiffness): T566 om-08 45 ; paper apparent density: T 220 sp-01 37 ; and brightness: T452 om-02. 46
Antibacterial activity
The antibacterial performance of the chitosan-treated and untreated paper samples was evaluated based on the zone of inhibition method. Figure 2 illustrates the methodology used for evaluating the antibacterial activity of the treated paper samples. Escherichia coli (a gram-negative bacterium) and Staphylococcus aureus (a gram-positive bacterium) were selected as model bacterial strains, owing to their ubiquitous presence in daily life. The chitosan-treated and untreated paper samples were cut into squares (1 cm × 1 cm) aseptically placed onto Luria-Bertani (LB) agar media in Petri dishes inoculated with the bacteria solution, with three replicates for each sample. The plates were then incubated at 37°C for 24 h. After the incubation period, the plates were examined visually to observe the presence of inhibition zones and bacterial growth around the paper samples. Diagram of the antibacterial activity testing method.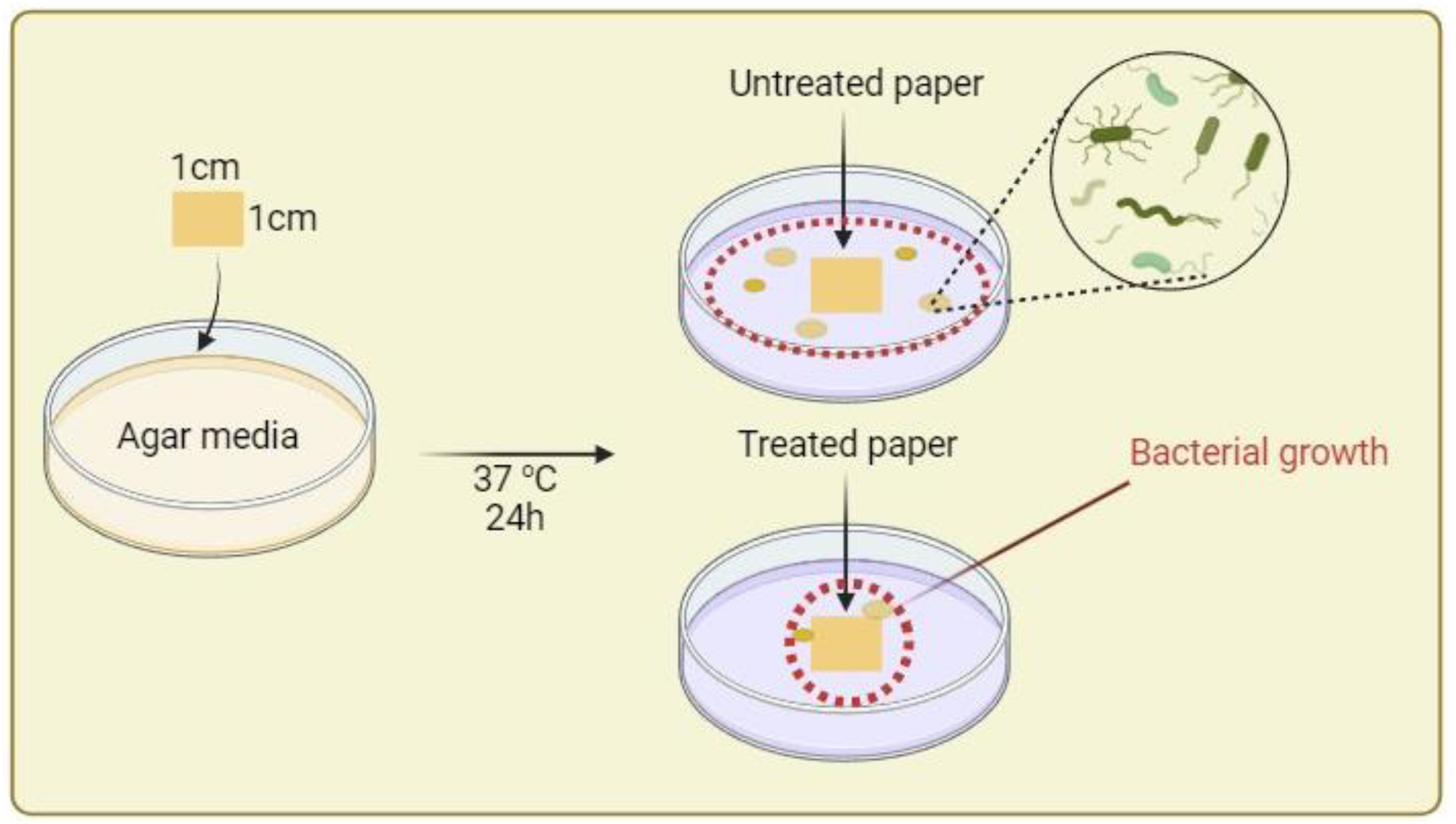
Result and discussion
FTIR analysis
The FTIR spectra presented in Figure 3 provide valuable insights into the structural changes induced by the glyoxal crosslinking of chitosan. The broad peaks observed at 3386 cm−1 (pure chitosan) and 3456 cm−1 (glyoxal-crosslinked chitosan) correspond to the stretching vibrations of the O-H and N-H groups present in the chitosan structure. The peak at 2866 cm−1 is attributed to the stretching vibrations of the C-H bonds in the chitosan backbone. A notable observation is the shift in the peak position related to the stretching vibrations of the C=O groups present in the N-acetyl groups of chitosan. The peak at 1670 cm−1 in pure chitosan is shifted to 1593 cm−1 in the glyoxal-crosslinked chitosan spectrum. This shift may be attributed to the formation of imine (C=N) bonds during the crosslinking reaction between the amino groups of chitosan and the aldehyde groups of glyoxal. The formation of these covalent imine bonds can lead to changes in the vibrational frequencies of the carbonyl groups, resulting in the observed peak shift. The peaks at 1423 cm−1 and 1380 cm−1 are associated with the bending vibrations of the C-H bonds in the chitosan structure and the N-acetyl groups, respectively. The peak at 1157 cm−1 corresponds to the stretching vibrations of the C-O-C glycosidic linkages in the chitosan backbone, while the peak at 1076 cm−1 is related to the stretching vibrations of the C-O bonds in the chitosan structure. The peak at 887 cm−1 is associated with the ring vibrations of the glucosamine units in the chitosan backbone, and the peak at 563 cm−1 is attributed to the bending vibrations of the N-H bonds. The observed shifts in the peak positions and the appearance of new peaks in the glyoxal-crosslinked chitosan spectrum can be attributed to the crosslinking reaction between the amino groups of chitosan and the aldehyde groups of glyoxal. In addition to the formation of imine (C=N) bonds, the potential acetal formation between the aldehyde groups of glyoxal and the hydroxyl groups of chitosan can lead to changes in the vibrational frequencies of the functional groups, resulting in the observed spectral differences. FTIR spectra of pure chitosan and glyoxal-crosslinked chitosan.
These spectral observations are consistent with the findings reported in other published studies investigating the crosslinking of chitosan with glyoxal. For example, Jawad et al. 30 observed similar shifts in the FTIR peaks related to the N-H, C=O, and C-N bonds upon crosslinking chitosan with glyoxal. Yang et al. 29 observed a similar shift in the amide II band (N-H bending vibrations) and a reduction in the intensity of the O-H and N-H stretching vibrations upon glyoxal modification of chitosan fibers, which they attributed to the formation of acetal linkages and potential crosslinking reactions. The FTIR analysis provides valuable insights into the structural changes induced by the glyoxal crosslinking of chitosan, which can contribute to the understanding of its reinforcing and functional properties in the context of paper applications.
Morphological analysis
The SEM micrograph of the control sample (Figure 4(a)) shows the typical fibrous structure of the recycled paper substrate without any additive treatment. The fibers appear loosely arranged, with visible gaps and spaces between the individual fibers. In contrast, the SEM image of the chitosan-treated paper (Figure 4(b)) reveals a more compact and cohesive fiber network. The fibers appear to be coated or bound together by a continuous film-like material, which can be attributed to the presence of chitosan. This chitosan film can act as a binding agent, promoting better fiber-fiber interactions and improving the overall structural integrity of the paper. The SEM micrograph of the Chi-Gly treated paper (Figure 4(c)) exhibits a distinct morphology compared to the other samples. The fibers appear more tightly bound together, with a smoother and more uniform surface texture. This observation suggests that the glyoxal crosslinking of chitosan has further enhanced the binding and film-forming properties of the biopolymer, resulting in a more compact and reinforced fiber network. These observations are consistent with the findings reported in other related studies. Rahmaninia et al.
24
investigated the effect of chitosan and bentonite microparticles on paper reinforcement. Their SEM analysis revealed a similar cohesive fiber network and improved fiber-fiber bonding in the presence of chitosan, attributed to the film-forming ability of the biopolymer. Li et al.
32
studied the grafting of nanocellulose with diethylenetriaminepentaacetic acid and chitosan as an additive for enhancing recycled pulp fibers. Their SEM images showed a more uniform and compact fiber structure in the presence of the chitosan-based additive, indicating improved fiber-fiber bonding and reinforcement. Campano et al.
4
observed a similar enhancement in fiber-fiber bonding and surface coverage in recycled paper treated with bacterial cellulose, which acted as a binding agent similar to chitosan. The observed morphological changes in the SEM images align with the expected behavior of chitosan as a reinforcing agent for paper substrates. The incorporation of chitosan, particularly in its glyoxal-crosslinked form, leads to improved fiber-fiber interactions and a more cohesive fiber network, which can contribute to the enhancement of mechanical properties and overall paper performance. SEM micrographs of (a) control, (b) chitosan-treated, and (c) Chi-Gly treated recycled papers.
Surface properties and wettability
Based on Figure 5, the water contact angle measurements provide insights into the wettability and surface properties of the paper samples treated with chitosan and chitosan-glyoxal (Chi-Gly) compared to the untreated control sample. For the control sample, the initial water contact angle is around 47°, typical for cellulosic materials like paper. As time progresses, the water droplet gradually spreads, and the contact angle decreases, indicating the hydrophilic nature of the paper surface. In the case of the chitosan-treated paper, the initial water contact angle is noticeably higher (around 108°) compared to the control. This increase in the initial contact angle can be attributed to the participation of the high hydroxyl groups of the fibers through the various cross-linked, ionic, and hydrogen bonds provided by chitosan, which offers less hydrophilicity. Furthermore, the water droplet spreads more slowly on the chitosan-treated paper, leading to a slower decrease in the contact angle over time. The Chi-Gly treated paper exhibits even higher initial water contact angles (around 114°) than the control and chitosan-treated samples. This observation suggests that the glyoxal crosslinking of chitosan further reduces the hydrophobicity of the paper surface. The water droplet spreads at a lower rate on the Chi-Gly treated paper, as evident from the slower decrease in the contact angle over time. These findings are consistent with the published literature on using chitosan and glyoxal-chitosan as hydrophobic modifiers for cellulosic materials. Jawad et al.
30
studied the effect of chitosan and glyoxal-crosslinked chitosan on the wettability of cellulose nanofiber films. They observed an extremely low swelling index for films treated with chitosan, which is caused by the blocking of hydrophilic groups such as hydroxyl groups, indicating reduced hydrophobicity. Similarly, Ashori et al.
31
reported a decrease in water contact angles for kenaf paper treated with chitosan, attributing the reduced hydrophilicity to the presence of hydrophilic functional groups and the formation of a uniform coating on the fabric surface. Qin et al.
10
investigated using chitosan and glyoxal-crosslinked chitosan as wet-end additives for pulp molded products. They found that the incorporation of these additives decreased the water absorption and wettability of the molded products and increased water contact angles which is the result of formation of hydrophobic covalent bonds in the paper. The enhanced wettability and hydrophilicity of the paper samples subjected to chitosan and Chi-Gly treatment offer several benefits for various applications. These include printing, coating, and filtration, where quick water absorption and spreading are desirable characteristics. Water contact angle values of control, chitosan-treated, and Chi-Gly treated paper samples.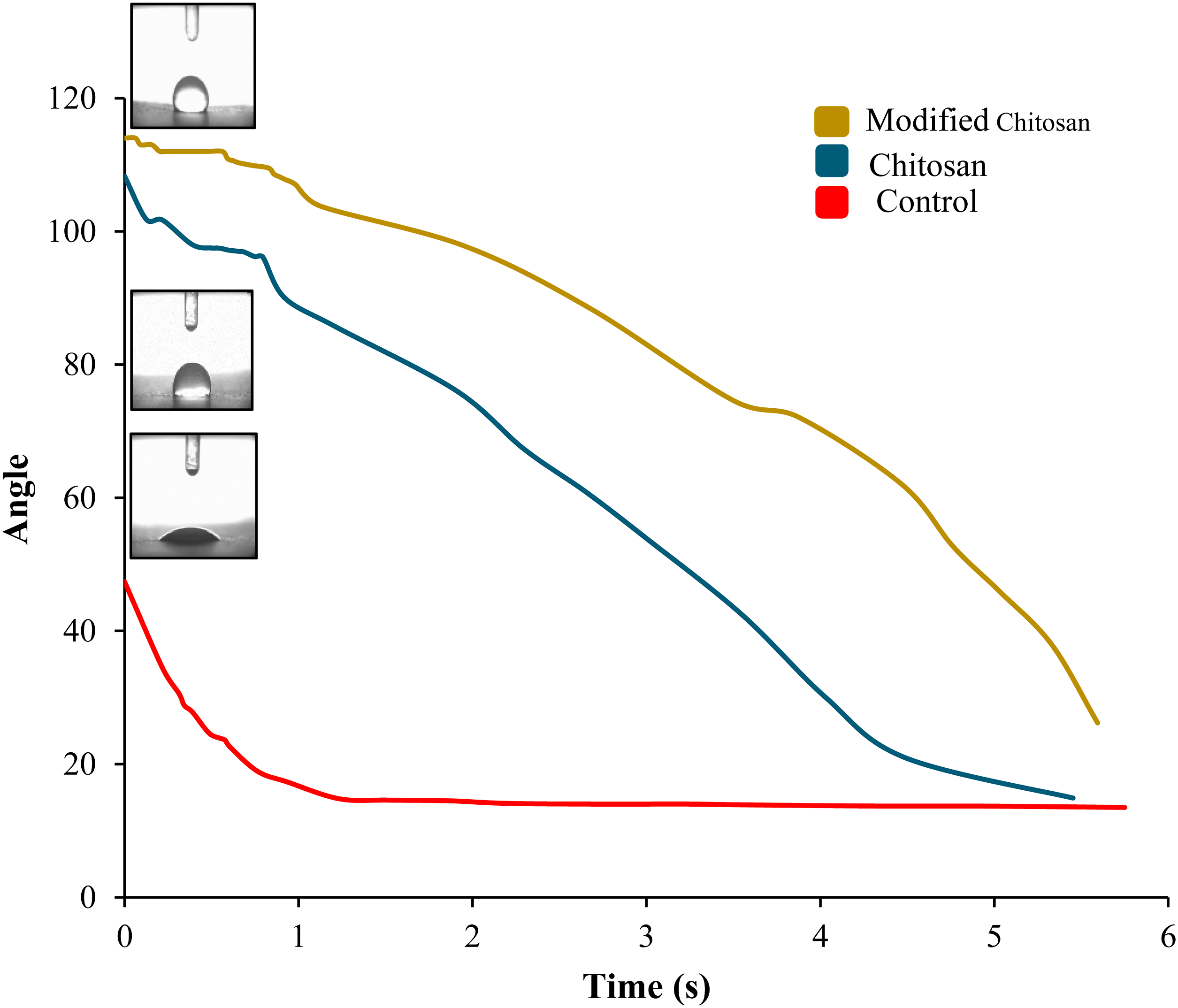
Mechanical properties
Figure 6 illustrates the effect of incorporating chitosan and Chi-Gly on the mechanical properties of recycled paper, including tensile index, burst index, and bending resistance. The dry tensile index represents the tensile strength per unit mass of the paper under dry conditions. From Figure 6, it is evident that the addition of both chitosan and Chi-Gly significantly enhances the dry tensile index compared to the untreated control sample. Moreover, the Chi-Gly treated paper exhibits the highest dry tensile index among all samples, suggesting a synergistic effect of glyoxal crosslinking on the reinforcing ability of chitosan. These findings are consistent with other published studies investigating using chitosan and its derivatives as paper-strength additives.47–49 For instance, Rahmaninia et al.
24
reported a substantial increase in the dry tensile strength of paper treated with a chitosan-bentonite system, attributing the improvement to the reinforcing and film-forming properties of chitosan. Similarly, Li et al.
33
observed enhanced dry tensile strength in recycled OCC pulp fibers treated with grafted nanocellulose and chitosan, further corroborating the effectiveness of chitosan in improving the dry tensile properties of paper. Effect of chitosan and Chi-Gly treatments on the mechanical properties of recycled papers: (a) Dry tensile index, (b) Wet tensile index, (c) Bending resistance, and (d) Burst index.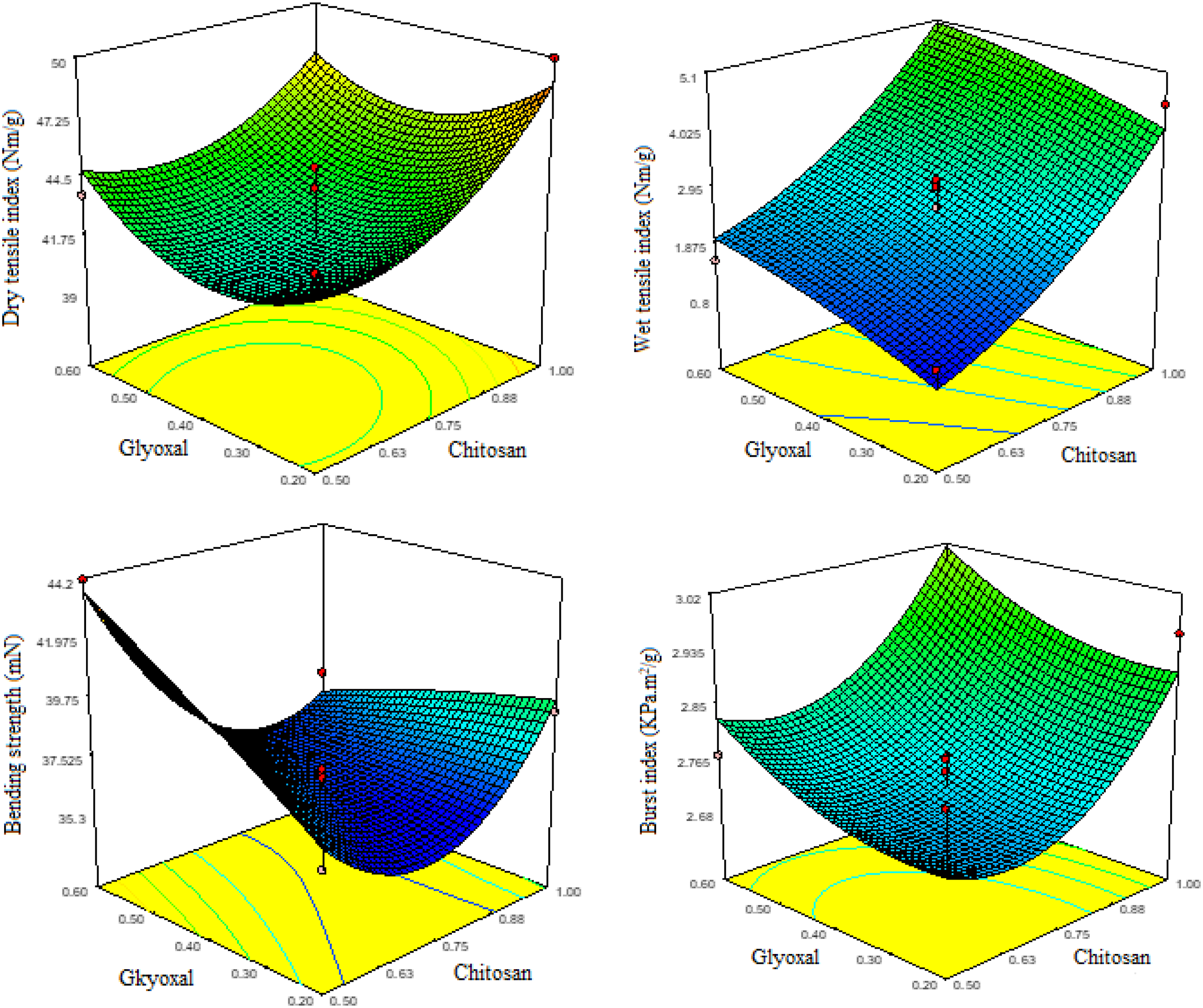
The wet tensile index measures the tensile strength per unit mass of the paper under wet conditions, which is a critical property for various applications, such as packaging and hygiene products. Figure 6 demonstrates that both chitosan and Chi-Gly treatments lead to a significant enhancement in the wet tensile index compared to the untreated control sample. Notably, the Chi-Gly treated paper exhibits the highest wet tensile index among all samples, indicating the superior performance of glyoxal-crosslinked chitosan in improving the wet strength of the paper. These observations align with the findings reported by Mangiante et al., 8 who investigated using chitosan as a wet-strength additive for biohybrid cellulose fibers. They observed a substantial increase in the wet tensile strength of the treated fibers, attributed to the improved fiber-fiber interactions and the formation of a more cohesive fiber network facilitated by chitosan. Additionally, Yun et al. 7 studied using aqueous adhesives containing chitosan for cellulose-based wet wipe substrates. They reported enhanced wet tensile strength, further highlighting the effectiveness of chitosan in imparting wet strength to cellulosic materials. It is worth noting that the improvement in wet tensile index observed for the Chi-Gly treated paper is more pronounced compared to the enhancement in dry tensile index. This behavior can be attributed to the crosslinking effect of glyoxal, which not only reinforces the fiber network but also contributes to forming more stable and water-resistant bonds within the paper structure, thereby enhancing the wet strength properties.
The burst index, which measures the resistance to rupture of the paper per unit mass, is significantly improved by adding chitosan and Chi-Gly compared to the control (untreated) sample. The Chi-Gly treated paper exhibits the highest burst index, indicating a synergistic effect of glyoxal crosslinking on the reinforcing ability of chitosan. This enhancement in burst index can be attributed to the improved fiber-fiber bonding and increased structural integrity of the paper, as observed in the SEM micrographs. These findings are consistent with other studies exploring using chitosan and its derivatives as paper-strength additives. For instance, Rahmaninia et al. 24 reported a substantial increase in the burst strength of paper treated with a chitosan-bentonite system, attributing the improvement to the reinforcing and film-forming properties of chitosan. Similarly, Li et al. 33 observed enhanced burst strength in recycled OCC pulp fibers treated with grafted nanocellulose and chitosan, further corroborating the effectiveness of chitosan in improving the paper’s resistance to rupture.
The bending resistance or stiffness of the paper is significantly enhanced by incorporating chitosan and Chi-Gly. The Chi-Gly treated paper exhibits the highest bending resistance, indicating a more rigid and less flexible structure than the control and chitosan-treated samples. This increased stiffness can be attributed to the crosslinking effect of glyoxal, which promotes stronger inter-fiber bonding and a more compact fiber network, as observed in the SEM micrographs. These findings are consistent with the results reported by Hu et al., 3 who observed improved bending resistance and stiffness in recycled paper treated with cellulose nanofibrils (CNFs) as reinforcing agents. Similarly, Song et al. 32 reviewed the application of chitin/chitosan and their derivatives in papermaking and highlighted their ability to enhance paper stiffness and dimensional stability.
Overall, the results presented in Figure 6 demonstrate the effectiveness of chitosan and glyoxal-crosslinked chitosan as reinforcing agents for recycled paper, leading to significant improvements in burst strength, tensile strength, and bending resistance. The glyoxal crosslinking of chitosan enhances its reinforcing ability, particularly in burst strength and bending resistance. These findings are supported by various published studies investigating the use of chitosan and its derivatives as paper-strength additives, which report similar trends and attribute the improvements to the film-forming, reinforcing, and crosslinking properties of these biopolymers.23,48
Physical properties
Based on Figure 7, it appears to present data on various physical properties of the recycled paper samples, including apparent density, brightness, and water absorption. I will analyze each property separately and compare the results with relevant published studies. Effect of chitosan and Chi-Gly treatments on the physical properties of recycled papers: (a) Apparent density, (b) Brightness, and (c) Water absorption.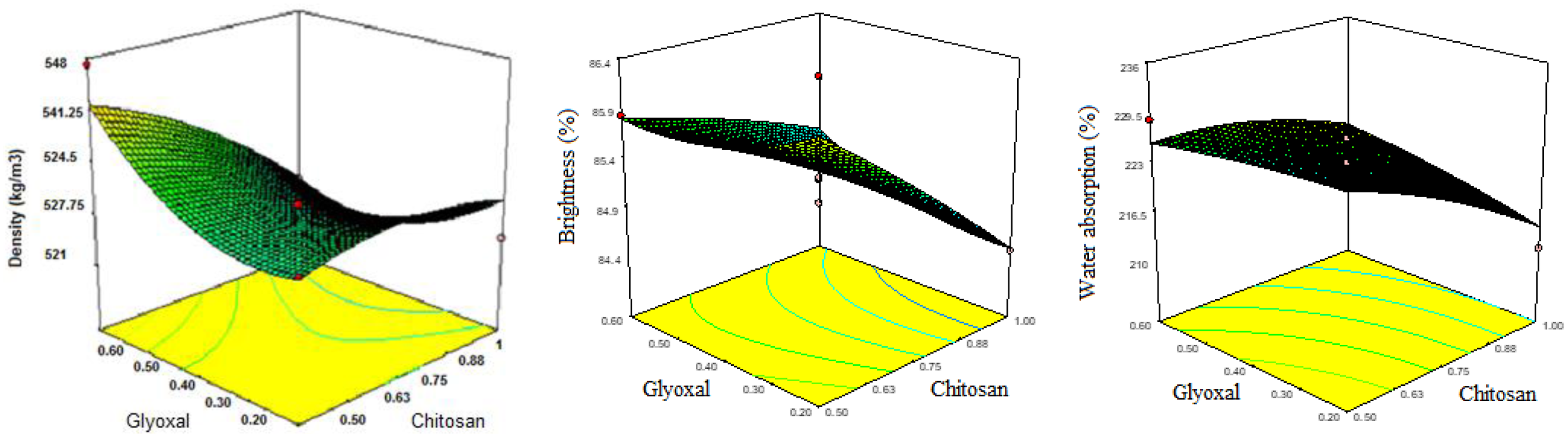
The apparent density of the paper samples, a measure of the mass per unit volume, exhibits a slight increase for the chitosan and Chi-Gly treated papers compared to the control.
This increase in apparent density can be attributed to forming a more compact and cohesive fiber network facilitated by the reinforcing and film-forming properties of chitosan and its glyoxal-crosslinked derivative. While there are limited studies specifically reporting the effect of chitosan on the apparent density of paper, the observed trend is consistent with the general understanding that the incorporation of additives and the formation of a more compact fiber structure can lead to an increase in the apparent density of the paper.
The brightness of the paper samples, measured on a scale with higher values indicating greater brightness, shows a slight decrease with the addition of chitosan and Chi-Gly (glyoxal-crosslinked chitosan) compared to the untreated control. However, the reduction in brightness is relatively small, suggesting that incorporating these biopolymers does not significantly affect the optical properties of the recycled paper. These findings are consistent with the observations reported by Rahmaninia et al., 24 who noted that the brightness of paper treated with a chitosan-bentonite system remained relatively unchanged compared to the untreated control. Similarly, Li et al. 33 found that the brightness of recycled OCC pulp fibers was not significantly impacted by adding grafted nanocellulose and chitosan.
The water absorption, which quantifies the amount of water absorbed per unit mass of the paper, shows a notable decrease for the chitosan and Chi-Gly treated samples compared to the control. The Chi-Gly treated paper exhibits the highest water absorption capacity, suggesting that the glyoxal crosslinking further reduces the water-absorbing ability of chitosan. These observations are consistent with the findings of Qin et al., 10 who investigated using chitosan and glyoxal-crosslinked chitosan as wet-end additives for pulp molded products. They reported decreased water absorption and wettability for the treated products, which they attributed to the block of hydrophilic functional groups.
Overall, the results presented in Figure 7 provide valuable insights into the physical properties of recycled paper treated with chitosan and glyoxal-crosslinked chitosan. The findings related to the apparent density and water absorption capacity are well-supported by other published studies investigating the use of these biopolymers as hydrophobic modifiers and reinforcing agents for cellulosic materials. The observed trends in brightness suggest that incorporating chitosan and its derivatives does not significantly impact these optical properties of the recycled paper.
Antibacterial properties
Figure 8 presents the antibacterial activity of the paper samples against E. coli (a gram-negative bacterium) and S. aureus (a gram-positive bacterium). The images show the presence or absence of bacterial growth around the paper samples placed on agar plates. For the control sample (untreated paper), there is no visible inhibition zone, indicating that bacterial growth occurred around the paper samples for both E. coli and S. aureus. This observation is expected since the control paper contains no antibacterial agents. In contrast, the chitosan-treated paper samples exhibit a clear inhibition zone around the paper samples for both bacterial strains. This inhibition zone indicates the prevention of bacterial growth in the surrounding area, which can be attributed to the well-known antibacterial properties of chitosan. The Chi-Gly treated paper samples also show distinct inhibition zones against both E. coli and S. aureus. Notably, the inhibition zones appear larger and more pronounced than those observed for the chitosan-treated samples. The antibacterial activity of chitosan is primarily due to its polycationic nature, which enables electrostatic interactions with the negatively charged bacterial cell membrane, leading to membrane disruption and leakage of intracellular components.
47
The superior antibacterial performance of the Chi-Gly treated samples can be attributed to the crosslinking effect of glyoxal, which may enhance the biocidal activity of chitosan by improving its stability and prolonging its release from the paper matrix. These findings are consistent with other published studies that have explored the antibacterial properties of chitosan and glyoxal-crosslinked chitosan in various applications. Lan et al.
35
reported durable antibacterial and antiviral activity against E. coli and influenza viruses for cellulosic fibers modified with chitosan oligosaccharides. They observed larger inhibition zones and longer-lasting antibacterial effects compared to untreated fibers. Jawad et al.
30
demonstrated the antibacterial efficacy of cellulose nanofiber films treated with chitosan and glyoxal-crosslinked chitosan against S. aureus and E. coli. They found that the glyoxal-crosslinked chitosan films exhibited superior antibacterial activity compared to pure chitosan films. Jie et al.
28
developed a chitosan-urushiol nanofiber membrane with enhanced antimicrobial properties against E. coli and S. aureus. They attributed the improved antibacterial performance to the synergistic effect of chitosan and urushiol, as well as the crosslinking effect of urushiol, which enhanced the stability and sustained release of the biocidal components. The observed antibacterial activity of the chitosan and Chi-Gly treated paper samples in this study highlights the potential of these biopolymers as eco-friendly antibacterial agents for various applications, including packaging, hygiene products, and medical materials, where preventing bacterial growth is crucial. Antibacterial activity against E. coli and S. aureus (a) for control, (b) chitosan-treated, and Chi-Gly treated papers.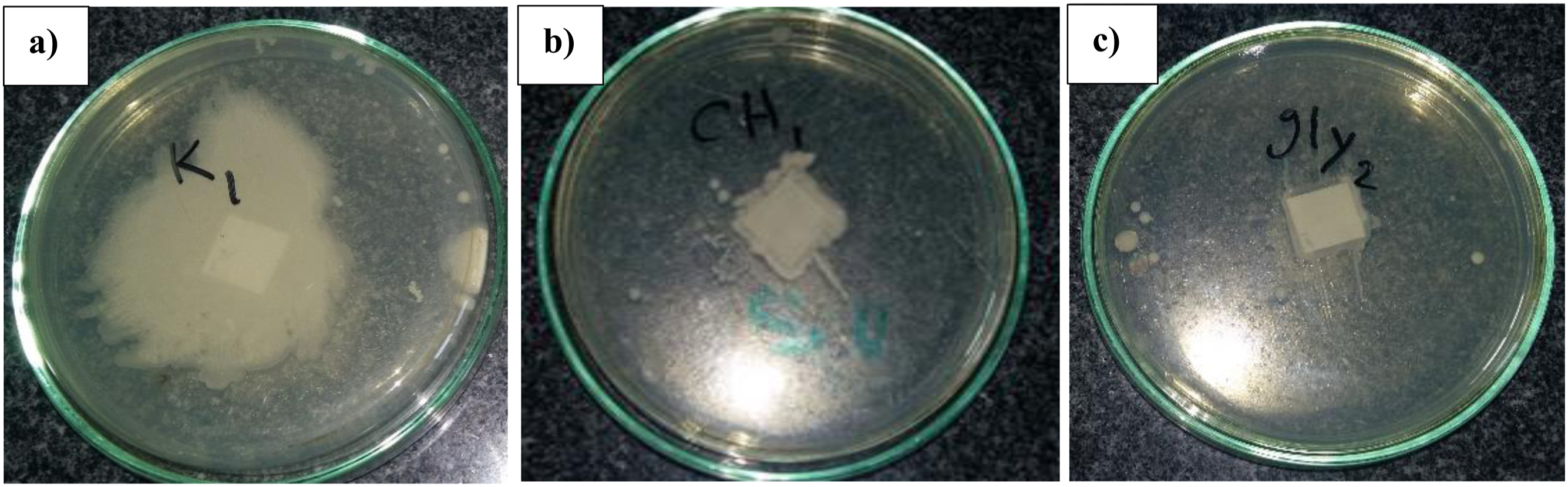
Conclusions
Incorporating chitosan and glyoxal-crosslinked chitosan (Chi-Gly) as reinforcing agents significantly improved the mechanical properties of recycled paper, including tensile strength, burst strength, and bending resistance. Notably, the Chi-Gly treated paper exhibited the highest improvement in these properties, indicating a synergistic effect of glyoxal crosslinking on the reinforcing ability of chitosan. The wet tensile strength of the recycled paper was substantially enhanced by adding chitosan and Chi-Gly, with the Chi-Gly treated paper exhibiting the highest wet tensile index. This improvement can be attributed to forming more stable and water-resistant bonds within the paper structure facilitated by glyoxal crosslinking. The scanning electron microscopy (SEM) analysis unveiled a denser and more interconnected network of fibers in the papers treated with chitosan and Chi-Gly. Notably, the Chi-Gly treated paper displayed the smoothest and most homogeneous surface texture, suggesting enhanced fiber-fiber bonding and reinforcement. The water contact angle measurements demonstrated improved hydrophobicity and wettability of the recycled paper treated with chitosan and Chi-Gly, with the Chi-Gly treated paper exhibiting the lowest initial water contact angle and fastest spreading rate. The chitosan and Chi-Gly treated papers exhibited distinct zones of inhibition against both gram-negative (Escherichia coli) and gram-positive (Staphylococcus aureus) bacteria, indicating effective antibacterial activity. The Chi-Gly treated paper displayed larger and more pronounced inhibition zones, suggesting enhanced antibacterial performance due to the cross-linking effect of glyoxal. The water absorption capacity of the recycled paper was significantly improved by incorporating chitosan and Chi-Gly, with the Chi-Gly treated paper exhibiting the highest water absorption capacity. The brightness of the recycled paper was not significantly affected by the addition of chitosan and Chi-Gly, indicating that these biopolymers do not negatively impact the optical properties of the paper.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
